Abstract
Chemokines and their receptors are part of polarized T helper 1 (Th1)- and Th2-mediated immune responses which control trafficking of immunogenic cells to sites of inflammation. The chemokine stromal cell-derived factor-1 CXCL-12 (SDF-1) and its ligand the CXCR4 chemokine receptor are important regulatory elements. CXCR4 is expressed on the surface of CD4+ T cells, dendritic cells and B lymphocytes. Levels of CXCR4 mRNA were increased in pancreatic lymph nodes (PLNs) of 4-week-old non-obese diabetic (NOD) mice in comparison to Balb/C mice. However, a significant reduction of CXCR4 was noticed at 12 weeks both at the mRNA and protein levels while expression increased in the inflamed islets. The percentage of SDF-1 attracted splenocytes in a transwell chemotaxis assay was significantly increased in NOD versus Balb/c mice. SDF-1 attracted T cells completely abolished the capacity of diabetogenic T cells to transfer diabetes in the recipients of an adoptive cell co-transfer. When T splenocytes from NOD females treated with AMD3100, a specific CXCR4 antagonist, were mixed with diabetogenic T cells during adoptive cell co-transfer experiments, prevalence of diabetes in the recipients rose from 33% to 75% (P < 0·001). This effect was associated with an increase of interferon (IFN)-γ mRNA and a reduction of interleukin (IL)-4 mRNA levels both in PLNs and isolated islets. AMD3100 also reduced IL-4 and IL-10 production of plate-bound anti-CD3 and anti-CD28-stimulated splenocytes. Immunofluorescence studies indicated that AMD3100 reduced the number of CXCR4+ and SDF-1 positive cells in the inflamed islets. We can conclude that the CXCL-12/CXCR4 pathway has protective effects against autoimmune diabetes.
Keywords: CXCR4, diabetes, NOD mouse, protection
Introduction
The non-obese diabetic (NOD) mouse is an established model of spontaneous autoimmune diabetes with many common genetic and immunological features with human type 1 diabetes, including autoreactive T cells which ultimately destroy beta cells [1,2]. Beside CD4+ and CD8+ T cell subsets, additional cells such as dendritic cells, monocytes and B lymphocytes are present in the islet infiltrates [3]. We and others [4,5] have underlined the importance of pancreatic lymph nodes (PLN) in NOD mice where both cytotoxic and regulatory T cells are activated in the presence of dendritic cells presenting beta cell autoantigens [6]. The balance between T helper 1 (Th1) and Th2 cytokine subsets in the pancreas and PLNs is one of the determining factors in diabetes development in the NOD mouse [7,8]. Chemokines direct the movement of circulating lymphocytes to sites of inflammation and target cell selectivity through specific chemokine receptors [9]. Among the huge battery of molecules that regulate inflammation and immunity, interferon (IFN)-γ-producing Th1 cells and cytotoxic type 1 T cells (Tc1) express the chemokine receptors CXCR3 and CCR5, whereas IL-4-producing Th2/Tc2 cells express mainly CCR3, CCR4 and CCR8 [10]. Unlike other chemokine receptors, CXCR4 molecule is expressed widely [9] and is involved in basal trafficking of naive lymphocytes [11]. IL-4 and IL-10 can significantly up- or down-regulate CXC chemokine receptor 4 (CXCR4) expression on CD4+ T lymphocytes [11]. We have observed previously an early increase in levels of CXCR4 RNA in the islets of NOD mice [12]. The aims of our study were to evaluate if activation of the CXCR4/SDF-1 pathway has a temporal correlation with insulitis and diabetes and whether this chemokine–chemokine receptor pair has a facilitating or protective role in beta cell autoimmunity.
Materials and methods
Mice and adoptive cell transfers
NOD Thy-1,2 mice were bred under standard conditions in our animal facility. In our colony, spontaneous diabetes starts at 12 weeks in females and the incidence of diabetes reaches 81% in females and 15–20% in males by 30 weeks. Diabetes was characterized by polydipsia, weight loss, glycosuria as assessed by urine chemstrips (Bayer Diagnostics, Dublin, Ireland) and persistent hyperglycaemia (above 200 mg/dl), which was determined with blood glucose chemstrips (Accu-chek active; Roche Diagnostics, Mannheim, Germany). Diabetic NOD females served as donors of autoreactive T cells during transfer experiments. BALB/c female mice were purchased from Charles River Laboratories (L'Arbresle, France).
