Abstract
Xenobiotics and drugs may lead to cholangiopathies and biliary fibrosis, but the underlying mechanisms are largely unknown. Therefore, we aimed to characterize the cause and consequences of hepatobiliary injury and biliary fibrosis in 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC)-fed mice as a novel model of xenobiotic-induced cholangiopathy. Liver morphology, markers of inflammation, cell proliferation, fibrosis, bile formation, biliary porphyrin secretion, and hepatobiliary transporter expression were studied longitudinally in DDC- and control diet-fed Swiss albino mice. DDC feeding led to increased biliary porphyrin secretion and induction of vascular cell adhesion molecule, osteopontin, and tumor necrosis factor-α expression in bile duct epithelial cells. This was associated with a pronounced pericholangitis with a significantly increased number of CD11b-positive cells, ductular reaction, and activation of periductal myofibroblasts, leading to large duct disease and a biliary type of liver fibrosis. After 4 weeks, we constantly observed intraductal porphyrin pigment plugs. Glutathione and phospholipid excretion significantly decreased over time. Expression of Ntcp, Oatp4, and Mrp2 was significantly reduced, whereas Bsep expression remained unchanged and adaptive Mrp3 and Mrp4 expression was significantly induced. We demonstrate that DDC feeding in mice leads to i) a reactive phenotype of cholangiocytes and bile duct injury, ii) pericholangitis, periductal fibrosis, ductular reaction, and consequently portal-portal bridging, iii) down-regulation of Mrp2 and impaired glutathione excretion, and iv) segmental bile duct obstruction. This model may be valuable to investigate the mechanisms of xenobiotic-induced chronic cholangiopathies and its sequels including biliary fibrosis.
Cholangiopathies such as primary sclerosing cholangitis, primary biliary cirrhosis, and drug-induced bile duct damage may result in ductopenia and a vanishing bile duct syndrome, which can progress to biliary cirrhosis and represents an important indication for liver transplantation and cause of liver-related death.1 Most of the present knowledge on the mechanisms of hepatic fibrosis is based on in vitro studies using cultured and activated hepatic stellate cells isolated from rodents or human liver and in vivo studies with bile duct-ligated or carbon tetrachloride (CCl4)-intoxicated rodents.2,3 The orchestrated interplay between activated proliferating cholangiocytes (bile duct epithelial cells; BECs), extracellular matrix-producing cells (eg, periductal/portal myofibroblasts, bone marrow-derived fibrocytes, and stellate cells), inflammatory cells (eg, neutrophils, macrophages, and lymphocytes), and smooth muscle cells localized in vessel walls may be pivotal in the pathogenesis of cholangiopathy-related biliary fibrosis.1,4 However, the precise mechanisms of how cholangiopathies—irrespective of their etiology—drive biliary fibrosis are still poorly understood which, at least in part, reflects the lack of well-defined, highly reproducible, and easy-to-perform animal models, allowing detailed longitudinal long-term studies.
Chronic feeding of 3,5-diethoxycarbonyl-1,4-dihydrocollidine (DDC) in mice is a well-established model to study formation of Mallory bodies, which are hepatocellular inclusion bodies characteristically associated with alcoholic and nonalcoholic steatohepatitis, metabolic liver diseases (eg, Wilson’s disease and other forms of copper toxicosis), and chronic cholestatic liver diseases.5,6,7 This model is also used to study the mechanisms of oval cell activation and proliferation8; moreover, serum markers for cholestasis are markedly induced.9 So far, the cholestatic phenotype and its pathobiology, however, remain poorly defined in DDC-fed mice. We therefore designed a longitudinal study to determine the mechanisms and time course of the cholestatic phenotype in DDC-fed mice serving as a new xenobiotic-induced mouse model for sclerosing cholangitis and biliary fibrosis. We herein demonstrate that DDC feeding in mice results in a reactive phenotype of BECs, leading to ductular reaction, periductal fibrosis, and portal-portal septa. This model is therefore useful to investigate the mechanisms of chronic cholangiopathies and their sequels, including liver fibrosis of the biliary type, and to test novel therapeutic strategies for these diseases.
Materials and Methods
Animals
Experiments were performed with 2-month-old male Swiss albino mice weighing 25 to 30 g. Mice were fed a 0.1% DDC-supplemented diet for 1 week, 4 weeks, and 8 weeks, housed with a 12-hour light/dark cycle, and permitted ad libitum consumption of water. To assess potential strain differences in the susceptibility to DDC feeding, liver injury was studied in four different mouse strains (Swiss albino, FVB/N, C57BL/6, and 129/Sv) in a pilot study. All mouse strains tested developed a comparable cholestatic phenotype (as outlined in Results). However, Swiss albino mice displayed the highest degree of large duct disease, and subsequent experiments were therefore performed in this strain. Controls were fed a standard mouse diet (Sniff, Soest, Germany). An additional group of animals was fed a 0.1% DDC-supplemented diet for 8 weeks and afterward allowed to recover for 4 weeks under standard mouse diet to study the potential reversibility of the cholestatic phenotype. The experimental protocols were approved by the local animal Care and Use Committee according to criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Sciences, as published by the National Institutes of Health (NIH publication 86-23, revised 1985). Serum biochemical analysis, liver histology, electron microscopy, immunofluorescence microscopy for keratins 8 and 18 (K8/18), and determination of bile flow and composition were performed as described.10,11
Determination of Biliary Porphyrin Concentration
Mice were fed a 0.1% DDC-supplemented diet for 1, 4, and 8 weeks. Bile was collected, and total porphyrins in bile were measured fluorometrically (using an F-2500 Fluorescence Spectrophotometer; Hitachi, Tokyo, Japan). For this purpose, bile was diluted 50-fold in 0.9 mol/L HClO4/ethanol [1:1 (v/v)], and fluorescence was determined at 400 nm extinction and 602 nm emission wavelengths using protoporphyrin IX (Sigma-Aldrich, Munich, Germany) as standard.
Bile Duct Plastination
To compare the gross bile duct morphology of standard diet-fed and 4-week-DDC-fed mice, plastination of the bile duct system was performed as described previously.11
Immunohistochemistry for Osteopontin
Immunohistochemistry for osteopontin was performed on microwave-treated (0.01 mmol/L citrate buffer, pH 6.0) paraffin sections (4 μm thick) using the monoclonal mouse anti-osteopontin antibody (catalog no. AF808, dilution 1:100; R&D Systems, Minneapolis, MN) (Table 1). Binding of the antibody was detected using the ABC system (Dako, Glostrup, Denmark), using β-amino-9-ethyl-carbazole (Dako) as substrate. Immunohistochemistry for CD11b, proliferation marker Ki-67, and keratin 19 (K19) were performed as described.12
Table 1.
