Abstract
Macrophage responses are regulated by multiple secreted factors as well as by cell surface receptors, including the inhibitory signals resulting from ligation of myeloid CD200 receptors (CD200R) by the widely distributed CD200. In the absence of CD200, animals display increased susceptibility to autoimmunity and earlier onset aggressive autoimmune disease. In these current experiments, an agonist monoclonal rat anti-mouse CD200R (DX109) antibody delivered a negative signal to bone marrow-derived macrophages, which suppressed interferon (IFN)γ-mediated nitric oxide (NO) and interleukin-6 production. Experimental autoimmune uveoretinitis (EAU) was used as a model of organ-specific autoimmunity in the eye, a tissue with extensive neuronal and endothelial CD200 expression. In mice lacking CD200 (CD200−/−), increased numbers of retina-infiltrating macrophages displaying heightened NO responses were observed during EAU. In addition, we aimed to suppress disease by maintaining tonic suppression of macrophage activation via CD200R. Systemically administered DX109 monoclonal antibody suppressed EAU despite maintained T-cell proliferation and IFNγ production. Furthermore, locally administered DX109 monoclonal antibody resulted in an earlier resolution of disease. These experiments demonstrate that promoting CD200R-mediated signaling can successfully prevent full expression of IFNγ-mediated macrophage activation and protect against tissue damage during autoimmune responses.
The versatility of macrophage responses is largely because of their ability to respond to both endogenous and exogenous signals via a multitude of cell surface receptors.1,2 Potential constitutive regulation of macrophage activation has recently been demonstrated in CD200−/− mice in which increased macrophage numbers, including microglial numbers in central nervous system and retina, are associated with increased expression of NOS2,3,4 increased susceptibility to autoimmune disease, and microglial activation to central nervous system degeneration.3,4,5,6 CD200 (originally OX2) is a membrane glycoprotein member of the Ig superfamily expressed on a wide variety of cell types, including lymphoid cells, neurons (including central nervous system and retina), and endothelial cells.6,7,8,9 The CD200 receptor (CD200R) is most highly expressed on leukocytes of myeloid lineage,7,8,10,11 mast cells,12 neutrophils, and dendritic cells but also on some T-cell subsets. Both interferon (IFN)γ- and interleukin (IL)-17-stimulated macrophage cytokine secretion has been shown to be modulated through CD200R.12 Mice receiving soluble CD200 protein are resistant to collagen-induced arthritis induction5,13 and show prolonged allo- and xenograft survival.5,14
Here, we show that regulating macrophage activity via CD200R ligation can suppress IFNγ-mediated macrophage activation, leading to reduced production of NO and IL-6, but lipopolysaccharide (LPS)-mediated activation was unaffected by CD200R ligation. A model of CD4+ T-cell organ-specific autoimmunity, experimental autoimmune uveoretinitis (EAU)15,16,17 was used in these studies because of extensive neuronal CD200 expression in the eye where tissue destruction is largely mediated via activated macrophages and NO damage. Systemic administration of a rat anti-mouse CD200R monoclonal antibody (mAb) (DX109) resulted in suppression of EAU and tissue damage even in the presence of systemic T-cell proliferation and IFNγ production. Local administration of DX109 resulted in an early resolution of disease. These in vivo data support the growing evidence that eliciting macrophage CD200R signaling in the presence of CD4+ T-cell responses is a novel route to restrict tissue damage in organ-specific autoimmunity.
Materials and Methods
Mice
C57BL/6 and B10.RIII mice were originally obtained from Harlan UK Limited (Oxford, UK), CD200-deficient mice (CD200−/−) of the background strain C57BL/6 were obtained from DNAX, Palo Alto, CA,6 and breeding colonies were established within the Animal Services Unit at Bristol University, Bristol, UK, for further experimentation. All mice were housed under specific pathogen-free conditions with continuously available food and water. Female mice immunized for disease induction were aged between 6 and 8 weeks. Treatment of animals conformed to the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research.
Reagents
Human (h)RBP-3 peptide 1-20 (GPTHLFQPSLVLDMAKVLLD) and hIRBP 161-180 (SGIPYIISYLHPGNTILHVD) were obtained from Sigma-Genosys Ltd. (Poole, UK). Peptide purity was determined by high-performance liquid chromatography. Peptide preparations were aliquotted and stored at −80°C. Complete media consisted of Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal calf serum, 100 U/ml penicillin-streptomycin, 2 mmol/L l-glutamine, 1 mmol/L sodium pyruvate, and 5 × 10−5 mol/L 2-mercaptoethanol (all from Invitrogen, Paisley, UK).
The agonist rat IgG1-anti-mouse CD200R mAb (DX109) was generated from rats using immunogenic fusion proteins consisting of the extracellular domains of CD200R gene fused to the Fc domain of human immunoglobulin. The control isotype mAb used was a rat anti-human IL-4. Both mAb preparations used contained <0.1 ng of endotoxin/mg of protein, as determined by the Limulus Amebocyte Lysate Pyrogen Testing kit, QCL-1000 (Cambrex, East Rutherford, NJ).