To evaluate the functional capacities of CXCR4 T cells, we performed a series of adoptive co-transfer experiments. Recipients were 8–12-week-old irradiated (750 rads) NOD males injected intravenously (i.v.) with 5 × 106 T lymphocytes from diabetic NOD females and according to the situation with either enriched CXCR4 T cell populations or inactivated CXCR4 T cells. Transwell chemotaxis assays in 24-well transwell plates (6·5 mm diameter, 5 µm pore size; Corning Inc., Life Sciences, Acton, MA, USA) were used to purify CXCR4 T splenocytes as described previously [13]. Briefly, the bottom chamber was filled with 0·6 ml of RPMI-1640 10% fetal bovine serum (FBS) medium containing or not 500 ng/ml of CXCL12 (SDF-1) (R&D Systems, Abingdon, UK). SDF-1-attracted T cells were collected in the lower chambers, numbered, characterized by cytometry analysis and injected i.v. at a concentration of 2 × 106 cells together with 5 × 106 diabetogenic T cells. Non-migrating T cells collected in the upper chambers were used as control. To inhibit the chemokine receptor CXCR4, we used AMD3100 (Sigma-Aldrich, St Louis, MO, USA), a potent and specific antagonist, administered subcutaneously (s.c.) at 10 mg/kg in 200 µl of phosphate-buffered saline (PBS), as described elsewhere [14]. Recipient mice of 5 × 106 T splenocytes from diabetic mice were treated s.c. with AMD3100 or PBS three times a week 1 week before cell transfer and every day for 2 weeks thereafter. To study T cell homing, a 5-mM solution of 5, 6-carboxy-succinimidyl-fluorescein-ester (CFSE) (Molecular Probes, Eugene, OR, USA) in dimethylsulphoxide (DMSO) was added to purified T cells at a final concentration of 10 µM. After an incubation of 10 min at 37°C, cells were washed and suspended in Hanks' balanced salt solution (HBSS) medium. All manipulations were carried out in such a manner in order to minimize exposure to light. CFSE-labelled T cells were transferred i.v. and the presence of labelled T cells in spleen, pancreatic and mesenteric lymph nodes was evaluated 3 days after cell transfer.
In vitro studies
Lymphocytes isolated from spleens of 12-week-old NOD females by negative selection columns (R&D Systems) were incubated for 30 min with or without 10 µg/ml AMD3100. Cytokine concentrations in cell-free supernatants were determined after cell stimulation with plate-bound anti-CD3 and anti-CD28 monoclonal antibodies (MoAbs) (10 µg/ml per clone, clone 145–2C11 and clone 37·51; BD Pharmingen, San Diego, CA, USA). Supernatants were collected after 48 h of culture and analysed for cytokine content using specific enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems), following the manufacturer's instructions. To evaluate the effects of AMD3100 on the capacity of SDF-1 to attract CXCR4+ T cells, we performed comparative transwell chemotaxis assays. A total of 106 lymphocytes purified from spleens of 4- and 12-week-old NOD or BALB/c mice resuspended in 0·1 ml of RPMI-1640 medium, 10% FBS, were added to the top chambers. After an incubation of 3 h at 37°C, cells from the bottom chambers were collected.
RNA isolation and quantitative reverse transcription–polymerase chain reaction (RT–PCR) assay
Total RNA was isolated from islets and PLN using TRIzol Reagent method (Invitrogen, Cergy Pontoise, France). Islets obtained after collagenase digestion of pancreatic glands were purified on a discontinuous Ficoll gradient (40%, 23%, 20%, 11%; Sigma-Aldrich) and hand-picked using a binocular microscope. Control examination of the final islet preparation revealed more than 80% purity. RNA was extracted with chloroform followed by isopropanol precipitation and washed with ethanol. Quantitative RT–PCR analysis was performed by reverse transcription followed by real-time PCR. One μg of total RNA was reverse-transcripted with 100 U Superscript II (Invitrogen) using random hexamer primers and oligo(dT) (Promega, Charbonnières, France), 6·25 mM deoxyribonucleoside triphosphate (dNTP) (Promega), 0·1 M dithiothreitol (DTT) and 5′ first-strand buffer (Invitrogen). Real-time RT–PCR on a LightCycler (Roche Diagnostics) was performed in a final volume of 20 µl containing 15 μl of reaction buffer FastStart DNA Master SYBR Green I kit (Roche Diagnostics), 10·5 pmol of each forward and reverse specific primers (Eurobio, Les Ulis, France) and 5 μl of 60-fold dilution of the RT product. Pairs of PCR primers were designed using the Beacon designer software. CXCR4 primer sequence: forward 5′-TCAGTGGCTGACCTCCTCTT-3′, reverse 5′-CTTGGCCTTTGACTGTTGGT-3′, SDF-1 alpha forward 5′-CAGAGCCAACGTCAAGCA-3′, reverse 5′-AGGTACTCTTGGATCCAC-3′, CXCR3 forward 5′-TGCTAGATGCCTCGGACTTT-3, reverse 5′-CGCTGACTCAGTAGCACAATT-3′. Real-time PCR was performed in glass capillaries. For sample quantification, standards (corresponding to each gene) of the purified DNA from agarose gel were prepared by serial dilutions from 10 attmol/µg to 0·0001 attmol/µg (representing 6·106−60 copies/µg). A calibration curve was generated to determine each mRNA concentration. Samples were quantified using the LightCycler relative quantification software (Roche Diagnostics). PCR products were loaded onto an agarose gel to verified product size. Specific mRNA levels were expressed as percentage relative to cyclophilin mRNA levels.
