Abstract
Early infantile epileptic encephalopathy with suppression-burst pattern (EIEE) is one of the most severe and earliest forms of epilepsy, often evolving into West syndrome; however, the pathogenesis of EIEE remains unclear. ARX is a crucial gene for the development of interneurons in the fetal brain, and a polyalanine expansion mutation of ARX causes mental retardation and seizures, including those of West syndrome, in males. We screened the ARX mutation and found a hemizygous, de novo, 33-bp duplication in exon 2, 298_330dupGCGGCA(GCG)9, in two of three unrelated male patients with EIEE. This mutation is thought to expand the original 16 alanine residues to 27 alanine residues (A110_A111insAAAAAAAAAAA) in the first polyalanine tract of the ARX protein. Although EIEE is mainly associated with brain malformations, ARX is the first gene found to be responsible for idiopathic EIEE. Our observation that EIEE had a longer expansion of the polyalanine tract than is seen in West syndrome is consistent with the findings of earlier onset and more-severe phenotypes in EIEE than in West syndrome.
Early infantile epileptic encephalopathy with suppression-burst pattern (EIEE) is one of the most severe and earliest forms of epilepsy and has been known as “Ohtahara syndrome” since Ohtahara et al. first reported it in 1976.1 Frequent tonic seizures or spasms start mainly in the neonatal period, with a specific finding of electroencephalography (EEG)—namely, suppression-burst pattern characterized by high-voltage bursts alternating with almost flat suppression phases at an approximately regular rate.2 Cerebral dysgenesis—including hemimegalencephaly, Aicardi syndrome, and porencephaly—is often accompanied by EIEE; however, the etiology is heterogeneous, and cryptogenic or idiopathic EIEE has been reported only in a small number of patients. Familial occurrence of EIEE has been reported only in a family with Leigh encephalopathy.3 The transition from EIEE to West syndrome, which is characterized by tonic spasms with clustering, arrest of psychomotor development, and hypsarrhythmia on EEG, occurs in 75% of patients with EIEE,4 and a common pathological mechanism between them has been suggested. Recently, two novel genes, ARX and CDKL5, have been found to be responsible for X-linked West syndrome or infantile spasms (ISSX [MIM 308350]).5–8
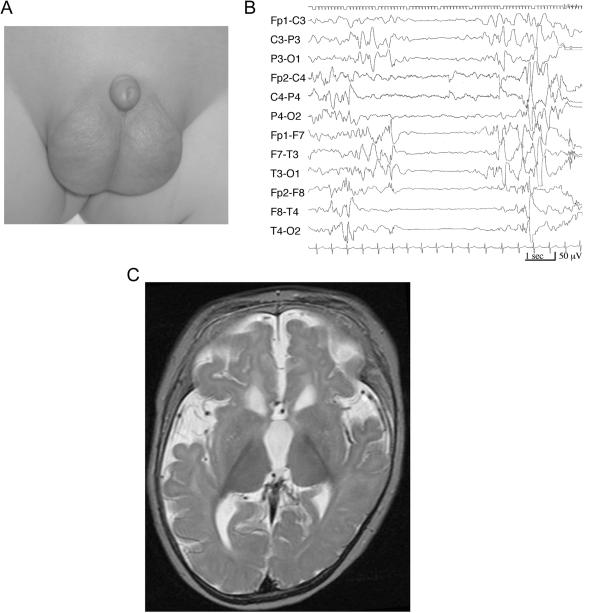
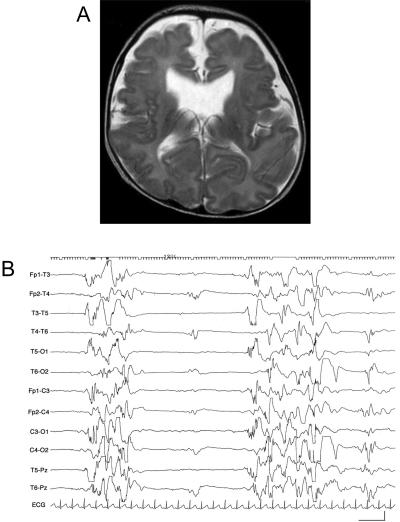
ARX, the aristaless-related homeobox gene (GenBank accession number NM_139058), is located in the human chromosome Xp22 region and as a transcription factor is essential for the development of cerebral interneurons.9 Mutation of ARX causes a wide variety of phenotypes, from severe brain malformations like hydranencephaly or lissencephaly lacking nonpyramidal or GABAergic interneurons to nonsyndromic mental retardation, with a strong correlation between genotype and phenotype.10,11 To identify the gene(s) underlying EIEE, we screened the ARX mutation in three male patients with EIEE defined by brief tonic seizures and a suppression-burst pattern on EEG with unknown etiology. Female patients were excluded, because even the null mutation of ARX causes less severe phenotypes than West syndrome.10 All patients showed a transition from EIEE to West syndrome and severe developmental delay. Clinical information regarding the patients in whom the ARX mutation was found was as follows: patient 1 was the first child of unrelated healthy parents. He was born normally at term without asphyxia. He suffered his first seizure 40 min after birth. His eyes deviated to the left with loss of consciousness, followed by brief clonic