Antibodies Used for Immunohistochemistry
| Antibody | Section | Pretreatment/fixation | Dilution | Company |
|---|---|---|---|---|
| Rat anti-VCAM | Cryo | Aceton | 1:50 | PharMingen, San Diego, CA |
| Rat anti-CD11b | Cryo | Formol-methanol Aceton | 1:50 | PharMingen |
| Rat anti-CK19 | Cryo | Aceton | 1:50 | Troma 3 Developmental Studies Hybridoma Bank, Iowa, IA |
| Mouse anti-α-SMA | Paraffin | Microwave 0.01 mol/L citrate buffer, pH 6.0 | 1:500 | Sigma-Aldrich, Munich, Germany |
| Goat anti-osteopontin | Paraffin | Microwave 0.01 mol/L citrate buffer, pH 6.0 | 1:100 | R&D Systems, Minneapolis, MN |
mRNA analysis and polymerase chain reaction of several genes was performed as described.12 Primers used for mRNA analysis of key genes for transporters, metabolic enzymes, inflammation, and fibrosis are summarized in Table 2.
Table 2.
Primers Used for Reverse Transcription-Polymerase Chain Reaction
| Gene | Forward primer | Reverse primer | Probe | Accession no. |
|---|---|---|---|---|
| 18sRNA | 5′-GTAACCCGTTGAACCCCATT-3′ | 5′-CCATCCAATCGGTAGTAGCG-3′ | * | X00686 |
| Bsep | 5′-GAGTGGTGGACAGAAGCAAA-3′ | 5′-TGAGGTAGCCATGTCCAGAA-3′ | 5′-CGCGCCCTCATACGGA- AACC-3′ | NM_021022 |
| Col 1a2 | 5′-GCAGGGTTCCAACGATGTTG-3′ | 5′-GCAGCCATCGACTAGGACAGA-3′ | * | NM_007743 |
| Cyp2b10 | 5′-CAATGGGGAACGTTGGAAGA-3′ | 5′-TGATGCACTGGAAGAGGAAC-3′ | 5′-TTCGTAGATTCTCTCTGG- CCACCATGAGA-3′ | NM_009998 |
| Cyp3a11 | 5′-CCACCAGTAGCACACTTTCC-3′ | 5′-TTCCATCTCCATCACAGT- ATCA-3′ | 5′-CTCTGCCCAACAAGGCA- CCTCC-3′ | NM_007818 |
| iNos | 5′-CAGAGCCCTCAGCAGCA- TCCA-3′ | 5′-CAGGTGTTCCCCAGGCAGGTA-3′ | 5′-TCCAGCCGCACCACCCT- CCTCGT-3′ | NM_010927 |
| Mdr1a | 5′-GCAGGTTGGCTAGACAGG- TTGT-3′ | 5′-GAGCGCCACTCCATGGATAA-3′ | * | NM_011076 |
| Mdr1b | 5′-GCTGGACAAGCTGTGCATGA-3′ | 5′-TGGCAGAATACTGGCTTC- TGCT-3′ | * | NM_011075 |
| Mdr2 | 5′-ATCCTATGCACTGGCCTT- CTGGT-3′ | 5′-GAAAGCATCAATACAGG- GGGCAG-3′ | * | NM_008830 |
| Mrp2 | 5′-GCTTCCCATGGTGATCTCTT-3′ | 5′-ATCATCGCTTCCCAGGTACT-3′ | 5′-CAGTCATCCAGGCCAGC- GTCTC-3′ | NM_013806 |
| Mrp3 | 5′-TGAGATCGTCATTGATGGGC-3′ | 5′-AGCTGAGAGCGCAGGTCG-3′ | 5′-CAACGTGGCACACATT- GGCCTCC-3′ | NM_029600 |
| Mrp4 | 5′-TTAGATGGGCCTCTGGTTCT-3′ | 5′-GCCCACAATTCCAACCTTT-3′ | 5′-ACTGCGCTCATCAAGTC- CAGGG-3′ | XM_908208 |
| Ntcp | 5′-CACCATGGAGTTCAGCAAGA-3′ | 5′-AGCACTGAGGGGCATGATAC-3′ | 5′-AGGCTCACTTCTGGAA- GCCCAAA-3′ | NM_011387 |
| Oatp4 | 5′-GATCCTTCACTTACCTGTTCAA-3′ | 5′-CCTAAAAACATTCCACTTG- CCATA-3′ | 5′-AGACAGCATCGCAGGCC- AACTTTCTG-3′ | NM_178235 |
| Ost-α | 5′-GTCTCAAGTGATGAACTGCCA-3′ | 5′-TTGAGTGCTGAGTCCAGGTC-3′ | * | NM_145932 |
| Ost-β | 5′-GACAAGCATGTTCCTCCTGAG-3′ | 5′-GATGCAGGTCTTCTGGTG- TTTC-3′ | 5′-TGCTTCTTTCGATTTCT- GTTTGCCAGGATGCTCC-3′ | NM_178933 |
| Sult2a1 | 5′-GGAAGGACCACGACTCATAAC-3′ | 5′-GATTCTTCACAAGGTTTGT- GTTACC-3′ | * | L02335 |
| Osteopontin | 5′-ACACTTTCACTCCAATCG- TCC-3′ | 5′-TGCCCTTTCCGTTGTTGTCC-3′ | * | NM_009263 |
| Tgf1-β | 5′-TGACGTCACTGGAGTTGT- ACGG-3′ | 5′-GGTTCATGTCATGGATGGTGC-3′ | 5′-TTCAGCGCTCACTGTC- TTGTGACAG-3′ | NM_011577 |
| Timp | 5′-CATGGAAAGCCTCTGTGGAT- ATG-3′ | 5′-AAGCTGCAGGCATTGATGTG-3′ | 5′-CTCATCACGGGCCGCCT- AAGGAAC-3′ | NM_011593 |
| Tnf-α | 5′-CATCTTCTCAAAATTCGAGT- GACAA-3′ | 5′-TGGGAGTAGACAAGGTACA- ACCC-3′ | 5′-CACGTCGTAGCAAACCA- CCAAGTGGA-3′ | NM_013693 |
Fluorogenic probes for TaqMan reverse transcription-polymerase chain reaction are 5′-FAM- and 3′-TAMRA-labeled.
Real-time polymerase chain reaction using the SYBR Green assay was performed.