Antibodies
Pancreatic glands were removed and frozen in optimum cutting temperature compound (Tissue-Tek, Euromedex, Souffelweyersheim, France). Sections were fixed in cold acetone for 5 min and blocked with 5% bovine serum albumin (BSA) (Sigma). For double immunofluorescence studies, primary antibodies were biotinylated CXCR4 antibody (BD Pharmingen), rabbit anti-mouse SDF1 (Cell Sciences, Canton, MA, USA), phycoerythrin (PE)-labelled rat anti-mouse CD4, CD8, CD19 CD11c (BD Pharmingen), rabbit antibody against porcine insulin (ICN Biomedicals/Cappel, Aurora, OH, USA) or rabbit anti-human von Willebrand factor polyclonal antibody (Chemicon International, Temecula, CA, USA). Bound antibodies were detected as appropriate with either streptavidin AlexaFluor 488 or 546 (Molecular Probes) or Alexa 594-conjugated goat anti-rabbit IgG (Molecular Probes). When the biotin–streptavidin system was used, sections were also blocked with an avidin–biotin blocking kit (Vector, Burlingame, CA, USA). Immunostained sections were examined using a Zeiss microscope equipped with a digital camera.
For flow cytometric analysis, single-cell suspensions from PLNs were prepared in Hanks' balanced salt solution (HBSS) medium 5% fetal calf serum (FCS) and incubated for 30 min at 4°C with fluorescein isothiocyanate (FITC)-conjugated rat anti-CD3 antibodies and either PE-conjugated anti-CCR5 (BD Pharmingen) or anti-CXCR3 antibodies (R&D Systems). CXCR4 positive cells were detected by using rat anti-CXCR4 antibodies (BD Pharmingen) and AlexaFluor 488-conjugated goat anti-rat IgG antibodies (Molecular Probes) simultaneously with PE-conjugated anti-CD3 or anti-CD19 antibodies.
Statistical analysis
Groups of animals were compared by Student's t-test analysis for independent samples with a value of P < 0·05 for statistical significance. Incidence curves for diabetes were compared using the Kaplan–Meier test.
Results
CXCR4 expression in PLN and islets of NOD mice
We quantified by real-time RT–PCR the levels of CXCR4 mRNA in the islets and PLNs from NOD females at 4 and 12 weeks and compared these results to age-matched Balb/c mice. As shown in Fig. 1, CXCR4 expression at 4 weeks was 3·4 times more elevated in the PLNs (Fig. 1a) and 1·5 times more elevated in the islets (Fig. 1b) of NOD mice versus control Balb/c mice and these differences were statistically significant (P < 0·001). As shown in Fig. 1a, a significant reduction in the ratio of CXCR4 over cyclophilin mRNA levels was observed in the PLN of NOD versus Balb/c mice at 12 weeks of age (10·2 ± 2·9 versus 26·5 + 7%, P < 0·001) while CXCR4 mRNA increased (Fig. 1b) in NOD versus Balb/c islets (7·4 ± 1·4 versus 0·9 ± 0·5%, P < 0·0001). Levels of CXCL-12 (SDF-1) mRNA decreased in the PLNs of NOD mice according to age (0·12 ± 0·003 at 4 weeks versus 0·09 ± 0·01% at 12 weeks, P < 0·01). On the other hand, levels of CXCR3 mRNA increased in the PLNs of NOD mice between 4 and 12 weeks of age (23·5 ± 17 versus 141 ± 6%, P < 0·001) and were very abundant in the infiltrated islets (data not shown).
Fig. 1.
Percentages of CXCR4/cyclophilin mRNA levels in non-obese diabetic female mice (n = 4, black bars) in comparison to Balb/C mice (n = 4, open bars) at 4 and 12 weeks of age in pancreatic lymph nodes (a) or islets (b).
We next examined the relative distribution of CXCR4, CXCR3 and CCR5 positive cells in the PLNs of NOD females at 4 or 12 weeks of age by flow cytometry analysis. As shown in Table 1, results from two independent experiments revealed that CD3+ T cells expressed all three chemokine receptors. Between 4 and 12 weeks of age, the percentages of CXCR4 +, CXCR3+ and CCR5+ CD3+ T cells decreased significantly in the PLNs (Table 1 and Fig. 2). CCR5+ CD3+ T cells were the predominant cell population with 12·4 ± 1·5% at 4 weeks of age. Among CXCR4+ CD3– cells, we found 16% of CD19+ and 14% of CD11c+ cells. No CXCR4+ cells were detected in the islets at 4 weeks of age in contrast to infiltrated islets where CXCR4+ cells were abundant.
Table 1.
Flow cytometry analysis of the percentages of chemokine receptor expressing cells in the pancreatic lymph nodes (PLNs) of non-obese diabetic female mice at 4 and 12 weeks of age. Results were pooled from two independent experiments.
| Age | CXCR4+CXCR4+ CD3+ | CXCR3+CXCR3+ CD3+ | CCR5+CCR5+ CD3+ |
|---|---|---|---|
| 4 weeks (n = 5) | 21·6 ± 4·11 | 10·4 ± 1·73 | 17·2 ± 1·15 |
| 6·8 ± 1·62 | 7·5 ± 0·84 | 12·4 ± 1·56 | |
| 12 weeks (n = 5) | 17·4 ± 1·31 | 6·3 ± 1·43 | 13·9 ± 1·65 |
| 4·9 ± 0·82 | 5·1 ± 1·14 | 8·6 ± 1·46 |
P-values
< 0·05
< 0·002.
