Abstract
Free radical formation in the cochlea plays a key role in the development of noise-induced hearing loss (NIHL). The amount, distribution, and time course of free radical formation have been defined, including a clinically significant formation of both reactive oxygen species and reactive nitrogen species 7–10 days following noise exposure. Reduction in cochlear blood flow as a result of free radical formation has also been described. Here we report that the antioxidant agents, vitamins A, C, and E, act in synergy with magnesium to effectively prevent noise-induced trauma. Neither the antioxidant agents nor magnesium reliably reduced NIHL or sensory cell death with the doses we used when these agents were delivered alone. In combination, however, they were highly effective in reducing both hearing loss and cell death even with treatment initiated just one hour prior to noise exposure. This study supports roles for both free radical formation and noise-induced vasoconstriction in the onset and progression of NIHL. Identification of this safe and effective antioxidant intervention that attenuates NIHL provides a compelling rationale for human trials in which free radical scavengers are used to eliminate this single major cause of acquired hearing loss.
Keywords: cochlea, free radical, noise, hearing, antioxidant, vasodilation
Mechanical destruction of cells in the organ of Corti was once assumed to be the primary cause of noise-induced hearing loss (NIHL) [1–8], with perhaps some effect of reduced blood flow to the inner ear [9–18]. We now know that another key factor is intense metabolic activity that results in production of excess free radicals [19–23] and lipid peroxidation products [24]. Noise-induced production of reactive oxygen species (ROS) in the cochlea has now been well characterized, and several recent reviews are available [25–27]. Mitochodrial dysfunction and ROS production have been implicated in numerous neurodegenerative syndromes and diseases [28–34, see 35, 36]. The use of antioxidant agents holds significant therapeutic promise for many neurodegenerative processes [32, 33, 37–42], and there is some suggestion that combinations of antioxidants are more effective than single agents [43]. The similarity of free radical production across multiple neurodegenerative diseases, including NIHL, and the putative efficacy of antioxidants in reducing neurodegenerative processes, provides a compelling rationale for study of antioxidants to prevent NIHL.
Consistent with antioxidant protection in neurodegenerative-cell death, many antioxidants are well known to partially protect sensory cells in the organ of Corti from stress-induced destruction. Upregulation of the endogenous antioxidant glutathione reduces NIHL and cell pathology [44, 45], whereas the opposite is observed with reduced endogenous antioxidants [20, 22, 23, 44]. That exogenous antioxidant agents reduce sensory cell death and NIHL has been well demonstrated in animal studies using a variety of free radical scavengers [24, 44–56], including several studies with dietary antioxidants [50, 57–60]. Magnesium supplements also reduce NIHL [61–69].
Given the potential for synergy of multiple agents in protection from neurodegenerative-cell death, in this investigation, we evaluated protection from NIHL and noise-induced sensory cell death with the combination of vitamins A, C, and E, or magnesium, or vitamins A, C, and E, plus magnesium. Each of these agents has a distinct mechanism of action. The primary antioxidant action of β-carotene (metabolized to vitamin A in vivo) is to scavenge singlet oxygen; because singlet oxygen reacts with lipids to form lipid hydroperoxides, the removal of singlet oxygen prevents lipid peroxidation [for review, see 70]. Vitamin E, present in lipids in cells [see 71], is a donor antioxidant that reacts with and reduces peroxyl radicals and, thus, inhibits the propagation cycle of lipid peroxidation [for review, see 70]. Vitamin C detoxifies free radicals by reducing them [for review, see 72]. Scavenging of oxygen radicals by vitamin C occurs in the aqueous phase [73, 74], which is in contrast to the site of action of vitamin E, within membranes. Given differences in mechanism and site of action, there are differences in antioxidant free radical scavenging ability [70, 75] as well as synergistic interaction of hydrophilic and lipophilic antioxidants [73, 74, 76–78]. Antioxidant scavenging of ROS also potentially reduces vasoconstriction that occurs with ROS production [46, for review see 27]. One effect of magnesium is reduction of noise-induced vasoconstriction [67, 79–81].
Demonstration of an additive efficacy of these agents would provide greater insight into their mechanisms of action. A treatment that prevents, or more effectively reduces, NIHL would be of significant clinical benefit to the millions of workers exposed to high levels of noise, military personnel, and millions of others exposed to high levels of noise during recreational activities.
Methods
Animals
Pigmented male guinea pigs (250–300g; Elm Hill Breeding Labs, Chelmsford, MA) with a normal Preyer’s reflex were used. Male guinea pigs were selected based on description of sex differences in ROS detoxification [82], activity of glutathione S-transferase in the cochlea [83], and susceptibility to NIHL [84]. The experimental protocol was reviewed and approved by the University Committee for the Care and Use of Animals (UCUCA) at the University of Michigan; all procedures conform to the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Experimental groups
Guinea pigs were divided into four groups. All groups received once daily treatments beginning one-hour prior to noise exposure and continuing once daily at 24-hour intervals until day 5 post-noise, for a total of 6 daily treatments. Control animals (N=9) received saline injections (1 cc, i.p.). The second group was treated with vitamins A (2.1 mg/kg beta-carotene, p.o.), C (71.4 mg/kg L-threoascorbic acid, s.c.), and E (26 mg/kg (±)-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid, “trolox”, s.c.) (“ACE”, N=8). Trolox is a cell-permeable, water-soluble derivative of vitamin E. The third group was treated with magnesium sulfate (“Mg”, 2.85 mmol/kg, equivalent to 343 mg/kg, s.c., N=6). The fourth group received a combination of ACE and Mg (at the same doses as groups 2 and 3, “ACEMg”, N=6). All test substances were purchased from Sigma-Aldrich (St. Louis, MO) (beta-carotene, #C9750, CAS 7235-40-7; L-threoascorbic acid, #A5960, CAS 50-81-7; Trolox, Fluka Chemika #56510, CAS 53188-07-1; magnesium sulfate, #M7506, CAS 7487-88-9).
Noise exposure
All subjects were exposed to octave-band noise (centered at 4 kHz, 120 dB SPL, 5 hours). This noise exposure is routinely used in our laboratory [85, 86]. As in those investigations animals were exposed, two at a time in separate cages, in a ventilated sound exposure chamber. The sound chamber was fitted with speakers (Model 2450H, JBL, Salt Lake City, UT) driven by a noise generator (ME 60 graphic equalizer, Rane, Mukilteo, WA) and power amplifier (HCA-1000 high current power amplifier, Parasound Products, San Francisco, CA). Sound levels were calibrated (Type 2203 precision sound level meter, Type 4134 microphone, Bruel and Kjar Instruments, Norcross, GA) at multiple locations within the sound chamber to ensure uniformity of the stimulus, using a fast Fourier transform network analyzer with a linear scale. The stimulus intensity varied by a maximum of 3 dB across measured sites within the exposure chamber. During noise exposure, noise levels were monitored using a sound level meter, a pre-amplifier, and a condenser microphone positioned in the center of the chamber at the level of the animal’s head.
Auditory brainstem responses
Auditory brainstem response (ABR) thresholds at 4, 8, and 16 kHz were measured for both right and left ears at two time points. Baseline ABR thresholds were established within 7 days prior to experimental day 1 (the first day of saline or micronutrient treatment, delivered on the day of noise exposure 1 hour prior to noise onset). Post-noise thresholds were established on day 10 post noise exposure. There is some recovery of ABR thresholds initially post-noise; both ABR threshold shift and loss of outer hair cells (OHCs) stabilize within 10 days of noise exposure [86].
Prior to ABR tests, animals were anesthetized with xylazine (10 mg/kg i.m.) and ketamine (40 mg/kg i.m.) and placed on a heating pad in one of two sound-isolated chambers (Industrial Acoustics Company, Bronx, New York, or C-A Tegnér, Bromma, Sweden). The external ear canals and tympanic membranes were inspected using an operating microscope to assure the ear canal was free of wax, there was no canal deformity, no inflammation of tympanic membrane, and no effusion of the middle ear [as in 85], then needle electrodes were placed subcutaneously below the test ear, at the vertex, and below the contralateral ear.
Acoustic stimuli were 4, 8, and 16 kHz tone bursts (15-ms duration; 1-ms rise-fall; 10/s) generated using Tucker-Davis Technology (TDT, Alachua, FL) software (SigGen 3.2, or SigGenRP) and TDT System II/III hardware (DA1, FT6-2, PA5, or RP2.1, PA5, HB7). Signals were presented to the external auditory meatus in a closed acoustic system through a tube connected to a transducer (Beyer DT-48, Beyer Dynamic, Farmingdale, NY). Starting levels were 80–85 dB during baseline tests, and 100–105 dB SPL during post-noise threshold tests. Sound intensity was initially decreased in 10 to 20 dB steps with 5-dB decrements presented near threshold. Evoked responses to 1024 tone presentations were amplified (100,000x) and filtered (300–3,000 Hz) (DB4 BioAmp, or RA4PA/RA4L1 Medusa) then digitized (AD1, or RA16 Medusa) and averaged (BioSig 3.2, or BioSigRP). Threshold, tested separately for each ear, was defined as the lowest intensity of stimulation that yielded a repeatable waveform based on an identifiable ABR wave III or IV. ABR wave III is the most robust component of the guinea pig waveform [87, 88].
