Abstract
Nocardia asteroides was isolated after prolonged culture from the pericardial fluid of a human immunodeficiency virus-infected patient. The lengthy duration required for culture growth and identification of this N. asteroides isolate affected both initial therapeutic decisions and patient management. A proposed algorithm for the microbiological workup of pericardial fluid for possible Nocardia spp. is described in an effort to improve the timeliness of results.
CASE REPORT
The patient was a 44-year-old Filipino male with a medical history significant for newly diagnosed human immunodeficiency virus (HIV) infection and AIDS. In March 2005, he was admitted to the hospital for persistent fever of unknown origin. He was found to have HIV on this admission, and the CD4 cell count was 42/mm3. The initial workup included sputum Gram stain, silver stain, and culture, which showed only normal flora. In addition, sputum acid-fast bacillus (AFB) stain and culture were negative for Mycobacterium spp. and routine urine and blood cultures were also negative. During this admission, he was noted to have a pericardial effusion, which prompted a pericardiocentesis. Direct Gram and AFB stains on the pericardial fluid were negative, and no significant growth was detected on routine bacterial cultures. After pericardiocentesis, a repeat echocardiogram showed no pericardial fluid and the patient was discharged in stable condition. However, the AFB culture of the pericardial fluid subsequently became positive at week 6. Both the AFB and modified AFB stains were positive, and the morphology at that time was interpreted as being consistent with Mycobacterium spp. The specimen was sent to a local reference laboratory for further workup. The patient was empirically treated with isoniazid, rifampin, pyrazinamide, and ethambutol for possible Mycobacterium tuberculosis, and he was discharged on this four-antimycobacterial-drug regimen, as well as trimethroprim-sulfamethoxazole (TMP-SMX) and azithromycin for empirical coverage of Pneumocystis jiroveci and Mycobacterium avium complex. Highly active antiretroviral therapy was not successfully initiated due to poor drug compliance.
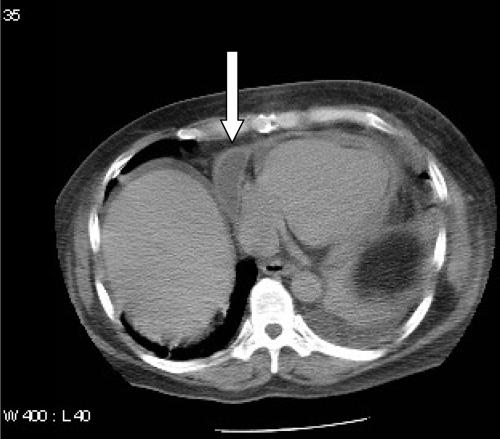
The patient continued to have persistent fever, fatigue, and weakness. Two weeks later, he was readmitted to the hospital with a fever of 102.6°F and severe tachycardia (heart rate of 144 beats/min). His blood pressure on admission was 128/93. A chest X-ray showed a widened mediastinum, and the echocardiogram showed a moderate pericardial effusion which appeared loculated on a subsequent computed tomography (CT) scan (Fig. 1). By this time, the additional workup performed by the local reference lab on the pericardial fluid taken during the first admission demonstrated a partially acid-fast-positive bacillus with infrequent branching. In addition, the culture of the same pericardial fluid showed tiny, dry, white colonies resembling Nocardia spp. on sheep blood agar. The specimen was then sent to a second reference lab for bacterial identification and susceptibility testing. The final identification at 9 weeks post-collection of the pericardial fluid isolate was Nocardia asteroides complex. Susceptibility testing was performed using broth microdilution. The isolate was imipenem resistant (16 μg/ml), clarithromycin intermediate (4 μg/ml), sulfamethoxazole sensitive (16 μg/ml), and TMP-SMX sensitive (<0.25/4.8 μg/ml). Sputum for AFB culture and smear collected 5 weeks after the pericardial fluid was also found to be positive for Nocardia asteroides complex. Based upon these results, antimycobacterial drugs were discontinued and intravenous TMP-SMX was initiated. The patient required surgical drainage of the pericardial effusion (approximately 800 μl) as well as a pericardial window. The patient recovered slowly but uneventfully and was discharged on oral TMP-SMX therapy.
FIG. 1.
A CT scan of the chest showing a large loculated pericardial effusion in the patient.
Microbiology.