Fig. 2.
Representative fluorescence activated cell sorter (FACS) analysis of pancreatic lymph nodes of non-obese diabetic female mice at 4 weeks (grey lines) or 12 weeks (dark lines) for the presence of CXCR4 (a), CXCR3 (b) and CCR5 (c).
In vivo studies of the CXCR4–CXCL12 (SDF-1) pathway
To determine the capacity of CXCR4+ T cells to interact with the immune process leading to beta cell destruction, we first decided to set an adoptive co-transfer experiment by injecting irradiated NOD males with the CXCR4+ T cell-enriched cell population and T splenocytes from diabetic females. Results illustrated in Fig. 3 indicated clearly that CXCR4+ T cells attracted in vivo by SDF-1 in transwell chemotaxis chambers completely abolished the capacity of diabetogenic T cells to transfer diabetes (none of six versus five of six diabetic mice, P < 0·001).
Fig. 3.
SDF-1 attracted CXCR4+ cells completely abolished the capacity of diabetogenic T cells to transfer diabetes (open circles) in contrast to cells from the upper chambers (closed circles). The incidence curve of mice transferred with diabetogenic T cells is illustrated as closed squares.
To characterize the potential role of the CXCL12–CXCR4 pathway in autoimmune diabetes, we treated mice with AMD3100, a specific CXCR4 inhibitor. Results shown in Fig. 4 indicated that AMD3100 significantly accelerated the capacity of diabetogenic T cells to transfer diabetes in comparison to sham-treated animals in two independent experiments (nine of 12 versus four of 12 diabetic mice at 30 days, Kaplan–Meier test P < 0·05). When PLNs from transferred animals were analysed for cytokine profiles, IFN-γ mRNA increased (14 ± 1·5 versus 6·4 ± 0·8, P < 0·01) and IL-4 mRNA decreased significantly (2·1 ± 0·4 versus 6·3 ± 2·5, P < 0·05). In this experiment, levels of IL-10 mRNA were identical to sham-treated animals.
Fig. 4.
In vivo administration of AMD3100 accelerates adoptive transfer of diabetes. The incidence curve for AMD3100-treated mice (closed circles) was significantly higher than sham-treated animals (open circles) (P < 0·05, Kaplan–Meier test).
To examine whether AMD3100 administration could modify T cell homing, we transferred intravenously CFSE-labelled diabetogenic T cells and analysed the recruitment of labelled T cells in spleen and lymph nodes. Mice treated with AMD3100 (n = 6) showed a significant increase in the percentage of labelled T cells 5 days after i.v. transfer in comparison with sham-treated animals (n = 6) in spleen (7·45 ± 1·9 versus 4·45 ± 0·8%, P < 0·01), PLNs (2·68 ± 1·6 versus 0·82 ± 0·3%, P < 0·05) and MLNs (8·78 ± 2·8 versus 4·52 ± 0·7%, P < 0·01). Immunofluorescence studies of pancreatic glands from transferred animals, as shown in Fig. 5, indicated that AMD3100 reduced the number of CXCR4+ cells in the islets but increased the number of autoreactive T cells and the severity of insulitis. Interestingly, mice treated with AMD3100 after cell transfer also had a reduction in the number of SDF-1 positive cells that were located preferentially in the islet periphery in sham-treated mice (Fig. 5).
Fig. 5.
Immunofluorescence studies of pancreatic glands of 12-week-old non-obese diabetic mice treated with phosphate-buffered saline (PBS) (a,c) or 10 mg/kg AMD3100 a specific CXCR4 antagonist (b,d). AMD3100 reduced the number of CXCR4+ cells (green, a,b) in the islets and the number of SDF-1 positive cells surrounding the islets (red) while the number of infiltrating CD3+ cells (green, c,d) increased.