In Situ Hybridization for Tumor Necrosis Factor-α
Labeled sense and antisense transcripts were synthesized in a 20-μl reaction mixture containing 1 μg of linearized plasmid, 10× transcription buffer [0.4 mol/L Tris-HCl, pH 8.0, 60 mmol/L MgCl2, 20 mmol/L spermidine, and 100 mmol/L dithiothreitol (DTT)], 20 mmol/L DTT, rNTP labeling mixture (1 mmol/L each of adenosine-triphosphate, cytosinetriphosphate, and guanosine-triphosphate; Roche, Basel, Switzerland), 40 U of ribonuclease inhibitor, 120 μCi of α-35S-UTP (GE Healthcare, Little Chalfont, Buckinghamshire, UK) and 20 U of either SP6 or T7 RNA polymerase (Roche). After an incubation period of 2 hours at 37°C, DNA was digested with 2 U of RNase-free DNase (Roche) for 10 minutes at 37°C, and the reaction was stopped with 2 μl of 0.5 mol/L ethylenediamine tetraacetic acid, pH 8.0. Unincorporated nucleotides were removed using a MicroSpin S-200 HR column (GE Healthcare). To 50 μl of the eluted probe, 50 μl of hydrolysis buffer (80 mmol/L NaHCO3, 120 mmol/L Na2CO3, and 120 mmol/L DTT) was added, and hydrolysis was performed at 60°C for 45 minutes to obtain an average probe size of 150 bp. After the addition of 5 μl of stopping solution (0.2 mol/L sodium acetate, 10 mmol/L DTT, and 1% glacial acetic acid), the probe was precipitated with LiCl/isopropanol. The washed pellet was resuspended in 100 μl of 50% formamide containing 25 mmol/L DTT. Preparation of paraffin sections as well as hybridization and autoradiography were performed as described previously by us.9
Preparation of liver membranes and analysis of transporter protein levels by Western blotting was performed as described.10 Antibodies used for the determination of several protein levels are listed in Table 3. Preparation of total liver protein and analysis for determination of vascular cell adhesion molecule (VCAM) protein was performed as described.12
Table 3.
Antibodies Used for Western Blotting
| Antibody | Provider | Dilution |
|---|---|---|
| Ntcp | Bruno Stieger, Institute for Clinical Pharmacology and Toxicology, Zurich, Switzerland | 1:2500 |
| Bsep | Renxue Wang, BC Cancer Research Centre Vancouver, BC, Canada | 1:7500 |
| Mrp2 | Bruno Stieger | 1:1000 |
| Mrp3 | James L. Boyer, Department of Internal Medicine, Yale, University New Haven, CT | 1:1000 |
| Mrp4 | J.D. Schütz, Department of Pharmaceutical Sciences, St. Jude Children’s Research Hospital, Memphis, TN | 1:1000 |
| β-Actin | Sigma-Aldrich, Munich, Germany | 1:5000 |
Bile Duct Morphometry
Morphometry of K19-expressing bile ducts and ductules was performed using a semiautomatic system for image analysis. This consisted of a microscope (Nikon E600; Tokyo, Japan), a video camera (Sony DXC-930P; Tokyo, Japan), a frame grabber (Intrigue Pro; Integral Technologies, Indianapolis, IN), and a personal computer with image analysis software (Optimas 6.51; Media Cybernetics Inc., Bethesda, MD). Approximate borders of a portal field were traced by the operator at ×20 objective magnification. A specially developed macro command served for automatic identification and measurement of the total area of cells positive for K19 within a given portal field. In addition, the area of the corresponding portal vein was also measured and used in statistical analysis for adjustment for the size of portal fields. All portal fields within each sample were measured (13 to 31 portal fields per mouse). Obvious artifacts, like nonspecific staining of necrotic areas as well as incomplete portal fields at the margins of the specimens, were excluded from analysis.
Measurement of Hepatic Hydroxyproline Content
To quantify liver fibrosis, hepatic hydroxyproline content was determined as described.12
Statistical Analysis
Data are reported as arithmetic means ± SD of 5 to 10 animals in each group. Statistical analysis was performed using Student’s t-test when appropriate or analysis of variance with Bonferroni posttesting when three or more groups were compared using the SigmaStat statistics (Jandel Scientific, San Rafael, CA). A P value <0.05 was considered significant. Morphometric data for K19 expressing cholangiocytes represented a hierarchical structure, where portal fields (level-one observation units) were nested within mice (level-two observation units). A linear regression model was fitted to calculate means and confidence intervals for K19-positive areas in each of the four study groups, as well as to test the significance of differences between them.
Results
DDC Feeding Leads to Sclerosing Cholangitis
After 1 week of DDC feeding, livers showed ductular reaction (Figure 1, B–D) that increased significantly over time as revealed by morphometry of K19-positive cells (Figure 2, B–E). At 4 weeks, ductules and small bile ducts frequently contained pigment plugs (Figures 1, C and D). Electron microscopy of these plugs revealed the picture of slender electron-dense crystals in a radiating pattern characteristic of porphyrin crystals (Supplemental Figure 1, available at http://ajp.amjpathol.org). In addition, these intraductal plugs showed characteristic autofluorescence (Supplemental Figure 2 is available at http:// ajp.amjpathol.org). Determination of biliary protoporphyrin secretion revealed that the formation of the pigment plugs was preceded by significantly increased biliary secretion already at 1 week of DDC feeding (Supplemental Figure 3, available at http://ajp.amjpathol.org). The liver lobules displayed single cell necrosis and some disseminated apoptotic bodies (data not shown). DDC-fed mice developed a characteristic sclerosing cholangitis of the onion skin-type (Figure 1, G and H). These morphological alterations where accompanied by a continuous increase in serum alanine aminotransferase levels as an indicator of hepatocyte injury followed by significant elevations of cholestasis parameters, such as alkaline phosphatase and bilirubin, as well as serum bile acid levels (Table 4). To visualize better the bile duct system, plastination casts of the bile ducts were prepared after 4 weeks of DDC treatment,11 revealing segmental bile duct strictures as well as slight focal dilatation of intrahepatic bile ducts (Figure 3E). In addition, some pigment plugs were found in the casts. Histological examination of the ductus hepatocholedocus (DHC) after in vivo formalin fixation and serial sectioning revealed a significantly increase of the wall thickness (Figure 3, F and G) with pronounced neutrophil infiltration (Figure 3H), clearly demonstrating involvement of the extrahepatic bile ducts. Taken together, these findings suggest that the cholestatic phenotype in DDC-fed mice is directly related to increased biliary porphyrin secretion and that segmental subtotal bile duct obstruction occurs by porphyrin pigment plugs in older DDC-fed mice. To study the potential reversibility of the cholestatic phenotype observed in 8-week DDC-fed mice, an additional group of animals was allowed to recover under standard chow for 4 weeks. The degree of ductular proliferation and sclerosing cholangitis was significantly reduced after 4 weeks recovery from DDC feeding (Supplemental Figure 4, B and D, available at http://ajp.amjpathol.org), suggesting partial reversibility of DDC-induced cholangiopathy and ductular proliferation.
Figure 1.
DDC feeding leads to ductular proliferation and cholangitis with onion skin type-like periductal fibrosis in mice. Livers of control mice (A and E) and mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (B and F), 4 weeks (C and G), and 8 weeks (D and H) are shown. Liver histology of DDC-fed mice reveals increasing ductular proliferation over time (outlined by arrowheads) (B–D). At 4 weeks, an increasing number of porphyrin plugs occluding the lumina of small bile ducts is observed (C and D). F–H: Increasing severity of cholangitis and periductal fibrosis of larger bile ducts over time. Hematoxylin and eosin staining: pv, portal vein; bd, bile duct. Original magnification, ×20 (A–H).