movement, predominantly of the left leg and arm. The seizure ceased after ∼1 min and recurred four times. Physical examination demonstrated a micropenis (fig. 1A). Findings from his blood and cerebrospinal fluid examinations were normal. A CT scan of his head also showed normal results. EEG examination demonstrated a suppression-burst pattern that was compatible with EIEE (fig. 1B). His seizure types shifted from (1) eye deviation followed by brief clonic movement to (2) brief tonic seizures and pedaling movements and then to (3) brief tonic spasms in clusters. He tended to cry with an opisthotonic posture. After 1 mo, EEG demonstrated a transition to hypsarrhythmia, suggesting West syndrome, but adrenocorticotropic hormone (ACTH) therapy was not effective. Combinations of multiple antiepileptic drugs, such as phenobarbital (PB), carbamazepine (CBZ), clobazam (CLB), valproic acid (VPA), vitamin B6, zonisamide (ZNS), and potassium bromide, failed to stop his seizures, and his development was profoundly delayed. He had not acquired eye contact or head control by the age of 18 mo. Consecutive brain magnetic resonance imaging (MRI) revealed progressive brain atrophy (fig. 1C). Patient 2 was the second child of unrelated healthy parents, and his elder sister was healthy. He was delivered spontaneously at term, without asphyxia. After birth, he showed blinking of the left eyelid and abrupt flexion of the right arm and left leg, suggesting seizures. At the age of 1 wk, he had brief tonic seizures in clusters, with a suppression-burst pattern on EEG (fig. 2B). His physical findings were normal except for a small penis at the age of 1 mo. His routine blood tests showed normal results except for a mild elevation in liver transaminase (aspartate aminotransferase [AST] 82 IU/liter, alanine aminotransferase [ALT] 34 IU/liter). His brain MRI scan demonstrated a defect of the cavum septi pellucidi and hypoplastic corpus callosum (fig. 2A). VPA, ZNS, PB, and clonazepam were given, but his EEG at the age of 7 mo showed transformation to hypsarrhythmia. With a diagnosis of West syndrome, he received ACTH injections, with no effect. His development was severely delayed.
Figure 1. .
A, Photograph of patient 1's external genitalia, showing a small penis with an apparently normal scrotum. B, Interictal EEG of patient 1, showing an asynchronous pattern, with high-voltage slow wave bursts mixed with spikes and sharp waves, and a low-voltage suppression background. C, Brain MRI of patient 1, demonstrating lateral ventricular dilatation with a frontal dominancy, suggesting a small caudate nucleus, thin corpus callosum (not shown), and delayed myelination in the white matter.
Figure 2. .
A, Brain MRI of patient 2 at the age of 8 WK shows dilatation of the anterior horns of the lateral ventricle and apparently normal gyri. B, Interictal EEG of patient 2 shows a marked depression of background activity, intermixed with asymmetric paroxysmal bursts.
Blood samples were collected from three patients with sporadic EIEE and from their parents, after both informed consent from the parents and approval from the Yamagata University School of Medicine Institutional Review Board were obtained. Genomic DNA was extracted according to standard procedures and was amplified for all coding exons and flanking introns of ARX. The primers and PCR conditions have been described elsewhere.10 Direct sequence analysis of the PCR product of the first half of exon 2 was performed on an automated DNA sequencer (ABI 310 [Applied Biosystems]). Other products were analyzed by denaturing high-performance liquid chromatography (DHPLC) performed using the Transgenomic WAVE system (Transgenomic).
