Abstract
We developed two solid-phase reagent systems for microscopic visualization and characterization of melanocyte-stimulating hormone (MSH) receptors of melanoma cells. Multiple copies of [Nle-4,d-Phe-7]-α-MSH, a potent analog of α-MSH, were conjugated to microspheres (latex beads) or macrospheres (polyamide beads) through a thioether or disulfide bond. Binding between the beads and mouse and human melanoma cells was examined by scanning electron microscopy and by light microscopy. Each mouse and human melanoma cell (of all cell lines) evinced binding to the beads. Binding of the melanotropin conjugates was not restricted to any one phase of the cell cycle. Specificity of binding was demonstrated by several studies. Negative controls included cell types of nonmelanocyte origin (e.g., mammary cancer cells) and beads that lacked the melanotropic ligand or had other attached ligands. Beads with a disulfide-linked melanotropin analog served as a direct control. Treatment of these beads with DTT during or before incubation of the beads with melanoma cells (resulting in release of the MSH analog from the beads) eliminated binding of the beads to melanoma cells. Binding interactions between melanoma cells and melanotropin-bound beads also could be abolished by prior incubation with unconjugated MSH analog. During these experiments, certain membrane receptor-hormone associated phenomena, such as capping (aggregation) of the receptor-ligand complex, also were observed. These results provide visual evidence that MSH receptors are a property common to melanoma cells. Normal human epidermal melanocytes and keratinocytes were also shown to express melanotropin receptors by the same criteria established for melanoma cells.
Keywords: α-melanocyte stimulating hormone, melanoma, melanotropin receptors, scanning electron microscopy
The search for a molecular marker for melanoma cancer cells is pivotal for the development of modalities for the early detection and management of melanoma. The identification of a specific universal antigen associated with melanoma has remained elusive. Usually, good antibody candidates specific for a particular melanoma specimen fail to recognize other tumor variants from different patients and sometimes even from the same individual (1, 2). The occurrence of melanotic as well as amelanotic tumors further illustrates the great degree of melanoma variance and the problem of unambiguous detection. A group of acidic cytosolic proteins, commonly referred to as S-100 proteins, have been claimed to be a useful marker for melanoma (3, 4). An immunoassay based on this protein has been used to screen formalin fixed melanoma tumors in vitro, stressing its importance as a good diagnostic tool for melanoma, especially in case of metastatic lymph node melanoma. Unfortunately, a large number of normal tissues also contain S-100 proteins in substantial amounts (5), making it a rather complex issue in distinguishing the truly positive from the truly negative specimens. Furthermore, the cytosolic localization of these proteins suggests that antibody-directed targeting of modalities of therapeutic relevance might not be feasible. For this purpose, the identification of specific cell membrane markers is better suited because of their rather facile accessibility by cell-specific targeting agents.
We recently described the development of melanotropin conjugates that could detect the presence of melanotropin receptors on a number of mouse and human melanoma cell lines, melanotic as well as amelanotic, that we investigated (6, 7). These conjugates were composed of multiple copies of both a potent melanotropin analog and a fluorophore that were conjugated to a biologically inert macromolecule in a pendant fashion. The multivalency of the ligand molecule in these conjugates afforded specific binding between the conjugate and the melanotropin receptors, presumably by establishing simultaneous interactions with a number of melanocyte-stimulating hormone (MSH) receptors on the cell membrane. In addition, these macromolecular composites also allowed visualization of certain phenomena, like capping and internalization, that are associated with certain ligand-receptor binding.
Here we describe another class of multivalent melanotropic ligands that can detect the presence or absence of specific MSH binding sites on melanoma cells in a very simple and straightforward manner. These methods provide simple alternatives to the use of fluorescence techniques previously described by Schwyzer and coworkers (8, 9, 10, 11, 12) and recently by us (6, 7). The specificity of these newer solid-phase reagents for MSH-binding sites is addressed and established.
MATERIALS AND METHODS
N,N-Dimethylaminopyridine was obtained from Aldrich. DTT, mercaptoethanol, 4-(p-maleimidophenyl)-butyric acid-N-hydroxysuccinimide ester, and 3-(2′-pyridyldithio)propionic acid N-hydroxysuccinimide ester were obtained from Sigma. Latex beads (microspheres, 1-μm diameter) derivatized with primary amino groups [0.125 milliequivalents (meq) of amine per g or 3.125 eq of amine per ml of the commercial bead suspension] were obtained from Polysciences. Aminoethyl polyamide beads (macrospheres, 40- to 100-μm diameter) also derivatized with primary amino groups (1 meq of amine per g of dry beads) were purchased from Bio-Rad (Aminoethyl Biogel P-2, 200–400 mesh). Tissue culture media and fetal calf serum were obtained from GIBCO. Protected amino acid derivatives were obtained from Bachem. The binding buffer for cell binding experiments was a low ionic strength (0.015 M) buffer made isotonic to the cells by the addition of glucose and sucrose (13). The composition of this buffer was: 0.015 M triethanolamine, 0.24 M glycine, 0.009 M sucrose, 0.025 M glucose, 0.004 M potassium acetate, and 0.0003 M calcium chloride. Acetic acid was used to adjust the pH of the buffer to 6.5, and, if necessary, the osmolality was adjusted to 0.310 osmol using sucrose.
Cell Cultures.