AMD3100 inhibits SDF-1 signalling and Th2-type cytokine production
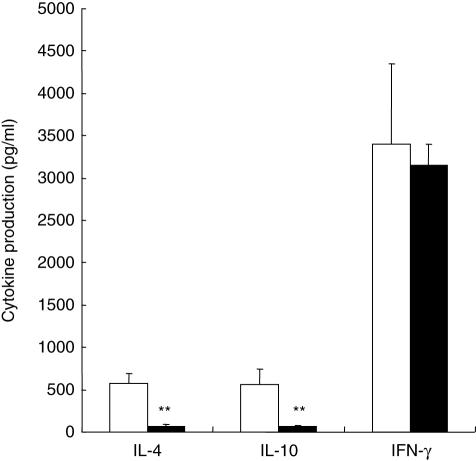
Using a transwell migration system, we observed a significantly higher rate of cell migration upon stimulation with CXCL-12 (SDF-1) using splenic NOD T cells as shown in Fig. 6. The number of migrating cells upon CXCL12 (SDF-1) stimulation remained identical between 4 and 12 weeks of age (Fig. 6a,b). Incubating T lymphocytes with AMD3100 resulted in the inhibition of cell transmigration using amounts of AMD3100 as little as 0·1 µg (Fig. 6c). Acceleration of diabetes transfer using AMD3100 prompted us to assess the phenotype of the chemoattracted cells using migration chambers. Flow cytometry analysis of transmigrated cells revealed an increase in the percentages of CD4+ CD62L+ T cells (11·4 versus 5%, P < 0·01). To examine the potential effect of AMD3100 on cytokine profiles, we measured the levels of IFN-γ, IL-4 and IL-10 cytokines in the supernatants of T lymphocytes from the spleen of 12-week-old NOD females preincubated or not with AMD3100 for 30 min. As shown in Fig. 7, AMD3100 did not modify IFN-γ production in comparison to controls (3·15 ± 0·24 versus 3·40 ± 0·94 ng/ml, P = 0·5), but decreased significantly the levels of both IL-4 (68·3 ± 17 versus 575·1 ± 112 pg/ml, P < 0·001) and IL-10 (70·7 ± 13 versus 555·5 ± 190 pg/ml, P < 0·001).
Fig. 6.
In vitro studies of cell trafficking using double chamber culture plates using splenocytes of 4-week-old (a) or 12-week-old (b) mice. SDF-1 (black bars) increased significantly the number of transmigrated lymphocytes in non-obese diabetic mice versus Balb/c mice at 4 weeks and this effect was blocked by adding AMD3100 in the upper chamber (c). **P-value < 0·01.
Fig. 7.
In vitro incubation with AMD3100 (closed bars) reduced significantly the levels of T helper 2 (Th2) cytokines produced by diabetogenic T cells in comparison to control (open bars) after plate-bound anti-CD3 and anti-CD28 stimulation in contrast to similar levels of interferon (IFN)-γ production.
Discussion
Our focus was to characterize the importance of the SDF-1/CXCR4 ligand chemokine receptor pair for development of insulitis and autoimmune diabetes in NOD mice. Our key findings showed that CXCR4 expression was increased in PLNs and inflamed islets that CXCR4 T cells blocked the capacity of diabetogenic T cells to transfer diabetes and that inhibition of CXCR4 by AMD3100 accelerated autoimmune diabetes by inhibiting Th2-mediated immune responses. These results suggest that the CXCR4/SDF-1 pathway serves beneficial regulatory functions in the NOD mouse model.
In this study, we have demonstrated an early wave of CXCR-4 expression in the PLNs and a late but progressive increase in the inflamed islets of NOD mice. In addition, SDF-1 expression was also increased in infiltrated islets, suggesting possible recruitment and local activation of CXCR4+ T cells. Blocking this specific cell population with AMD3100 resulted in the decrease of CXCR4+ T cell homing to the islets and the increase in the Th1/Th2 cytokine ratio both in PLNs and islets. These changes were associated with an acceleration of autoimmune diabetes in T cell-transferred recipients. On the other hand, we provided evidence that SDF-1 attracted CXCR4+ T cells that had the capacity to protect recipient mice from an adoptive cell transfer of diabetes. Those data indicate collectively that CXCR4+ T cells belong to Th2-type cells and are recruited to the pancreas of NOD females by SDF-1. This conclusion is in sharp contrast with the common view that SDF-1, expressed constitutively in a broad range of tissues, is involved in basal trafficking of naïve lymphocytes [15]. CXCR4+ T cells may down-regulate Th1-type T cells transiently in the islet infiltrates, which may explain the time lag between insulitis and diabetes onset during the pre-diabetic phase. The fact that AMD3100 administration was not able to increase significantly the spontaneous incidence of spontaneous diabetes is not contradictory and was due probably to the short duration of treatment. It has been shown that upon IL-2 activation, expression of CXCR4 was reduced in contrast to CXCR3 which may target T, natural killer (NK) and NK T cells to sites of inflammation [16]. Those results correlate with our results in the PLNs from 12-week-old females with a decrease in CXCR4 while CXCR3 expression increased. These patterns are of interest as a close temporal correlation between T cell activation in the PLNs and islet infiltration has been demonstrated, supporting the hypothesis that beta cell antigens are transported specifically to PLNs where they trigger reactive T cells to invade the islets [17]. In line with this hypothesis, our CFSE findings indicated that homing to lymphoid organs and especially pancreatic lymph nodes was increased significantly in AMD3100-treated animals, which may predispose to diabetes. Development of diabetes results from a breakdown in tolerance to pancreatic islet antigens and the inability of CD4+ CD25+ regulatory T cells (Tregs) to counteract pathogenic T cells [18]. These Tregs specifically express l-selectin (CD62L), delay adoptive transfer of diabetes [19] and mediate oral tolerance [20]. It has been shown recently that CXCR4 is required for migration of forkhead box P3 (FoxP3)+ CD4+ CD25+ thymic T cells to secondary lymphoid tissues [21]. It can be hypothesized that AMD3100 inhibits this migration through trafficking receptor interactions. The observation that CXCR4 is down-regulated while a number of memory/effector type homing receptors are up-regulated for migration to non-lymphoid tissues upon antigen priming is in accordance with our findings that CXCR4 expression is decreased in pancreatic lymph nodes at 12 weeks of age. We have shown here that SDF-1 chemoattracts CD62L positive CD4+ T cells using a transwell migration system. The demonstration that the number of CXCR4+ splenocytes chemoattracted in vitro did not increased in pre-diabetic versus young animals was against any spontaneous recruitment of this T cell subset which may fit with the concept that NOD mouse has a general defect in T cell suppression. Whether CD4+ CXCR4+ CD62L+ cells during procedures that protect NOD mice from diabetes may influence functional cellular responses is under investigation.