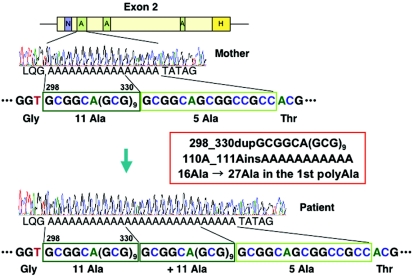
A hemizygous 33-bp duplication in exon 2, 298_330dupGCGGCA(GCG)9, which is thought to expand the original 16 alanine residues to 27 alanine residues (A110_A111insAAAAAAAAAAA) in the first polyalanine tract of the ARX protein, was found in patient 1 (fig. 3). Interestingly, the same mutation of the ARX gene was also found in patient 2. No mutation was found in their parents, suggesting that these mutations are in fact de novo. The mothers, however, can still be gonadal mosaics, because gonadal or somatic mosaicism was documented for the ARX gene.12 Mutation screening of ARX showed normal results in another patient.
Figure 3. .
The structure of exon 2 of the ARX gene and representative electropherograms in patient 1 and his parents. Both parents have the wild-type sequence encoding 16 alanine residues, whereas the patient has a hemizygous mutant sequence with a duplication of 33 bp, which encodes 11 alanine residues and results in an expanded repeat of 27 alanine residues. Polyalanine tract (A), nuclear localization sequence (N), and homeobox (H) are illustrated in the structure of exon 2.9
ARX comprises five exons encoding a protein of 562 aa, including a paired-class homeodomain and four repeats of 7–16 alanine residues called “polyalanine tracts.”5,9 In nature, the maximum length of alanine repeats is 20.13 Mutation of the second polyalanine tract with eight expanded alanine residues, which is the most common mutation in ARX, results in nonsyndromic mental retardation; Partington syndrome (PRTS [MIM 309510]), which is characterized by mental retardation and dystonic movements of the hands; and West syndrome or infantile spasms in males.6,14 An expansion of 1–3 alanine residues in the first polyalanine tract containing the original 16 alanine residues has been reported in patients with mental retardation, though the expansion of 1 alanine residue has been found in 1 of 188 control males.15,16 An expansion of seven alanine residues in the first polyalanine tract causes West syndrome more severe than that caused by the second polyalanine tract expansion mutation of the ARX gene.14,17 So far, nine genes with expansion of the polyalanine tract have been found to be responsible for human diseases, and clinical observations demonstrate a correlation between the length of the repeat and the severity of the clinical phenotype and penetrance in HOXD13 for synpolydactyly (SPD1 [MIM 186000]),18 PABPN1 for oculopharyngeal muscular dystrophy (OPMD [MIM 164300]),19 and PHOX2B for congenital central hypoventilation syndrome (CCHS [MIM 209880]).20 Longer expansion of the polyalanine tract causes a more severe phenotype, as well as other diseases caused by triplet repeat expansion, such as fragile X syndrome (MIM 300624) by FMR1 mutation, Huntington disease (HD [MIM 143100]) by IT15 mutation, and so on.21 As seen in both of our patients, the onset of epileptic seizures in EIEE is usually within the 1st mo after birth, which is distinctly earlier than 3–9 mo of age as seen in West syndrome, whereas the development outcome and life expectancy in EIEE are worse than those in West syndrome.4 Our observation that EIEE had longer expansion of the polyalanine tract than West syndrome may explain why EIEE starts earlier and is more severe than West syndrome.
Although the function of polyalanine tracts remains unknown, repeats of several alanine residues are frequent in transcription factors and other proteins with nuclear localization.13 Extended alanine repeats of Hoxd13, Hoxa13, Runx2, and Sox3 spontaneously form aggregates in the cytoplasm.22 The smallest alanine-repeat expansion (+7) of Hoxd13 was predominantly located in the nucleus and was well tolerated by the cells, whereas long expansions (⩾+10 alanine residues) were cytotoxic and mainly located in the cytoplasm.23 Therefore, the size of the alanine expansions correlates with the severity of the cytoplasmic aggregation. In vitro transfection experiments of the polyalanine-expanded constructs of Arx cause nuclear aggregation, filamentous nuclear inclusions, and an increase in cell death, whereas cortical neurons transfected with expanded polyalanine using whole-brain electroporation form neuronal nuclear inclusions in vivo.24 Recently, the human ARX protein was shown to be a potent transcriptional repressor.25 Although the domain encompassing polyalanine tracts 1 and 2 of ARX seems not to be significantly contributing to the repression activity, the expansions of either polyalanine tract 1 or 2 enhance transcriptional repression activity in a manner dependent on the length of the alanine expansion.25 These experimental data suggest that the expansion of ARX polyalanine tracts is harmful to neurons in a size-dependent manner.