Histological examinations
On day 14, after ABR measurement, the deeply anesthetized animals were decapitated and the cochleae were immediately removed for immunohistochemical staining with rhodamine phalloidin and hair cell counts. Upon removal, cochleas were transferred into 4% paraformaldehyde in 0.1M phosphate-buffered saline (PBS, pH 7.4). Under a dissecting microscope, the bone nearest the apex and the round and oval windows was opened, followed by gentle local perfusion from the apex. The tissue was kept in fixative for 12 hours, then the bony capsule and the lateral wall tissues were removed, and the modiolar core was carefully removed from the temporal bone. Following permeabilization with Triton X-100 (0.3%, 30 min), the organ of Corti was stained for f-actin using rhodamine phalloidin (1%, 60–120 min) to outline hair cells and their stereocilia [89]. After washing the tissues with PBS, the organ of Corti was dissected and surface preparations were mounted on glass slides. The tissues were observed under fluorescence microscopy, and the number of missing inner hair cells and outer hair cells were counted from the apex to the base in 0.19 mm segments [as described in 86]. Counting was begun approximately 0.76–1.14 mm from the apex, thus omitting the initial irregular most-apical part of the cochlear spiral. Percentages of hair cell loss in each 0.19 mm length of tissue were plotted along the cochlear length.
Statistical analysis
All data values in the text and figures are mean ± S.E.M.; all statistical comparisons were performed using SPSS. Statistical reliability of group differences in threshold and threshold shift were via ANOVA; frequency (4, 8, and 16 kHz) and ear (left, right) were treated as within-subject factors and treatment (saline, ACE, Mg, and ACEMg) was the between-subjects factor. Adjustment for multiple comparisons was accomplished using the Bonferroni correction. Inner and outer hair cell loss in different treatment groups were also compared via ANOVA; cochlear place (0–4.99 mm, 5–9.99 mm, 10–14.99 mm, and 15–20 mm from the apex) and ear (left, right) were treated as within-subject factors and treatment (saline, ACE, Mg, and ACEMg) was the between-subjects factor. Specific analyses are described below.
Results
Noise-induced threshold deficits were significantly smaller in animals treated with a combination of antioxidant agents and magnesium (see Figure 1). ABR threshold shift measured 10-days post-noise varied with treatment group (F=15.289; df=3,25; p<0.001) as well as frequency (F=43.844; df=1.854, 46.338; p<0.001); there was no effect of side (right/left), and there were no significant interactions of frequency x treatment, side x treatment, frequency x side, or frequency x treatment x side (all p’s > 0.12). Adjusted pair-wise comparisons revealed that ABR thresholds were reliably lower in the group treated with vitamins A, C, E and magnesium compared to all other groups (all p’s<0.001); lack of statistically reliable interactions among factors indicates that protection was equivalent across all test frequencies.
Figure 1.
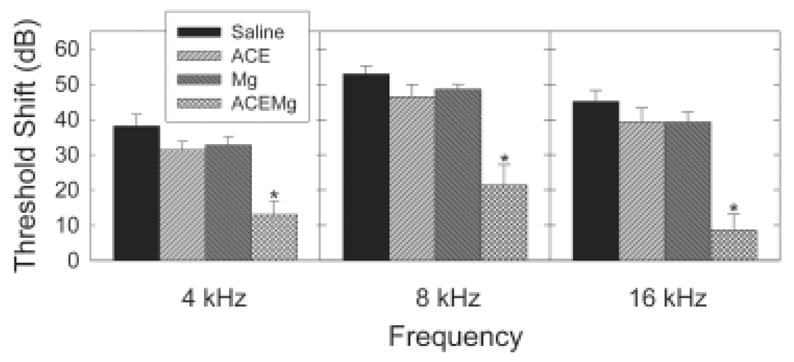
Noise-induced hearing loss, estimated using auditory brainstem response thresholds prior to and 10 days post noise (octave-band noise centered at 4 kHz, 120-dB SPL, 5 hours), was reduced at 4, 8, and 16 kHz by treatment with a combination of vitamins A, C, E and magnesium (ACEMg), but not by treatment with the antioxidants (ACE) or magnesium (Mg). Asterisks indicate statistically reliable differences (p’s < 0.001) between ACEMg and all other groups.
The significant reduction in noise-induced hearing loss in animals treated with both dietary antioxidants and magnesium reflects synergistic effects of these agents. ABR thresholds for this group of animals were lower post-noise, with no systematic group differences in threshold sensitivity prior to noise exposure. To verify that the group differences in noise-induced threshold shift were not a consequence of systematic group differences other than assigned treatment, threshold data used to calculate shift measures were also compared. Baseline ABR thresholds varied with frequency (F=427.011; df=1.763, 44.071; p<0.001) but were not reliably different based on side (right/left) (F=0.217; df=1, 25; p=0.645) and did not vary as a function of group (F=1.430; df=3, 25; p=0.258). There were no statistically reliable interactions for frequency x group, side x group, or frequency x side x group (all p’s > 0.15). In contrast, ABR thresholds measured 10-days post-noise varied with treatment group (F=15.383; df=1,25; p<0.001) as well as frequency (F=111.660; df=1.380, 34.506; p<0.001), and there were significant interactions of frequency x treatment (F=2.840; df=4.141, 34.506; p=0.037) and frequency x treatment x side (F=2.456; df=5.7, 47.499; p=0.40) but no reliable differences associated with side, or interactions of side x group or frequency x side (all p’s > 0.2). Adjusted pair-wise comparisons revealed that post-noise ABR thresholds were reliably lower in the group treated with vitamins A, C, E and magnesium compared to all other groups (all p’s<0.01). There were no statistically reliable differences between post-noise ABR thresholds in control animals and animals treated with either the antioxidant agents or magnesium (all p’s > 0.4). Additional analysis revealed that the threshold differences between animals treated with vitamins A, C, and magnesium, and the other groups, were statistically significant at all test frequencies (all p’s ≤ 0.011), with no effects of side and no interactions for side x group comparisons (all p’s >0.13).
Sensory cell death was observed primarily within the basal half of the cochlea, i.e., 10–20 mm from the apex of the cochlea (see Figure 2), which corresponds to frequencies of approximately 3 kHz and above [90]. Within the basal half of the cochlea, treatment-based differences in outer hair cell survival exceeded the standard criteria limiting the probability that observed results are due to chance alone to 5% or less (rows 1–3, p=0.111). However, statistical power for this between-group comparison was 0.511 and, thus, was not sufficient to detect mean differences of this magnitude given the observed variability (see error bars, figure 2). To reduce the number of pair-wise comparisons, and maintain statistical power, we compared outer hair cell loss in the 10–15 and 15–20 mm from the apex segments of the cochlea in the two groups most crucial to testing our hypothesis: saline and combination treatment with antioxidants and magnesium (see Figure 3). Loss of row 1 outer hair cells was location dependant (F=5.577; df=1, 13; p=0.034) and the reliability of group differences approached the accepted definition of statistical significance (F=4.134; df=1, 13; p=0.063). Loss of row 2 outer hair cells was more homogenous across segments (F=3.057; df=1, 13; p=0.104) and the group difference was statistically reliable (F=4.829; df=1, 13; p=0.047). Outer hair cell loss in row 3 was generally equivalent to row 2 (location: F=3.364; df=1, 13; p=0.090; treatment: F=4.550; df=1, 13; p=0.053). Both the mean and the median hair cell loss for all 24 of the 0.19 mm segments of the cochlea in which hair cells were counted were less for the combined treatment group than for the saline control group; the probability of this outcome occurring by chance is less than 0.0001. Taken together, the combination treatment appears to preserve not only threshold sensitivity but also hair cell survival.
Figure 2.
