Abstract
The highly related transcription factors Sox4 and Sox11 are both expressed in oligodendrocyte precursors. Yet whether they have a function in oligodendrocyte development is unknown. By overexpressing Sox4 under the control of 3.1 kb of 5′ flanking sequences of the myelin basic protein gene in transgenic mice, we extended Sox4 expression in the oligodendrocyte lineage from oligodendrocyte precursors to cells undergoing terminal differentiation. As a consequence of transgene expression, mice develop the full spectrum of phenotypic traits associated with a severe hypomyelination during the first postnatal weeks. Myelin gene expression was severely reduced, and myelin dramatically thinned in several central nervous system (CNS) regions. Despite these disturbances in CNS myelination, the number of oligodendrocytic cells remained unaltered. Considering that apoptosis rates were normal and proliferation only slightly increased, oligodendrocytes likely persist in a premyelinating to early myelinating state. This shows that prolonged Sox4 expression in cells of the oligodendrocyte lineage is incompatible with the acquisition of a fully mature phenotype and argues that the presence of Sox4, and possibly Sox11, in oligodendrocyte precursors may normally prevent premature differentiation.
Oligodendrocytes are the myelin-forming glial cells of the vertebrate central nervous system (CNS). They express several Sox transcription factors during their development. This includes Sox8, Sox9, and Sox10 as members of group E; Sox5 and Sox6 as members of group D; Sox4 and Sox11 as members of group C; and Sox17 as a member of group F (12, 19, 21-24).
Several of these Sox proteins have been shown to be important for defined aspects of oligodendrocyte development in vivo. Sox9, for instance, is needed for the specification of oligodendrocyte precursors, whereas Sox10 is required for terminal differentiation, at least partially by activating several myelin genes (2, 16, 22, 23). The least amount of information is known about the function of the group C proteins Sox4 and Sox11, which in primary cultures are transiently expressed in oligodendrocyte precursors and shut off before terminal differentiation (12).
Taking into account that Sox4 and Sox11 are closely related and exhibit significant similarity on the amino acid level, both proteins are expected to have similar biochemical activities and therefore be able to compensate for each other's loss in tissues where they are coexpressed. The CNS is such a tissue of largely overlapping Sox4 and Sox11 expression. Accordingly, neither Sox4-deficient embryos nor Sox11-deficient embryos exhibit any overt defect in CNS development up until their time of death (5, 18). Group C Sox protein function in the oligodendrocyte lineage may thus only be amenable by loss-of-function studies in mice that are deficient for both Sox4 and Sox11. Unfortunately, constitutive double-deficient mouse embryos cannot be obtained because of lethality of the compound heterozygotes (E. Sock and M. Wegner, unpublished observations).
We therefore decided to address the role of group C Sox proteins by gain-of-function studies in which we prolonged Sox4 expression in the oligodendrocyte lineage using an MBP-Sox4 transgene. The 5′ flanking region of the myelin basic protein (MBP) gene spanning positions −3109 to +31 was successfully used in the past to drive transgene expression during the first postnatal weeks in terminally differentiating oligodendrocytes. It is, however, not sufficient to maintain expression in already differentiated oligodendrocytes in older animals (9).
Mice which expressed the MBP-Sox4 transgene developed a phenotype typical of mouse myelin mutants and exhibited significant hypomyelination. We conclude that expression of the group C protein Sox4 needs to be downregulated for oligodendrocytes to undergo full maturation. Sox4 and likely Sox11 therefore appear to be involved in keeping cells of the oligodendrocyte lineage in an immature state and timing the onset of oligodendrocyte differentiation.
MATERIALS AND METHODS
Construction of transgene, generation of transgenic animals, and determination of transgene copy numbers.
A 3.1-kb DNA fragment spanning positions −3109 to +31 of the mouse MBP promoter (gift of A. Roach, Samuel Lunenfeld Research Institute, Toronto, Canada) and the internal ribosome entry site 2 (IRES2)-enhanced green fluorescent protein (EGFP)-poly(A) cassette from pIRES2-EGFP (Clontech, Heidelberg, Germany) were successively inserted between two tandemly arranged H19 insulators in the context of a pWHERE plasmid backbone (Invivogen, San Diego, CA), before 2 kb of rat Sox4 sequences including the complete open reading frame was placed as an EcoRI fragment between the MBP promoter and IRES-EGFP-poly(A) cassette (see Fig. 3A). Rat Sox4 protein and mouse Sox4 protein are 98% identical and thus expected to have identical function. In agreement, both Sox4 proteins robustly activated the 3xSX-luc reporter plasmid that has previously been used to characterize Sox4 transcriptional activity (12). The transgene was separated from the vector backbone by a PacI digest, purified, and microinjected into the male pronucleus of fertilized FVB/N oocytes. Transgenic mice were generated from injected oocytes according to standard techniques.