All cells were grown in Falcon 25-cm2 tissue culture flasks at 37°C in a humidified atmosphere of 5% CO2 and 95% air. The cell lines were maintained in monolayer culture and were grown in Ham’s F-10 medium with NaHCO3 (1.2 g/liter) supplemented with 10% horse serum and 2% fetal calf serum (both heat-inactivated at 56°C for 30 min) and 1% penicillin/streptomycin (100 units/ml and 100 μg/ml, respectively).
Melanoma cells.
The human melanotic cell line A375P was obtained through the American Type Culture Collection. The other human melanoma cells were obtained from Arizona Cancer Center Tissue Culture Core Facility, University of Arizona, Tucson. Cells were subcultured weekly and were maintained in monolayer culture for only 10 passages to avoid phenotypic drift, which is often observed in long-term cultures.
The B16/F10 mouse melanoma cell line was obtained from Stanley P. L. Leong of the Laboratory of Surgical Oncology at the Arizona Cancer Center. The cell line was originally provided by A. Overjera of the Frederick Cancer Center, Frederick, MD, and has been well characterized by Fidler. Cells were subcultured weekly and were maintained in monolayer culture for only 10 passages to avoid phenotypic drift, which is often observed in long-term cultures.
Other cell lines.
A human small cell lung cancer cell line (NCI-N592) was obtained from Tom Davis, Department of Pharmacology, University of Arizona. The NCI-N592 cell line grows as floating aggregates in Ham’s F-10 medium supplemented with 10% heat-inactivated fetal calf serum and 1% penicillin/streptomycin. The human breast cancer (MCF-7) cell line was obtained from D. Blask, Department of Anatomy, University of Arizona. The mammary cancer cells were maintained in monolayer culture and grown in Ham’s F-10 medium supplemented with 10% heat-inactivated fetal calf serum and 1% penicillin/streptomycin.
Normal human epidermal melanocytes and growth medium were a gift from Abdel Malek, Department of Dermatology, University of Cincinnati. The cells were grown in monolayer in medium consisting of Ham’s F-10 medium, 8% newborn calf serum, 1% penicillin/streptomycin, 0.1 mM 3-isobutyl-1-methylxanthine (IBMX), 1.6 × 10−8 M 12-O-tetradecanoyl-phorbol 13-acetate (TPA), and 10 nM cholera toxin. Normal human keratinocytes and keratinocyte growth medium were purchased from Clonetics (San Diego, CA). The keratinocyte growth medium is serum-free and supplemented with recombinant epidermal growth factor, insulin, hydrocortisone, antimicrobial agents, and bovine pituitary extract.
Peptide Synthesis.
The two peptides used in this study, namely [Nα-des-acetyl,Nα-3′-mercaptopropionyl,Nle-4,d-Phe-7]-α-MSH (peptide 1) and [Nα-3′-mercaptopropionyl]-substance P (peptide 2), were synthesized by solid-phase methods of peptide synthesis (14) on p-methylbenzhydrylamine resin (substitution of 0.51–0.72 meq of amine per g of resin) on a Vega 250 semi-automated peptide synthesizer. A 4-fold excess of the appropriate Nα-Boc-protected amino acid (with side chain protected as needed) was used at each coupling step. Couplings were performed by using diisopropylcarbodiimide-N-hydroxybenzotriazole (DIC-HOBt) as coupling reagent and were monitored by the Kaiser test (15) in all cases, except when the incoming amino acid was coupled to a proline residue. In this instance, the chloranil test (16) was performed to ascertain the progress of the coupling. A mixture of trifluoroacetic acid/dichloromethane/anisole (50:48:2) was used to remove the Boc group after each of the coupling steps. The following three fully protected peptide resins corresponding to peptides 1–2 were synthesized: Nα-Boc-Ser(Bzl)-Tyr(Cl2Bzl)-Ser(Bzl)-Nle-Glu(OBzl)-His(Bom)-D-Phe-Arg(Tos)-Trp-Gly-Lys(Z-Cl)-Pro-Val-resin (peptide 3) and Nα-Boc-Arg(Tos)-Pro-Lys(Z-Cl)-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-resin (peptide 4). The Nα-Boc groups from peptides 3 and 4 were cleaved by treatment with trifluoroacetic acid/dichloromethane/anisole (50:48:2). To each of the resulting peptide resins, 3′-(S-p-methylbenzyl)-mercaptopropionic acid was individually coupled in the usual fashion. The dried peptide–resins were individually treated with HF-thioanisole (9:1) for 45 min at O°C, and the resulting crude peptides purified by HPLC and characterized by fast atom bombardment mass spectrometry and amino acid analysis. Analytical HPLC was performed on a C18 column (model 218TP104, 25 cm × 4.6 mm; Vydac, Hesperia, CA). TLC was performed on Baker 250-nm analytical silica gel glass plates in the following solvent systems: (i) 1-butanol/acetic acid/pyridine/water (5:5:1:4 vol/vol); (ii) 2-propanol/25% aqueous ammonia/water (3:1:1 vol/vol); (iii) ethyl acetate/pyridine/acetic acid/water (5:5:1:3 vol/vol); and (iv) 1-butanol/acetic acid/water (4:1:5 vol/vol). The peptides were visualized by ninhydrin and iodine vapor. Analytical data for peptides were as follows. Peptide 1, mass (M)= 1692.6 (calculated 1692.97); αD23 = −50.6° (c, 0.41, 10% aqueous AcOH); HPLC K′ = 7.02 (gradient of 10% to 40% acetonitrile in 0.1% aqueous trifluoroacetic acid completed in 30 min at 1.5 ml/min); TLC Rf values = 0.33 (i), 0.61 (ii), 0.0 (iii), 0.02 (iv). Peptide 2, mass = 1434.9 (calculated 1434.78); αD23 = −37.2° (c, 0.41, 10% aqueous AcOH); HPLC K′ = 5.6 (gradient of 10% to 40% acetonitrile in 0.1% aqueous trifluoroacetic acid completed in 30 min at 1.5 ml/min); TLC Rf values = 0.53 (i), 0.75 (ii), 0.64 (iii), 0.36 (iv).