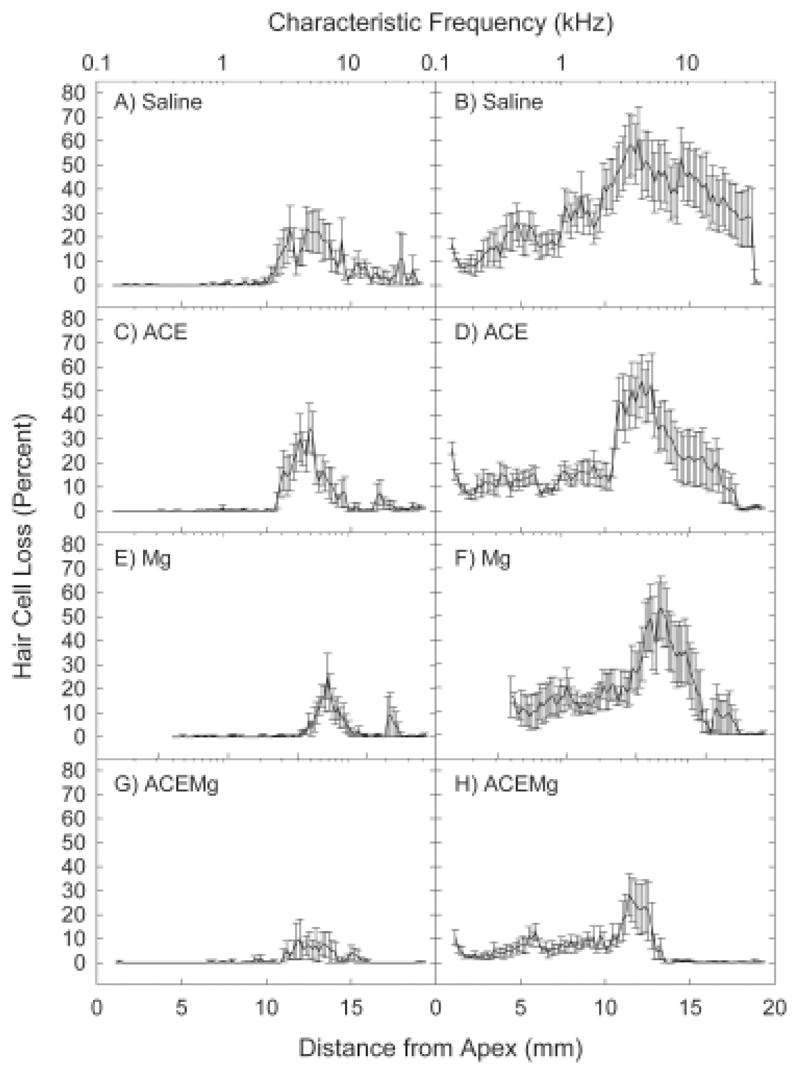
Sensory cell death was observed primarily within the basal half of the cochlea, i.e., 10–20 mm from the apex of the cochlea, and corresponding to frequencies of approximately 3 kHz and above [90] for both inner hair cells (A, C, E, and G) and outer hair cells (B, D, F, and H). Both the mean and the median hair cell loss for all 24 of the 0.19 mm segments of the cochlea in which hair cells were counted were less for the combined treatment group (ACEMg) than for the saline group; the probability of this outcome occurring by chance is less than 0.0001.
Figure 3.
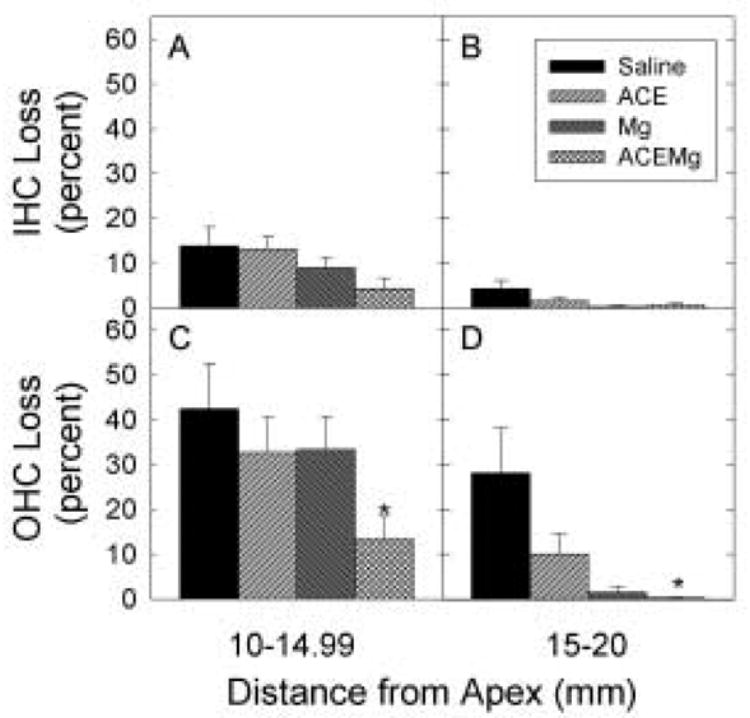
Percent inner (A, B) and outer (C, D) hair cell loss in the 10–15 and 15–20 mm from the apex segments of the cochlea is illustrated. Treatment with a combination of vitamins and magnesium reduced outer hair cell loss by approximately 30% in both the 10–15 mm region (Saline: 42%; ACEMg: 13%) and the 15–20 mm region (Saline: 28%; ACEMg: <1%). This protection was statistically reliable when group comparisons were limited (to reduce the number of pair-wise comparisons, and increase statistical power) to the two groups most crucial to testing our hypothesis: saline and combination treatment with antioxidants and magnesium.
Discussion
Synergistic Effects
Treatment with a combination of vitamins A, C, E, and magnesium, initiated 1 hour prior to noise exposure, produced a compelling reduction in NIHL and cell death. Effects of either the antioxidant agents (vitamins A, C, and E), or magnesium, were very small and not statistically reliable. Thus, the combination of agents was clearly more effective than any single category of agents. High oral doses of some nutrients can have adverse effects in some populations; for example, high-dose vitamin A is associated with an increased rate of lung cancer in smokers [91–93]. However, the Institute of Medicine’s Food and Nutrition Board has defined the highest level of daily nutrient intake that is likely to pose no risks of adverse health effects to almost all individuals in the healthy population (upper limit). The lack of significant adverse effects with long-term high-dose intake of several of these micronutrients has now been confirmed in several investigations [94, 95]. In addition, the US Food and Drug Administration has established recommended daily allowances that are safe for human use; these are readily available ‘over-the-counter’.
Mechanism of Action
We know that noise induces free radical formation; there is a nearly 4-fold increase in hydroxyl (OH) radicals within 1–2 hours of noise exposure [21], and a similarly significant early increase in superoxide (O2−) with reaction products evident at 5 min and 2 hours post noise [19]. There is also a significant late formation of ROS and RNS, occurring 7–10 days post noise [86]. We also know that noise induces lipid peroxidation; significant noise-induced lipid peroxidation and peroxynitrite (ONOO−) formation have been described 15–30 min post-noise [25, 96], and lipid peroxidation products increase in level from hour 1 to hour 5 of noise exposure [24]. We assume that pre-treatment with a variety of scavengers (including vitamins A, C, and E) reduced the early formation of free radicals that has been well characterized by Ohinata, Ohlemiller, Yamane, Nicotera, and their colleagues [19, 21, 24, 25, 96]. Daily treatment that continued through day 5 post-noise presumably reduced the late forming radicals revealed in the studies of Yamashita et al. [86]. Unlike most tissues, where increased metabolism increases blood flow to provide additional oxygen to stressed cells, reduced blood vessel diameter and red blood cell velocity [97, 98] and decreased blood flow [9, 10, 16, 26, 99] are observed in the cochlea post-noise. This noise-induced vasoconstriction is a direct consequence of noise-induced formation of 8-isoprostane-F2α, a vasoactive by-product of free radicals [46], and thus, antioxidant agents that reduce free radical formation may eliminate noise-induced vasoconstriction. The combination of vitamins A, C, and E, delivered one hour pre-noise presumably did not adequately preserve inner ear blood flow, as prevention of noise-induced deficits was achieved only by the combination of vitamins A, C, E, and magnesium.
Together, the agents used here scavenge singlet oxygen (Vitamin A), react with and reduce peroxyl radicals in cell membranes (Vitamin E), detoxify free radicals by reducing them in the aqueous phase (Vitamin C), and reduce noise-induced vasoconstriction (magnesium). Although there are well-characterized differences in the primary mechanism and site of action of these agents, there is also potential for overlap in their effects. Specifically, antioxidant scavenging of ROS may reduce the vasoconstriction that occurs with ROS production. In addition to well known effects of magnesium on blood flow, other biochemical mechanisms may further contribute to the protective effects of magnesium. For example, magnesium modulates calcium channel permeability, influx of calcium into cochlear hair cells, and glutamate release [100, 101], each of which may reduce NIHL. Regardless of the specific mechanism of action of vitamins A, C, and E, and magnesium, a combination of these agents clearly attenuates NIHL even when treatment is initiated very near the time of noise exposure (i.e., 1 hour prior to noise).
Individual Effects
Given that vitamins A, C, E, and magnesium, have been shown to attenuate NIHL and hearing loss from other stressors, it was somewhat surprising that protection was not observed for either the antioxidant (ACE) or magnesium treatments. Deficits were reduced to some small extent, but these effects were not statistically reliable. Stable plasma and tissue levels of vitamin C are obtained (in humans) approximately 3 weeks after beginning dietary treatment [102], and vitamin E levels similarly stabilize over an initial month-long window [for review, see 103]. Thus, it may be the case that dietary treatments must be provided on a daily basis for some longer period of time pre-noise for single-agent therapies to be effective. Indeed, the best protection against NIHL obtained with vitamin C [L--2-pascorbylolyphosphate, delivered as 5,000 mg per kg chow for 35 days, 60] included a 1-month pre-treatment protocol. Treatment initiated shortly before noise exposure failed to prevent cell death [500 mg/kg ascorbic acid, i.p., 48 h, 24 h, and 5 min prior to noise exposure, 104]. In contrast, vitamin E (alpha-tocopherol, 10–50 mg/kg, i.p.) reduced NIHL with treatment initiated 3 days pre-noise [50], and vitamin A reduced NIHL with treatment initiated two days pre-noise [all-trans retinoic acid, 1 mg/kg p.o., in sesame oil, 105].