Figure 2.
DDC feeding induces ductular reaction in mice. Immunohistochemistry for keratin 19 (K19) in control liver (A) and mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (B), 4 weeks (C), and 8 weeks (D) is shown. In contrast to control liver (A), there is a significantly increased number of K19-postive cholangiocytes/portal field in DDC-fed animals (B–D). E: For morphometric analysis, the bile duct mass was measured by a semiautomatic system and normalized to the size of portal veins showing significantly increased K19-positive area/portal field in DDC-fed mice. *P < 0.05 (controls versus 1- to 4-week old DDC-fed mice). pv, portal vein. Original magnification, ×10 (A–D).
Table 4.
Serum Biochemistry of 1-, 4-, and 8-Week DDC-Intoxicated Mice
| Control n = 7 | 1w DDC n = 10 | 4w DDC n = 8 | 8w DDC n = 9 | |
|---|---|---|---|---|
| ALT (U/L) | 81 + 40 | 1692 + 987* | 2654 + 1624* | 1780 + 524* |
| AP (U/L) | 108 + 29 | 779 + 650* | 1293 + 908* | 1045 + 761* |
| Bili tot (mg/dl) | 0.2 + 0.1 | 0.8 + 0.4* | 1.8 + 2.4* | 0.5 + 0.7 |
| SBA (μmol/L) | 7.1 + 6.9 | 51.6 + 39.8 | 351.3 + 305.6* | 228.8 + 134.6* |
ALT, alanine aminotransferase; AP, alkaline phosphatase; Bili tot, total bilirubin; SBA, serum bile acids.
Values are expressed as mean ± SD.
P < 0.05 compared with standard diet-fed controls (analysis of variance with Bonferroni post-testing).
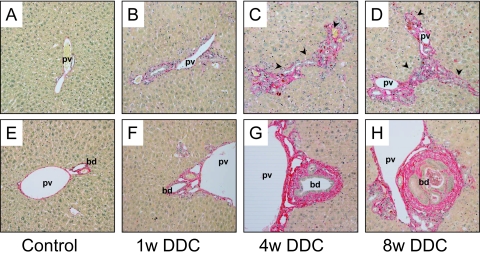
Figure 3.
DDC feeding induces segmental subtotal mechanical bile duct obstruction via porphyrin plugs and large duct disease in mice. Plastination of the bile duct system followed by maceration of the liver was performed in (A) standard diet-fed and (E) 4-week old DDC-fed mice. A: The bile duct system was filled with plastogen G after cannulation of the gallbladder (GB). In contrast to the delicately structured bile duct system in the control diet-fed mouse (A), the DDC-fed mouse (E) shows slight dilatation of bile ducts (highlighted by white arrows), showing dilatation of the bile duct and some porphyrin plugs (black dots) within the plastogen. B: Histological examination of the DHC shows a slim duct from the liver hilus (L) down to the pancreas (P) in a control diet-fed mouse. F: In contrast, in 8-week-old DDC-fed mice, the DHC wall is markedly thickened. G and H: The bile duct epithelium shows reactive changes and substantial neutrophil infiltration of the DHC wall. GB, gall bladder, L, liver; P, pancreas. Original magnification: ×4 (B and F); ×20 (C and G); ×40 (D and H).
DDC Feeding Leads to a Reactive Cholangiocyte Phenotype
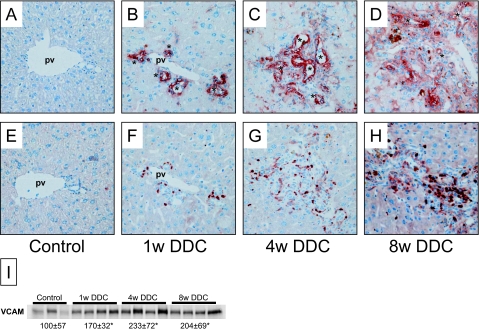
Because the transformation of BECs into a reactive phenotype (characterized by secretion of proinflammatory and/or chemotactic cytokines) is pivotal in the pathobiology of cholangiopathies, we studied these parameters in our model system. DDC-fed mice developed a dense neutrophil granulocytic infiltrate around small (Figure 4, F–H) and large interlobular bile ducts (Figure 1, F–G), which increased with time of DDC feeding. The finding of numerous CD11b-positive periductal cells (ie, neutrophil granulocytes) in DDC-fed mice was paralleled by a significantly increased VCAM expression by BECs as revealed by immunohistochemistry and Western blotting indicating their transformation to a reactive phenotype (Figure 4, B–D). In addition, activated BECs represented a major source of tumor necrosis factor (TNF)-α in DDC-fed mice as demonstrated by in situ hybridization performed in 1-week DDC-fed mice (Figure 5C). In contrast, in lipopolysaccharide-challenged mouse liver (50 μg for 6 hours) (serving as a positive control), Kupffer cells were the source for TNF-α mRNA (Figure 5B). Taken together, these findings suggest that DDC feeding leads to the activation of BECs and that the development of a reactive phenotype of BECs (ie, significantly induced VCAM and TNF-α expression) is pivotal for the development of sclerosing cholangitis in this model. This assumption is further supported by the finding of significantly reduced VCAM expression in BECs and reduced number of infiltrating CD11-positive cells in mice recovered from DDC intoxication (Supplemental Figure 5, B and D, available at http://ajp.amjpathol.org).
Figure 4.
DDC feeding induces a reactive phenotype of cholangiocytes and pericholangitis in mice. Immunohistochemistry for VCAM (A–D) and neutrophil marker CD11b (E–H) in control liver (A and E) and mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (B and F), 4 weeks (C and G), and 8 weeks (D and H). In contrast to control liver (A), proliferating bile ducts of DDC-fed mice (B–D) express VCAM. F–G: VCAM overexpression of bile ducts is paralleled by the occurrence of a dense neutrophil infiltrate. I: Protein quantification of VCAM by Western blotting reveals significantly increased VCAM hepatic protein level in DDC-fed animals. Densitometric values are the means from three to four animals in each group. *P < 0.05 compared with standard diet-fed controls. pv, portal vein. Original magnification, ×20.
Figure 5.
DDC feeding induces TNF-α mRNA expression in cholangtiocytes. In situ hybridization for TNF-α mRNA in control liver (A), lipopolysaccharide-injected mouse (positive control) (B), and 1-week DDC-fed mouse livers (C). Note the significant increase of the TNF-α signal in Kupffer cells in lipopolysaccharide-treated mouse liver (B), serving as a positive control. C: In contrast, in DDC-fed mice cholangiocytes of bile duct proliferates represent the main source for TNF-α. pv, portal vein; bd, bile duct. Original magnification, ×40.
