In three recent papers, one published last year in the Journal of Virology by Jiang et al. (6) and earlier studies by Yamagishi et al. in 2003 (11) and Iwanaga et al. in 2004 (5), RNA was isolated from insect cells infected with the prototypical baculovirus Autographa californica multiple nucleopolyhedrovirus (AcMNPV) and converted to labeled cDNA, which was then hybridized to microarrays bearing the entire AcMNPV genome complement of ∼150 open reading frames (ORFs) in the form of tailored double-stranded DNA fragments generated by PCR. In the two most recent reports (5, 6), transcription kinetics are represented by numerical data for each ORF, albeit with unmatched sampling time points and different units to display transcript levels. For reasons discussed below, these data may create a highly distorted impression of the kinetics of viral protein synthesis, and they should therefore be interpreted cautiously.
Jiang et al. (6) and Iwanaga et al. (5) both performed real-time PCR experiments for a selection of ORFs to validate their array data, and I do not question here the reliability of any of this information. Curiously, though, Jiang et al. (6) failed to cite the earlier study, perhaps because it is impossible to superimpose the two array data sets and compare them quantitatively. Qualitative graphical comparisons can be made using two y axes, as illustrated in Fig. 1 for ORF32 (see below); by making such comparisons for 10 randomly selected genes (ORFs 25, 28, 31, 33, 77, 104, 120, 125, 139, and 148), I find that in six cases, the time of the onset of transcription differs by more than 8 hours and that in three cases, the time of peak transcription differs by more than 8 hours (data not shown). Such variability may be attributable in part to differences between the AcMNPV genotypes or the cell lines used in the two studies (see legend to Fig. 1). A more substantial difficulty arises, however, when these data are invoked to describe the progression of viral infection in terms of gene expression: any general assumption that these transcriptional profiles are synonymous with the translational profiles of corresponding ORFs is probably invalid.
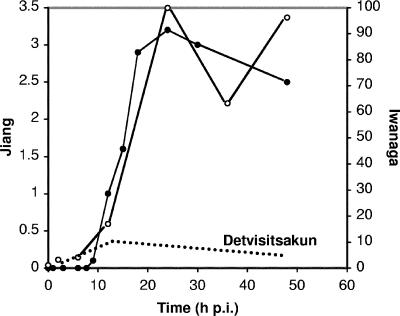
FIG. 1.
Transcription profiles for AcMNPV ORF32 (vfgf) in Spodoptera frugiperda cells. Microarray data from Jiang et al. (6) (filled circles) and Iwanaga et al. (5) (open circles) are plotted against time after infection of Sf21 cells with Bac-PH-EGFP (an AcMNPV recombinant lacking polh) at a multiplicity of infection (MOI) of 10 and infection of Sf9 cells with AcMNPV E2 at the same MOI, respectively. Transcription levels (y axes) are represented using the authors' explicit or implicit tabular designations of “Log (normalized expression level)” (left axis, Jiang et al. [6]) and percent maximum level (right axis, Iwanaga et al. [5]). The dotted line near the bottom of the plot area superimposes on these array data my assessment of transcript levels for monocistronic 0.6-kb ORF32 mRNA, as depicted in the Northern blot autoradiograph shown in Fig. 2B of Detvisitsakun et al. (2) (Sf21 cells were infected with AcMNPV L1 at an MOI of 20). The downward slope of this line begins at 12 hpi, when the intensity of the 0.6-kb band is maximal, and ends at 48 hpi, the last sampling time point, reflecting the “slight decrease in transcription at 24 and 48 hpi” noted by Detvisitsakun et al.; its peak is linked to the 12-hpi datum point of Iwanaga et al. in the sense that little more than half of the combined Northern-analysis band intensities at 12 hpi (2) is judged to comprise monocistronic vfgf mRNA. Since my assertion in the text is that only this latter mRNA can yield vFGF polypeptide, the divergent trajectories of the dotted and solid lines from this time point onward contrast, impressionistically, the abundance of ORF32-containing polycistronic transcripts that cannot generate vFGF and the paucity of monocistronic transcripts that can.