Taken together, there may be differences in the pharmacodynamics of different agents, and protection may be observed with antioxidants or magnesium alone if treatment is initiated 48 hours (or longer) prior to noise exposure to produce higher serum levels of the agents in the bloodstream. Agents that are effective shortly post-treatment have greater clinical utility. While the demonstration of significant protection with vitamins A, C, E and magnesium, delivered within 1-hour of noise exposure, suggests a compelling new clinical strategy to prevent NIHL, several of the active agents were injected. Thus, it is possible that the most efficacious treatment window, assuming oral administration for all agents, will be somewhat longer than 1 hour pre-noise exposure. Indeed, treatments based on other antioxidant combinations are the most effective when treatment begins several days prior to noise exposure [85]. Although increasing total daily doses of single agents might reduce the need for lengthy pre-treatment, clinical strategies tested in human subjects must not exceed the upper limits established by the Institute of Medicine’s Food and Nutrition Board.
Efficacy of delayed treatment?
The compelling protection observed when treatment with vitamins A, C, and E, and magnesium was initiated shortly prior to noise exposure raises the possibility that treatments initiated post-noise may also be effective. Ohlemiller et al. [21] were among the first to suggest oxidative stress begins early and becomes substantial over time. Free radical insult that builds over time would explain hair cell death that continues up to 14 days post-noise [106]. Using 4-hydroxynonenal and nitryotyrosine, Yamashita et al. [86] confirmed that peak ROS and RNS production in cells in the organ of Corti occurs 7–10 days post-noise, and that noise-induced hair cell death is similarly delayed. As a consequence of delayed ROS production, ROS and RNS scavengers reduce NIHL not only when administered prior to noise, but also when treatment begins on days 1 or 3 post-noise [85]. Treatment delayed 5 days relative to noise insult was not effective, suggesting the therapeutic “window of opportunity” for successful antioxidant-based intervention against NIHL occurs within the first three days post-noise. Determining the efficacy of vitamins A, C, E, and magnesium when treatment is delayed relative to the noise insult is a compelling topic for future research.
Alternative Strategies
Multiple groups have pursued preservation of auditory function using glutathione (GSH) -based therapeutic strategies in recent years. Ebselen is a potent glutathione peroxidase (GPx) mimic; GPx catalyzes the anti-oxidant actions of GSH [107]. Pretreatment with ebselen reduces NIHL [108–111]. Given that one of the major determinants of GSH levels is bioavailable cysteine, and that cysteine can be derived from methionine, other studies have evaluated the potential for protection by pre-treatment with D-methionine [45]. D-methionine specifically alters the cochlear oxidative state; superoxide dismutase, catalase, GPx, and glutathione reductase levels all increase in animals treated with D-Methionine [112].
N-L-Acetylcysteine (L-NAC), which increases intracellular GSH and acts as a ROS scavenger, has also been used singly [47], and in combination with salicylate [113] to effectively attenuate NIHL. The utility of comparisons across investigations is compromised by variation in dose schedule and duration, species, and noise exposure. However, the significant reduction of NIHL (up to 35 dB at 16 kHz, with treatment initiated 1 hour prior to noise exposure), with vitamins A, C, E, and magnesium, suggests that this combination will prove to be one of the most effective therapies when different agents are compared under identical test conditions. Relatively greater efficacy could be explained by the combination of antioxidants reducing both ROS and RNS production, with additional protection as a consequence of magnesium-mediated reductions in noise-induced vasoconstriction and excitotoxicity.
Identifying treatments to reduce neurodegenerative disease
Mitochondrial dysfunction and ROS production have been implicated in many, if not all, neurodegenerative processes, and also in association with changes in local blood flow, such as stroke [28–36]. The use of antioxidant agents to prevent neurodegeneration has been widely described [32, 33, 37–42]. As in our investigation, combinations of antioxidants have been shown to be more effective than single agents in at least some cases [43]. Vasoregulators are also clearly of potential benefit given observations of reduced cortical blood flow in both Alzheimer’s and Parkinson’s disease [114–118]. This similarity of free radical production and reduced blood flow in neurodegenerative processes and NIHL, and the demonstrated efficacy of antioxidants plus magnesium in reducing sensory cell death and neurodegeneration in the cochlea, provides a compelling rationale for study of combinations of antioxidants, with and without magnesium, to ameliorate neurodegeneration from other stresses and disease processes, at other sites of the nervous system, of more heterogeneous origins, such as Alzheimer’s disease, Parkinson’s disease, and stroke.
Conclusions
NIHL is a significant clinical issue [for recent review, see 26]. We now know many of the molecular pathways leading to apoptotic cell death, which are triggered by noise and other environmentally mediated trauma such as aminoglycoside antibiotics and chemotherapeutics, as well as aging; and there is increasing evidence for their similarity. Free radical formation has been implicated in all. Interventions can be directed at preventing initial ROS formation and maintaining cochlear blood flow (as reported here and by others); alternative therapeutic interventions and strategies include neurotrophic growth factors [119–122], steroids [123, 124], calcineurin inhibitors [125, 126], caspase inhibitors [127–132], and Src protein tyrosine kinase (Src-PTK) inhibitors [133]. Each of these are at least partially effective in prevention of hearing loss and hair cell death; none of these strategies have alone been sufficiently effective. Given the various points of intervention along the cell death pathways, there are clearly an abundance of potential therapeutic targets. For clinical purposes, the most effective strategy may include targeting initiating events as well as early molecular processes, thus maintaining a cell in a relatively ‘normal’ physiological state. Although additional pre-clinical investigation is essential to the task of defining the most effective combination of agents, given that safe and effective interventions are now available, we should no longer delay the initiation of systematic human clinical trials to demonstrate the potential for pharmaceutical treatment of the inner ear in man.
Acknowledgments
Support for this research was provided by the National Institutes of Health (NIH-NIDCD R01 DC03820 and P30 DC005188), the General Motors Corporation/United Automotive Workers Union, and the Ruth and Lynn Townsend Professor of Communication Disorders. We thank Susan DeRemer, Chris Ellinger, Alice Mitchell, Diane Prieskorn, and Hollie Valentine for technical assistance.
Footnotes
Disclosure. Dr. Miller is a founding member and Chairman of the Board of Directors at OtoMedicine, Inc., a company that has an option to license the intellectual property described in this report. Dr. Le Prell is a paid consultant to OtoMedicine, Inc. Drs. Miller and Le Prell have disclosed these relationships to the Medical School Conflict of Interest Board at the University of Michigan; conflict management plans including semi-annual review by the Board are in place.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Spoendlin H. Primary structural changes in the organ of corti after acoustic overstimulation. Acta Otolaryngol (Stockh) 1971;71:166–176. doi: 10.3109/00016487109125346. [DOI] [PubMed] [Google Scholar]