Synthesis of Micro-(and Macro-)Sphere–Peptide Conjugates.
Two types of bead–peptide conjugates were synthesized: (i) conjugates in which the bond linking the peptide to the bead was a thioether bond; and (ii) conjugates in which the bond linking the peptide to the bead was a disulfide bond. The second type of conjugate allowed the cleavage of the peptide from the bead by reduction of the disulfide bond under mild reducing conditions. This provided an opportunity to conduct control binding experiments to establish the specificity of the interaction between these beads and MSH receptor-positive cells.
Micro-(and macro-)sphere–[maleimidophenyl butyrate]n (conjugates 5 and 6, respectively).
The amino groups on both micro- and macrospheres were fully derivatized with 4-(p-maleimidophenyl)-butyric acid-N-hydroxysuccinimide ester to introduce the maleimido group for eventual synthesis of the thioether-linked conjugates. Beads obtained after centrifugation of 1 ml of latex microspheres or 25 mg of macrospheres (2.27 × 10−7 mol of NH2 groups in either case) were suspended in 9 ml of 50 mM Hepes buffer (pH 7.5). The beads were added to a mixture of 4-(p-maleimidophenyl)-butyric acid-N-hydroxysuccinimide ester (4.02 mg, 1.13 × 10−5 mol) and N,N-dimethylaminopyridine (1.38 mg, 1.13 × 10−5 mol) dissolved in 1 ml of N,N-dimethlyformamide. The resultant reaction mixture was shaken for 12–18 hr. In both cases, the beads were collected by centrifugation and washed further with the 1:1 ethanol/Hepes buffer (5 times with 1 ml) by repeated centrifugation and resuspension by gentle sonication. The degree of derivatization was measured after reacting a small measured amount of this product with a known excess of thiophenol (5-fold molar excess) for 5 hr, followed by extensive washings by centrifugation and resuspension of the beads and spectrophotometric measurements of the amount of unreacted thiophenol present in the combined washings [λ269 (ɛ = 700) (17)].
Micro-(and macro-)sphere–[pyridyldithiopropionate]n (conjugates 7 and 8, respectively).
The amino groups on the beads of either type were completely derivatized with 3-(2′-pyridyldithio)-propionic acid-N-hydroxysuccinimide ester to introduce the dithiopyridyl group. The general method described above for the synthesis of conjugates 5 and 6 was followed using similar amounts of 3-(2′-pyridyldithio)-propionic acid-N-hydroxysuccinimide ester as the derivatizing reagent. The degree of substitution in this case was also followed in a similar fashion by reacting small amounts of extensively washed beads with a known excess of thiophenol (5-fold molar excess) for 5 hr, then followed by centrifugation of the reaction mixture and subjection of the supernatant to direct spectrophotometric measurements at λ343 (ɛ = 8080) for the amounts of 2-thiopyridine that were released in this reaction (18).
Micro-(and macro-)sphere–[S-MSH]n (conjugates 9 and 10, respectively) and micro-(and macro-)sphere–[substance P]n (conjugates 11 and 12, respectively).
A sample of derivatized beads (conjugates 5, 6, 7, or 8) suspended in 1.5–2.0 ml of 50 mM Hepes (pH 7.5) was mixed with a 2- to 4-fold molar excess of peptide 1 or 2 dissolved in N,N-dimethlyformamide (0.3 ml) at room temperature for 5 hr. The MSH analog- or substance P analog-conjugated beads were then recovered from the reaction mixture by centrifugation and washed further with the 1:1 ethanol/Hepes buffer (5 times with 1 ml) by repeated centrifugation and resuspension by gentle sonication. The degree of derivatization was measured by spectrophotometric measurements for the unreacted peptide present in the combined washings. For the synthetic disulfide-linked peptide-conjugated beads, direct measurements of the supernatant from the centrifuged reaction mixture at λ343 (ɛ = 8080) for the amounts of 2-thiopyridine released during the coupling of the ligand (18) gave the degree of substitution directly. In each case, this matched very closely the original substitution of the commercially obtained amino-derivatized beads.
Scanning Electron Microscopic Evaluation of Interactions Between Cells and Peptide-Conjugated Microspheres 9 and 11.