EAU Induction and Scoring
C57BL/6 and CD200−/− mice were immunized subcutaneously in one flank with 500 μg/mouse hRBP-3 1-20 peptide in phosphate (PBS) (2% dimethyl sulfoxide), in emulsion with complete Freud’s adjuvant [1 mg/ml; 1:1 (v/v)] supplemented with 1.5 mg/ml Mycobacterium tuberculosis complete H37 RA (BD Biosciences) and 1.5 μg of Bordetella pertussis toxin (Sigma-Aldrich, Dorset, UK) intraperitoneally (i.p.). B10.RIII mice were immunized subcutaneously in one flank with 50 μg/mouse IRBP 161-180 peptide in PBS (2% dimethyl sulfoxide), in complete Freud’s adjuvant as described earlier, with an additional i.p. injection of 1 μg of Bordetella pertussis toxin. At various time points after immunization, eyes were enucleated and carefully snap-frozen, oriented in optimal cutting temperature compound (R. Lamb Ltd., East Sussex, UK). Serial 12-μm sections were thawed at room temperature and fixed in acetone for 10 minutes. Sections were stained with rat anti-mouse monoclonal anti-CD45 antibody (Serotec, Oxford, UK) and counterstained with hematoxylin (ThermoShandon, Pittsburgh, PA). Sections were scored for inflammatory infiltrate (presence of CD45-positive cells) and structural disease (disruption of morphology), as described previously.18
Generation of Bone Marrow-Derived Macrophages
Bone marrow-derived macrophages (BM-MΦ) were generated as previously described.19 In brief, bone marrow cells were washed in DMEM media and resuspended at 1 × 105 cells/ml in complete media supplemented with 5% horse serum (Invitrogen) and 50 ng/ml macrophage-colony stimulating factor. Cell suspension (50 ml) was transferred to Teflon-coated tissue culture bags (supplied by Dr. M. Munder, University of Heidelberg, Heidelberg, Germany) and incubated for 8 days at 37°C in 5% CO2. Cells were removed from the Teflon bags and plated at a concentration of 1 × 105 cells/well in 96-well, flat-bottomed plates (Corning-Costar, Corning, NY) in complete media. After 2 hours, BM-MΦ were stimulated with recombinant IFNγ cytokine (100 U/ml; Peprotech, London, UK), LPS (1 μg/ml to 0.1 pg/ml; Sigma-Aldrich), or left unstimulated.
Quantification of NO Synthesis
Accumulated NO production was measured after 36 hours in culture by assaying culture supernatants for the stable reaction product of NO (nitrite) against a sodium nitrite standard on the same plate. A 50-μl total volume of each cell-free supernatant was incubated (v/v) of Griess reagent [0.5% sulfanilamide and 0.05% N-(1-napthyl) ethylenediamine dihydrochloride in 2.5% phosphoric acid] in 96-well flat-bottomed plates for 10 minutes at room temperature. The optical densities were measured at 540 nm, with a reference filter of 630 nm.
Isolation of Retinal Myeloid Cells
Eyes were enucleated from immunized mice, and the retinas of each animal were dissected microscopically in ice-cold 1% bovine serum albumin (Sigma-Aldrich) in PBS (1% PBSA). Retinas were then mechanically disrupted through a 40-μm cell strainer to obtain a single-cell suspension. A sample was removed to determine the purity and percentage of F4/80-positive macrophages by staining cells with fluorescein isothiocyanate-conjugated anti-mouse F4/80 (Serotec) before flow cytometric analysis. Cells were plated at 5 × 105 F4/80-positive cells/ml for 2 hours, to allow the isolation of macrophages from other cell contaminants via plastic adherence. The medium was replaced and assays performed.
Splenocyte Proliferation
Spleens were collected from immunized mice at various time points and mechanically disrupted through a 40-μm cell strainer and red blood cells lysed. Cells were then stimulated with the immunizing peptide and incubated for 2 days at 37°C in 5% CO2, in a humidified atmosphere. Supernatants were aspirated at 48 hours for cytokine assays and frozen at −80°C, and the plates were pulsed with 18.5-kBq triturated thymidine (GE Healthcare, Bucks, UK) per well for the last 18 hours of incubation. Cells were harvested with a 24-well harvester and thymidine uptake [measured in counts per minute (cpm)] was determined by liquid scintillation with a microbeta liquid scintillation counter (Wallac 1450; PerkinElmer Life Sciences, Cambridge, UK).
Cytokine Assays
Cytokine production (IFNγ, IL-2, IL-4, and IL-10) in culture supernatants was assayed by capture enzyme-linked immunosorbent assay (ELISA). Briefly, on day 0, capture antibody (in carbonate buffer, pH 9.6) was applied to flat-bottomed, 96-well plates (Nunc Immuno Plate; VWR, Lutterworth, UK) and left overnight at 4°C. On day 1, nonspecific binding sites were blocked using 1% PBSA for 1 hour at 37°C. The supernatants were then added for 1 hour at 37°C, and the plates washed with 0.5% Tween 20 (Sigma-Aldrich) in PBS. The detection antibody was added for 1 hour at room temperature, and the plates washed as just described. Extra-avidin peroxidase (Sigma-Aldrich) was applied to the plates for 30 minutes at room temperature, plates were washed, and chromogen substrate [3,3′,5,5′-tetramethylbenzidine and hydrogen peroxide (H2O2); BD Biosciences, Oxford, UK] were added. The reaction was stopped using 2 N sulfuric acid (H2SO4) (Sigma-Aldrich). The monoclonal antibody pairs for capture and detection and were obtained from BD Pharmingen. Recombinant cytokines (BD Pharmingen) were used as standards, with curves generated from doubling dilutions used to calculate concentrations of cytokine in the test sample.
Therapeutic Intervention
Systemic administration of mAb was performed by intravenous (i.v.) tail injection in B10.RIII mice immunized as described above. In brief, mice were placed under heat lamp for 5 minutes in order for tail vein to dilate and placed in a restrainer, and 120 μg of mAb/100 μl of PBS was injected using a 25-gauge needle. Injections were performed on days 10, 14, and 18 after immunization.
Local administration of mAb was performed by intravitreal injection in anesthetized mice. The left eye was proptosed and held in position with a pair of forceps, while 10 μg of mAb/5 μl of PBS was injected using a 33-gauge hypodermic needle (Hamilton, Bonaduz, Switzerland). The injection site was treated with chloramphenicol ointment and globe reposited. Injections were performed on days 10 and 14 after immunization.
