Abstract
Three commonly used Mycobacterium bovis BCG vaccine strains elicited different magnitudes of T-cell activation and gamma interferon production in vitro in healthy BCG-vaccinated individuals. Glaxo 1077 exhibited the greatest stimulatory capacity, followed by Pasteur 1173 and then Danish 1331. These differences may affect in vitro stimulation and vaccination-induced immunogenicity.
Mycobacterium bovis bacillus Calmette-Guérin (BCG) is the only vaccine currently available to prevent tuberculosis. It is administered to over 100 million people per year (25), although its efficacy against pulmonary TB worldwide is variable (11). M. bovis BCG is an attenuated form of M. bovis obtained by 13 years of serial passage in vitro, following which it was found to provide protective immunity to challenge with virulent M. tuberculosis (3). M. bovis BCG was maintained by a process of serial passage for 40 years before methods for stable long-term storage became available, and this culminated in a family of approximately 13 daughter strains which are collectively known today by the generic term BCG. These strains have been shown to differ genetically (2, 5, 6, 22) and phenotypically (1, 14, 20, 21), including their induction of immune responses and protection in animal models (16, 18). Three strains represent nearly 60% of the total number of the BCG doses currently produced per year (12): BCG Danish 1331 is currently in use in the United Kingdom vaccination program, although BCG Glaxo 1077 was used until 2002. BCG Pasteur 1173 is the BCG reference strain lyophilized at the Institute Pasteur in 1961.
Despite the numerous differences previously found among BCG strains, and despite BCG stimulation being routinely used to study mycobacterium-specific immune responses, T-cell responses elicited by different BCG strains have not been compared in human cultures and were therefore investigated in this study.
The BCG vaccine strains Danish 1331, Pasteur 1173, and Glaxo 1077 were grown in liquid cultures of Middlebrook 7H9 medium (Difco) supplemented with 10% Bacto Middlebrook ADC (Scientific Laboratory Supplies) and 0.025% Tween 80 (Difco), quantified by growth on Middlebrook 7H10 agar plates for 3 weeks, and kept frozen at −80°C for long-term storage. All three strain preparations were found to be 90 to 95% viable, as determined by fluorescein diacetate/ethidium bromide labeling. Peripheral blood mononuclear cells (PBMC) were isolated by density gradient centrifugation from seven healthy BCG-vaccinated donors, at least five of whom were likely to have been vaccinated with Glaxo 1077, and were cultured for 6 days either in the absence of antigenic stimulation or in the presence of one of the live M. bovis BCG strains titrated between 0.01 and 1 CFU per PBMC: if monocytes are estimated to constitute 10% of total PBMC, this is the equivalent of multiplicity of infection (MOI) ratios (M. bovis BCG bacilli to monocytes) of 0.1:1 to 10:1. For flow cytometry, cells were stained with fluorescently labeled monoclonal antibodies against CD3 (allophycocyanin conjugated), CD8 (peridinin chlorophyll protein conjugated), and CD25 (fluorescein isothiocyanate conjugated), fixed and permeabilized using reagents purchased from Caltag, and then stained intracellularly for IFN-γ (phycoerythrin conjugated). Antibodies were all from BD Biosciences (Cowley, United Kingdom). Flow cytometric data were acquired (40,000 events within lymphocyte gate) using a FACSCalibur flow cytometer (BD Biosciences) and analyzed using CellQuest (BD Biosciences). CD3+ CD8− cells were 95% CD4+ and are referred to herein as CD4+ T cells. Ethical permission was obtained from the London School of Hygiene & Tropical Medicine Ethics committee, and written informed consent was gained from the donors.