Cells (0.3 × 105) were incubated with about 7.5 × 106 melanotropin-conjugated microspheres (9 and 11) in 0.5 ml of the binding buffer described above for 30 min with occasional mixing (every 5 min) at room temperature. To perform control experiments involving reductive cleavage of the disulfide linkage between the peptide and the bead (thereby releasing the peptide from the bead), the beads were first treated with 50 mM DTT or 2-mercaptoethanol for 30 min at room temperature and then washed three times with the buffer by cycles of centrifugation and resuspension by sonication, followed by incubation with the cells. At the end of the incubation period, the cell and bead suspension was filtered through a membrane filter with pores of either 0.45-μm or 5-μm diameter. The cell suspension was filtered with a gentle suction from a water aspirator. The cell pellet was washed on the filter three times with the binding buffer. The filter with a smaller diameter pore size (0.45 μm) retained the unbound microspheres in addition to the cells with associated microspheres, whereas the filter with 5-μm diameter pores retained only the cells with microspheres bound to the cells and allowed the unbound microspheres to pass through it. Another membrane filter was placed on the top of the original filter, and the filters were secured between two plastic clamps, thereby securing the cell specimen during further processing. The clamps bearing the cell specimens were immersed in 2.5% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) at 0°C overnight for fixation. After washing three times with the same buffer, the samples were postfixed with 1% osmium tetroxide, followed by three washings with distilled water. The resulting samples were dehydrated successively in 30%, 50%, 70%, 90%, and finally 100% ethanol. The dehydrated samples were dried using a Polaron (Energy Beam Sciences, Agawam, MA) critical point drier and coated with 30 nm of gold in a Hummer Sputter Coater (Anatech, Alexandria, VA). The binding of the peptide-conjugated microsphere to the cells was visualized by an I.S.I. scanning electron microscope (model DS 130; Topcon Technologies, Paramus, NJ) at a voltage of 20 kV. Positive binding was documented as + (Table 1) when beads were observed (see Fig. 1 A, C, and D) bound to the cells. Complete absence of any beads bound to cells was termed as no interaction and was designated as − (Table 1; and see Fig. 2 A, B, and D). A set of three specimens was prepared and examined in each case, and the results from only one set are shown in the figures.
Table 1.
Specific binding of MSH microspheres and macrospheres to various cells
| Cell lines | Binding |
|---|---|
| Epidermal melanocytes (human) | + |
| Keratinocytes (human) | + |
| Melanoma | |
| B16 (mouse, positive control) | + |
| HD (human, melanotic) | + |
| VS (human, amelanotic) | + |
| A375P (human, melanotic) | + |
| A375M (human, melanotic) | + |
| C8161 (human, amelanotic) | + |
| Breast cancer cell MCF-7 (human negative control) | − |
| Small cell lung cancer (NC1-N592) | − |
Figure 1.
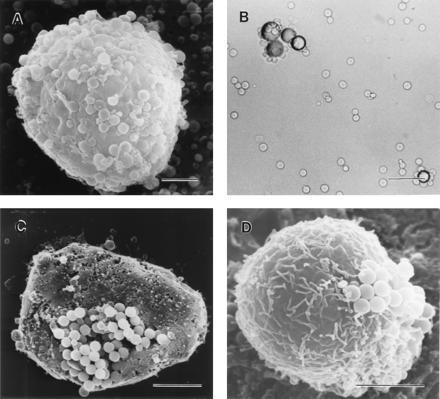
Binding of MSH beads with melanoma cells. (A) Scanning electron micrograph showing MSH microspheres bound to C-8161 human amelanotic melanoma cells. (Bar = 5 μm.) (B) Light microscopic visualization of specific binding of B16 mouse melanoma cells to MSH macrospheres. (Bar = 100 μm.) (C) Clustering of microspheres observed during binding to B-16 mouse melanoma cells. (Bar = 5 μm.) (D) Human amelanotic melanoma cells (VS). (Bar = 5 μm.)
Figure 2.
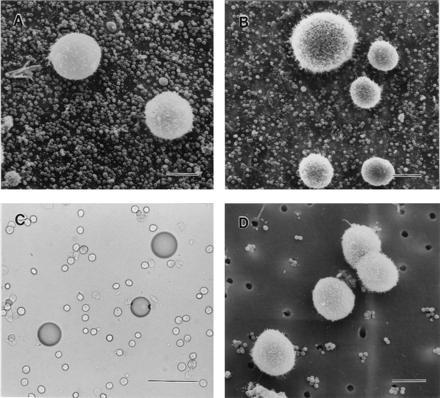
Negative control experiments to determine specificity of binding of melanoma cells to melanotropin-conjugated beads. (A) Blockade of binding of human A375P melanoma cells with MSH beads (microspheres) due to pretreatment of cells with an unconjugated (free) MSH ligand, [Nle-4,d-Phe-7]-α-MSH. (Bar = 10 μm.) (B) Naive (no MSH present) microspheres fail to bind to B-16 mouse melanoma cells. (Bar = 10 μm.) (C) Substance P-conjugated beads (macrospheres) do not bind to B16 mouse melanoma cells. (Bar = 100 μm.) (D) MCF-7 breast cancer cells do not bind to MSH-conjugated microspheres. (Bar = 10 μm.)
Light Microscopic Evaluation of Interactions Between Cells and Peptide-Conjugated Macrospheres 10 and 12.
Cells (0.5 × 105) were incubated with ≈12,000 melanotropin-conjugated macrospheres (10 and 12) in 0.5 ml of the binding buffer for 30 min with occasional mixing (every 5 min). An aliquot (≈50 μl) of this incubation medium was then placed in a small well made on a microscope slide with a wax pencil. After placing a coverslip on the well containing the cell and bead suspension, the slides were immediately examined under the light microscope to observe any interaction between the macrospheres and the cells. Control experiments as described above for microspheres also were performed with macrospheres. A positive interaction, termed + (Table 1), was specified if beads were bound to all cells on a slide (see Fig. 1B). On the other hand, if all the cells were found free of any beads attached to them, the interaction was termed as − (Table 1; and see Fig. 2C). A set of three slide specimens was prepared and evaluated for each assay.