Statistical Analyses
Using Prism4 software (GraphPad Software Inc., San Diego, CA), comparisons of statistical significance between groups were assessed using the Mann-Whitney U-test.
Results
Increased Myeloid Infiltrate and Early Nitrite Response during EAU in the Absence of CD200
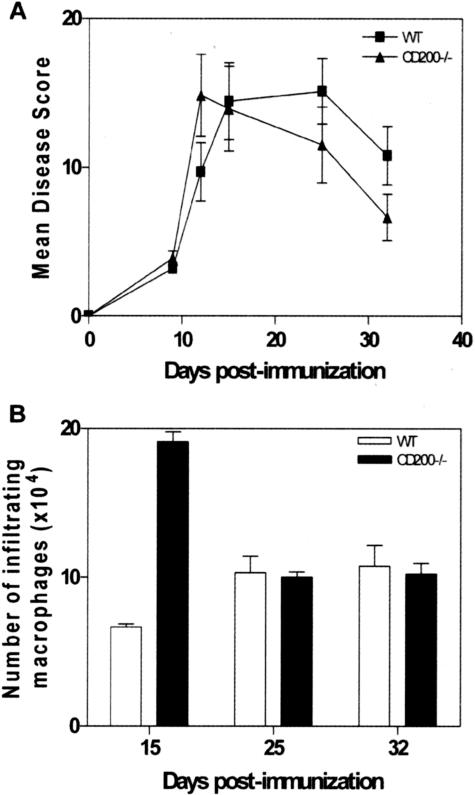
We have previously shown that inducing EAU, a model of autoimmunity in which tissue damage is dependent on macrophage-derived nitrite production,20,21 in CD200−/− mice leads to earlier onset and increased severity of disease.3 Here, we confirm the kinetics of the disease course in CD200−/− mice and clearly show the early onset of EAU in mice lacking CD200 (Figure 1A). The overall severity of the disease in terms of histological assessment is no different between the strains, suggesting other regulatory mechanisms may be involved. The absence of CD200, however, does lead to a marked increase in the numbers of infiltrating macrophages before the peak of disease compared with wild-type mice (Figure 1B). Cells isolated from the retina on day 15 after immunization show a significant increase in the number of F4/80-positive cells as confirmed by flow cytometry [6.67 × 10−4 and 19.11 × 10−4 macrophages, C57BL/6 and CD200−/−, respectively (P < 0.0002)]. F4/80+ retinal-derived macrophage activation was determined by the level of NO production. On day 15, before disease onset, macrophages isolated from CD200−/− mice display increased levels of NO compared with wild-type mice. By the peak of disease (day 25 after immunization), NO levels were reduced in CD200−/− mice and could not be further stimulated with IFNγ in vitro, unlike wild-type C57BL/6 retinal-derived myeloid cells (Table 1). These results are consistent with our previous data of NOS2 expression in F4/80+ retinal macrophages during EAU in CD200−/− mice.3
Figure 1.
CD200−/− mice demonstrate earlier EAU onset with increased numbers of infiltrating macrophages. A: C57BL/6 and CD200−/− mice were immunized with hRBP-3 1-20, eyes were harvested at day 9, 12, 15, 25, and 32 after immunization, and average total disease score was calculated and plotted. n = 5/time point, data representative of two separate experiments. B: C57BL/6 and CD200−/− mice were immunized and eyes taken at 15, 25, and 32 days after immunization. The number of F4/80-positive infiltrating macrophages isolated from each eye was determined by flow cytometry. n = 3/time point, data representative of two separate experiments.
Table 1.
NO Production from Retinal-Derived Macrophages
| Day | WT
|
CD200−/−
|
||
|---|---|---|---|---|
| Control | IFNγ | Control | IFNγ | |
| 15 | 1028 (±500) | 1568 (±209) | 2271 (±215) | 2597 (±452) |
| 25 | 1280 (±116) | 2558 (±558) | 1542 (±193) | 1509 (±711) |
Eyes from WT and CD200−/− animals were harvested at days 15 and 25 after immunization, and F4/80-positive macrophages were isolated. Cells were plated at 5 × 105 cells/ml; after 2 hours, media alone or recombinant IFNγ were added to give final concentration of 20 U/ml. After 24 hours, NO production (nM) in culture supernatants was analyzed using the Griess reaction. Results are calculated as mean ± SEM of eight wells.
CD200R Expression Unaffected by IFNγ/LPS Stimulation
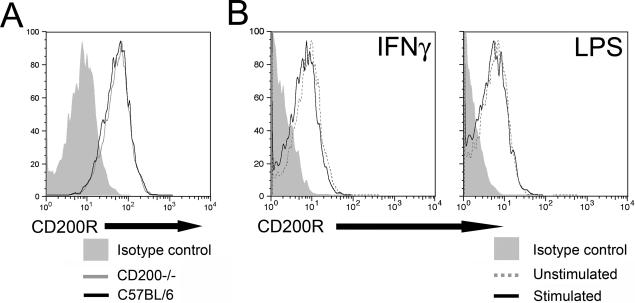
Uncoupling CD200:CD200R interaction through the deletion of CD200 results in earlier onset EAU with increased cellular infiltrate. In addition, we have previously shown a similar effect using blocking antibodies that prevent signaling through CD200R.6,7 Therefore, we predicted that disease may be ameliorated by increased inhibitory signaling in macrophages through CD200R using an agonist antibody. We have previously shown that in rat EAU there is persistent myeloid-CD200R expression during peak disease.8 To use CD200R as a therapeutic target, it was important to confirm the stability of CD200R expression during murine macrophage activation.