Consider the case of AcMNPV ORF32, which encodes a viral ortholog of a fibroblast growth factor (vFGF). vFGF is secreted from infected cells and can attract host hemocytes (2, 7, 8), a phenotype whose significance is not yet well understood; a Bombyx mori NPV mutant lacking vfgf also produced less budded virus and killed infected insects more slowly than did the wild type (9), although an equivalent AcMNPV mutant displayed no such effects (3). In a Northern blot experiment, Detvisitsakun et al. (2) revealed that a 0.6-kb transcript representing monocistronic AcMNPV ORF32 mRNA is synthesized at early times during infection, with rates of synthesis peaking around 12 h postinfection (hpi) and declining thereafter (Fig. 1). However, a second and longer (1.4-kb) transcript appears at 9 hpi and a third, longer still (3.1 kb), at 12 hpi. By 24 hpi, these two larger transcripts constitute the overwhelming majority of mRNA hybridizing to the ORF32-specific probe. The second transcript appears to comprise ORF32 and the adjacent ORF33, and although the boundaries of the third were not predicted by Detvisitsakun et al. (2), they presumably encompass an additional ORF(s).
ORF32 and ORF33 lie in the same orientation on the AcMNPV genome, so that the 5′ half of the 1.4-kb transcript is occupied by ORF33 and the 3′ half by ORF32 (vfgf). A critical question therefore arises: is vFGF polypeptide translated from such an mRNA?
The answer is likely to be no. Data in the literature to support this contention are lamentably scant, but in 1991 Doerfler and colleagues (4) published the last in a series of papers characterizing a region of the AcMNPV genome spanning five ORFs. Experiments shown in Fig. 2 of that study (4) indicate that only the 5′-proximal ORF of a multi-ORF polycistronic transcript is translated. The five ORFs are oriented clockwise on the genome, and, although Happ et al. (4) were working with an AcMNPV genotype (isolate E) whose ORF arrangement in this segment differs from that of the genome sequence (isolate C6) deposited in GenBank (accession number L22858) (1), the fourth and fifth ORFs correspond to ORFs 132 and 133 of AcMNPV C6. The pattern of transcripts synthesized from this genomic region appears to be a common one for AcMNPV, and it fits with that described above for the less-complex vfgf region. Nine mRNAs with identical 3′ but distinct 5′ termini were mapped, and restriction fragments were used by Happ et al. (4) to select subpopulations of various sizes for in vitro translation. Notably, in the present context, an EcoRI-HpaI fragment that selected the four longest transcripts, each spanning three or more complete ORFs, yielded translation products including one that was expected from the central ORF adjacent to ORF132 but lacking those expected from ORF132 or ORF133. While the three largest selectable transcripts have short 5′-proximal ORFs whose translation products are indistinct, the fourth (3,208 nucleotides) possesses the central ORF at its 5′ end and ORF132 and ORF133 towards its 3′ end. The polypeptide profile obtained after the translation of this four-component mRNA population is most simply explained by a model in which the 5′-proximal ORF of a polycistronic transcript, but no other ORF(s), is translatable.
The existence of “overlapping” baculovirus transcripts of various configurations, which can include variable numbers and relative orientations of ORFs as well as variable 5′ or 3′ termini, was first reported in the Journal of Virology 23 years ago (10). However, apart from the experiments by Happ et al. (4), described above, little attention seems to have been paid to the implications of these structures for translation. Jiang et al. (6) appear to have been unaware of this early work: while they note that members of contiguous ORF “clusters” have similar transcription profiles, their discussion of this observation is framed in terms of putative “operon-like” regulatory behavior and is presented as though the idea were new to baculovirology.