RESULTS
Synthesis of Peptide-Conjugated Beads.
Both the microspheres and the macrospheres were prepared by similar chemistry. The substitution level in each case closely matched the original commercial level of derivatization of the beads. This was expected, because a large excess of the derivatizing reagents was employed at every stage to ensure complete derivatization. The microsphere conjugate preparations (9 and 11) having thioether-linked peptide were generally single bead-suspended preparations. On the other hand, in the preparations where the peptide was linked to the microspheres via a disulfide bridge, a small population of beads were grouped into aggregates of two or more beads. It was observed that, upon treatment with DTT or 2-mercaptoethanol, these aggregates would disperse, suggesting that the aggregation might have been due to disulfide interchange, as earlier observed for tobacco mosaic virus disulfide conjugates (8). Use of a lower pH (pH 6) during the conjugation reaction described under the synthesis of 10 and 12 helped to minimize this cross-linking. The presence of a small population of these aggregates did not hamper the ability of the MSH analog-conjugated beads to bind with the cells which contain hormone receptors (21). The synthesis of peptide-conjugated macrospheres (10 and 12), on the other hand, were free of any such effects.
Binding of Peptide-Conjugated Beads to Melanoma Cells.
Human melanoma cells as well as B16/F10 mouse melanoma cells are ≈10–15 μm in diameter. The latex microspheres used in these investigations were of 1-μm diameter, and the diameter of the polyamide macrospheres ranged from 40 to 100 μm. It, therefore, was not unexpected to see that many MSH analog-conjugated microspheres were bound to a single melanoma cell (Fig. 1A). On the other hand, because of the larger dimensions (4–6 times larger) of the polyamide macrospheres compared with the melanoma cells, the positive interaction was observed in terms of clustering of many of the melanoma cells around a single MSH analog-conjugated macrosphere (Fig. 1B). This binding pattern could be described as similar to a rosette, and this configuration proved to be a convenient way to demonstrate binding by light microscopy.
The binding of the MSH analog-containing beads (both micro- and macrospheres belonging to both the thioether and the disulfide class of preparations) to cell membrane MSH receptors was established within 30 min of incubation. This binding undoubtedly resulted from the multiplicity of the peptide ligand on these solid-phase probes, such that a single assembly of the bead–peptide conjugate is most likely able to interact simultaneously with more than one receptor on the cell membrane. This behavior has earlier been reported on interaction of tobacco mosaic virus–MSH conjugates with mouse melanoma cells (8, 9, 10, 11, 12). Similarly, certain classes of polyvalent antibodies that also are capable of establishing multiple interactions exhibit much higher binding affinities (22).
Homogeneity of Conjugate Binding to Melanoma Cells and Capping of the Ligand.
During the binding experiments using MSH conjugates, it was noted that all the cells of a particular melanoma cell line (melanotic as well as amelanotic) bound to the microspheres. This is suggestive of cellular homogeneity of each cell line with respect to melanotropin receptor expression. Every mouse melanoma cell also bound to these microspheres. Some of the cells that were oriented favorably to the observer also appeared to exhibit clustering of the microspheres, which might be suggestive of capping (receptor aggregation in preparation for internalization; Fig. 1 C and D). In this respect, these results are similar to electron microscopic visualization of internalized receptor–ligand complex for a glucagon–serum albumin preparation labeled with 17 nanometer gold particles (23). These results are consistent with those obtained earlier using fluorescent tobacco mosaic virus–MSH conjugates (8, 9, 10, 11, 12) and fluorescent polyvinyl alcohol–MSH conjugates (6, 7).
Specificity of Binding.
The specificity of binding of human melanoma cells to both the small (microsphere) and large (macrosphere) macromolecular conjugates was determined by several different means.
Disulfide linkage disruption (by 2-mercaptoethanol or DTT).
The micro- and macrospheres in which the melanotropin analog was linked to the beads by a disulfide linkage (10) provided a control experiment to establish the specificity of the interactions between MSH beads and melanoma cells as described by Schwyzer and coworkers (8, 10). Reducing agents such as DTT and 2-mercaptoethanol were able to reduce the S–S bond and therefore release the hormone analog. The beads (10) were incubated with DTT (final concentration, 50 mM) for 30 min at room temperature with constant shaking. The DTT or 2-mercaptoethanol-treated beads were exhaustively washed and, when incubated with B16/F10 mouse melanoma cells or other human melanoma cells, now failed to bind these cells (not shown). These experiments provide strong evidence for specific binding between the MSH analog-conjugated beads with MSH receptors on the melanoma cells.
Preincubation in the presence of unconjugated free MSH ligand.
Binding established between MSH-containing beads to melanoma cells was antagonized by [Nle-4,d-Phe-7]-MSH as competitor. However, when the cells were preincubated with the free ligand (1 × 10−6 M) for 1 hr before their exposure to the MSH analog-containing beads, a very dramatic loss of binding was observed (Fig. 2A). This is suggestive of either blocked (occupied) receptors or lack of receptors on the cell membrane due to their internalization (24). These results lend further support for the specificity of the binding of the MSH-conjugated beads for melanoma melanotropin receptors.
Binding studies using naive micro-(and macro-)spheres.