Analysis of wild-type C57BL/6 and CD200−/− BM-MΦ demonstrated that both strains express the CD200R at equivalent levels (Figure 2A). Therefore, we sought to determine the stability of CD200R expression on activated BM-MΦ from C57BL/6 mice. Cells were generated as described and plated at a concentration of 1 × 105 cells/well, rested for 2 hours before stimulation with 100 U/ml of recombinant IFNγ or 0.1 μg/ml LPS. The macrophages were analyzed after an additional 48 hours by flow cytometry for their expression of CD200R. Neither IFNγ nor LPS stimulation altered the cell surface expression of CD200R when compared with unstimulated macrophages (Figure 2B) or freshly isolated BM-MΦ (data not shown). In addition, no differences in the level of CD200R expression following stimulation for 24 hours or stimulation with IFNγ at 20 U/ml or LPS at 0.01 and 1 μg/ml were observed (data not shown).
Figure 2.
Expression of CD200R on BM-MΦ. A: C57BL/6 and CD200−/− BM-MΦ were cultured in 96-well plates in complete DMEM at a concentration of 1 × 105 cells/well for 24 hours. Cells were stained with either Alexa 647-conjugated anti-CD200R mAb or Alexa 647-conjugated relevant isotype control antibody (Serotec). Macrophages were then analyzed by flow cytometry. Graph shows the cell-surface expression of CD200R on macrophages from C57BL/6 (black line) or CD200−/− (gray line), with isotype control staining shown as filled gray. Data are representative of two independent experiments. B: C57/BL6 BM-MΦ were cultured in 96-well plates in complete DMEM at a concentration 1 × 105 cells/well. After 2 hours, LPS or recombinant IFNγ was added to give final concentrations of 0.1 μg/ml LPS or 100 U/ml IFNγ. Following a further 48 hours of culture, cells were stained with either Alexa 647-conjugated anti-CD200R mAb or Alexa 647-conjugated relevant isotype control antibody, before analysis by flow cytometry. Graphs show the cell-surface expression of CD200R by macrophages stimulated with LPS or IFNγ (black lines) compared with unstimulated macrophages (gray lines). Isotype control staining on stimulated macrophages is depicted as filled gray and is similar to isotype staining of unstimulated cells. Data are representative of three independent experiments.
Suppression of EAU with Systemic Administration of DX109
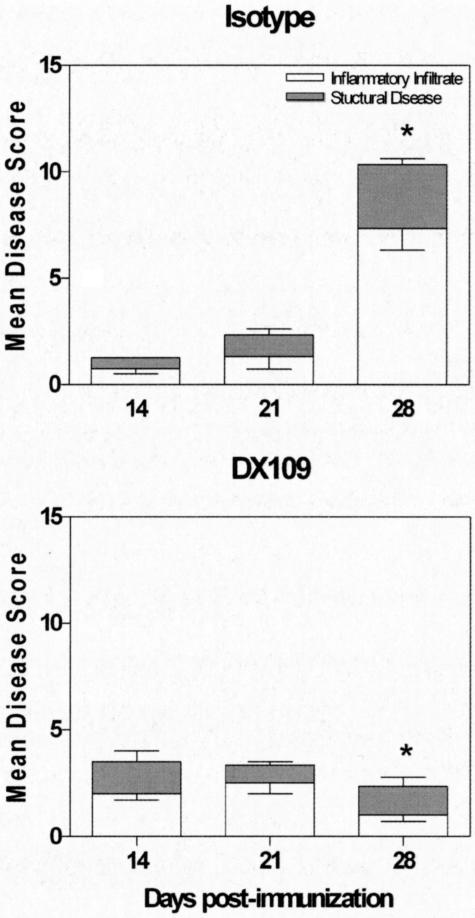
Given the stability of CD200R expression in the face of macrophage activation with LPS or IFNγ, we sought to determine whether or not CD200R signaling using an agonist DX109 mAb could suppress EAU. Our initial experiments were performed in C57BL/6 mice, a strain that is moderately susceptible to EAU induction with low severity when immunized subcutaneously with 500 μg each of hRBP-3 1-20 emulsified in complete Freud’s adjuvant with pertussis toxin intraperitoneally. Systemic administration of either DX109 or an isotype control mAb was performed via a tail vein (i.v.) injection on days 10, 14, and 18 after immunization. On days 14, 21, and 28, eyes were snap-frozen, sectioned, and stained with anti-CD45 antibody. Three sections per treatment per time point were scored for both inflammatory infiltrate and structural morphology.18 Both groups of mice display similar low-level disease scores for inflammatory infiltrate and structural damage on days 14 and 21 (start of peak disease) (Figure 3). However, by day 28, systemic administration of DX109 significantly reduced the overall disease score when compared with isotype controls (P = 0.0022). This observation suggests that administration of DX109 in the C57BL/6 model does confirm a trend toward EAU suppression. To confirm and demonstrate further the efficacy of DX109-induced suppression, we went on to use B10.RIII mice, a strain that is highly susceptible to EAU induction, and demonstrates greater disease severity. Mice were immunized subcutaneously with 50 μg each of hIRBP 161-180 emulsified in complete Freud’s adjuvant with pertussis toxin intraperitoneally. Systemic administration with either DX109 or an isotype control mAb and processing of eyes for histological assessment were performed as described previously.
Figure 3.
Systemic administration of DX109 suppresses EAU in C57BL/6 mice. Eyes were harvested at the days after immunization indicated, and average disease score ± SD of inflammatory infiltrate and structural disease is shown. n = 3/time point; data are representative of two separate experiments. Mann-Whitney U-test was performed for total disease score on day 28. *P = 0.0022.