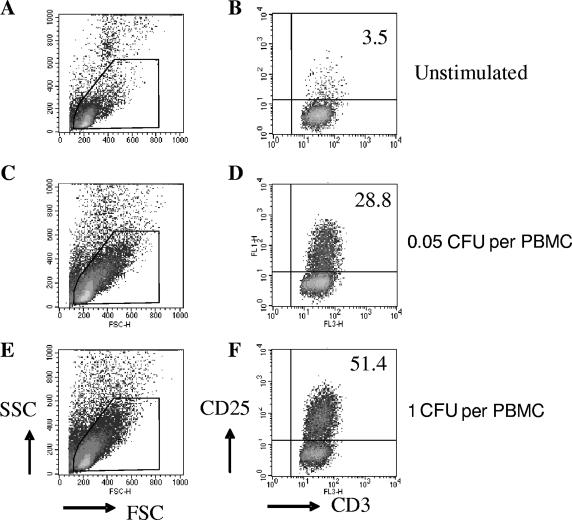
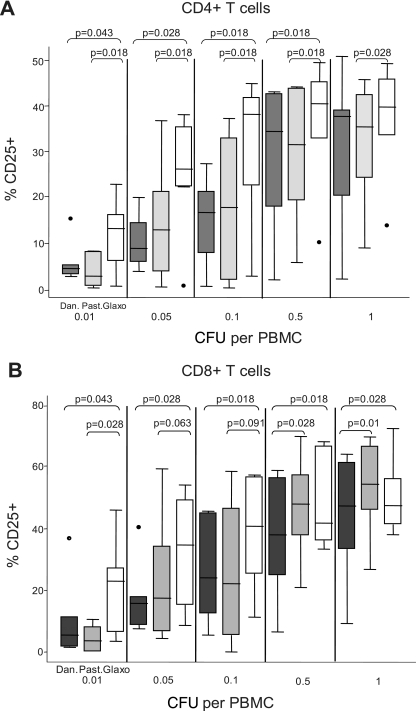
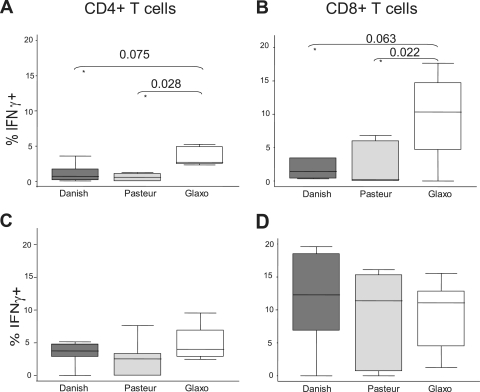
T cells were activated by in vitro stimulation with all three strains of BCG. A progressive increase of both light scatter parameters was observed as the dose of M. bovis BCG increased (Fig. 1, left). Expression of the activation marker CD25 also increased gradually with BCG dose (Fig. 1, right). These data are concordant with numerous previous studies (8, 10, 19, 23, 24). The three BCG strains tested differed significantly in the percentage of activated CD25+ T cells they elicited in vitro. Danish 1331 induced the lowest activation response overall, whereas Glaxo 1077 induced the highest, with statistically significant increases at most doses in both the CD4+ and CD8+ T-cell populations (Fig. 2A and B). The average rank of CD25+ response for each strain for each donor was determined across the BCG doses, and the overall difference between Glaxo 1077 and Danish 1331 was highly significant (P = 0.001316, paired Student's t test), as was the difference between Glaxo 1077 and Pasteur 1173 (P = 0.000121), within the CD4+-T-cell compartment. The proportions of CD8+ T cells which were CD25+ were also significantly higher with Glaxo 1077 stimulation than with Danish 1331 stimulation (P = 0.00028) in the BCG strain rank analysis. Interestingly, statistical significance (P = 0.013853) was observed between Pasteur 1173 and Glaxo 1077 only when the lowest three doses were included, and the effect was diminished with inclusion of the higher doses. Similarly, the BCG strains varied in their induction of IFN-γ expression. At a low dose, Glaxo 1077 elicited the highest number of IFN-γ-producing T cells and Danish 1331 and Pasteur 1173 induced lower responses (Fig. 3A and B).