Phenotypes caused by the ARX mutation are largely classified into two groups, a malformation group and a nonmalformation group.10 Premature termination mutations and missense mutations within the homeobox of the ARX gene mainly cause malformation of the brain and external genitalia, such as X-linked lissencephaly with abnormal genitalia or hydranencephaly with abnormal genitalia (XLAG [MIM 300215]) and Proud syndrome (MIM 300004), which consists of X-linked mental retardation, agenesis of the corpus callosum, and abnormal genitalia.10 Missense mutations outside the homeobox and expansions of polyalanine tracts lead to the nonmalformation group, as mentioned above.5,15 No apparent brain or genital malformations have been reported in patients with expansion of the polyalanine tracts, except for a boy showing transsphenoidal encephalocele and agenesis of the corpus callosum in a family affected with X-linked mental retardation26; however, both our patients with an expansion of 11 alanine residues in the first polyalanine tract showed micropenis. Among patients with XLAG, missense mutations within the homeobox of ARX lead to less-severe genital malformation than premature termination mutations, which are thought to cause loss of function.10 Another patient without the ARX mutation in this report did not have genital malformation. Similar genital findings between a malformation group and EIEE caused by the longest expansion of the polyalanine tract so far reported in ARX bridges the gap between the malformation and nonmalformation groups and suggests a common pathological mechanism between them.
Suppression-burst pattern has also been reported in a patient with XLAG caused by a premature termination mutation in ARX.27 The neuropathology of XLAG and Arx-knockout mice indicates a defect in GABAergic interneurons.9,11 Interestingly, an absence of GABA in the cerebrospinal fluid has been reported in a girl with EIEE and migrational disorder.28 We hypothesize that an error of GABAergic system is critical to the suppression-burst pattern on EEG in patients with EIEE via dysfunction of interneurons due to the ARX mutation. EIEE could be designated “interneuronopathy” as well as West syndrome.7
The sequence of polyalanine expansions we found is an imperfect repeat of GCG including a triplet of GCA, 298_330dupGCGGCA(GCG)9. The first 11 bp (GCGGCAGCGGC) recurrently appear just after the sequence of duplication from nucleotides 331 to 341. This suggests a predisposition to recombination between the repeats and that unequal crossing-over could be a mutational mechanism of the expansion, as suggested for the second polyalanine expansion of ARX and other polyalanine expansions.6,29
Knowledge of the genetic background of epilepsy is expanding, and, to our knowledge, this is the first report to reveal the genetic cause of EIEE. Mutation screening of ARX should be considered in patients with EIEE and in other patients showing suppression burst on EEG or infantile seizures with developmental delay.
Acknowledgments
This study was supported by research grants 18A-5 and 17A-11 for Nervous and Mental Disorders from the Ministry of Health, Labor, and Welfare of Japan (to M.K.). We thank Keiko Tanaka for technical assistance.
Web Resources
Accession numbers and URLs for data presented herein are as follows:
- GenBank, http://www.ncbi.nlm.nih.gov/Genbank/(for ARX [accession number NM_139058])
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/Omim/ (for ISSX, PRTS, SPD1, OPMD, CCHS, fragile X syndrome, HD, XLAG, Proud syndrome)
References
- 1.Ohtahara S, Ishida T, Oka E, Yamatogi Y, Inoue H, Karita S, Ohtsuka Y (1976) [On the specific age dependent epileptic syndrome: the early-infantile epileptic encephalopathy with suppression-burst.] No to Hattatsu 8:270–279 [Google Scholar]