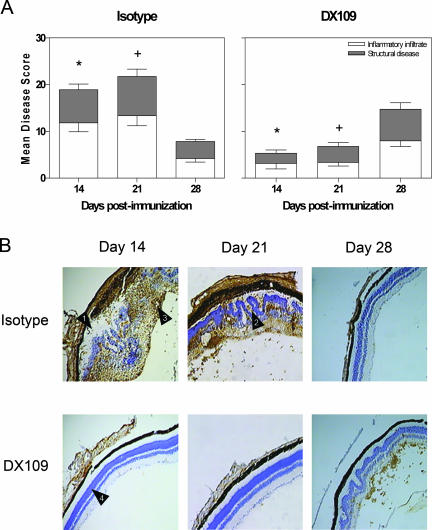
By the first time point on day 14 (start of peak disease), mice treated with DX109 displayed significantly reduced overall disease score when compared with the isotype controls (*P = 0.0075) (Figure 4A). The extensive CD45-positive cellular infiltrate and accompanying structural damage (including retinal disruption, chronic folding, and vasculitis) were clearly visible in the isotype-treated group (Figure 4B). In contrast, the retinas from the DX109-treated group seemed normal and healthy, with maintained photoreceptor layers and minimal infiltrate. This reduction in disease severity in the DX109-treated group compared with the isotype-treated group remains significant (+P = 0.0043) and is clearly evident until day 21 after immunization (at the end of peak disease phase). The difference between the two groups was not significant at day 28 after immunization (resolution phase of disease), with increased disease scores in DX109-treated animals and substantial CD45-positive infiltrate, as well as retinal disruption.
Figure 4.
Systemic administration of DX109 suppresses EAU. A: Eyes were harvested at the days postimmunization indicated, and average disease score ± SD of inflammatory infiltrate and structural disease is shown. n = 6/time point. Data representative of two independent experiments n = 6. Mann-Whitney U-test was performed total disease score. *P = 0.0075, +P = 0.0043. B: Representative 12-μm sections from two separate experiments at days 14, 21, and 28 after immunization. Photographs show sections stained with anti-CD45 antibody counterstained with hematoxylin, from mice either treated with an isotype control or DX109 administered on days 7, 14, and 18 after immunization. In isotype mAb-treated animals, there was marked retinal destruction as indicated by arrows, with retinal detachment (1), chronic retinal folding (2), and retinal vasculitis (3). In the majority of DX109-treated animals, the retina appeared normal with maintained photoreceptors (4), although by day 28, retinal folds and low-level infiltrate were present. Original magnifications, ×100.
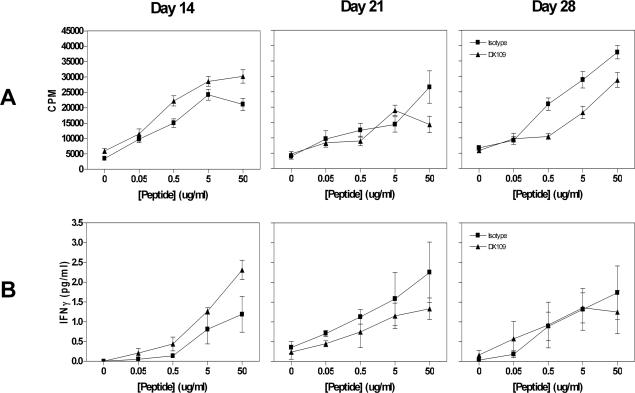
Since DX109 was administered systemically, it may have additional adverse effects on the peripheral immune system. To determine whether or not this was the case, splenocytes from mice at each time point were stimulated with the immunizing peptide, and T-cell proliferation and cytokine production were assessed. The proliferative response of splenocytes to reactivation with hIRBP 161-180 from both the DX109 and isotype-treated groups is similar and maintained at each time point (Figure 5A). Furthermore, treatment does not suppress the production of IFNγ (Figure 5B) or IL-2 (data not shown). At all time points no IL-4 or IL-10 was detected in supernatants by enzyme-linked immunosorbent assay.
Figure 5.
Maintained splenocyte proliferation and cytokine production. A: Proliferation of splenocytes taken from isotype- or DX109-treated B10.RIII mice 14, 21, and 28 days after immunization. Data are representative of two separate experiments, and each condition was plated in triplicate. B: Cytokine (IFNγ) production of splenocytes stimulated with hRBP-3 161-180 taken from isotype- or DX109-treated B10.RIII mice at the indicated time points.
CD200R Signaling Is Independent of NOS2 through Toll-Like Receptor Signaling
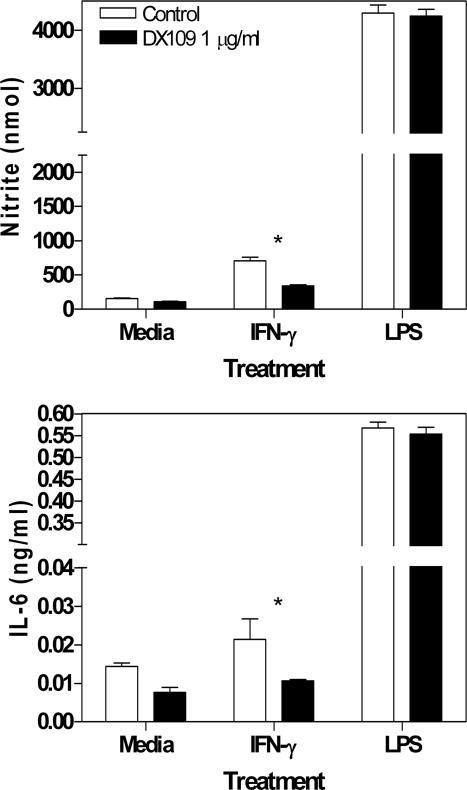
Administration of the CD200R-specific agonist mAb DX109 clearly suppresses the development of EAU. Previously, IFNγ-mediated macrophage NO production has been linked to EAU; therefore, we investigated whether or not DX109 treatment can inhibit such classical macrophage activation. C57BL/6 BM-MΦ were preincubated with DX109 for 1 hour before stimulation with IFNγ or LPS. After 36 hours, culture supernatants were analyzed for NO and IL-6 production. DX109 mAb treatment was able to suppress IFNγ-mediated NO production and IL-6 production when compared with isotype (Figure 6). However, pretreatment of macrophages with DX109 was unable to inhibit LPS-mediated macrophage NO or IL-6 production (Figure 6). Preincubation with DX109 had no effect on IFNγ-induced MHCII or CD40 cell surface expression (data not shown).