FIG. 1.
Scatter characteristics and CD25 expression in PBMC cultures stimulated with live M. bovis BCG. Cultured cells are shown according to both forward and side light scatter parameters (left). Representative examples from one of the donors tested when cells were cultured in the absence of stimulation (A, B) or in the presence of titrated amounts of Danish 1331 (C to F) are shown. CD3+ cells were selected within the live-lymphocyte gate (R1). Expression of the activation marker CD25 is shown for the CD3+ CD8+ T-cell population (right), with the numbers showing the percentage of CD8+ T cells which were activated.
FIG. 2.
CD25 expression induced in vitro by different BCG strains in CD4+ and CD8+ T cells. Percentages of CD4+ (A) and CD8+ (B) T cells expressing the activation marker CD25, as a result of in vitro BCG stimulation, are shown. The horizontal axis is divided into the five BCG doses tested. Within each of the five doses, the results for each of the three BCG strains tested are shown: Danish 1331 (Dan., black), Pasteur 1173 (Past., gray) and Glaxo 1077 (Glaxo, white). Lines within the boxes represent median values. Upper and lower hinges of the boxes represent 75th and 25th percentiles, respectively. Adjacent lines represent the adjacent values, and dots represent outliers. Statistically significant differences (P < 0.05) and borderline differences (0.05 < P < 0.1) between strains are indicated by their Wilcoxon signed-rank test P values.
FIG. 3.
IFN-γ expression induced by BCG stimulation. Percentages of IFN-γ-producing T cells following BCG stimulation with a low dose of 0.01 CFU/PBMC (A, B) or intermediate dose of 0.1 CFU/PBMC (C, D) are shown for the CD4+ (A, C) and CD8+ (B, D) T-cell compartments. The horizontal axes show the three strains tested. Lines within the boxes represent median values, and upper and lower hinges of the boxes represent 75th and 25th percentiles, respectively. Adjacent lines represent adjacent values, and dots represent outliers. Statistically significant differences (P < 0.05) and borderline differences (0.05 < P < 0.1) between strains are indicated by their Wilcoxon signed-rank test P values.
Activation and IFN-γ production showed a trend to plateau with increasing MOIs of BCG, in both the CD4+- and CD8+-T-cell compartments. At the intermediate dose of 0.1 CFU/PBMC, similar IFN-γ responses were elicited by all three BCG strains (Fig. 3C and D), and responses to each strain reached a similar maximum level. This suggests that with higher MOIs, the BCG-specific immune response is saturated but that other factors might be critical at lower doses when differences between strains are more pronounced.
Glaxo 1077 and Pasteur 1173 have been found to provide higher levels of acquired protection than Danish 1331 and Japanese strains in mice (13), although another study did not find a significant difference between Glaxo and Pasteur strains (9). Comparing vaccination studies in humans is difficult, since not only have they used different BCG strains but also most were carried out in different geographical locations and populations. At least five of the donors studied here were likely to have been immunized with Glaxo 1077. When Danish 1331 and Glaxo 1077 were compared in the United Kingdom during the 1960s and 1970s, no significant differences in protective efficacy were found (7), although the Danish strain gave consistently higher mean tuberculin skin test reactions and greater vaccination scar sizes (17). In more recent studies carried out with United Kingdom schoolchildren, Danish 1331 induced consistently lower skin test reactions, scar sizes, and IFN-γ production in a whole-blood assay than Glaxo 1077, although these differences were not statistically significant (15). Thus, although there is no epidemiological evidence for strain differences in the induction of protection against tuberculosis infection and no evidence of genetic differences between the Glaxo and Danish strains (6), the BCG strain used for vaccination may affect the extent of the BCG-induced immune response induced.