Microspheres with amino groups that were never exposed to the MSH ligand completely failed to bind any type of the cells under any of the binding conditions (Fig. 2B). This negative control established the inertness of the microspheres (latex) to MSH receptors. The amino ethyl polyamide beads, on the other hand, exhibited a great degree of nonspecific binding to all types of the cells (melanoma as well as others). These nonspecific interactions occur because of a much higher positive charge density on macrospheres (amine concentration, 1 meq/g) than microspheres (amine concentration, 0.125 meq/g), establishing electrostatic interactions with predominantly negatively charged cell membranes.
Other Peptide (Substance P)-Conjugated Beads.
Substance P-conjugated beads were chosen as negative controls to demonstrate that the binding seen with MSH-conjugated beads was a peptide-specific event. Both types of substance P-containing micro- as well as macrospheres did not bind to any of the melanoma cells (Fig. 2C).
Cellular Specificity (Other Cell Types).
A few cell types of nonmelanogenetic origin (Table 1) were included in this study to act as negative controls from the receptor aspect. Human small cell lung cancer (NCl-N592) and breast cancer (MCF-7) cells did not exhibit any affinity toward the MSH-conjugated beads (Fig. 2D). Not surprisingly, both normal human melanocytes and keratinocytes also bound to the [Nle-4,d-Phe-7]-α-MSH conjugates (19, 20). Specificity of binding of the melanotropin-bound beads to the melanocytes and keratinocytes was established by the same criteria as discussed here for melanoma cells. These results are extensively documented elsewhere (19, 20).
DISCUSSION
Melanoma is derived from cells of melanocytic origin (usually epidermal melanocytes). MSHs—e.g., α-MSH—stimulate adenylate cyclase activity and cAMP formation in melanoma cells (25). A number of recent studies have therefore concentrated on determining the presence or absence of MSH receptors on various melanoma cells. Several groups of investigators have used radioreceptor binding assays and autoradiography techniques to identify these receptor in mouse (26, 27, 28, 29, 30, 31) and in several human melanoma cell lines (32, 33, 34, 35). These studies failed to detect the existence of MSH receptors on all human melanoma cell lines. Most interestingly, the presence of MSH receptors appeared to be dependent on the type of radiolabeled melanotropin analog used. For example, Libert et al. (32) found that binding of radioiodinated α-MSH could be demonstrated in only 4 of 10 human melanoma cell lines. On the other hand, when radioiodinated [Nle-4,d-Phe-7]-α-MSH was used, the presence of MSH receptors in 6 of the same group of 10 cell lines was observed. It may be possible that this behavior could be due to heterogeneity of MSH receptors (i.e., subtypes of the MSH receptors) present among these cell types, imparting a differential recognition among radioiodinated α-MSH and [Nle-4,d-Phe-7]-α-MSH. Nonetheless, these results also stress the need to develop a MSH ligand or a multivalent macromolecule as reported here that would recognize all of these receptor subtypes (i.e., a nonreceptor subtype selective ligand).
Our studies using multiple copies of an MSH analog conjugated to both micro- and macrospheres, in conjunction with scanning electron microscope and light microscope visualization methods, respectively, demonstrate the specificity of these solid-phase reagents with a variety of melanoma cells that have been investigated by us (Table 1). The specificity of the receptor-mediated binding was demonstrated by the following observations. (i) The positive control, mouse B16/F10 melanoma cells (known to express MSH receptors) bound to the MSH-containing beads. (ii) The negative control cells, namely the MCF-7 human breast cancer cells and the NCI-N592 small cell lung cancer cells (both assumed not to express MSH receptors), failed to bind either type of conjugate. (iii) Incubation of naive microspheres alone (no hormone attached) or incubation with substance P-conjugated beads did not result in binding of the beads to the melanoma cells. (iv) Pretreatment of a disulfide-linked melanotropin conjugate bead preparation with reducing agents (DTT or 2-mercaptoethanol) resulted in cleavage of the bond between the bead and the hormone, and this abolished the ability of the resulting beads to bind both B-16 mouse and various human melanoma cells. (v) If binding is specific to melanotropin receptors, one would expect that the presence of the conjugated analog would compete with the unbound ligand ([Nle-4,d-Phe-7]-α-MSH) for receptor binding. Preincubation of the melanoma cells with high concentrations of [Nle-4,d-Phe-7]-α-MSH abolished the binding ability of the MSH analog-conjugated beads to the melanoma cells (Fig. 2A).
Pretreatment of the cells in the presence of free (unconjugated) ligand may have resulted in receptor occupation or down-regulation before addition of the melanotropin-bound conjugates. We have documented in numerous other publications that [Nle-4,d-Phe-7]-substituted melanotropins bind to melanocortin receptors for very prolonged periods of time. Radiolabeling of murine melanoma cells using [Nle-4,d-Phe-7]-α-MSH, for example, results in a very slow off-rate of the ligand from the cells (36).
The present results are in complete agreement with our earlier results on fluorescence labeling of these cells using a water-soluble class of fluorescent melanotropic macromolecular reagents (6, 7) as well as with those of Schwyzer and coworkers, who developed conjugates of MSH with tobacco mosaic virus (8, 9, 10, 11, 12). Using the melanotropin fluorescein conjugate, we demonstrated that both melanotic as well as amelanotic human melanoma cells expressed membrane melanocortin receptors (6, 7). Six of these cell lines are in addition to the four of the cell lines used in the present study. In addition, we recently demonstrated that several human melanoma cell lines responded to [Nle-4,d-Phe-7]-α-MSH by increased tyrosinase activity (37), again suggesting the existence of human melanoma cell melanocortin receptors.