Figure 6.
DX109 can inhibit NO and IL-6 production from IFNγ-stimulated BM-MΦ. C57BL/6 bone marrow-derived macrophages were plated in 96-well plates in complete DMEM at a concentration 1 × 105 cells/well. After 2 hours, DX109 was added at 1 μg/ml for 1 hour, before recombinant IFNγ or LPS was added to give final concentrations of 50 U/ml IFNγ or 0.1 μg/ml LPS. All treatments were performed in triplicate. After 36 hours, NO and IL-6 production in culture supernatants was analyzed using the Griess reaction and enzyme-linked immunosorbent assays, respectively. Data are representative of three independent experiments. The effect of DX109 treatment on NO and IL-6 production was assessed by using a Mann-Whitney U-test (*P = 0.05).
Earlier Resolution of EAU with Local Administration of DX109
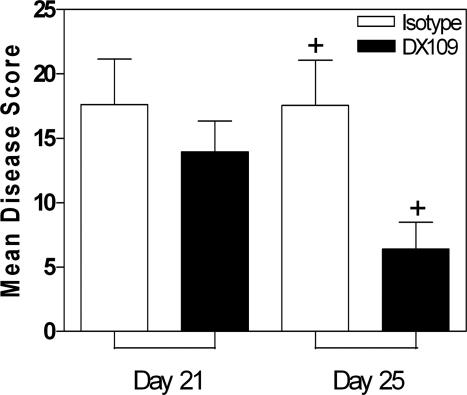
Our data suggest that the persistent suppression of EAU achieved when DX109 is systemically administered requires maintained tonic signaling through CD200R. This signaling is likely to be more consistently maintained when DX109 is delivered via a local route. Such local delivery may also limit any adverse systemic effects of peripheral macrophage deactivation. To this end, DX109 or the isotype control mAb was administered to B10.RIII mice via an intravitreal injection on days 10 and 14 after immunization. The peak of disease in this model is on day 14, thus treating at these times ensures DX109 saturation of the target tissue during disease peak. On days 21 and 25, eyes were snap-frozen, sectioned, and stained with anti-CD45 antibody. Three sections per treatment per time point were scored for total EAU disease (Figure 7). Total disease score was slightly reduced at day 21 in treated mice and significantly lower by d25 (+P = 0.0224), as demonstrated by a 55% reduction in disease when compared with the isotype.
Figure 7.
Local administration of DX109. Eyes were harvested at the days after immunization indicated, and average total disease score ± SD is shown. n = 8/time point. Data are mean of two independent experiments, n = 8. Mann-Whitney U-test was performed for total disease score on day 25. +P = 0.0224.
Discussion
The ability to target inhibitory receptors is emerging as an effective mechanism to “switch-off” antigen nonspecific effector cells during inflammatory disease. CD200 and its myeloid receptor CD200R is one inhibitory pathway that seems to regulate autoimmune disease as well as graft rejection. In this study, we show that inducing myeloid CD200R signaling via the systemic administration of an agonist rat anti-mouse mAb, DX109 successfully suppresses an archetypal organ-specific T-cell-mediated disease, EAU. Under normal conditions, CD200 is expressed abundantly within the eye, on retinal vascular endothelium and glial fibrillary acidic protein (GFAP)-negative neurons in retina and optic nerve, and on a subpopulation of CD45-positive perivascular and juxtavascular cells, and during EAU, CD200R expression is observed on infiltrating macrophages.8 A role for myeloid CD200R in the control of macrophage activation in experimental models of autoimmunity was indicated by studies that showed that CD200−/− mice display earlier onset of EAU.3 Here, we directly confirm that CD200R signaling can control autoimmunity. As the disease resolves, the number of infiltrating macrophages is equivalent in both CD200−/− and wild-type mice, with equivalent disease severity. Previous studies have alluded to a protection against chronically exacerbated disease in CD200−/− mice by demonstrating that in later disease there is an increase in Th2-associated cytokines and IL-10 in response to recall antigen, an increase in the spleen of IL-10-secreting cells, and the emergence of splenic CD3+CD4+CD25+ cells.22 The apparent inability of the retina to control trafficking of the number of myeloid infiltrating leukocytes could also be due to a direct effect of no down-regulatory signal of CD200 interaction with CD200R at the level of the retinal vascular endothelium.