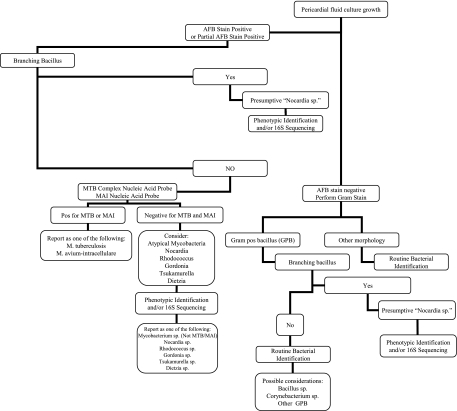
Nocardia spp. can be presumptively identified using a variety of methods including Gram stain, modified AFB stain, and culture morphology. Usually the Gram stain shows branching gram-positive bacilli, which along with a positive modified AFB stain supports a preliminary identification of Nocardia spp. If the organism is weakly acid fast and does not exhibit branching, other bacteria such as Rhodococcus, Gordonia, Tsukamurella, and Dietzia spp. should be considered (32) (Fig. 2). In the present case, Mycobacterium sp. was suspected due to the robustness of the acid-fast staining and the initial absence of branching. Nocardia sp. was initially cultured using a BacT/ALERT (Organon Teknika Corp., bioMerieux, Durham, NC) mycobacterial identification system. Bacterial identification by the second reference laboratory included high-pressure liquid chromatography profiling and biochemical testing (i.e., decomposition of casein, tyrosine, and xanthine).
FIG. 2.
Algorithm for the identification of Nocardia spp., mycobacteria, and other bacteria. Abbreviations: pos, positive; MTB, M. tuberculosis; MAC, M. avium complex; MAI, M. avium-intracellulare.
A report by Isner et al. (12) suggests that the Gram stain is the most sensitive method to visualize and recognize Nocardia spp. based upon the authors’ high percentage (78%) of detection in expectorated sputum from non-HIV patients using the Gram stain (23). However, in reviewing the literature, we noted that 6 out of 15 cases (40%) of confirmed nocardial pericarditis in non-HIV patients did not exhibit a positive result when direct Gram staining of the pericardial fluid was performed. Several circumstances other than a negative Gram stain may contribute to a delay in the laboratory diagnosis of this infection. For instance, culture may require a prolonged period due to the presence of a low bacterial load. Nocardia spp. also exhibit a longer generation time than most bacteria, usually requiring 5 to 21 days for detection. Therefore, they can easily be missed on most routine culture workups if the cultures are discarded after 48 to 72 h of incubation. Some isolates may also show a negative partial acid-fast smear or lack of branching upon examination of the Gram stain, further complicating laboratory diagnosis. In addition, phenotypic (e.g., biochemical) identification of Nocardia spp. is complex and time-consuming. Molecular methods can shorten the identification turnaround time but are not used routinely in the average microbiology laboratory. Molecular detection methods may include restriction endonuclease analysis of amplified PCR products (e.g., hsp gene) or nucleic acid sequencing (e.g., 16S rRNA) (5, 16, 22, 23, 31). However, neither phenotypic nor genotypic identification of Nocardia spp. is commonly offered outside of reference laboratories or large teaching medical centers. It is important for optimum patient management to at least presumptively identify Nocardia spp. in HIV-infected patients as soon as possible. A proposed diagnostic algorithm for the microbiology workup of pericardial fluid in HIV-infected patients is outlined in Fig. 2 in support of more efficient and expeditious bacterial identification.
Discussion.
Nocardiosis is an uncommon human infection. It occurs primarily in immunocompromised hosts, such as patients with autoimmune disease or malignancy and those infected with HIV. However, Nocardia sp. is considered an unusual opportunistic pathogen among HIV-infected patients, partly due to prophylaxis for Pneumocystis jiroveci pneumonia with TMP-SMX. However, if infection occurs, the most common site is the lung and pericarditis due to this organism is a very rare event. An extensive literature review demonstrated only five other reported cases of Nocardia asteroides pericarditis associated with HIV type 1 infection prior to this report (10, 17, 20, 21).
Pericarditis in HIV-infected patients can manifest as either an infectious or a noninfectious etiology. In Western countries, pericardial effusions are often idiopathic in HIV patients (9). In regions where M. tuberculosis is endemic, tuberculosis has been reported as the major cause of pericarditis in more than 80% of the cases in this population (4). Other microorganisms that may infect the pericardium include other bacteria, viruses, and fungi. Cytomegalovirus is a frequent cause of opportunistic infection in patients with AIDS and has been reported to cause cardiac tamponade. There are also reports of pericarditis in AIDS patients caused by herpes simplex virus (4, 7, 8, 13, 29). Bacterial pathogens causing pericarditis include Staphylococcus aureus (6, 26), Streptococcus pneumoniae (14), and Klebsiella pneumoniae (24). Fungal pericarditis secondary to Cryptococcus neoformans has also been reported (2, 25). Other cases in HIV-infected patients have been linked to Kaposi's sarcoma and non-Hodgkin's B-cell lymphoma (1, 11, 12, 18, 27, 28, 33).