Interestingly, an inverse relationship between passage number before cryopreservation of seed lot (given as the strain number) and activation response was suggested in this study. These results fit with the observation that laboratory adaptation of bacteria is associated with attenuation of virulence, and the lower T-cell immunogenicity of Danish 1331 might be a consequence of this attenuation. A relationship between in vitro passage number and vaccine efficacy was suggested previously (4). Another factor that might affect T-cell activation is the conditions of BCG culture: we chose to passage all three strains in the same ADC-supplemented Middlebrook 7H9 broth to minimize this effect on our T-cell stimulation assays. The number of dead bacilli in BCG preparations may also affect CD4+-T-cell responses, but their effect on CD8+ T cells would be less pronounced (24).
In conclusion, different BCG vaccine strains elicited quantitatively different T-cell responses in human in vitro assays when healthy BCG-vaccinated individuals were tested. The magnitude of T-cell responses, measured as the proportion of T cells that expressed the activation marker CD25 and IFN-γ in both CD4+ and CD8+ T cells, elicited by the three BCG strains tested was ranked in decreasing order as follows: Glaxo 1077, Pasteur 1173, and Danish 1331. These and other observations suggest that there may be strain-dependent differences between commonly used BCG vaccines which may affect the immune responses they induce in vitro and those they induce in vivo following immunization.
Acknowledgments
This work was supported by an Action TB grant from GlaxoSmithKline.
We thank Carolynne Stanley for phlebotomy assistance and Sian Floyd for statistical advice.
Editor: J. L. Flynn
Footnotes
Published ahead of print on 26 March 2007.
REFERENCES
- 1.Abou-Zeid, C., I. Smith, J. Grange, J. Steele, and G. Rook. 1986. Subdivision of daughter strains of bacille Calmette-Guerin (BCG) according to secreted protein patterns. J. Gen. Microbiol. 132:3047-3053. [DOI] [PubMed] [Google Scholar]
- 2.Bedwell, J., S. K. Kairo, M. A. Behr, and J. A. Bygraves. 2001. Identification of substrains of BCG vaccine using multiplex PCR. Vaccine 19:2146-2151. [DOI] [PubMed] [Google Scholar]
- 3.Behr, M. A. 2002. BCG—different strains, different vaccines? Lancet Infect. Dis. 180:141-149. [DOI] [PubMed] [Google Scholar]
- 4.Behr, M. A., and P. M. Small. 1997. Has BCG attenuated to impotence? Nature 389:133-134. [DOI] [PubMed] [Google Scholar]
- 5.Behr, M. A., and P. M. Small. 1999. A historical and molecular phylogeny of BCG strains. Vaccine 17:915-922. [DOI] [PubMed] [Google Scholar]
- 6.Behr, M. A., M. A. Wilson, W. P. Gill, H. Salamon, G. K. Schoolnik, S. Rane, and P. M. Small. 1999. Comparative genomics of BCG vaccines by whole-genome DNA microarray. Science 284:1520-1523. [DOI] [PubMed] [Google Scholar]
- 7.Brewer, T. F. 2000. Preventing tuberculosis with bacillus Calmette-Guerin vaccine: a meta-analysis of the literature. Clin. Infect. Dis. 31(Suppl. 3):S64-S67. [DOI] [PubMed] [Google Scholar]
- 8.Canaday, D. H., C. Ziebold, E. H. Noss, K. A. Chervenak, C. V. Harding, and W. H. Boom. 1999. Activation of human CD8+ alpha beta TCR+ cells by Mycobacterium tuberculosis via an alternate class I MHC antigen-processing pathway. J. Immunol. 162:372-379. [PubMed] [Google Scholar]
- 9.Collins, F. M. 1985. Protection to mice afforded by BCG vaccines against an aerogenic challenge by three mycobacteria of decreasing virulence. Tubercle 66:267-276. [DOI] [PubMed] [Google Scholar]
- 10.Esin, S., G. Batoni, G. Kallenius, H. Gaines, M. Campa, S. B. Svenson, R. Andersson, and H. Wigzell. 1996. Proliferation of distinct human T cell subsets in response to live, killed or soluble extracts of Mycobacterium tuberculosis and Myco. avium. Clin. Exp. Immunol. 104:419-425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fine, P. E. 1995. Variation in protection by BCG: implications of and for heterologous immunity. Lancet 346:1339-1345. [DOI] [PubMed] [Google Scholar]
- 12.Fine, P. E. M., I. A. M. Carneiro, J. B. Milstien, and J. Clements. 1999. Issues relating to the use of BCG in immunization programmes. Department of Vaccines and Biologicals, World Health Organization, Geneva, Switzerland.