During EAU, resident microglia followed at peak disease by infiltrating macrophages are activated in response to T-cell-derived IFNγ secretion.20,21 We have shown that in CD200−/− mice retinal macrophages (whether resident or infiltrating) have a higher constitutive NO production, which is not suppressed by the addition of transforming growth factor β (data not shown). Both the increased production of the effector molecule NO, coupled with the increased number of infiltrating macrophages in CD200−/− mice, support our previous findings of increased NOS2 expression.3 In addition, the observation that macrophages from CD200−/− mice are unable to respond to transforming growth factor β, which is constitutively produced by the retinal pigment epithelium, may also contribute to the disease outcome, either via unrestrained NO production or increased monocyte chemotaxis. The ability to control NO-induced tissue destruction is one avenue of controlling inflammatory eye disease23 and plausibly via down-regulating macrophage activation through CD200R signaling. In vitro suppression of bone marrow macrophage NO and IL-6 production was observed when the CD200R agonist, DX109 was preincubated with cells before IFNγ treatment. These current results support other data of DX109 agonist CD200R mAb inhibition of mast cell activation through a Dok-MAPK-Erk pathway24 and that CD200R signaling suppresses IFNγ and IL-17-stimulated cytokine secretion including tumor necrosis factor α from mouse peritoneal macrophages.12 However, as with previous reports the in vitro suppression of LPS-induced macrophage activation was not complete, with no suppression of NO or IL-6. We have previously determined that IFNγ-mediated NO production during EAU is reduced in TNFR1−/− mice, which are primed but remain resistant to EAU.25 Conversely, in vitro studies showed that stimulation of TNFR1−/− macrophages by LPS does induce an NO response. These results indicate that pathogen-associated molecular patterns are present at priming and are sufficient to induce an immune response in the absence of TNFR1. However, pathogen-associated molecular pattern signaling via TLR-4 is not present within the retina, or within this tissue TLR-4 interactions with endogenous ligands are protective, or signals that deactivate the immune response, such as those delivered by CD200R, are dominant. Either way in the current context of these data, Toll-like receptor signaling bypassing CD200R either may be advantageous therapeutically, reducing unwanted adverse effects, or may indeed accentuate Dok-1 and Dok-2 signaling and effect of CD200R inhibitory signals.26 Given the myeloid specificity of DX109 binding to CD200R, the effect of DX109 will not be via directly suppressing the efferent limb of the immune response but may possibly reduce effector T-cell function in the efferent limb via suppression of myeloid derived cytokines such as IL-23 and IL-6.
Given the success of these and previous in vitro experiments showing suppression of macrophage activation with DX109, we have now been able to show that systemic delivery suppressed tissue destruction during EAU in the presence of T-cell responses and IFNγ production. Direct local administration of DX109 before and during peak disease resulted in a 50% reduction of disease. The increasing evidence of possible therapeutic potential can be evaluated well in EAU, where discrimination between inflammatory infiltrate and tissue destruction can be made. Moreover, there remains the possibility to investigate further the local delivery and efficacy at the site of inflammation as has been previously highlighted in experimental usage of gene therapy suppression of EAU27 and, more recently, in the control of angiogenesis with antivascular endothelial growth factor agents in age-related macular degeneration.28
Acknowledgments
We thank Carly Guyver and Debatri Banerjee for technical assistance.
Footnotes
Address reprint requests to Professor Andrew D. Dick, Department of Clinical Sciences South Bristol, Bristol Eye Hospital, Lower Maudlin St., Bristol BS1 2LX, UK. E-mail: a.dick@bristol.ac.uk.
See related commentary on page 396
Supported by the National Eye Research Centre.
Current address of C.J.C.: Department of Medical Biochemistry and Immunology, Cardiff University, Cardiff, United Kingdom.
Current address of J.D.S.: Lilly Research Laboratories, Indianapolis, Indiana.
References
- Gordon S. Pattern recognition receptors: doubling up for the innate immune response. Cell. 2002;111:927–930. doi: 10.1016/s0092-8674(02)01201-1. [DOI] [PubMed] [Google Scholar]
- Gordon S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3:23–35. doi: 10.1038/nri978. [DOI] [PubMed] [Google Scholar]
- Broderick C, Hoek RM, Forrester JV, Liversidge J, Sedgwick JD, Dick AD. Constitutive retinal CD200 expression regulates resident microglia and activation state of inflammatory cells during experimental autoimmune uveoretinitis. Am J Pathol. 2002;161:1669–1677. doi: 10.1016/S0002-9440(10)64444-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banerjee D, Dick AD. Blocking CD200-CD200 receptor axis augments NOS-2 expression and aggravates experimental autoimmune uveoretinitis in Lewis rats. Ocul Immunol Inflamm. 2004;12:115–125. doi: 10.1080/09273940490895326. [DOI] [PubMed] [Google Scholar]
- Gorczynski RM. Transplant tolerance modifying antibody to CD200 receptor, but not CD200, alters cytokine production profile from stimulated macrophages. Eur J Immunol. 2001;31:2331–2337. doi: 10.1002/1521-4141(200108)31:8<2331::aid-immu2331>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- Hoek RM, Ruuls SR, Murphy CA, Wright GJ, Goddard R, Zurawski SM, Blom B, Homola ME, Streit WJ, Brown MH, Barclay AN, Sedgwick JD. Down-regulation of the macrophage lineage through interaction with OX2 (CD200). Science. 2000;290:1768–1771. doi: 10.1126/science.290.5497.1768. [DOI] [PubMed] [Google Scholar]