- 2.Ohtahara S, Yamatogi Y (2006) Ohtahara syndrome: with special reference to its developmental aspects for differentiating from early myoclonic encephalopathy. Epilepsy Res Suppl 70:S58–S67 10.1016/j.eplepsyres.2005.11.021 [DOI] [PubMed] [Google Scholar]
- 3.Miyake S, Yamashita S, Yamada M, Iwamoto H (1987) [Therapeutic effect of ACTH and gamma-globulin in 8 cases with the early-infantile epileptic encephalopathy with suppression-burst (EIEE).] Shonika Rinsho (Tokyo) 40:1681–1688 [Google Scholar]
- 4.Yamatogi Y, Ohtahara S (2002) Early-infantile epileptic encephalopathy with suppression-bursts, Ohtahara syndrome; its overview referring to our 16 cases. Brain Dev 24:13–23 10.1016/S0387-7604(01)00392-8 [DOI] [PubMed] [Google Scholar]
- 5.Stromme P, Mangelsdorf ME, Shaw MA, Lower KM, Lewis SM, Bruyere H, Lutcherath V, Gedeon AK, Wallace RH, Scheffer IE, et al (2002) Mutations in the human ortholog of Aristaless cause X-linked mental retardation and epilepsy. Nat Genet 30:441–445 10.1038/ng862 [DOI] [PubMed] [Google Scholar]
- 6.Kato M, Das S, Petras K, Sawaishi Y, Dobyns WB (2003) Polyalanine expansion of ARX associated with cryptogenic West syndrome. Neurology 61:267–276 [DOI] [PubMed] [Google Scholar]
- 7.Kato M (2006) A new paradigm for West syndrome based on molecular and cell biology. Epilepsy Res Suppl 70:S87–S95 10.1016/j.eplepsyres.2006.02.008 [DOI] [PubMed] [Google Scholar]
- 8.Kalscheuer VM, Tao J, Donnelly A, Hollway G, Schwinger E, Kubart S, Menzel C, Hoeltzenbein M, Tommerup N, Eyre H, et al (2003) Disruption of the serine/threonine kinase 9 gene causes severe X-linked infantile spasms and mental retardation. Am J Hum Genet 72:1401–1411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kitamura K, Yanazawa M, Sugiyama N, Miura H, Iizuka-Kogo A, Kusaka M, Omichi K, Suzuki R, Kato-Fukui Y, Kamiirisa K, et al (2002) Mutation of ARX causes abnormal development of forebrain and testes in mice and X-linked lissencephaly with abnormal genitalia in humans. Nat Genet 32:359–369 10.1038/ng1009 [DOI] [PubMed] [Google Scholar]
- 10.Kato M, Das S, Petras K, Kitamura K, Morohashi K, Abuelo DN, Barr M, Bonneau D, Brady AF, Carpenter NJ, et al (2004) Mutations of ARX are associated with striking pleiotropy and consistent genotype-phenotype correlation. Hum Mutat 23:147–159 10.1002/humu.10310 [DOI] [PubMed] [Google Scholar]
- 11.Bonneau D, Toutain A, Laquerriere A, Marret S, Saugier-Veber P, Barthez MA, Radi S, Biran-Mucignat V, Rodriguez D, Gelot A (2002) X-linked lissencephaly with absent corpus callosum and ambiguous genitalia (XLAG): clinical, magnetic resonance imaging, and neuropathological findings. Ann Neurol 51:340–349 10.1002/ana.10119 [DOI] [PubMed] [Google Scholar]
- 12.Gecz J, Cloosterman D, Partington M (2006) ARX: a gene for all seasons. Curr Opin Genet Dev 16:308–316 10.1016/j.gde.2006.04.003 [DOI] [PubMed] [Google Scholar]
- 13.Lavoie H, Debeane F, Trinh QD, Turcotte JF, Corbeil-Girard LP, Dicaire MJ, Saint-Denis A, Page M, Rouleau GA, Brais B (2003) Polymorphism, shared functions and convergent evolution of genes with sequences coding for polyalanine domains. Hum Mol Genet 12:2967–2979 10.1093/hmg/ddg329 [DOI] [PubMed] [Google Scholar]
- 14.Stromme P, Mangelsdorf ME, Scheffer IE, Gecz J (2002) Infantile spasms, dystonia, and other X-linked phenotypes caused by mutations in Aristaless related homeobox gene, ARX. Brain Dev 24:266–268 10.1016/S0387-7604(02)00079-7 [DOI] [PubMed] [Google Scholar]
- 15.Bienvenu T, Poirier K, Friocourt G, Bahi N, Beaumont D, Fauchereau F, Ben Jeema L, Zemni R, Vinet MC, Francis F, et al (2002) ARX, a novel Prd-class-homeobox gene highly expressed in the telencephalon, is mutated in X-linked mental retardation. Hum Mol Genet 11:981–991 10.1093/hmg/11.8.981 [DOI] [PubMed] [Google Scholar]