- 2.Hawkins JE, Jr, Johnsson LG, Stebbins WC, Moody DB, Coombs SL. Hearing loss and cochlear pathology in monkeys after noise exposure. Acta Otolaryngol (Stockh) 1976;81:337–343. doi: 10.3109/00016487609119971. [DOI] [PubMed] [Google Scholar]
- 3.Hamernik RP, Henderson D. Impulse noise trauma. A study of histological susceptibility. Arch Otolaryngol. 1974;99:118–121. doi: 10.1001/archotol.1974.00780030124010. [DOI] [PubMed] [Google Scholar]
- 4.Hamernik RP, Henderson D, Crossley JJ, Salvi RJ. Interaction of continuous and impulse noise: Audiometric and histological effects. J Acoust Soc Am. 1974;55:117–121. doi: 10.1121/1.1928141. [DOI] [PubMed] [Google Scholar]
- 5.Hunter-Duvar IM, Bredberg G. Effects of intense auditory stimulation: Hearing losses and inner ear changes in the chinchilla. J Acoust Soc Am. 1974;55:795–801. doi: 10.1121/1.1914602. [DOI] [PubMed] [Google Scholar]
- 6.Hunter-Duvar IM, Elliott DN. Effects of intense auditory stimulation: Hearing losses and inner ear changes in the squirrel monkey. Ii. J Acoust Soc Am. 1973;54:1179–1183. doi: 10.1121/1.1914364. [DOI] [PubMed] [Google Scholar]
- 7.Hunter-Duvar IM, Elliott DN. Effects of intense auditory stimulation: Hearing losses and inner ear changes in the squirrel monkey. J Acoust Soc Am. 1972;52:1181–1192. doi: 10.1121/1.1913230. [DOI] [PubMed] [Google Scholar]
- 8.Mulroy MJ, Henry WR, McNeil PL. Noise-induced transient microlesions in the cell membranes of auditory hair cells. Hear Res. 1998;115:93–100. doi: 10.1016/s0378-5955(97)00181-0. [DOI] [PubMed] [Google Scholar]
- 9.Lipscomb DM, Roettger RL. Capillary constriction in cochlear and vestibular tissues during intense noise stimulation. Laryngoscope. 1973;83:259–263. doi: 10.1288/00005537-197302000-00008. [DOI] [PubMed] [Google Scholar]
- 10.Perlman HB, Kimura R. Cochlear blood flow and acoustic trauma. Acta Otolaryngol (Stockh) 1962;54:99–119. doi: 10.3109/00016486209126927. [DOI] [PubMed] [Google Scholar]
- 11.Hawkins JE., Jr The role of vasoconstriction in noise-induced hearing loss. Ann Otol Rhinol Laryngol. 1971;80:903–913. doi: 10.1177/000348947108000617. [DOI] [PubMed] [Google Scholar]
- 12.Hawkins JE, Jr, Johnsson LG, Preston RE. Cochlear microvasculature in normal and damaged ears. Laryngoscope. 1972;82:1091–1104. doi: 10.1288/00005537-197207000-00001. [DOI] [PubMed] [Google Scholar]
- 13.Scheibe F, Haupt H, Ludwig C. Intensity-related changes in cochlear blood flow in the guinea pig during and following acoustic exposure. Eur Arch Otorhinolaryngol. 1993;250:281–285. doi: 10.1007/BF00186226. [DOI] [PubMed] [Google Scholar]
- 14.Axelsson A, Dengerink H. The effects of noise on histological measures of the cochlear vasculature and red blood cells: A review. Hear Res. 1987;31:183–191. doi: 10.1016/0378-5955(87)90125-0. [DOI] [PubMed] [Google Scholar]
- 15.Axelsson A, Vertes D. Histological findings in cochlear vessels after noise. In: Hamernik R, Anderson D, Salvi R, editors. New perspectives in noise-induced hearing loss. New York: Raven; 1981. pp. 49–68. [Google Scholar]
- 16.Miller JM, Ren TY, Dengerink HA, Nuttall AL. Cochlear blood flow changes with short sound stimulation. In: Axelsson A, Borchgrevink HM, Hamernik RP, Hellstrom PA, Henderson D, Salvi RJ, editors. Scientific basis of noise-induced hearing loss. New York: Thieme Medical Publishers; 1996. pp. 95–109. [Google Scholar]
- 17.Duvall AJ, 3rd, Robinson KS. Local vs systemic effects of acoustic trauma on cochlear structure and transport. Arch Otolaryngol Head Neck Surg. 1987;113:1066–1071. doi: 10.1001/archotol.1987.01860100044019. [DOI] [PubMed] [Google Scholar]
- 18.Santi PA, Duvall AJ., 3rd Stria vascularis pathology and recovery following noise exposure. Otolaryngology. 1978 Mar-Apr;86 doi: 10.1177/019459987808600229. [DOI] [PubMed] [Google Scholar]
- 19.Yamane H, Nakai Y, Takayama M, Iguchi H, Nakagawa T, Kojima A. Appearance of free radicals in the guinea pig inner ear after noise-induced acoustic trauma. Eur Arch Otorhinolaryngol. 1995;252:504–508. doi: 10.1007/BF02114761. [DOI] [PubMed] [Google Scholar]
- 20.Ohlemiller KK, McFadden SL, Ding DL, Lear PM, Ho YS. Targeted mutation of the gene for cellular glutathione peroxidase (gpx1) increases noise-induced hearing loss in mice. J Assoc Res Otolaryngol. 2000;1:243–254. doi: 10.1007/s101620010043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ohlemiller KK, Wright JS, Dugan LL. Early elevation of cochlear reactive oxygen species following noise exposure. Audiol Neurootol. 1999;4:229–236. doi: 10.1159/000013846. [DOI] [PubMed] [Google Scholar]
- 22.McFadden SL, Ding DL, Ohlemiller KK, Salvi RJ. The role of superoxide dismutase in age-related and noise-induced hearing loss: Clues from sod1 knockout mice. In: Willot JF, editor. Handbook of mouse auditory research; from behavior to molecular biology. New York: CRC Press; 2001. pp. 489–504. [Google Scholar]
- 23.Ohlemiller KK, McFadden SL, Ding DL, Flood DG, Reaume AG, Hoffman EK, Scott RW, Wright JS, Putcha GV, Salvi RJ. Targeted deletion of the cytosolic cu/zn-superoxide dismutase gene (sod1) increases susceptibility to noise-induced hearing loss. Audiol Neurootol. 1999;4:237–246. doi: 10.1159/000013847. [DOI] [PubMed] [Google Scholar]
- 24.Ohinata Y, Miller JM, Altschuler RA, Schacht J. Intense noise induces formation of vasoactive lipid peroxidation products in the cochlea. Brain Res. 2000;878:163–173. doi: 10.1016/s0006-8993(00)02733-5. [DOI] [PubMed] [Google Scholar]
- 25.Henderson D, Bielefeld EC, Harris KC, Hu BH. The role of oxidative stress in noise-induced hearing loss. Ear Hear. 2006;27:1–19. doi: 10.1097/01.aud.0000191942.36672.f3. [DOI] [PubMed] [Google Scholar]
- 26.Miller JM, Yamashita D, Minami S, Yamasoba T, Le Prell CG. Mechanisms and prevention of noise-induced hearing loss. Otol Jpn. 2006;16:139–153. [Google Scholar]
- 27.Le Prell CG, Yamashita D, Minami S, Yamasoba T, Miller JM. Mechanisms of noise-induced hearing loss indicate multiple methods of prevention. Hear Res. 2006 doi: 10.1016/j.heares.2006.1010.1006. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moosmann B, Behl C. Antioxidants as treatment for neurodegenerative disorders. Expert Opinion on Investigational Drugs. 2002;11:1407–1435. doi: 10.1517/13543784.11.10.1407. [DOI] [PubMed] [Google Scholar]
- 29.Barnham KJ, Masters CL, Bush AI. Neurodegenerative diseases and oxidative stress. Nature Reviews Drug Discovery. 2004;3:205–214. doi: 10.1038/nrd1330. [DOI] [PubMed] [Google Scholar]
- 30.Vina J, Lloret A, Orti R, Alonso D. Molecular bases of the treatment of alzheimer's disease with antioxidants: Prevention of oxidative stress. Mol Aspects Med. 2004;25:117–123. doi: 10.1016/j.mam.2004.02.013. [DOI] [PubMed] [Google Scholar]
- 31.Stavrovskaya IG, Kristal BS. The powerhouse takes control of the cell: Is the mitochondrial permeability transition a viable therapeutic target against neuronal dysfunction and death? Free Radic Biol Med. 2005;38:687–697. doi: 10.1016/j.freeradbiomed.2004.11.032. [DOI] [PubMed] [Google Scholar]
- 32.Calabrese V, Lodi R, Tonon C, D'Agata V, Sapienza M, Scapagnini G, Mangiameli A, Pennisi G, Stella AM, Butterfield DA. Oxidative stress, mitochondrial dysfunction and cellular stress response in friedreich's ataxia. J Neurol Sci. 2005;233:145–162. doi: 10.1016/j.jns.2005.03.012. [DOI] [PubMed] [Google Scholar]
- 33.Mayo JC, Sainz RM, Tan DX, Antolin I, Rodriguez C, Reiter RJ. Melatonin and parkinson's disease. Endocrine. 2005;27:169–178. doi: 10.1385/ENDO:27:2:169. [DOI] [PubMed] [Google Scholar]
- 34.Beal MF. Mitochondria take center stage in aging and neurodegeneration. Ann Neurol. 2005;58:495–505. doi: 10.1002/ana.20624. [DOI] [PubMed] [Google Scholar]