The differential diagnosis of acid-fast organisms causing pericarditis in HIV patients includes Mycobacterium tuberculosis and a variety of other nontuberculosis bacteria, including Mycobacterium avium, Mycobacterium intracellulare, Mycobacterium kansasii, and the partially acid-fast Nocardia spp. (27). A prompt and accurate diagnostic workup of the infecting pathogen is important as optimal therapy differs among these potential pathogens.
In reference to the five other cases of nocardial pericarditis in HIV-infected patients that have been reported, the characteristics of these cases are hereby described. This comparison shows a male predominance (five/six cases) and CD4+ cell counts of less than 250 cells/mm3 (48, 66, 91, 239, and 42/mm3). None of the patients were on highly active antiretroviral therapy when they developed nocardial pericarditis. Three of the patients had a history of intravenous drug use. One of the patients had a concomitant tuberculosis infection. Two of the five male cases admitted to having sex with men. There were no underlying diseases or other risks reported, such as diabetes mellitus, autoimmune diseases, cancer, or steroid use. All of the patients were treated with TMP-SMX. Four of six patients received follow-up treatment of pericardiectomy and drainage. One patient received repeated pericardial aspirations with a three-lumen central line. All patients survived with therapy and were discharged with outpatient follow-up. Only one of the six patients had died by 3 months after hospital discharge, and death was not directly related to the nocardial pericarditis.
Pericardial disease may be clinically silent or may present with chest discomfort, dyspnea, or fever. Physical examination may reveal tachycardia, tachypnea, or friction rub. Cardiac tamponade usually presents with hypotension, pulsus paradoxus, and tachypnea. An electrocardiogram may demonstrate ST-T wave elevation indicative of pericarditis. As pericardial effusion increases, low QRS voltage and electrical alternans may appear. Echocardiography is a useful diagnostic tool for detecting and monitoring pericardial effusion and tamponade. CT and magnetic resonance imaging are indicated when loculated, hemorrhagic effusions are clinically suspected (3, 8, 12).
TMP-SMX is considered the standard of therapy for nocardiosis, and high-dose TMP-SMX should be started in immunocompromised patients. Based on previous reports, successful therapy of nocardial pericarditis usually consists of antimicrobial drugs in combination with appropriate surgical drainage (15, 18). Our case indicates that treatment of both mycobacterial and nocardial infection should be started when the AFB smear is positive and it should be continued until the culture identification is known. Low-dose maintenance therapy should be continued for life in HIV patients with Nocardia pericarditis (19, 30).
Although uncommon, nocardial pericarditis can be seen in AIDS patients, especially those with CD4+ cell counts of less than 100 cells/mm3 (i.e., the median is 66 with a range of 42 to 239 cells/mm3 for reported cases). The Gram stain is the most rapid means of diagnosis but may suffer from low sensitivity, potentially resulting in missed or delayed diagnoses. High-dose TMP-SMX should be started if clinical suspicion for Nocardia spp. is high, and attempts should be made to expeditiously support the laboratory identification of Nocardia spp. This case reinforces the consideration of Nocardia spp. as etiological agents of pericarditis in HIV-infected patients even in the absence of a positive AFB stain or branching organism morphology.
The sixth case of Nocardia asteroides in a newly diagnosed HIV patient suffering from pericarditis is reported in this paper. Treatment for this patient was delayed due to a preliminary report of a slow-growing, possibly atypical AFB microorganism. This case was also complicated by purulent pericarditis that required extensive surgical drainage. Because of its high incidence of morbidity and mortality, we propose a diagnostic workup for Nocardia spp. to include 16S rRNA sequencing for critical cases. Also empirical treatment for nocardial pericarditis should be considered until the final identification of the acid-fast organism is confirmed.
Acknowledgments
We thank Alan D. Tice, FACP Consultant in Infectious Diseases, John A. Burns School of Medicine, Honolulu, HI, and Randall T. Hayden, St. Jude Children's Research Hospital, Memphis, TN, for their critical review of the manuscript. We also thank Terrie Koyamatsu, Microbiology Manager, and Rodney Nagai at Diagnostic Laboratory Services, Inc., and The Queen's and Kuakini Health Systems for their expert microbiology review and comments on the manuscript.
Footnotes
Published ahead of print on 16 May 2007.