DDC Feeding Leads to a Biliary Type Liver Fibrosis
In later stages, cholangitis in DDC-fed mice was accompanied by the development of portal-portal septa (Figure 6, B–D) and periductal onion skin-like fibrosis of large bile ducts (Figure 6, F–H) concomitant with proliferation and activation of periductal myofibroblasts as demonstrated by a significantly increased number of Ki-67-positive (data not shown) and α-SMA-positive periductal myofibroblasts (Figure 7). These are characteristic morphological features of biliary type liver fibrosis. Evolving biliary fibrosis in DDC-fed mice over time was, in addition, confirmed biochemically by significantly increased hepatic hydroxyproline content (Figure 8). Moreover, progressive biliary fibrosis in mice was accompanied by significantly enhanced expression of key fibrosis genes such as collagen-1a2, TIMP-1, and TGF-β genes as shown in Table 5. The development of biliary fibrosis in DDC-fed mice was paralleled by pronounced ductular reaction as demonstrated by a significantly increased number of K19-positive cells (Figure 2E). Reactive ductules and hepatocytes in acinar zone 1 showed a strong immunoreactivity for the profibrogenic cytokine osteopontin (Figure 9). Most interestingly, immunoreactivity of hepatocytes along the margins of the liver acinus for osteopontin preceded the development of portal-portal fibrous bridges (Figure 9, E and F). These findings therefore clearly demonstrate that DDC-fed mice develop all histological and biochemical characteristics of liver fibrosis of the biliary type. Liver fibrosis was less pronounced after 4 weeks recovery from DDC intoxication suggesting partial resolution of biliary fibrosis after discontinuation the drug (Supplemental Figure 4, B and D, available at http://ajp.amjpathol.org). Of interest, decreased liver fibrosis in DDC-recovered mouse liver was paralleled by significantly reduced osteopontin expression (Supplemental Figure 8, B and D, available at http://ajp.amjpathol.org).
Figure 6.
Biliary fibrosis DDC-fed mice. Sirius red staining (A–H) in control liver (A and E) and livers of mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (B and F), 4 weeks (C and G), and 8 weeks (D and H). B–D: Ductular proliferation leading to portal-portal bridges is paralleled by an increase in extracellular matrix deposition (ie, Sirius-red stain-positive collagen fibers indicated by arrowheads) in DDC-fed mice over time. F–G: Increasing periductal onion skin-like fibrosis of larger ducts in DDC-fed mice. H: Note that there is also subtotal obstruction of the bile duct by a porphyrin plug. pv, portal vein, bd, bile duct. Original magnification, ×20.
Figure 7.
Proliferation of periductal myofibroblasts in DDC-fed mouse liver. Immunohistochemistry of α-SMA-positive cells (A) in control liver and (B–C) livers of mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (B), 4 weeks (C), and 8 weeks (D). A: In the control liver, only smooth muscle cells of hepatic artery (ha) and portal vein (pv) branches are α-SMA-positive. B–D: In contrast, there is an increasing number of α-SMA-positive myofibroblasts surrounding the bile ducts (bd) in DDC-fed mice. Note that hepatic arteries and portal veins of the control liver and DDC-fed mice are of the same size. pv, portal vein; bd, bile duct. Original magnification, ×20.
Figure 8.
DDC feeding increases hepatic hydroxyproline content in mice. Hepatic hydroxyproline content was determined in liver homogenates from control diet-fed mice (Control) and mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (1w DDC), 4 weeks (4w DDC), and 8 weeks (8w DDC). Hepatic hydroxyproline content was significantly increased in 4- and 8-week-DDC-fed mice compared with controls. Values are mean ± SD from five animals per group. *P < 0.05.
Table 5.
Hepatic mRNA Expression of Several Genes in 1-, 4-, and 8-Week DDC-Intoxicated Mice
| Gene | Control | 1w DDC | 4w DDC | 8w DDC |
|---|---|---|---|---|
| Transport | ||||
| Bsep | 1.1 ± 0.3 | 0.9 ± 0.3 | 0.8 ± 0.2 | 0.7 ± 0.1 |
| Ntcp | 0.8 ± 0.3 | 0.2 ± 0.1* | 0.3 ± 0.1* | 0.3 ± 0.1* |
| Mrp2 | 0.9 ± 0.1 | 1.3 ± 0.2 | 1.2 ± 0.4 | 1.2 ± 0.2 |
| Mrp3 | 0.6 ± 0.3 | 2.9 ± 0.6* | 2.5 ± 0.7* | 3.6 ± 0.6* |
| Mrp4 | 0.9 ± 0.3 | 9.0 ± 3.7 | 15.8 ± 3.8* | 66.8 ± 25.4* |
| Mdr1a | 0.8 ± 0.5 | 15.1 ± 5.3* | 18.7 ± 7.4* | 11.5 ± 3.8 |
| Mdr1b | 0.4 ± 0.4 | 2.8 ± 2.4 | 1.5 ± 1.4 | 2.0 ± 0.7 |
| Mdr2 | 0.9 ± 0.2 | 2.8 ± 0.9* | 2.4 ± 0.8 * | 1.9 ± 0.3 |
| Oatp4 | 0.8 ± 0.2 | 0.4 ± 0.2* | 0.4 ± 0.2* | 0.5 ± 0.04* |
| Ost-α | 1.3 ± 0.9 | 1.8 ± 0.5 | 1.3 ± 1.0 | 1.6 ± 0.4 |
| Ost-β | 1.0 ± 0.4 | 8.0 ± 2.7 | 14.3 ± 7.8* | 8.1 ± 2.8 |
| Metabolism | ||||
| Cyp3a11 | 1.1 ± 0.2 | 1.2 ± 0.4 | 1.0 ± 0.2 | 1.0 ± 0.2 |
| Cyp2b10 | 0.6 ± 0.5 | 149.4 ± 114.8* | 148.5 ± 93.1* | 45.0 ± 8.9* |
| Inflammation | ||||
| TNF-α | 0.6 ± 0.2 | 3.1 ± 3.0 | 4.3 ± 3.1* | 10.9 ± 4.1* |
| INOS | 1.7 ± 1.4 | 4.4 ± 1.5 | 4.9 ± 3.6 | 3.1 ± 0.2 |
| Osteopontin | 1.6 ± 1.2 | 17.9 ± 7.7* | 20.5 ± 16.4* | 8.9 ± 3.6 |
| Fibrosis | ||||
| TGF-β | 0.9 ± 0.1 | 2.2 ± 0.4* | 2.0 ± 0.9 | 2.3 ± 0.04* |
| TIMP1 | 1.1 ± 1.2 | 25.9 ± 14.6* | 31.1 ± 14.5* | 26.7 ± 12.2* |
| Col 1a2 | 0.9 ± 0.2 | 4.0 ± 0.7* | 3.7 ± 1.1* | 3.3 ± 0.8* |
Values are expressed as mRNA arbitrary units (mean ± SD) and normalized to 18S rRNA expression expressed as mean.