- 35.Miquel J, Economos AC, Fleming J, Johnson JE., Jr Mitochondrial role in cell aging. Exp Gerontol. 1980;15:575–591. doi: 10.1016/0531-5565(80)90010-8. [DOI] [PubMed] [Google Scholar]
- 36.Casetta I, Govoni V, Granieri E. Oxidative stress, antioxidants and neurodegenerative diseases. Curr Pharm Des. 2005;11:2033–2052. doi: 10.2174/1381612054065729. [DOI] [PubMed] [Google Scholar]
- 37.Banaclocha MM. Therapeutic potential of n-acetylcysteine in age-related mitochondrial neurodegenerative diseases. Med Hypotheses. 2001;56:472–477. doi: 10.1054/mehy.2000.1194. [DOI] [PubMed] [Google Scholar]
- 38.Russo A, Palumbo M, Aliano C, Lempereur L, Scoto G, Renis M. Red wine micronutrients as protective agents in alzheimer-like induced insult. Life Sci. 2003;72:2369–2379. doi: 10.1016/s0024-3205(03)00123-1. [DOI] [PubMed] [Google Scholar]
- 39.Heo HJ, Lee CY. Protective effects of quercetin and vitamin c against oxidative stress-induced neurodegeneration. J Agric Food Chem. 2004;52:7514–7517. doi: 10.1021/jf049243r. [DOI] [PubMed] [Google Scholar]
- 40.Kontush K, Schekatolina S. Vitamin e in neurodegenerative disorders: Alzheimer's disease. Ann N Y Acad Sci. 2004;1031:249–262. doi: 10.1196/annals.1331.025. [DOI] [PubMed] [Google Scholar]
- 41.Tucker JM, Townsend DM. Alpha-tocopherol: Roles in prevention and therapy of human disease. Biomed Pharmacother. 2005;59:380–387. doi: 10.1016/j.biopha.2005.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Feng Z, Qin C, Chang Y, Zhang JT. Early melatonin supplementation alleviates oxidative stress in a transgenic mouse model of alzheimer's disease. Free Radic Biol Med. 2006;40:101–109. doi: 10.1016/j.freeradbiomed.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 43.Dhitavat S, Ortiz D, Rogers E, Rivera E, Shea TB. Folate, vitamin e, and acetyl-l-carnitine provide synergistic protection against oxidative stress resulting from exposure of human neuroblastoma cells to amyloid-beta. Brain Res. 2005;2:114–117. doi: 10.1016/j.brainres.2005.05.074. [DOI] [PubMed] [Google Scholar]
- 44.Yamasoba T, Nuttall AL, Harris C, Raphael Y, Miller JM. Role of glutathione in protection against noise-induced hearing loss. Brain Res. 1998;784:82–90. doi: 10.1016/s0006-8993(97)01156-6. [DOI] [PubMed] [Google Scholar]
- 45.Kopke RD, Coleman JK, Liu J, Campbell KC, Riffenburgh RH. Candidate's thesis: Enhancing intrinsic cochlear stress defenses to reduce noise-induced hearing loss. Laryngoscope. 2002;112:1515–1532. doi: 10.1097/00005537-200209000-00001. [DOI] [PubMed] [Google Scholar]
- 46.Miller JM, Brown JN, Schacht J. 8-iso-prostaglandin f(2alpha), a product of noise exposure, reduces inner ear blood flow. Audiol Neurootol. 2003;8:207–221. doi: 10.1159/000071061. [DOI] [PubMed] [Google Scholar]
- 47.Ohinata Y, Miller JM, Schacht J. Protection from noise-induced lipid peroxidation and hair cell loss in the cochlea. Brain Res. 2003;966:265–273. doi: 10.1016/s0006-8993(02)04205-1. [DOI] [PubMed] [Google Scholar]
- 48.Duan M, Qiu J, Laurell G, Olofsson A, Counter SA, Borg E. Dose and time-dependent protection of the antioxidant n-l-acetylcysteine against impulse noise trauma. Hear Res. 2004;192:1–9. doi: 10.1016/j.heares.2004.02.005. [DOI] [PubMed] [Google Scholar]
- 49.Hight NG, McFadden SL, Henderson D, Burkard RF, Nicotera T. Noise-induced hearing loss in chinchillas pre-treated with glutathione monoethylester and r-pia. Hear Res. 2003;179:21–32. doi: 10.1016/s0378-5955(03)00067-4. [DOI] [PubMed] [Google Scholar]
- 50.Hou F, Wang S, Zhai S, Hu Y, Yang W, He L. Effects of alpha-tocopherol on noise-induced hearing loss in guinea pigs. Hear Res. 2003;179:1–8. doi: 10.1016/s0378-5955(03)00065-0. [DOI] [PubMed] [Google Scholar]
- 51.Seidman M, Babu S, Tang W, Naem E, Quirk WS. Effects of resveratrol on acoustic trauma. Otolaryngol Head Neck Surg. 2003;129:463–470. doi: 10.1016/S0194-59980301586-9. [DOI] [PubMed] [Google Scholar]
- 52.Ohinata Y, Yamasoba T, Schacht J, Miller JM. Glutathione limits noise-induced hearing loss. Hear Res. 2000;146:28–34. doi: 10.1016/s0378-5955(00)00096-4. [DOI] [PubMed] [Google Scholar]
- 53.Cassandro E, Sequino L, Mondola P, Attanasio G, Barbara M, Filipo R. Effect of superoxide dismutase and allopurinol on impulse noise-exposed guinea pigs--electrophysiological and biochemical study. Acta Otolaryngol (Stockh) 2003;123:802–807. [PubMed] [Google Scholar]
- 54.Hu BH, Zheng XY, McFadden SL, Kopke RD, Henderson D. R-phenylisopropyladenosine attenuates noise-induced hearing loss in the chinchilla. Hear Res. 1997;113:198–206. doi: 10.1016/s0378-5955(97)00143-3. [DOI] [PubMed] [Google Scholar]
- 55.Seidman MD, Shivapuja BG, Quirk WS. The protective effects of allopurinol and superoxide dismutase on noise-induced cochlear damage. Otolaryngol Head Neck Surg. 1993;109:1052–1056. doi: 10.1177/019459989310900613. [DOI] [PubMed] [Google Scholar]
- 56.Quirk WS, Shivapuja BG, Schwimmer CL, Seidman MD. Lipid peroxidation inhibitor attenuates noise-induced temporary threshold shifts. Hear Res. 1994;74:217–220. doi: 10.1016/0378-5955(94)90189-9. [DOI] [PubMed] [Google Scholar]
- 57.Biesalski HK, Wellner U, Weiser H. Vitamin a deficiency increases noise susceptibility in guinea pigs. J Nutr. 1990;120:726–737. doi: 10.1093/jn/120.7.726. [DOI] [PubMed] [Google Scholar]
- 58.Rabinowitz PM, Pierce Wise J, Sr, Hur Mobo B, Antonucci PG, Powell C, Slade M. Antioxidant status and hearing function in noise-exposed workers. Hear Res. 2002;173:164–171. doi: 10.1016/s0378-5955(02)00350-7. [DOI] [PubMed] [Google Scholar]
- 59.Derekoy FS, Koken T, Yilmaz D, Kahraman A, Altuntas A. Effects of ascorbic acid on oxidative system and transient evoked otoacoustic emissions in rabbits exposed to noise. Laryngoscope. 2004;114:1775–1779. doi: 10.1097/00005537-200410000-00019. [DOI] [PubMed] [Google Scholar]
- 60.McFadden SL, Woo JM, Michalak N, Ding D. Dietary vitamin c supplementation reduces noise-induced hearing loss in guinea pigs. Hear Res. 2005;202:200–208. doi: 10.1016/j.heares.2004.10.011. [DOI] [PubMed] [Google Scholar]
- 61.Attias J, Weisz G, Almog S, Shahar A, Wiener M, Joachims Z, Netzer A, Ising H, Rebentisch E, Guenther T. Oral magnesium intake reduces permanent hearing loss induced by noise exposure. Am J Otolaryngol. 1994;15:26–32. doi: 10.1016/0196-0709(94)90036-1. [DOI] [PubMed] [Google Scholar]
- 62.Joachims Z, Netzer A, Ising H, Rebentisch E, Attias J, Weisz G, Gunther T. Oral magnesium supplementation as prophylaxis for noise-induced hearing loss: Results of a double blind field study. Schriftenr Ver Wasser Boden Lufthyg. 1993;88:503–516. [PubMed] [Google Scholar]
- 63.Attias J, Sapir S, Bresloff I, Reshef-Haran I, Ising H. Reduction in noise-induced temporary threshold shift in humans following oral magnesium intake. Clin Otolaryngol Allied Health Sci. 2004;29:635–641. doi: 10.1111/j.1365-2273.2004.00866.x. [DOI] [PubMed] [Google Scholar]
- 64.Attias J, Bresloff I, Haupt H, Scheibe F, Ising H. Preventing noise induced otoacoustic emission loss by increasing magnesium (mg2+) intake in guinea-pigs. J Basic Clin Physiol Pharmacol. 2003;14:119–136. doi: 10.1515/jbcpp.2003.14.2.119. [DOI] [PubMed] [Google Scholar]
- 65.Haupt H, Scheibe F, Mazurek B. Therapeutic efficacy of magnesium in acoustic trauma in the guinea pig. ORL J Otorhinolaryngol Relat Spec. 2003;65:134–139. doi: 10.1159/000072250. [DOI] [PubMed] [Google Scholar]
- 66.Scheibe F, Haupt H, Ising H, Cherny L. Therapeutic effect of parenteral magnesium on noise-induced hearing loss in the guinea pig. Magnes Res. 2002;15:27–36. [PubMed] [Google Scholar]
- 67.Haupt H, Scheibe F. Preventive magnesium supplement protects the inner ear against noise-induced impairment of blood flow and oxygenation in the guinea pig. Magnes Res. 2002;15:17–25. [PubMed] [Google Scholar]
- 68.Scheibe F, Haupt H, Ising H. Preventive effect of magnesium supplement on noise-induced hearing loss in the guinea pig. Eur Arch Otorhinolaryngol. 2000;257:10–16. doi: 10.1007/pl00007505. [DOI] [PubMed] [Google Scholar]