REFERENCES
- 1.Aboulafia, D. M., R. Bush, and V. J. Picozzi. 1994. Cardiac tamponade due to primary pericardial lymphoma in a patient with AIDS. Chest 106:1295-1299. [DOI] [PubMed] [Google Scholar]
- 2.Brivet, F., J. Livartowski, P. Herve, B. Rain, and J. Dormont. 1987. Pericardial cryptococcal disease in acquired immune deficiency syndrome. Am. J. Med. 82:1273. [DOI] [PubMed] [Google Scholar]
- 3.Bull, R. K., P. D. Edwards, and A. K. Dixon. 1998. CT dimensions of the normal pericardium. Br. J. Radiol. 71:923-925. [DOI] [PubMed] [Google Scholar]
- 4.Cegielski, J. P., J. Lwakatare, C. S. Dukes, L. E. Lema, G. J. Lallinger, J. Kitinya, L. B. Reller, and F. Sheriff. 1994. Tuberculous pericarditis in Tanzanian patients with and without HIV infection. Tuber. Lung Dis. 75:429-434. [DOI] [PubMed] [Google Scholar]
- 5.Conville, P. S., S. H. Fischer, C. P. Cartwright, and F. G. Witebsky. 2000. Identification of Nocardia species by restriction endonuclease analysis of an amplified portion of the 16S rRNA gene. J. Clin. Microbiol. 38:158-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Decker, C. F., and C. U. Tuazon. 1994. Staphylococcus aureus pericarditis in HIV-infected patients. Chest 105:615-616. [DOI] [PubMed] [Google Scholar]
- 7.Freedberg, R. S., A. J. Gindea, D. T. Dieterich, and J. B. Greene. 1987. Herpes simplex pericarditis in AIDS. N. Y. State J. Med. 87:304-306. [PubMed] [Google Scholar]
- 8.Gowda, R. M., I. A. Khan, N. J. Mehta, M. R. Gowda, T. J. Sacchi, and B. C. Vasavada. 2003. Cardiac tamponade in patients with human immunodeficiency virus disease. Angiology 54:469-474. [DOI] [PubMed] [Google Scholar]
- 9.Heidenreich, P. A., M. J. Eisenberg, L. L. Kee, C. A. Somelofski, H. Hollander, N. B. Schiller, and M. D. Cheitlin. 1995. Pericardial effusion in AIDS. Incidence and survival. Circulation 92:3229-3234. [DOI] [PubMed] [Google Scholar]
- 10.Holtz, H. A., D. P. Lavery, and R. Kapila. 1985. Actinomycetales infection in the acquired immunodeficiency syndrome. Ann. Intern. Med. 102:203-205. [DOI] [PubMed] [Google Scholar]
- 11.Ioachim, H. L., M. C. Cooper, and G. C. Hellman. 1985. Lymphomas in men at high risk for acquired immune deficiency syndrome (AIDS). A study of 21 cases. Cancer 56:2831-2842. [DOI] [PubMed] [Google Scholar]
- 12.Isner, J. M., B. L. Carter, M. S. Bankoff, M. A. Konstam, and D. N. Salem. 1982. Computed tomography in the diagnosis of pericardial heart disease. Ann. Intern. Med. 97:473-479. [DOI] [PubMed] [Google Scholar]
- 13.Joshi, V. V. 1990. Pathology of AIDS and other manifestations of HIV infection. Igaku-Shoin, New York, NY.
- 14.Kan, B., J. Ries, B. H. Normark, F. Y. Chang, C. Feldman, W. C. Ko, J. Rello, D. R. Snydman, V. L. Yu, and A. Ortqvist. 2006. Endocarditis and pericarditis complicating pneumococcal bacteraemia, with special reference to the adhesive abilities of pneumococci: results from a prospective study. Clin. Microbiol. Infect. 12:338-344. [DOI] [PubMed] [Google Scholar]
- 15.Kessler, R., F. Follis, D. Daube, and J. Wernly. 1991. Constrictive pericarditis from Nocardia asteroides infection. Ann. Thorac. Surg. 52:861-862. [DOI] [PubMed] [Google Scholar]
- 16.Laurent, F. J., F. Provost, and P. Boiron. 1999. Rapid identification of clinically relevant Nocardia species to genus level by 16S rRNA gene PCR. J. Clin. Microbiol. 37:99-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leang, B., L. Lynen, K. Lim, G. Jacques, M. Van Esbroeck, and M. Zolfo. 2004. Disseminated nocardiosis presenting with cardiac tamponade in an HIV patient. Int. J. STD AIDS 15:839-840. [DOI] [PubMed] [Google Scholar]
- 18.Leung, W. H., K. L. Wong, C. P. Lau, and C. K. Wong. 1990. Purulent pericarditis and cardiac tamponade caused by Nocardia asteroides in mixed connective tissue disease. J. Rheumatol. 17:1237-1239. [PubMed] [Google Scholar]
- 19.Mandell, G. L., R. G. Douglas, J. E. Bennett, and R. Dolin. 2005. Mandell, Douglas, and Bennett's principles and practice of infectious diseases, 6th ed. Elsevier/Churchill Livingstone, New York, NY.