In the present studies we found that our melanotropin conjugate beads bound to every individual mouse or human melanoma cell. Since the cells were not synchronized, our results suggest that binding (30-min incubation) to the cells occurred during each phase of the cell cycle. While some groups have suggested that the expression of MSH receptors occur predominantly in the G2 phase of the cell cycle (38, 39), other investigations were unable to confirm such G2 restriction (40). Similarly, it was also reported that at least some of the responses to MSH (increased cAMP production and tyrosinase activity) occur in the G2 phase of the cell cycle (41). It has also been claimed that melanoma cells “regulate their response to MSH by the discontinuous appearance of receptors for the hormonal signal” (42). Although our results do not restrict the ability of cells to bind MSH to the G2 phase of the cycle, the data provides no information on the functional ability (to transduce signals and effect second messenger formation) of melanotropin receptors throughout the cycle.
In summary, we have designed a new class of solid-phase melanotropic reagents which can serve as powerful diagnostic tools. These reagents complement our previously reported fluorescent polyvinyl alcohol-conjugated (6, 7) MSH (8, 9, 10, 11, 12) constructs for use in receptor identification. Most importantly, every cell of every melanoma cell line possessed melanotropin receptors as visualized by these reagents. Only one cell type of nonmelanocyte origin (keratinocytes) exhibited such receptors. These cell specific melanotropin receptors may serve as cell surface markers for melanoma (Table 1).
Acknowledgments
This work was supported by U.S. Public Health Service Grant DK 17420 (V.J.H.) and by the National Science Foundation (V.J.H.).
Footnotes
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. §1734 solely to indicate this fact.
Abbreviations: MSH, melanocyte-stimulating hormone; eq, equivalent(s).
References
- 1.Albino A P, Lloyd K O, Houghton A N, Oettgen H F, Old L J. J Exp Med. 1981;154:1764–1778. doi: 10.1084/jem.154.6.1764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Natali P G, Bigotti A, Cavaliere R, Liao S K, Taniguchi M, Matsui M, Ferrone S. Cancer Res. 1985;45:2883–2889. [PubMed] [Google Scholar]
- 3.Cochran A J, Wen D R, Herschman H R, Gaynor R B. Int J Cancer. 1982;30:295–297. doi: 10.1002/ijc.2910300307. [DOI] [PubMed] [Google Scholar]
- 4.Hachisuka H, Sakamoto F, Nomura H, Mori O, Sasai Y. Acta Histochem. 1986;80:215–223. doi: 10.1016/S0065-1281(86)80066-6. [DOI] [PubMed] [Google Scholar]
- 5.Haimoto H, Hosoda S, Kato K. Lab Invest. 1987;57:489–498. [PubMed] [Google Scholar]
- 6.Sharma S D, Hruby V J, Hadley M E, Granberry M E, Leong S P. In: Peptides: Chemistry and Biology, Proceedings of the Twelfth American Peptide Symposium. Smith J A, Rivier J E, editors. Leiden, The Netherlands: ESCOM; 1992. pp. 599–600. [Google Scholar]
- 7.Sharma S D, Granberry M E, Jiang J, Leong S P L, Hadley M E, Hruby V J. J Bioconjugate Chem. 1994;5:591–601. doi: 10.1021/bc00030a015. [DOI] [PubMed] [Google Scholar]
- 8.Wunderlin R, Sharma S D, Minakakis P, Schwyzer R. Helv Chim Acta. 1985;86:12–22. [Google Scholar]
- 9.Wunderlin R, Minakakis P, Tun-Kyi A, Sharma S D, Schwyzer R. Helv Chim Acta. 1985;68:1–11. [Google Scholar]
- 10.Schwyzer R, Kriwaczek V M, Wunderlin R. Naturwissenschaften. 1981;68:95–96. doi: 10.1007/BF01047233. [DOI] [PubMed] [Google Scholar]
- 11.Schwyzer R, Kriwaczek V M. Biopolymers. 1981;20:2011–2020. doi: 10.1002/bip.1981.360200923. [DOI] [PubMed] [Google Scholar]
- 12.Kriwaczek V M, Bristow A F, Eberle A N, Gleed C, Schulster D, Schwyzer R. Mol Cell Biochem. 1981;40:49–59. doi: 10.1007/BF00230187. [DOI] [PubMed] [Google Scholar]
- 13.Zeiller K, Pascher G, Hannig K. Prep. Biochem. 1972;2:21–37. doi: 10.1080/00327487208061450. [DOI] [PubMed] [Google Scholar]
- 14.Hruby V J, Wilkes B C, Hadley M E, Al-Obeidi F, Sawyer T K, Staples D J, deVaux A E, Dym O, Castrucci A M L, Hintz M F, Riehm J R, Rao R. J Med Chem. 1987;30:2126–2130. doi: 10.1021/jm00394a033. [DOI] [PubMed] [Google Scholar]