P < 0.05 compared with standard diet-fed controls (analysis of variance with Bonferroni post-testing).
Figure 9.
DDC feeding induces hepatic osteopontin expression. Immunohistochemistry for osteopontin in (A) control liver and in livers of mice fed 0.1% (w/w) DDC-supplemented diet for (B, E, and F) 1 week, (C) 4 weeks, and (D and H) 8 weeks. A: In the control liver, bile ducts show some osteopontin expression (arrowheads). B–C: In contrast, in DDC-fed mice osteopontin expression is induced in hepatocytes (as indicated by the white arrowhead) in acinar zone 1 (zone of cholate stasis) and proliferating reactive cholangiocytes (indicated by black arrowheads). E: Note the immunoreactivity of hepatocytes along margin of the liver lobule (highlighted in red) with osteopontin preceding the development of portal-portal fibrous bridges. For better orientation, portal fields are framed in green. F: Higher magnification of E demonstrating the cytoplasmic osteopontin-staining pattern of hepatocytes along the margin of the liver acinus margin. pv, portal vein. Original magnifications: ×20 (A–D), ×10 (E), ×60 (F).
DDC Feeding Leads to Reduced Biliary Glutathione and Phospholipid Excretion and an Adaptive Hepatocellular Transporter Expression in Mice
To study the functional significance of the described cholestatic phenotype, we determined bile flow and composition in DDC-fed mice at the various time points (Table 6). Biliary excretion of cholesterol remained unaltered, and the reduction in biliary bile acid output did not reach statistical significance. Analysis of biliary bile acid composition by gas chromatography/mass spectrometry revealed no significant differences between controls and DDC-fed animals (data not shown), arguing against a major pathogenetic role of potentially toxic bile acids in the cholestatic phenotype. However, glutathione excretion was significantly reduced in DDC-fed mice, suggesting that reduced bile flow, at least in part, was related to its reduced secretion. The significance of these findings was further underlined by decreased Mrp2 expression suggesting that reduced biliary glutathione output might be related to reduced Mrp2 expression (Figure 10). In addition, we observed significantly reduced Ntcp expression, which was reciprocally accompanied by enhanced Mrp3 and Mrp4 expression as previously observed in other mouse models of cholestasis (Figure 10). The changes in transporter protein levels were accompanied by respective changes in mRNA expression levels (Table 5), indicating that these changes may be regulated, at least in part, also at a transcriptional level. In line with the unaffected bile acid output, canalicular Bsep expression was unaltered in DDC-intoxicated mice. Because bile flow only showed a slight reduction (without reaching statistical significance), the significantly increased serum bile acid levels in DDC-fed mice (Table 4) might primarily result from the pronounced reciprocal changes in Ntcp and Mrp3/4 expression.12,13,14,15
Table 6.
Bile Flow and Biliary Output Rates in 1-, 4-, and 8-Week DDC-Intoxicated Mice
| Bile flow μL/gLW/minute | Cholesterol nmol/gLW/minute | Phospholipids nmol/gLW/minute | Glutathione nmol/gLW/minute | Bile acids nmol/gLW/minute | |
|---|---|---|---|---|---|
| Control (n = 9) | 1.6 ± 0.2 | 0.5 ± 0.2 | 4.1 ± 0.6 | 6.3 ± 1.8 | 19.3 ± 10.0 |
| 1w DDC (n = 9) | 1.7 ± 0.8 | 0.5 ± 0.3 | 5.1 ± 2.6 | 2.7 ± 1.5* | 16.0 ± 13.6 |
| 4w DDC (n = 7) | 1.4 ± 0.8 | 0.4 ± 0.3 | 3.9 ± 2.8 | 2.0 ± 1.3* | 12.9 ± 15.1 |
| 8w DDC (n = 7) | 1.1 ± 0.4 | 0.4 ± 0.2 | 3.1 ± 1.5* | 1.6 ± 1.0* | 7.1 ± 3.8 |
Values are expressed as mean ± SD. gLW, gram liver weight.
P < 0.05 compared with standard diet-fed controls (analysis of variance with Bonferroni post-testing).
Figure 10.
Effects of DDC feeding on hepatic Ntcp, Mrp2, Bsep, and Mrp3 protein levels. Liver membranes were isolated from control diet-fed mice (control) and mice fed 0.1% (w/w) DDC-supplemented diet for 1 week (1w DDC), 4 weeks (4w DDC), and 8 weeks (8w DDC) and analyzed by Western blotting. Densitometry data are expressed as the fold change relative to control diet-fed animals. Values are the means from three to four animals in each group. There is a significant decrease in Ntcp and Mrp2 concomitant with a significant increase in Mrp3 and Bsep protein levels in DDC-fed mice, whereas β-actin expression remained unchanged. *P < 0.05.
Discussion
Our results show that the feeding of DDC to mice within a few weeks induces sclerosing cholangitis and a biliary-type of liver fibrosis in a highly reproducible manner, allowing longitudinal in vivo studies to uncover the mechanisms of chronic xenobiotic-induced cholangitis and its sequels including biliary fibrosis. We herein demonstrate that the cholestatic phenotype in this model is related to increased biliary porphyrin secretion and the induction of a reactive phenotype of BECs with development of bile duct injury leading to i) pericholangitis and ductular reaction resulting in onion skin-like periductal, portal-portal fibrosis, and large duct disease; ii) down-regulation of canalicular Mrp2 with impaired glutathione excretion; and iii) ultimately some degree of mechanical segmental bile duct obstruction by porphyrin-containing pigment plugs (summarized in Figure 11).
Figure 11.
Suggested pathobiology of DDC-induced cholestatic liver disease. (1) DDC leads to a reactive phenotype of BECs with overexpression of proinflammatory and profibrogenetic cytokines and adhesion molecules, and consequently to (2) infiltration of the portal field with neutrophils (ie, pericholangitis). Activation and injury of BECs results in ductular reaction triggering (3) the formation of portal-portal septa. (4) Activation and proliferation of periductal myofibroblasts leads to increased production of extracellular matrix components and consequently to sclerosing cholangitis. (5) The formation of intraductal porphyrin plugs further promotes the development of sclerosing cholangitis and biliary fibrosis. (6) At the hepatocellular level, down-regulation of bile acid uptake systems (Ntcp, Oatp4) and up-regulation of export pumps (Mrp3, Mrp4) may represent an adaptive response of hepatocytes and, at least in part, explain pronounced increase of serum bile acid levels.