- 13.Gheorghiu, M., and P. H. Lagrange. 1983. Viability, heat stability and immunogenicity of four BCG vaccines prepared from four different BCG strains. Ann. Immunol. (Paris) 134C:125-147. [DOI] [PubMed] [Google Scholar]
- 14.Gobin, J., D. K. Wong, B. W. Gibson, and M. A. Horwitz. 1999. Characterization of exochelins of the Mycobacterium bovis type strain and BCG substrains. Infect. Immun. 67:2035-2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gorak-Stolinska, P., R. E. Weir, S. Floyd, M. K. Lalor, S. Stenson, K. Branson, R. Blitz, S. Luke, B. Nazareth, A. Ben-Smith, P. E. Fine, and H. M. Dockrell. 2006. Immunogenicity of Danish-SSI 1331 BCG vaccine in the UK: comparison with Glaxo-Evans 1077 BCG vaccine. Vaccine 24:5726-5733. [DOI] [PubMed] [Google Scholar]
- 16.Grange, J. M., and J. A. Gibson. 1986. Strain to strain variation in the immunogenicity of BCG. Dev. Biol. Stand. 58:37-41. [PubMed] [Google Scholar]
- 17.Horwitz, O., and K. Bunch-Christensen. 1972. Correlation between tuberculin sensitivity after 2 months and 5 years among BCG vaccinated subjects. Bull. W. H. O. 47:49-58. [PMC free article] [PubMed] [Google Scholar]
- 18.Lagranderie, M., A. Balazuc, E. Deriaud, C. Leclerc, and M. Gheorghiu. 1996. Comparison of immune responses of mice immunized with five different Mycobacterium bovis BCG vaccine strains. Infect. Immun. 64:1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lewinsohn, D. M., M. R. Alderson, A. L. Briden, S. R. Riddell, S. G. Reed, and K. H. Grabstein. 1998. Characterization of human CD8+ T cells reactive with Mycobacterium tuberculosis-infected antigen-presenting cells. J Exp. Med. 187:1633-1640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li, H., J. C. Ulstrup, T. O. Jonassen, K. Melby, S. Nagai, and M. Harboe. 1993. Evidence for absence of the MPB64 gene in some substrains of Mycobacterium bovis BCG. Infect. Immun. 61:1730-1734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miura, K., S. Nagai, M. Kinomoto, S. Haga, and T. Tokunaga. 1983. Comparative studies with various substrains of Mycobacterium bovis BCG on the production of an antigenic protein, MPB70. Infect. Immun. 39:540-545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mostowy, S., A. G. Tsolaki, P. M. Small, and M. A. Behr. 2003. The in vitro evolution of BCG vaccines. Vaccine 21:4270-4274. [DOI] [PubMed] [Google Scholar]
- 23.Smith, S. M., A. S. Malin, T. Pauline, Lukey, S. E. Atkinson, J. Content, K. Huygen, and H. M. Dockrell. 1999. Characterization of human Mycobacterium bovis bacille Calmette-Guerin-reactive CD8+ T cells. Infect. Immun. 67:5223-5230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Turner, J., and H. M. Dockrell. 1996. Stimulation of human peripheral blood mononuclear cells with live Mycobacterium bovis BCG activates cytolytic CD8+ T cells in vitro. Immunology 87:339-342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.World Health Organization. 2004. Vaccine-preventable diseases: monitoring system global summary. World Health Organization, Geneva, Switzerland.