Blocking CXCR4+ receptor/ligand interactions with AMD3100 accelerated the rate of diabetes incidence in the recipients of diabetogenic T cells. This is in apparent contrast with a previous report indicating that antibodies to SDF-1 delayed average diabetes onset by 10 weeks, suppressed insulitis and led to a 30% reduction of spontaneous diabetes at 30 weeks of age [22]. Although SDF-1 and its receptor CXCR4 were shown to bind only to each other [15,23], co-operation between inducible CXCR3 ligands and constitutive SDF-1/CXCL12 has also been observed. This may regulate recruitment of DCs either in lymph nodes or at peripheral sites of inflammation [24]. Prevention of autoimmune diabetes by antibodies to SDF-1 may result from a reduction in DC activation and homing to PLNs. Antibodies to SDF-1 may also alter the binding to glycosaminoglycans on endothelial cells (ECs) which is hypothesized to be relevant to regulation of leucocyte diapedesis and neoangiogenesis during inflammatory responses [25]. Rheumatoid arthritis (RA) synovial tissues had higher levels of CXCL12 on the endothelium, which was co-localized to heparan sulphate proteoglycans and high endothelial venules to promote angiogenesis and inflammatory cell infiltration [26]. In contrast to our results in NOD mice, AMD3100 was shown to reduce autoimmune collagen-induced arthritis and to inhibit inflammatory responses to exogenous SDF-1 injection in periarthritic tissues [27]. This suggests strongly that the CXCL12/CXCR4 axis acts differently in these two autoimmune models. The capacity of the islet endothelium to express SDF-1 to assure successful homing and adhesion of immune competent cells is indeed a critical step in autoimmunity. Alpha4–integrin/MadCAM interactions are important in the inflamed islets. In NOD pancreata, alpha(4) beta(7) is expressed on most infiltrating cells at all stages of insulitis [28]. It has been suggested that up-regulation by SDF-1alpha of lymphocyte adhesion mediated by alpha(4) beta(7) could contribute to lymphocyte homing to secondary lymphoid tissues [29]. In addition, l-selectin stimulation has been shown to significantly increase surface CXCR4 expression and function [30]. In this study, blockade of SDF-1-induced CXCR4 internalization by l-selectin increased SDF-1-induced lymphocyte adhesion and transendothelial migration. In constrast, we observed that CXCR3 T cells accumulated both in PLNs and islets of pre-diabetic and diabetic mice. In line with our results, all human T cell clones detected in the islets of NOD SCID mice after cell transfer expressed CXCR3 [31]. AMD3100 is also an effective mobilizer of haematopoietic progenitor cells [32]. However, this effect cannot be responsible for the aggravation of diabetes incidence, because bone marrow stem cells of T cell recipients had a low capacity to proliferate in our experiments of cell transfer due to sublethal irradiation.
Direct involvement of the SDF-1/CXCR4 ligand/chemokine receptor pair in the recruitment and differentiation of stem cells has also been reported including cardiomyocytes [33] and pancreatic islets [34]. This system appears to be activated after tissue injury. Neutralization with CXCR4 antibody has been shown to elicit a fourfold increase in the proportion of apoptotic ductal cells. We cannot exclude that regeneration of beta cells after administration of AMD3100 had been hampered either through defective mobilization of haematopoietic stem cells or migration of ductal cells upon SDF-1 gradient. This hypothesis may explain why few CXCR4+ cells were seen at the proximity of ducts and ductal infiltrates (data not shown). However, we were unable to demonstrate CXCR4 expression at the surface of insulin-containing cells by indirect immunofluorescence. Thus, although the precise pathophysiological role of CXCR4 chemokine receptor expression on beta cell differentiation needs to be clarified further, it is likely to play an important role during insulitis.
In conclusion, the results presented here indicate the importance of the CXCL12/CXCR4 pair as a tool to examine Th2-cell-dependent immunity and its ability to prevent autoimmune diabetes. Increasing the capacity and numbers of CD4+ CXCR4+ cells may be beneficial.
Acknowledgments
We are indebted to Annie Durand for excellent technical assistance and to Christiane Aubry for managing the NOD colony.