An emerging concept in the pathobiology of cholangiopathies is that BECs actively participate in the pathogenesis of cholestatic liver diseases by transformation into a reactive phenotype characterized by secretion of proinflammatory and/or chemotactic cytokines (eg, TNF-α, interleukin-6, and monocyte chemoattractant protein-1) together with profibrogenic factors (eg, platelet-derived growth factor, transforming growth factor-β, and osteopontin).1,4 The interaction of VCAM with its ligand integrin α4β1 is involved in the recruitment of lymphocytes from the gut and participates in the adhesion and migration of monocytes and lymphocytes to activated endothelial cells.16 We show that DDC feeding significantly induced VCAM expression in BECs within days, which correlated well with the transmigration of neutrophils into portal fields causing acute cholangitis. On the other hand, 4 weeks of recovery from DDC intoxication resulted in significantly reduced VCAM expression and reduced transmigration of neutrophils. In addition, DDC significantly induced cholangiocellular TNF-α expression as demonstrated by in situ hybridization. This is of particular interest because TNF-α was previously shown to directly impair the barrier function of BECs.17 It is therefore attractive to speculate that pericholangitis in DDC-fed mice is directly related to the induction of a reactive BEC phenotype by DDC via increased biliary porphyrin secretion leading to leaky bile ducts (eg, physically and/or chemically via the formation of porphyrin crystals). This assumption is further supported by unchanged biliary bile acid composition but significantly increased biliary porphyrin secretion in DDC-fed mice. Because we found no evidence for a biliary enrichment of hydrophobic, potentially toxic bile acids in this model, biliary bile acids may not represent a critical initiator for the observed reactive BEC phenotype. Likewise, biliary excretion of DDC metabolites may be relevant and remains to be explored in further studies.
Another intriguing finding of this study was the pronounced induction of osteopontin expression in BECs but also in hepatocytes in acinar zone 1 and particularly at the margins of the liver lobules in DDC-fed mice, preceding the formation of portal-portal bridges. Osteopontin, a pleiotropic cytokine with potent chemoattractant and profibrogenic properties, was previously shown to be engaged in the pathogenesis of lung fibrosis but was also found to be overexpressed in human cholestatic liver disease.18 In addition, profibrogenic properties of osteopontin were demonstrated in vitro.19,20 However, its definitive role in the development of biliary fibrosis is still under debate since osteopontin was shown to be overexpressed in rodent liver after carbon tetrachloride (CCl4) treatment and in response to common bile duct ligation. However, osteopontin knock-out mice were more susceptible to CCl4 treatment and showed increased fibrosis in one previous study.19 Of interest, the induction of osteopontin expression in hepatocytes of DDC-fed mice was primarily observed in acinar zone 1 and hepatocytes at the margins of the liver lobules (ie, regions with presumably highest intralobular bile acid concentrations). The lobular distribution of osteopontin expression in DDC-fed mice makes it most attractive to speculate that this cytokine may be directly engaged in the induction of ductular reaction and associated portal-portal bridging. Furthermore, recovery from DDC feeding resulted in significant decreased osteopontin expression paralleled by partial resolution of biliary fibrosis. Taken together, these findings would be in line with Desmet’s18 hypothesis that “ductular proliferates are pace-maker of biliary fibrosis” and the proposed role of osteopontin in other fibrotic conditions, such as in lung. In addition, bile duct proliferation in the DDC model may also be triggered by locally increased biliary pressure (known to represent a potent inductor for bile duct proliferation) as a result of partial bile duct obstruction due to porphyrin plugs.
DDC feeding in mice resulted in onion skin-like periductal and portal-portal fibrosis with ductular reaction and significantly increased serum bile acid levels, representing phenotypical characteristics of a chronic cholangiopathy. However, we observed no significant reduction in bile flow together with preserved biliary bile acid secretion, which parallels previous findings in Mdr2 knockout mice with macroscopic and histological characteristics of human sclerosing cholangitis.11,21,22 Therefore, both models do not fulfill the term cholestasis in the pathophysiological sense of a “lack of sufficient amount of bile reaching the duodenum” as postulated by Erlinger.23 However, we do not know whether this is the case in any human prototypic cholestatic liver disease (eg, primary biliary cirrhosis and primary sclerosing cholangitis) with the exception of complete bile duct obstruction. The herein observed reciprocal regulation of the basolateral bile acid uptake system Ntcp and export systems Mrp3/4 may, at least in part, explain increased serum bile acid levels in the DDC model. In addition, we show that DDC feeding significantly reduced hepatic Mrp2 protein concentration probably representing the cause of reduced biliary glutathione excretion, which could partially explain the trend of decreased bile flow rates. However, reduced biliary glutathione concentration in DDC-fed mice may also result from increased hydrolysis by the induction of hepatic γ-glutamyl-transpeptidase. Finally, the cholestatic phenotype in regard to alterations in hepatocellular transport protein expression may again, at least in part, be related to partial biliary obstruction by porphyrin plugs as revealed by morphological and bile duct plastination studies.
In summary, we provide data on the mechanisms involved in the development of sclerosing cholangitis and biliary fibrosis in DDC-fed mice, making this a novel attractive model for further in vivo studies on the complicated interplay between hepatocytes, BECs, and mesenchymal cells in the pathogenesis of cholangiopathies and biliary fibrosis. In addition, our findings further underline the pivotal role of reactive BECs and ductular reaction for the development of biliary fibrosis.
Supplementary Material
Acknowledgments
We gratefully acknowledge Dr. W. Erwa (Graz) and colleagues for performing biochemical analyses of serum liver tests, and Judith Gumhold and Dagmar Silbert for excellent technical assistance.
Footnotes
Address reprint requests to Michael Trauner, M.D., Laboratory of Experimental and Molecular Hepatology, Division of Gastroenterology and Hepatology, Department of Medicine, Medical University Graz, Auenbruggerplatz 15; A-8036 Graz, Austria. E-mail: michael.trauner@meduni-graz.at.
Supported by grants P-18613 (to M.T.) from the Austrian Science Foundation and a GEN-AU project grant from the Austrian Ministry for Science (to M.T.).
P.F. and U.S. contributed equally to this work.
Supplemental material for this article can be found on http://ajp. amjpathol.org.