- 15.Kaiser E, Colescott R L, Bossinger C D, Cook P I. Anal Biochem. 1970;34:595–598. doi: 10.1016/0003-2697(70)90146-6. [DOI] [PubMed] [Google Scholar]
- 16.Christensen T. In: Peptides, Structure and Biological Function. Gross E, Meienhofer J, editors. Rockford, IL: Pierce Chemical Co.; 1979. pp. 385–388. [Google Scholar]
- 17.Silverstein R M, Bassler G C, Morrill T C. Spectrometric Identification of Organic Compounds. 4th Ed. New York: Wiley; 1981. p. 322. [Google Scholar]
- 18.Stuchbury T, Shipton M, Norris R, Malthouse J P G, Brocklehurst K, Herbert J A L, Suschitzky H. Biochem J. 1975;151:417–432. doi: 10.1042/bj1510417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jiang, J., Sharma, S. D., Hruby, V. J. & Hadley, M. E. (1996) Exp. Dermatol., in press. [DOI] [PubMed]
- 20.Jiang, J., Sharma, S. D., Fink, J. L., Hadley, M. E. & Hruby, V. J. (1996) Exp. Dermatol., in press. [DOI] [PubMed]
- 21.Cutrecasas P, Hollenberg M D. Adv Protein Chem. 1976;30:251–430. doi: 10.1016/s0065-3233(08)60481-7. [DOI] [PubMed] [Google Scholar]
- 22.Greenbury C L, Moore D H, Nunn L A C. Immunology. 1965;8:420–431. [PMC free article] [PubMed] [Google Scholar]
- 23.Ackerman G A, Yang J, Wolken K W. J Histochem Cytochem. 1983;31:433–440. doi: 10.1177/31.3.6186734. [DOI] [PubMed] [Google Scholar]
- 24.Pawelek J M, Chakraborty M P, Osker M P, Orlow S J, Min K K. Pigm. Cell Res. 1992;5:348–356. doi: 10.1111/j.1600-0749.1992.tb00561.x. [DOI] [PubMed] [Google Scholar]
- 25.Hadley M E, editor. The Melanotropic Peptides: Source, Synthesis, Chemistry, Secretion, and Metabolism. Vol. 1. Boca Raton, FL: CRC; 1988. [Google Scholar]
- 26.Panasci L C, McQuillan A, Kaufman M. J Cell. Physiol. 1987;132:97–103. doi: 10.1002/jcp.1041320113. [DOI] [PubMed] [Google Scholar]
- 27.Siegrist W, Solca F, Stutz S, Giuffre L, Carrel S, Girard J, Eberle A N. Cancer Res. 1989;49:6352–6358. [PubMed] [Google Scholar]
- 28.Siegrist W, Ostreicher M, Stutz S, Girard J, Eberle A. J Recept. Res. 1988;8:323–343. doi: 10.3109/10799898809048996. [DOI] [PubMed] [Google Scholar]
- 29.Solca F, Siegrist W, Drozdz R, Girard J, Eberle A N. J Biol Chem. 1989;264:14277–14281. [PubMed] [Google Scholar]
- 30.Tatro J B, Watson M L, Lester B R, Reichlin S. Cancer Res. 1990;50:1237–1242. [PubMed] [Google Scholar]
- 31.Tatro J B, Reichlin S. Endocrinology. 1987;121:1900–1907. doi: 10.1210/endo-121-5-1900. [DOI] [PubMed] [Google Scholar]
- 32.Libert A, Ghanem G, Arnould R, Lejeune F. Pigment Cell Res. 1989;2:510–518. doi: 10.1111/j.1600-0749.1989.tb00247.x. [DOI] [PubMed] [Google Scholar]
- 33.Legros F, Coel J, Doyen A, Hanson P, Van Tieghem N, Vercammon G A, Fruhling J, Lejeune F J. Cancer Res. 1980;41:1539–1544. [PubMed] [Google Scholar]
- 34.Ghanem G E, Communale A, Libert A, Vercammon G A, Lejeune F. Int J Cancer. 1988;41:248–255. doi: 10.1002/ijc.2910410216. [DOI] [PubMed] [Google Scholar]
- 35.Tatro J B, Atkins M, Mier J W, Hardarson S, Wolfe H, Smith T, Entwistle M L, Reichlin S. J Clin Invest. 1990;85:1825–1832. doi: 10.1172/JCI114642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Haskell-Luevano C, Boteju L W, Miwa H, Dickinson C, Gantz I, Yamata T, Hadley M E, Hruby V J. J Med Chem. 1995;38:4720–4729. doi: 10.1021/jm00023a012. [DOI] [PubMed] [Google Scholar]
- 37.Jiang J, Sharma S D, Nakamura S, Lai J, Fink J L, Hruby V J, Hadley M E. Pigm. Cell Res. 1995;8:314–323. doi: 10.1111/j.1600-0749.1995.tb00680.x. [DOI] [PubMed] [Google Scholar]
- 38.Varga J B, Dipasquale A, Pawelek J, McGuire J S, Lerner A B. Proc Natl Acad Sci USA. 1974;71:1590–1593. doi: 10.1073/pnas.71.5.1590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.McLane J A, Pawelek J M. Biochemistry. 1988;27:3743–3747. doi: 10.1021/bi00410a033. [DOI] [PubMed] [Google Scholar]
- 40.Fuller B B, Brooks B A. Exp Cell Res. 1980;126:183–190. doi: 10.1016/0014-4827(80)90484-x. [DOI] [PubMed] [Google Scholar]
- 41.Wong G, Pawelek J, Sansone M, Morowitz J. Nature (London) 1974;248:351–354. doi: 10.1038/248351a0. [DOI] [PubMed] [Google Scholar]
- 42.Pawelek J. J Invest Dermatol. 1976;66:201–209. doi: 10.1111/1523-1747.ep12482134. [DOI] [PubMed] [Google Scholar]