References
- 1.Yang Y, Santamaria P. Lessons on autoimmune diabetes from animal models. Clin Sci (Lond) 2006;110:627–39. doi: 10.1042/CS20050330. [DOI] [PubMed] [Google Scholar]
- 2.Atkinson MA, Leiter EH. The NOD mouse model of type 1 diabetes: as good as it gets? Nat Med. 1999;5:601–4. doi: 10.1038/9442. [DOI] [PubMed] [Google Scholar]
- 3.O'Reilly LA, Hutchings PR, Crocker PR, et al. Characterization of pancreatic islet cell infiltrates in NOD mice: effect of cell transfer and transgene expression. Eur J Immunol. 1991;21:1171–80. doi: 10.1002/eji.1830210512. [DOI] [PubMed] [Google Scholar]
- 4.Fabien N, Bergerot I, Maguer-Satta V, et al. Pancreatic lymph nodes are early targets of T cells during adoptive transfer of diabetes in NOD mice. J Autoimmun. 1995;8:323–34. doi: 10.1006/jaut.1994.0025. [DOI] [PubMed] [Google Scholar]
- 5.Gagnerault MC, Luan JJ, Lotton C, Lepault F. Pancreatic lymph nodes are required for priming of beta cell reactive T cells in NOD mice. J Exp Med. 2002;5:369–77. doi: 10.1084/jem.20011353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tang Q, Adams JY, Tooley AJ, et al. Visualizing regulatory T cell control of autoimmune responses in nonobese diabetic mice. Nat Immunol. 2006;7:83–92. doi: 10.1038/ni1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liblau RS, Singer RM, McDewitt HO. Th1 and Th2 CD4+ T cells in the pathogenesis of organ specific autoimmune diseases. Immunol Today. 1995;16:34–8. doi: 10.1016/0167-5699(95)80068-9. [DOI] [PubMed] [Google Scholar]
- 8.Bradley LM, Asensio VC, Schioetz LK, et al. Islet-specific Th1, but not Th2, cells secrete multiple chemokines and promote rapid induction of autoimmune diabetes. J Immunol. 1999;162:2511–20. [PubMed] [Google Scholar]
- 9.Charo IF, Ransohoff RM. The many role of chemokines and chemokine receptors in inflammation. N Engl J Med. 2006;354:610–21. doi: 10.1056/NEJMra052723. [DOI] [PubMed] [Google Scholar]
- 10.Debes GF, Dahl ME, Mahiny AJ, et al. Chemotactic responses of IL-4-, IL-10-, and IFN-gamma-producing CD4+ T cells depend on tissue origin and microbial stimulus. J Immunol. 2006;176:557–66. doi: 10.4049/jimmunol.176.1.557. [DOI] [PubMed] [Google Scholar]
- 11.Jinquan T, Quan S, Jacobi HH, et al. CXC chemokine receptor 4 expression and stromal cell-derived factor-1alpha-induced chemotaxis in CD4+ T lymphocytes are regulated by interleukin-4 and interleukin-10. Immunology. 2000;99:402–10. doi: 10.1046/j.1365-2567.2000.00954.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Aspord C, Rome S, Thivolet C. Early events in islets and pancreatic lymph nodes in autoimmune diabetes. J Autoimmun. 2004;23:27–35. doi: 10.1016/j.jaut.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 13.Harrison JK, Fong AM, Swain PA, et al. Mutational analysis of the fractalkine chemokine domain. Basic amino acid residues differentially contribute to CX3CR1 binding, signalling and cell adhesion. J Biol Chem. 2001;276:21632–41. doi: 10.1074/jbc.M010261200. [DOI] [PubMed] [Google Scholar]
- 14.Fricker SP, Anastassov V, Cox J, et al. Characterization of the molecular pharmacology of AMD3100: a specific antagonist of the G-protein coupled chemokine receptor, CXCR4. Biochem Pharmacol. 2006;72:588–96. doi: 10.1016/j.bcp.2006.05.010. [DOI] [PubMed] [Google Scholar]
- 15.Bleul CC, Fuhlbrigge RC, Casasnovas JM, et al. A highly efficacious lymphocyte chemoattractant, stromal cell-derived factor 1 (SDF-1) J Exp Med. 1996;184:1101–9. doi: 10.1084/jem.184.3.1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Beider K, Nagler A, Wald O, et al. Involvement of CXCR4 and IL-2 in the homing and retention of human NK and NK T cells to the bone marrow and spleen of NOD/SCID mice. Blood. 2003;102:1951–8. doi: 10.1182/blood-2002-10-3293. [DOI] [PubMed] [Google Scholar]
- 17.Hoglund P, Mintern J, Waltzinger C, et al. Initiation of autoimmune diabetes by developmentally regulated presentation of islet cell antigens in the pancreatic lymph nodes. J Exp Med. 1999;189:331–9. doi: 10.1084/jem.189.2.331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bour-Jordan H, Salomon BL, Thompson HL, et al. Costimulation controls diabetes by altering the balance of pathogenic and regulatory T cells. J Clin Invest. 2004;114:979–87. doi: 10.1172/JCI20483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Szanya V, Ermann J, Taylor C, et al. The subpopulation of CD4+ CD25+ splenocytes that delays adoptive transfer of diabetes express L-Selectin and high levels of CCR7. J Immunol. 2002;169:2461–5. doi: 10.4049/jimmunol.169.5.2461. [DOI] [PubMed] [Google Scholar]