- 16.Gronskov K, Hjalgrim H, Nielsen IM, Brondum-Nielsen K (2004) Screening of the ARX gene in 682 retarded males. Eur J Hum Genet 12:701–705 10.1038/sj.ejhg.5201222 [DOI] [PubMed] [Google Scholar]
- 17.Wohlrab G, Uyanik G, Gross C, Hehr U, Winkler J, Schmitt B, Boltshauser E (2005) Familial West syndrome and dystonia caused by an Aristaless related homeobox gene mutation. Eur J Pediatr 164:326–328 10.1007/s00431-005-1622-2 [DOI] [PubMed] [Google Scholar]
- 18.Goodman FR, Mundlos S, Muragaki Y, Donnai D, Giovannucci-Uzielli ML, Lapi E, Majewski F, McGaughran J, McKeown C, Reardon W, et al (1997) Synpolydactyly phenotypes correlate with size of expansions in HOXD13 polyalanine tract. Proc Natl Acad Sci USA 94:7458–7463 10.1073/pnas.94.14.7458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hill ME, Creed GA, McMullan TF, Tyers AG, Hilton-Jones D, Robinson DO, Hammans SR (2001) Oculopharyngeal muscular dystrophy: phenotypic and genotypic studies in a UK population. Brain 124:522–526 10.1093/brain/124.3.522 [DOI] [PubMed] [Google Scholar]
- 20.Trochet D, Hong SJ, Lim JK, Brunet JF, Munnich A, Kim KS, Lyonnet S, Goridis C, Amiel J (2005) Molecular consequences of PHOX2B missense, frameshift and alanine expansion mutations leading to autonomic dysfunction. Hum Mol Genet 14:3697–3708 10.1093/hmg/ddi401 [DOI] [PubMed] [Google Scholar]
- 21.Di Prospero NA, Fischbeck KH (2005) Therapeutics development for triplet repeat expansion diseases. Nat Rev Genet 6:756–765 10.1038/nrg1690 [DOI] [PubMed] [Google Scholar]
- 22.Albrecht A, Mundlos S (2005) The other trinucleotide repeat: polyalanine expansion disorders. Curr Opin Genet Dev 15:285–293 10.1016/j.gde.2005.04.003 [DOI] [PubMed] [Google Scholar]
- 23.Albrecht AN, Kornak U, Boddrich A, Suring K, Robinson PN, Stiege AC, Lurz R, Stricker S, Wanker EE, Mundlos S (2004) A molecular pathogenesis for transcription factor associated poly-alanine tract expansions. Hum Mol Genet 13:2351–2359 10.1093/hmg/ddh277 [DOI] [PubMed] [Google Scholar]
- 24.Nasrallah IM, Minarcik JC, Golden JA (2004) A polyalanine tract expansion in Arx forms intranuclear inclusions and results in increased cell death. J Cell Biol 167:411–416 10.1083/jcb.200408091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McKenzie O, Ponte I, Mangelsdorf M, Finnis M, Colasante G, Shoubridge C, Stifani S, Gécz J, Broccoli V (2007) Aristaless-related homeobox gene, the gene responsible for West syndrome and related disorders, is a Groucho/transducin-like enhancer of split dependent transcriptional repressor. Neuroscience 146:236–247 10.1016/j.neuroscience.2007.01.038 [DOI] [PubMed] [Google Scholar]
- 26.Van Esch H, Poirier K, de Zegher F, Holvoet M, Bienvenu T, Chelly J, Devriendt K, Fryns JP (2004) ARX mutation in a boy with transsphenoidal encephalocele and hypopituitarism. Clin Genet 65:503–505 10.1111/j.1399-0004.2004.00256.x [DOI] [PubMed] [Google Scholar]
- 27.Kato M, Dobyns WB (2005) X-linked lissencephaly with abnormal genitalia as a tangential migration disorder causing intractable epilepsy: proposal for a new term, “interneuronopathy.” J Child Neurol 20:392–397 [DOI] [PubMed] [Google Scholar]
- 28.Miller SP, Dilenge ME, Meagher-Villemure K, O’Gorman AM, Shevell MI (1998) Infantile epileptic encephalopathy (Ohtahara syndrome) and migrational disorder. Pediatr Neurol 19:50–54 10.1016/S0887-8994(98)00009-5 [DOI] [PubMed] [Google Scholar]
- 29.Brown LY, Brown SA (2004) Alanine tracts: the expanding story of human illness and trinucleotide repeats. Trends Genet 20:51–58 10.1016/j.tig.2003.11.002 [DOI] [PubMed] [Google Scholar]