References
- Lazaridis KN, Strazzabosco M, LaRusso NF. The cholangiopathies: disorders of biliary epithelia. Gastroenterology. 2004;127:1565–1577. doi: 10.1053/j.gastro.2004.08.006. [DOI] [PubMed] [Google Scholar]
- Bataller R, Brenner DA. Liver fibrosis. J Clin Invest. 2005;115:209–218. doi: 10.1172/JCI24282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rippe RA, Brenner DA. From quiescence to activation: gene regulation in hepatic stellate cells. Gastroenterology. 2004;127:1260–1262. doi: 10.1053/j.gastro.2004.08.028. [DOI] [PubMed] [Google Scholar]
- Strazzabosco M, Fabris L, Spirli C. Pathophysiology of cholangiopathies. J Clin Gastroenterol. 2005;39:S90–S102. doi: 10.1097/01.mcg.0000155549.29643.ad. [DOI] [PubMed] [Google Scholar]
- Denk H, Stumptner C, Zatloukal K. Mallory bodies revisited. J Hepatol. 2000;32:689–702. doi: 10.1016/s0168-8278(00)80233-0. [DOI] [PubMed] [Google Scholar]
- Fickert P, Trauner M, Fuchsbichler A, Stumptner C, Zatloukal K, Denk H. Mallory body formation in primary biliary cirrhosis is associated with increased amounts and abnormal phosphorylation and ubiquitination of cytokeratins. J Hepatol. 2003;38:387–394. doi: 10.1016/s0168-8278(02)00439-7. [DOI] [PubMed] [Google Scholar]
- Fickert P, Trauner M, Fuchsbichler A, Stumptner C, Zatloukal K, Denk H. Bile acid-induced Mallory body formation in drug-primed mouse liver. Am J Pathol. 2002;161:2019–2026. doi: 10.1016/S0002-9440(10)64480-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakubowski A, Ambrose C, Parr M, Lincecum JM, Wang MZ, Zheng TS, Browning B, Michaelson JS, Baetscher M, Wang B, Bissell DM, Burkly LC. TWEAK induces liver progenitor cell proliferation. J Clin Invest. 2005;115:2330–2340. doi: 10.1172/JCI23486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fickert P, Trauner M, Fuchsbichler A, Stumptner C, Zatloukal K, Denk H. Cytokeratins as targets for bile acid-induced toxicity. Am J Pathol. 2002;160:491–499. doi: 10.1016/S0002-9440(10)64868-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fickert P, Zollner G, Fuchsbichler A, Stumptner C, Pojer C, Zenz R, Lammert F, Stieger B, Meier PJ, Zatloukal K, Denk H, Trauner M. Effects of ursodeoxycholic and cholic acid feeding on hepatocellular transporter expression in mouse liver. Gastroenterology. 2001;121:170–183. doi: 10.1053/gast.2001.25542. [DOI] [PubMed] [Google Scholar]
- Fickert P, Zollner G, Fuchsbichler A, Stumptner C, Weiglein AH, Lammert F, Marschall HU, Tsybrovskyy O, Zatloukal K, Denk H, Trauner M. Ursodeoxycholic acid aggravates bile infarcts in bile duct-ligated and Mdr2 knockout mice via disruption of cholangioles. Gastroenterology. 2002;123:1238–1251. doi: 10.1053/gast.2002.35948. [DOI] [PubMed] [Google Scholar]
- Fickert P, Fuchsbichler A, Marschall HU, Wagner M, Zollner G, Krause R, Zatloukal K, Jaeschke H, Denk H, Trauner M. Lithocholic acid feeding induces segmental bile duct obstruction and destructive cholangitis in mice. Am J Pathol. 2006;168:410–422. doi: 10.2353/ajpath.2006.050404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner M, Fickert P, Zollner G, Fuchsbichler A, Silbert D, Tsybrovskyy O, Zatloukal K, Guo GL, Schuetz JD, Gonzalez FJ, Marschall HU, Denk H, Trauner M. Role of farnesoid X receptor in determining hepatic ABC transporter expression and liver injury in bile duct-ligated mice. Gastroenterology. 2003;125:825–838. doi: 10.1016/s0016-5085(03)01068-0. [DOI] [PubMed] [Google Scholar]
- Denson LA, Bohan A, Held MA, Boyer JL. Organ-specific alterations in RAR α:RXRα abundance regulate rat Mrp2 (Abcc2) expression in obstructive cholestasis. Gastroenterology. 2002;123:599–607. doi: 10.1053/gast.2002.34758. [DOI] [PubMed] [Google Scholar]
- Lee J, Azzaroli F, Wang L, Soroka CJ, Gigliozzi A, Setchell KD, Kramer W, Boyer JL. Adaptive regulation of bile salt transporters in kidney and liver in obstructive cholestasis in the rat. Gastroenterology. 2001;121:1473–1484. doi: 10.1053/gast.2001.29608. [DOI] [PubMed] [Google Scholar]
- Rangaswami H, Bulbule A, Kundu GC. Osteopontin: role in cell signaling and cancer progression. Trends Cell Biol. 2006;16:79–87. doi: 10.1016/j.tcb.2005.12.005. [DOI] [PubMed] [Google Scholar]
- Hanada S, Harada M, Koga H, Kawaguchi T, Taniguchi E, Kumashiro R, Ueno T, Ueno Y, Ishii M, Sakisaka S, Sata M. Tumor necrosis factor-α and interferon-γ directly impair epithelial barrier function in cultured mouse cholangiocytes. Liver Int. 2003;23:3–11. doi: 10.1034/j.1600-0676.2003.01707.x. [DOI] [PubMed] [Google Scholar]
- O’Regan A. The role of osteopontin in lung disease. Cytokine Growth Factor Rev. 2003;14:479–488. doi: 10.1016/s1359-6101(03)00055-8. [DOI] [PubMed] [Google Scholar]
- Lorena D, Darby IA, Gadeau AP, Leen LL, Rittling S, Porto LC, Rosenbaum J, Desmouliere A. Osteopontin expression in normal and fibrotic liver. Altered liver healing in osteopontin-deficient mice. J Hepatol. 2006;44:383–390. doi: 10.1016/j.jhep.2005.07.024. [DOI] [PubMed] [Google Scholar]
- Lee SH, Seo GS, Park YN, Yoo TM, Sohn DH. Effects and regulation of osteopontin in rat hepatic stellate cells. Biochem Pharmacol. 2004;68:2367–2378. doi: 10.1016/j.bcp.2004.08.022. [DOI] [PubMed] [Google Scholar]
- Van Nieuwkerk CM, Elferink RP, Groen AK, Ottenhoff R, Tytgat GN, Dingemans KP, Van Den Bergh Weerman MA, Offerhaus GJ. Effects of Ursodeoxycholate and cholate feeding on liver disease in FVB mice with a disrupted mdr2 P-glycoprotein gene. Gastroenterology. 1996;111:165–171. doi: 10.1053/gast.1996.v111.pm8698195. [DOI] [PubMed] [Google Scholar]
- Fickert P, Fuchsbichler A, Wagner M, Zollner G, Kaser A, Tilg H, Krause R, Lammert F, Langner C, Zatloukal K, Marschall HU, Denk H, Trauner M. Regurgitation of bile acids from leaky bile ducts causes sclerosing cholangitis in Mdr2 (Abcb4) knockout mice. Gastroenterology. 2004;127:261–274. doi: 10.1053/j.gastro.2004.04.009. [DOI] [PubMed] [Google Scholar]
- Erlinger S. What is cholestasis in 1985? J Hepatol. 1985;1:687–693. doi: 10.1016/s0168-8278(85)80012-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.