- 69.Joachims Z, Babisch W, Ising H, Gunther T, Handrock M. Dependence of noise-induced hearing loss upon perilymph magnesium concentration. J Acoust Soc Am. 1983;74:104–108. doi: 10.1121/1.389726. [DOI] [PubMed] [Google Scholar]
- 70.Schafer FQ, Wang HP, Kelley EE, Cueno KL, Martin SM, Buettner GR. Comparing beta-carotene, vitamin e and nitric oxide as membrane antioxidants. Biol Chem. 2002;383:671–681. doi: 10.1515/BC.2002.069. [DOI] [PubMed] [Google Scholar]
- 71.Burton GW, Joyce A, Ingold KU. Is vitamin e the only lipid-soluble, chain-breaking antioxidant in human blood plasma and erythrocyte membranes? Arch Biochem Biophys. 1983;221:281–290. doi: 10.1016/0003-9861(83)90145-5. [DOI] [PubMed] [Google Scholar]
- 72.Evans P, Halliwell B. Free radicals and hearing. Cause, consequence, and criteria. Ann N Y Acad Sci. 1999;884:19–40. doi: 10.1111/j.1749-6632.1999.tb08633.x. [DOI] [PubMed] [Google Scholar]
- 73.Niki E. Interaction of ascorbate and alpha-tocopherol. Ann N Y Acad Sci. 1987;498:186–199. doi: 10.1111/j.1749-6632.1987.tb23761.x. [DOI] [PubMed] [Google Scholar]
- 74.Niki E. Lipid antioxidants: How they may act in biological systems. Br J Cancer Suppl. 1987;8:153–157. [PMC free article] [PubMed] [Google Scholar]
- 75.Soares DG, Andreazza AC, Salvador M. Sequestering ability of butylated hydroxytoluene, propyl gallate, resveratrol, and vitamins c and e against abts, dpph, and hydroxyl free radicals in chemical and biological systems. J Agric Food Chem. 2003;51:1077–1080. doi: 10.1021/jf020864z. [DOI] [PubMed] [Google Scholar]
- 76.Niki E, Noguchi N, Tsuchihashi H, Gotoh N. Interaction among vitamin c, vitamin e, and beta-carotene. Am J Clin Nutr. 1995;62:1322S–1326S. doi: 10.1093/ajcn/62.6.1322S. [DOI] [PubMed] [Google Scholar]
- 77.Tsuchihashi H, Kigoshi M, Iwatsuki M, Niki E. Action of beta-carotene as an antioxidant against lipid peroxidation. Arch Biochem Biophys. 1995;323:137–147. doi: 10.1006/abbi.1995.0019. [DOI] [PubMed] [Google Scholar]
- 78.Niki E. Action of ascorbic acid as a scavenger of active and stable oxygen radicals. Am J Clin Nutr. 1991;54:1119S–1124S. doi: 10.1093/ajcn/54.6.1119s. [DOI] [PubMed] [Google Scholar]
- 79.Scheibe F, Haupt H, Vlastos GA. Preventive magnesium supplement reduces ischemia-induced hearing loss and blood viscosity in the guinea pig. Eur Arch Otorhinolaryngol. 2000;257:355–361. doi: 10.1007/s004050000252. [DOI] [PubMed] [Google Scholar]
- 80.Altura BH, Altura BM. Role of mg ions in contractility of blood vessels and skeletal muscles. Magnesium-B. 1981;1a:102–114. [Google Scholar]
- 81.Altura BM, Altura BT, Gebrewold A, Ising H, Gunther T. Noise-induced hypertension and magnesium in rats: Relationship to microcirculation and calcium. J Appl Physiol. 1992;72:194–202. doi: 10.1152/jappl.1992.72.1.194. [DOI] [PubMed] [Google Scholar]
- 82.Julicher RH, Sterrenberg L, Haenen GR, Bast A, Noordhoek J. Sex differences in the cellular defense system against free radicals from oxygen or drug metabolites in rat. Arch Toxicol. 1984;56:83–86. doi: 10.1007/BF00349076. [DOI] [PubMed] [Google Scholar]
- 83.El Barbary A, Altschuler RA, Schacht J. Glutathione s-transferases in the organ of corti of the rat: Enzymatic activity, subunit composition, and immunohistochemical localization. Hear Res. 1993;71:80–90. doi: 10.1016/0378-5955(93)90023-t. [DOI] [PubMed] [Google Scholar]
- 84.McFadden SL, Henselman LW, Zheng XY. Sex differences in auditory sensitivity of chinchillas before and after exposure to impulse noise. Ear Hear. 1999;20:164–174. doi: 10.1097/00003446-199904000-00007. [DOI] [PubMed] [Google Scholar]
- 85.Yamashita D, Jiang H-Y, Le Prell CG, Schacht J, Miller JM. Post-exposure treatment attenuates noise-induced hearing loss. Neuroscience. 2005;134:633–642. doi: 10.1016/j.neuroscience.2005.04.015. [DOI] [PubMed] [Google Scholar]
- 86.Yamashita D, Jiang H, Schacht J, Miller JM. Delayed production of free radicals following noise exposure. Brain Res. 2004;1019:201–209. doi: 10.1016/j.brainres.2004.05.104. [DOI] [PubMed] [Google Scholar]
- 87.Le Prell CG, Yagi M, Kawamoto K, Beyer LA, Atkin G, Raphael Y, Dolan DF, Bledsoe SCJ, Moody DB. Chronic excitotoxicity in the guinea pig cochlea induces temporary functional deficits without disrupting otoacoustic emissions. J Acoust Soc Am. 2004;116:1044–1056. doi: 10.1121/1.1772395. [DOI] [PubMed] [Google Scholar]
- 88.Puel JL, Saffiedine S, Gervais d'Aldin C, Eybalin M, Pujol R. Synaptic regeneration and functional recovery after excitotoxic injury in the guinea pig cochlea. C R Acad Sci III. 1995;318:67–75. [PubMed] [Google Scholar]
- 89.Raphael Y, Altschuler RA. Reorganization of cytoskeletal and junctional proteins during cochlear hair cell degeneration. Cell Motil Cytoskeleton. 1991;18:215–227. doi: 10.1002/cm.970180307. [DOI] [PubMed] [Google Scholar]
- 90.Tsuji J, Liberman MC. Intracellular labeling of auditory nerve fibers in guinea pig: Central and peripheral projections. J Comp Neurol. 1997;381:188–202. [PubMed] [Google Scholar]
- 91.The Alpha-Tocopherol Beta Carotene Cancer Prevention Study Group. The effect of vitamin e and beta carotene on the incidence of lung cancer and other cancers in male smokers. N Engl J Med. 1994;330:1029–1035. doi: 10.1056/NEJM199404143301501. [DOI] [PubMed] [Google Scholar]
- 92.Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Jr, Valanis B, Williams JH, Jr, Barnhart S, Cherniack MG, Brodkin CA, Hammar S. Risk factors for lung cancer and for intervention effects in caret, the beta-carotene and retinol efficacy trial. J Natl Cancer Inst. 1996;88:1550–1559. doi: 10.1093/jnci/88.21.1550. [DOI] [PubMed] [Google Scholar]
- 93.Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Valanis B, Williams JH, Barnhart S, Hammar S. Effects of a combination of beta carotene and vitamin a on lung cancer and cardiovascular disease. N Engl J Med. 1996;334:1150–1155. doi: 10.1056/NEJM199605023341802. [DOI] [PubMed] [Google Scholar]
- 94.Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins c and e, beta carotene, and zinc for age-related macular degeneration and vision loss: Areds report no. 8. Arch Ophthalmol. 2001;119:1417–1436. doi: 10.1001/archopht.119.10.1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Heart Protection Study Collaborative Group. Mrc/bhf heart protection study of antioxidant vitamin supplementation in 20,536 high-risk individuals: A randomised placebo-controlled trial. Lancet. 2002;360:23–33. [Google Scholar]
- 96.Nicotera TM, Henderson D, Zheng XY, Ding DL, McFadden SL. Reactive oxygen species, apoptosis, and necrosis in noise-exposed cochleas of chinchillas. Abs Assoc Res Otolaryngol. 1999;22:159. [Google Scholar]
- 97.Quirk WS, Avinash G, Nuttall AL, Miller JM. The influence of loud sound on red blood cell velocity and blood vessel diameter in the cochlea. Hear Res. 1992;63:102–107. doi: 10.1016/0378-5955(92)90079-3. [DOI] [PubMed] [Google Scholar]
- 98.Quirk WS, Seidman MD. Cochlear vascular changes in response to loud noise. Am J Otol. 1995;16:322–325. [PubMed] [Google Scholar]
- 99.Thorne PR, Nuttall AL. Laser doppler measurements of cochlear blood flow during loud sound exposure in the guinea pig. Hear Res. 1987;27:1–10. doi: 10.1016/0378-5955(87)90021-9. [DOI] [PubMed] [Google Scholar]
- 100.Cevette MJ, Vormann J, Franz K. Magnesium and hearing. J Am Acad Audiol. 2003;14:202–212. [PubMed] [Google Scholar]
- 101.Gunther T, Ising H, Joachims Z. Biochemical mechanisms affecting susceptibility to noise-induced hearing loss. Am J Otol. 1989;10:36–41. [PubMed] [Google Scholar]
- 102.Levine M, Conry-Cantilena C, Wang Y, Welch RW, Washko PW, Dhariwal KR, Park JB, Lazarev A, Graumlich JF, King J, Cantilena LR. Vitamin c pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc Natl Acad Sci USA. 1996;93:3704–3709. doi: 10.1073/pnas.93.8.3704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kappus H, Diplock AT. Tolerance and safety of vitamin e: A toxicological position report. Free Radic Biol Med. 1992;13:55–74. doi: 10.1016/0891-5849(92)90166-e. [DOI] [PubMed] [Google Scholar]