Because the in vitro translation data presented by Happ et al. (4) are not incompatible with a view of baculovirus infection in which only the 5′-proximal ORF of a transcript spanning two or more ORFs can be translated, and because a monocistronic mRNA may be in decline well before the total population of transcripts overlapping its ORF has peaked (Fig. 1), the discussion of baculovirus array profiles in terms of gene expression should be restricted carefully to transcription. The discrepancy between this circumscribed interpretative framework on the one hand and, on the other, any broader notion that currently available array data reflect the translation of a particular baculovirus gene, is potentially a major one.
Acknowledgments
I thank Kimiko Hasegawa for her assistance in constructing the graph.
REFERENCES
- 1.Ayres, M. D., S. C. Howard, J. Kuzio, M. Lopez-Ferber, and R. D. Possee. 1994. The complete DNA sequence of Autographa californica nuclear polyhedrosis virus. Virology 202:586-605. [DOI] [PubMed] [Google Scholar]
- 2.Detvisitsakun, C., M. F. Berretta, C. Lehiy, and A. L. Passarelli. 2005. Stimulation of cell motility by a viral fibroblast growth factor homolog: proposal for a role in viral pathogenesis. Virology 336:308-317. [DOI] [PubMed] [Google Scholar]
- 3.Detvisitsakun, C., E. L. Hutfless, M. F. Berretta, and A. L. Passarelli. 2006. Analysis of a baculovirus lacking a functional viral fibroblast growth factor homolog. Virology 346:258-265. [DOI] [PubMed] [Google Scholar]
- 4.Happ, B., J. Li, and W. Doerfler. 1991. Proteins encoded in the 81.2- to 85.0-map-unit fragment of Autographa californica nuclear polyhedrosis virus DNA can be translated in vitro and in Spodoptera frugiperda cells. J. Virol. 65:89-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Iwanaga, M., K. Takaya, S. Katsuma, M. Ote, S. Tanaka, S. G. Kamita, W. K. Kang, T. Shimada, and M. Kobayashi. 2004. Expression profiling of baculovirus genes in permissive and nonpermissive cell lines. Biochem. Biophys. Res. Commun. 323:599-614. [DOI] [PubMed] [Google Scholar]
- 6.Jiang, S. S., I.-S. Chang, L.-W. Huang, P.-C. Chen, C.-C. Wen, S.-C. Liu, L.-C. Chien, C.-Y. Lin, C. A. Hsiung, and J.-L. Juang. 2006. Temporal transcription program of recombinant Autographa californica multiple nucleopolyhedrosis virus. J. Virol. 80:8989-8999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Katsuma, S., T. Shimada, and M. Kobayashi. 2004. Characterization of the baculovirus Bombyx mori nucleopolyhedrovirus gene homologous to the mammalian FGF gene family. Virus Genes 29:211-217. [DOI] [PubMed] [Google Scholar]
- 8.Katsuma, S., T. Daimon, K. Mita, and T. Shimada. 2006. Lepidopteran ortholog of Drosophila Breathless is a receptor for the baculovirus fibroblast growth factor. J. Virol. 80:5474-5481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Katsuma, S., S. Horie, T. Daimon, M. Iwanaga, and T. Shimada. 2006. In vivo and in vitro analyses of a Bombyx mori nucleopolyhedrovirus mutant lacking functional vfgf. Virology 355:62-70. [DOI] [PubMed] [Google Scholar]
- 10.Lübbert, H., and W. Doerfler. 1984. Transcription of overlapping sets of RNAs from the genome of Autographa californica nuclear polyhedrosis virus: a novel method for mapping RNAs. J. Virol. 52:255-265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamagishi, J., R. Isobe, T. Takebuchi, and H. Bando. 2003. DNA microarrays of baculovirus genomes: differential expression of viral genes in two susceptible insect cell lines. Arch. Virol. 148:587-597. [DOI] [PubMed] [Google Scholar]