- 20.Ramanathan, P., and A. R. Rahimi. 2000. Nocardia asteroides pericarditis in association with HIV. AIDS Patient Care STDs 14:621-625. [DOI] [PubMed] [Google Scholar]
- 21.Rivero, A., A. Esteve, J. Santos, and M. Marquez. 2000. Cardiac tamponade caused by Nocardia asteroides in an HIV-infected patient. J. Infect. 40:206-207. [DOI] [PubMed] [Google Scholar]
- 22.Roth, A., S. Andrees, R. M. Kroppenstedt, D. Harmsen, and H. Mauch. 2003. Phylogeny of the genus Nocardia based on reassessed 16S rRNA gene sequences reveals underspeciation and division of strains classified as Nocardia asteroides into three established species and two unnamed taxons. J. Clin. Microbiol. 41:851-856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saubolle, M. A., and D. Sussland. 2003. Nocardiosis: review of clinical and laboratory experience. J. Clin. Microbiol. 41:4497-4501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Scerpella, E. G., A. A. Fatmi, and M. A. Brito. 1995. Bacterial pericarditis and cardiac tamponade in patients infected with human immunodeficiency virus type 1: case report and review. Clin. Infect. Dis. 21:1518-1519. [DOI] [PubMed] [Google Scholar]
- 25.Schuster, M., F. Valentine, and R. Holzman. 1985. Cryptococcal pericarditis in an intravenous drug abuser. J. Infect. Dis. 152:842. [DOI] [PubMed] [Google Scholar]
- 26.Stechel, R. P., D. J. Cooper, J. Greenspan, R. A. Pizzarello, and M. J. Tenenbaum. 1986. Staphylococcal pericarditis in a homosexual patient with AIDS-related complex. N. Y. State J. Med. 86:592-593. [PubMed] [Google Scholar]
- 27.Steigman, C. K., D. W. Anderson, A. M. Macher, J. D. Sennesh, and R. Virmani. 1988. Fatal cardiac tamponade in acquired immunodeficiency syndrome with epicardial Kaposi's sarcoma. Am. Heart J. 116:1105-1107. [DOI] [PubMed] [Google Scholar]
- 28.Stotka, J. L., C. B. Good, W. R. Downer, and W. N. Kapoor. 1989. Pericardial effusion and tamponade due to Kaposi's sarcoma in acquired immunodeficiency syndrome. Chest 95:1359-1361. [DOI] [PubMed] [Google Scholar]
- 29.Toma, E., M. Poisson, M. R. Claessens, C. Vega, and R. Morisset. 1989. Herpes simplex type 2 pericarditis and bilateral facial palsy in a patient with AIDS. J. Infect. Dis. 160:553-554. [DOI] [PubMed] [Google Scholar]
- 30.Uttamchandani, R. B., G. L. Daikos, R. R. Reyes, M. A. Fischl, G. M. Dickinson, E. Yamaguchi, and M. R. Kramer. 1994. Nocardiosis in 30 patients with advanced human immunodeficiency virus infection: clinical features and outcome. Clin. Infect. 18:348-353. [DOI] [PubMed] [Google Scholar]
- 31.Wilson, R. W., V. A. Steingrube, B. A. Brown, and R. J. Wallace, Jr. 1998. Clinical application of PCR-restriction enzyme pattern analysis for rapid identification of aerobic actinomycete isolates. J. Clin. Microbiol. 36:148-152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Winn, W. C., and E. W. Koneman. 2006. Koneman's color atlas and textbook of diagnostic microbiology, 6th ed. Lippincott Williams & Wilkins, Philadelphia, PA.
- 33.Ziegler, J. L., K. Bragg, D. Abrams, J. Beckstead, M. Cogan, P. Volberding, D. Baer, L. Wilkinson, E. Rosenbaum, K. Grant, et al. 1984. High-grade non-Hodgkin's lymphoma in patients with AIDS. Ann. N. Y. Acad. Sci. 437:412-419. [DOI] [PubMed] [Google Scholar]