- 104.Branis M, Burda H. Effect of ascorbic acid on the numerical hair cell loss in noise exposed guinea pigs. Hear Res. 1988;33:137–140. doi: 10.1016/0378-5955(88)90026-3. [DOI] [PubMed] [Google Scholar]
- 105.Ahn JH, Kang HH, Kim YJ, Chung JW. Anti-apoptotic role of retinoic acid in the inner ear of noise-exposed mice. Biochem Biophys Res Commun. 2005;335:485–490. doi: 10.1016/j.bbrc.2005.07.114. [DOI] [PubMed] [Google Scholar]
- 106.Bohne BA, Harding GW, Nordmann AS, Tseng CJ, Liang GE, Bahadori RS. Survival-fixation of the cochlea: A technique for following time-dependent degeneration and repair in noise-exposed chinchillas. Hear Res. 1999;134:163–178. doi: 10.1016/s0378-5955(99)00082-9. [DOI] [PubMed] [Google Scholar]
- 107.Kil J, Pierce C, Tran H, Gu R, Lynch ED. Ebselen treatment reduces noise induced hearing loss via the mimicry and induction of glutathione peroxidase. Hear Res. 2006 doi: 10.1016/j.heares.2006.08.006. in press. [DOI] [PubMed] [Google Scholar]
- 108.Lynch ED, Kil J. Compounds for the prevention and treatment of noise-induced hearing loss. Drug Discov Today. 2005;10:1291–1298. doi: 10.1016/S1359-6446(05)03561-0. [DOI] [PubMed] [Google Scholar]
- 109.Lynch ED, Gu R, Pierce C, Kil J. Ebselen-mediated protection from single and repeated noise exposure in rat. Laryngoscope. 2004;114:333–337. doi: 10.1097/00005537-200402000-00029. [DOI] [PubMed] [Google Scholar]
- 110.Pourbakht A, Yamasoba T. Ebselen attenuates cochlear damage caused by acoustic trauma. Hear Res. 2003;181:100–108. doi: 10.1016/s0378-5955(03)00178-3. [DOI] [PubMed] [Google Scholar]
- 111.Yamasoba T, Pourbakht A, Sakamoto T, Suzuki M. Ebselen prevents noise-induced excitotoxicity and temporary threshold shift. Neurosci Lett. 2005;380:234–238. doi: 10.1016/j.neulet.2005.01.047. [DOI] [PubMed] [Google Scholar]
- 112.Campbell KC, Meech RP, Rybak LP, Hughes LF. The effect of d-methionine on cochlear oxidative state with and without cisplatin administration: Mechanisms of otoprotection. J Am Acad Audiol. 2003;14:144–156. [PubMed] [Google Scholar]
- 113.Kopke RD, Weisskopf PA, Boone JL, Jackson RL, Wester DC, Hoffer ME, Lambert DC, Charon CC, Ding DL, McBride D. Reduction of noise-induced hearing loss using l-nac and salicylate in the chinchilla. Hear Res. 2000;149:138–146. doi: 10.1016/s0378-5955(00)00176-3. [DOI] [PubMed] [Google Scholar]
- 114.Nakano S, Yamashita F, Matsuda H, Kodama C, Yamada T. Relationship between delusions and regional cerebral blood flow in alzheimer's disease. Dement Geriatr Cogn Disord. 2006;21:16–21. doi: 10.1159/000089215. [DOI] [PubMed] [Google Scholar]
- 115.Schreiber SJ, Doepp F, Spruth E, Kopp UA, Valdueza JM. Ultrasonographic measurement of cerebral blood flow, cerebral circulation time and cerebral blood volume in vascular and alzheimer's dementia. J Neurol. 2005;252:1171–1177. doi: 10.1007/s00415-005-0826-8. [DOI] [PubMed] [Google Scholar]
- 116.Girouard H, Iadecola C. Neurovascular coupling in the normal brain and in hypertension, stroke, and alzheimer disease. J Appl Physiol. 2006;100:328–335. doi: 10.1152/japplphysiol.00966.2005. [DOI] [PubMed] [Google Scholar]
- 117.Maalikjy Akkawi N, Borroni B, Agosti C, Magoni M, Broli M, Pezzini A, Padovani A. Volume cerebral blood flow reduction in pre-clinical stage of alzheimer disease: Evidence from an ultrasonographic study. J Neurol. 2005;252:559–563. doi: 10.1007/s00415-005-0689-z. [DOI] [PubMed] [Google Scholar]
- 118.Kasama S, Tachibana H, Kawabata K, Yoshikawa H. Cerebral blood flow in parkinson's disease, dementia with lewy bodies, and alzheimer's disease according to three-dimensional stereotactic surface projection imaging. Dement Geriatr Cogn Disord. 2005;19:266–275. doi: 10.1159/000084551. [DOI] [PubMed] [Google Scholar]
- 119.Sugahara K, Shimogori H, Yamashita H. The role of acidic fibroblast growth factor in recovery of acoustic trauma. Neuroreport. 2001;12:3299–3302. doi: 10.1097/00001756-200110290-00030. [DOI] [PubMed] [Google Scholar]
- 120.Ylikoski J, Pirvola U, Virkkala J, Suvanto P, Liang XQ, Magal E, Altschuler R, Miller JM, Saarma M. Guinea pig auditory neurons are protected by glial cell line-derived growth factor from degeneration after noise trauma. Hear Res. 1998;124:17–26. doi: 10.1016/s0378-5955(98)00095-1. [DOI] [PubMed] [Google Scholar]
- 121.Yamasoba T, Schacht J, Shoji F, Miller JM. Attenuation of cochlear damage from noise trauma by an iron chelator, a free radical scavenger and glial cell line-derived neurotrophic factor in vivo. Brain Res. 1999;815:317–325. doi: 10.1016/s0006-8993(98)01100-7. [DOI] [PubMed] [Google Scholar]
- 122.Shoji F, Miller AL, Mitchell A, Yamasoba T, Altschuler RA, Miller JM. Differential protective effects of neurotrophins in the attenuation of noise-induced hair cell loss. Hear Res. 2000;146:134–142. doi: 10.1016/s0378-5955(00)00106-4. [DOI] [PubMed] [Google Scholar]
- 123.Takemura K, Komeda M, Yagi M, Himeno C, Izumikawa M, Doi T, Kuriyama H, Miller JM, Yamashita T. Direct inner ear infusion of dexamethasone attenuates noise-induced trauma in guinea pig. Hear Res. 2004;196:58–68. doi: 10.1016/j.heares.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 124.Lamm K, Arnold W. The effect of prednisolone and non-steroidal anti-inflammatory agents on the normal and noise-damaged guinea pig inner ear. Hear Res. 1998;115:149–161. doi: 10.1016/s0378-5955(97)00186-x. [DOI] [PubMed] [Google Scholar]
- 125.Minami SB, Yamashita D, Schacht J, Miller JM. Calcineurin activation contributes to noise-induced hearing loss. J Neurosci Res. 2004;78:383–392. doi: 10.1002/jnr.20267. [DOI] [PubMed] [Google Scholar]
- 126.Uemaetomari I, Tabuchi K, Hoshino T, Hara A. Protective effect of calcineurin inhibitors on acoustic injury of the cochlea. Hear Res. 2005;209:86–90. doi: 10.1016/j.heares.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 127.Okuda T, Sugahara K, Takemoto T, Shimogori H, Yamashita H. Inhibition of caspases alleviates gentamicin-induced cochlear damage in guinea pigs. Auris Nasus Larynx. 2005;32:33–37. doi: 10.1016/j.anl.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 128.Nakagawa T, Kim TS, Murai N, Endo T, Iguchi F, Tateya I, Yamamoto N, Naito Y, Ito J. A novel technique for inducing local inner ear damage. Hear Res. 2003;176:122–127. doi: 10.1016/s0378-5955(02)00768-2. [DOI] [PubMed] [Google Scholar]
- 129.Cheng AG, Huang T, Stracher A, Kim A, Liu W, Malgrange B, Lefebvre PP, Schulman A, Van De Water TR. Calpain inhibitors protect auditory sensory cells from hypoxia and neurotrophin-withdrawal induced apoptosis. Brain Res. 1999;850:234–243. doi: 10.1016/s0006-8993(99)01983-6. [DOI] [PubMed] [Google Scholar]
- 130.Wei X, Zhao L, Liu J, Dodel RC, Farlow MR, Du Y. Minocycline prevents gentamicin-induced ototoxicity by inhibiting p38 map kinase phosphorylation and caspase 3 activation. Neuroscience. 2005;131:513–521. doi: 10.1016/j.neuroscience.2004.11.014. [DOI] [PubMed] [Google Scholar]
- 131.Corbacella E, Lanzoni I, Ding D, Previati M, Salvi R. Minocycline attenuates gentamicin induced hair cell loss in neonatal cochlear cultures. Hear Res. 2004;197:11–18. doi: 10.1016/j.heares.2004.03.012. [DOI] [PubMed] [Google Scholar]
- 132.Zhang M, Liu W, Ding D, Salvi R. Pifithrin-alpha suppresses p53 and protects cochlear and vestibular hair cells from cisplatin-induced apoptosis. Neuroscience. 2003;120:191–205. doi: 10.1016/s0306-4522(03)00286-0. [DOI] [PubMed] [Google Scholar]
- 133.Harris KC, Hu B, Hangauer D, Henderson D. Prevention of noise-induced hearing loss with src-ptk inhibitors. Hear Res. 2005;208:14–25. doi: 10.1016/j.heares.2005.04.009. [DOI] [PubMed] [Google Scholar]