- Wright GJ, Puklavec MJ, Willis AC, Hoek RM, Sedgwick JD, Brown MH, Barclay AN. Lymphoid/neuronal cell surface OX2 glycoprotein recognizes a novel receptor on macrophages implicated in the control of their function. Immunity. 2000;13:233–242. doi: 10.1016/s1074-7613(00)00023-6. [DOI] [PubMed] [Google Scholar]
- Dick AD, Broderick C, Forrester JV, Wright GJ. Distribution of OX2 antigen and OX2 receptor within retina. Invest Ophthalmol Vis Sci. 2001;42:170–176. [PubMed] [Google Scholar]
- Preston S, Wright GJ, Starr K, Barclay AN, Brown MH. The leukocyte/neuron cell surface antigen OX2 binds to a ligand on macrophages. Eur J Immunol. 1997;27:1911–1918. doi: 10.1002/eji.1830270814. [DOI] [PubMed] [Google Scholar]
- Barclay AN, Wright GJ, Brooke G, Brown MH. CD200 and membrane protein interactions in the control of myeloid cells. Trends Immunol. 2002;23:285–290. doi: 10.1016/s1471-4906(02)02223-8. [DOI] [PubMed] [Google Scholar]
- Nathan C, Muller WA. Putting the brakes on innate immunity: a regulatory role for CD200? Nat Immunol. 2001;2:17–19. doi: 10.1038/83124. [DOI] [PubMed] [Google Scholar]
- Jenmalm MC, Cherwinski H, Bowman EP, Phillips JH, Sedgwick JD. Regulation of myeloid cell function through the CD200 receptor. J Immunol. 2006;176:191–199. doi: 10.4049/jimmunol.176.1.191. [DOI] [PubMed] [Google Scholar]
- Gorczynski RM, Chen Z, Yu K, Hu J. CD200 immunoadhesin suppresses collagen-induced arthritis in mice. Clin Immunol. 2001;101:328–334. doi: 10.1006/clim.2001.5117. [DOI] [PubMed] [Google Scholar]
- Gorczynski G. Role of CD200 in graft rejection. Mod Asp Immunobiol. 2002;2:130–133. [Google Scholar]
- Hankey DJ, Lightman SL, Baker D. Interphotoreceptor retinoid binding protein peptide-induced uveitis in B10.RIII mice: characterization of disease parameters and immunomodulation. Exp Eye Res. 2001;72:341–350. doi: 10.1006/exer.2000.0957. [DOI] [PubMed] [Google Scholar]
- Caspi RR, Roberge FG, Chan CC, Wiggert B, Chader GJ, Rozenszajn LA, Lando Z, Nussenblatt RB. A new model of autoimmune disease. Experimental autoimmune uveoretinitis induced in mice with two different retinal antigens. J Immunol. 1988;140:1490–1495. [PubMed] [Google Scholar]
- Avichezer D, Silver PB, Chan CC, Wiggert B, Caspi RR. Identification of a new epitope of human IRBP that induces autoimmune uveoretinitis in mice of the H-2b haplotype. Invest Ophthalmol Vis Sci. 2000;41:127–131. [PubMed] [Google Scholar]
- Dick AD, Cheng YF, Liversidge J, Forrester JV. Immunomodulation of experimental autoimmune uveoretinitis: a model of tolerance induction with retinal antigens. Eye. 1994;8:52–59. doi: 10.1038/eye.1994.10. [DOI] [PubMed] [Google Scholar]
- Munder M, Eichmann K, Moran JM, Centeno F, Soler G, Modolell M. Th1/Th2-regulated expression of arginase isoforms in murine macrophages and dendritic cells. J Immunol. 1999;163:3771–3777. [PubMed] [Google Scholar]
- Robertson MJ, Erwig LP, Liversidge J, Forrester JV, Rees AJ, Dick AD. Retinal microenvironment controls resident and infiltrating macrophage function during uveoretinitis. Invest Ophthalmol Vis Sci. 2002;43:2250–2257. [PubMed] [Google Scholar]
- Dick AD, Carter D, Robertson M, Broderick C, Hughes E, Forrester JV, Liversidge J. Control of myeloid activity during retinal inflammation. J Leukoc Biol. 2003;74:161–166. doi: 10.1189/jlb.1102535. [DOI] [PubMed] [Google Scholar]
- Taylor N, McConachie K, Calder C, Dawson R, Dick A, Sedgwick JD, Liversidge J. Enhanced tolerance to autoimmune uveitis in CD200-deficient mice correlates with a pronounced Th2 switch in response to antigen challenge. J Immunol. 2005;174:143–154. doi: 10.4049/jimmunol.174.1.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoey S, Grabowski PS, Ralston SH, Forrester JV, Liversidge J. Nitric oxide accelerates the onset and increases the severity of experimental autoimmune uveoretinitis through an IFN-γ-dependent mechanism. J Immunol. 1997;159:5132–5142. [PubMed] [Google Scholar]
- Zhang S, Cherwinski H, Sedgwick JD, Phillips JH. Molecular mechanisms of CD200 inhibition of mast cell activation. J Immunol. 2004;173:6786–6793. doi: 10.4049/jimmunol.173.11.6786. [DOI] [PubMed] [Google Scholar]
- Calder CJ, Nicholson LB, Dick AD. A selective role for the TNF p55 receptor in autocrine signaling following IFN-γ stimulation in experimental autoimmune uveoretinitis. J Immunol. 2005;175:6286–6293. doi: 10.4049/jimmunol.175.10.6286. [DOI] [PubMed] [Google Scholar]
- Shinohara H, Inoue A, Toyama-Sorimachi N, Nagai Y, Yasuda T, Suzuki H, Horai R, Iwakura Y, Yamamoto T, Karasuyama H, Miyake K, Yamanashi Y. Dok-1 and Dok-2 are negative regulators of lipopolysaccharide-induced signaling. J Exp Med. 2005;201:333–339. doi: 10.1084/jem.20041817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broderick CA, Smith AJ, Balaggan KS, Georgarias A, Buch PK, Trittibach PC, Barker SE, Sarra GM, Thrasher AJ, Dick AD, Ali RR. Local administration of an adeno-associated viral vector expressing IL-10 reduces monocyte infiltration and subsequent photoreceptor damage during experimental autoimmune uveitis. Mol Ther. 2005;12:369–373. doi: 10.1016/j.ymthe.2005.03.018. [DOI] [PubMed] [Google Scholar]
- Cunningham ET, Jr, Adamis AP, Altaweel M, Aiello LP, Bressler NM, D’Amico DJ, Goldbaum M, Guyer DR, Katz B, Patel M, Schwartz SD. A phase II randomized double-masked trial of pegaptanib, an anti-vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology. 2005;112:1747–1757. doi: 10.1016/j.ophtha.2005.06.007. [DOI] [PubMed] [Google Scholar]