- 20.Aspord C, Czerkinsky C, Durand A, et al. alpha4 integrins and 1-selectin differently orchestrate T-cell activity during diabetes prevention following oral administration of CTB-insulin. J Autoimmun. 2002;19:223–32. doi: 10.1006/jaut.2002.0610. [DOI] [PubMed] [Google Scholar]
- 21.Lee JH, Kang SG, Kim CH. FoxP3+ T cells undergo conventional first switch to lymphoid tissue homing receptors in thymus but accelerated second switch to nonlymphoid tissue homing receptors in secondary lymphoid tissues. J Immunol. 2007;178:301–11. doi: 10.4049/jimmunol.178.1.301. [DOI] [PubMed] [Google Scholar]
- 22.Matin K, Salam MA, Akhter J, et al. Role of stromal-cell derived factor-1 in the development of autoimmune diseases in non-obese diabetic mice. Immunology. 2002;107:222–32. doi: 10.1046/j.1365-2567.2002.01478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ganju RK, Brubaker SA, Meyer J, et al. The alpha-chemokine, stromal cell-derived factor 1alpha, binds to the transmembrane G-protein coupled CXCR-4 receptor and activates multiple signal transduction pathways. J Biol Chem. 1998;273:23169–75. doi: 10.1074/jbc.273.36.23169. [DOI] [PubMed] [Google Scholar]
- 24.Vanbervliet B, Bendriss-Vermare N, Massacrier C, et al. The inducible CXCR3 ligands control plasmacytoid dendritic cell responsiveness to the constitutive chemokine stromal cell-derived factor 1 (SDF-1) /CXCL12. J Exp Med. 2003;198:823–30. doi: 10.1084/jem.20020437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Okada T, Ngo VN, Ekland EH, et al. Chemokine requirements for B cell entry to lymph nodes and Peyer's patches. J Exp Med. 2002;196:65–75. doi: 10.1084/jem.20020201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pablos JL, Santiago B, Galindo M, et al. Synoviocyte-derived CXCL12 is displayed on endothelium and induces angiogenesis in rheumatoid arthritis. J Immunol. 2003;170:2147–52. doi: 10.4049/jimmunol.170.4.2147. [DOI] [PubMed] [Google Scholar]
- 27.Matthys P, Hatse S, Vermeire K, et al. AMD3100, a potent and specific antagonist of the stromal cell-derived factor-1 chemokine receptor CXCR4, inhibits autoimmune joint inflammation in IFN-gamma receptor-deficient mice. J Immunol. 2001;167:4686–92. doi: 10.4049/jimmunol.167.8.4686. [DOI] [PubMed] [Google Scholar]
- 28.Hanninen A, Taylor C, Streeter PR, et al. Vascular addressins are induced on islet vessels during insulitis in nonobese diabetic mice and are involved in lymphoid cell binding to islet endothelium. J Clin Invest. 1993;92:2509–15. doi: 10.1172/JCI116859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wright N, Hidalgo A, Rodriguez-Frade JM, et al. The chemokine stromal cell-derived factor-1 alpha modulates alpha 4 beta 7 integrin-mediated lymphocyte adhesion to mucosal addressin cell adhesion molecule-1 and fibronectin. J Immunol. 2002;168:5268–77. doi: 10.4049/jimmunol.168.10.5268. [DOI] [PubMed] [Google Scholar]
- 30.Ding Z, Issekutz TB, Downey GP, Waddell TK. 1-selectin stimulation enhances functional expression of surface CXCR4 in lymphocytes: implications for cellular activation during adhesion and migration. Blood. 2003;101:4245–52. doi: 10.1182/blood-2002-06-1782. [DOI] [PubMed] [Google Scholar]
- 31.Van Halteren AG, Kardol MJ, Mulder A, Roep BO. Homing of human autoreactive T cells into pancreatic tissue of NOD-SCID mice. Diabetologia. 2005;48:75–82. doi: 10.1007/s00125-004-1613-2. [DOI] [PubMed] [Google Scholar]
- 32.Capoccia BJ, Shepherd RM, Link DC. G-CSF and AMD3100 mobilize monocytes into the blood that stimulate angiogenesis in vivo through a paracrine mechanism. Blood. 2006;108:2438–45. doi: 10.1182/blood-2006-04-013755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abbott JD, Huang Y, Liu D, et al. Stromal cell-derived factor-1alpha plays a critical role in stem cell recruitment to the heart after myocardial infarction but is not sufficient to induce homing in the absence of injury. Circulation. 2004;110:3300–5. doi: 10.1161/01.CIR.0000147780.30124.CF. [DOI] [PubMed] [Google Scholar]
- 34.Kayali AG, Van Gunst K, Campbell IL, et al. 1alpha/CXCR4 ligand-receptor axis is critical for progenitor survival and migration in the pancreas. J Cell Biol. 2003;163:859–69. doi: 10.1083/jcb.200304153. [DOI] [PMC free article] [PubMed] [Google Scholar]