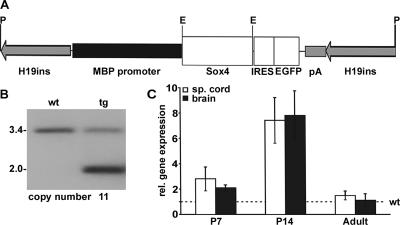
FIG. 3.
Generation of MBP-Sox4 transgenic mice. (A) Schematic representation of the MBP-Sox4 transgene consisting of mouse MBP promoter (positions −3109 to +31), rat Sox4 open reading frame, and IRES-EGFP-poly(A) cassette flanked by H19 insulator sequences. Landmark restriction sites in the construct are indicated by the letters E (EcoRI) and P (PacI). (B) Southern blot analysis of genomic DNA from wild-type (wt) and MBP-Sox4 transgenic (tg) mice after EcoRI digestion. The 3.4-kb band is indicative of the wild-type Sox4 gene, and the 2.0-kb band represents the Sox4 transgene. The copy number was determined using a phosphorimager and is given below the panel. (C) Quantitative RT-PCR. Amounts of Sox4 transcripts were determined in total RNA prepared from spinal cord (sp. cord) and brain of transgenic and wild-type progeny of the female founder at postnatal day 7, postnatal day 14, and in adulthood. Values were normalized to β-actin for each sample. Transcript amounts in the transgenic samples were expressed as multiples of the wild-type values (indicated by the dashed line). rel., relative.
Founder mice were identified by PCR on DNA prepared from tail biopsies (see below) and confirmed by Southern blotting with a 600-bp probe spanning the region of Sox4 behind the coding sequences for the high-mobility group domain. This probe hybridized in genomic DNA digested with EcoRI to a 3.4-kb fragment of the endogenous Sox4 gene and a 2-kb fragment of the Sox4 transgene. As a consequence, Southern blots also revealed the number of integrated transgene copies as the ratio of band intensities for the Sox4 gene versus the MBP-Sox4 transgene after quantification on a phosphorimager. Two female founders with 11 and 13 tandemly arranged copies were obtained and used for further analysis.
Animal husbandry, genotyping, tissue preparation, histology, electron microscopy, and g ratio determination.
All mice were kept under standard housing conditions. Transgenic founders were mated to wild-type C57BL6/J mice, and transgenic offspring were identified by PCR analysis of DNA prepared from tail biopsies using 5′-CCCTCTAGGCCTCGTACAGG-3′ and 5′-GAAAGCGACATCGTCTCTAGC-3′ as primers. A 566-bp PCR product was indicative of the transgene. The transgene was exclusively transmitted from one generation to the next through the female germ line to prevent epigenetic silencing.
Male mice with a Sox11lacZ allele (18) were crossed with either Sox11+/lacZ females to generate Sox11lacZ/lacZ embryos or Sox4+/− females (gift of H. Clevers, Netherlands Institute for Developmental Biology, Utrecht) (15) to generate double-heterozygous mice. Sox10::Cre transgenic mice (14, 24) were crossed with yellow fluorescent protein (YFP)-expressing Rosa26+/stop-YFP mice (20) to obtain Rosa26+/stop-YFP Sox10::Cre mice. Genotyping was performed as described previously (15, 18, 20, 24).
Embryos at 18.5 days postcoitum (dpc) were obtained from staged pregnancies and underwent overnight fixation at 4°C in phosphate-buffered saline containing 4% paraformaldehyde. At postnatal days 1, 7, 14, and 29, transgenic mice and wild-type littermates were anesthetized and perfused transcardially using 0.9% NaCl solution followed by 1% glutaraldehyde and 2.5% paraformaldehyde in 0.1 M sodium cacodylate buffer (pH 7.5). Brain, spinal cord, and sciatic nerve were dissected and for immunohistochemistry underwent overnight fixation at 4°C in phosphate-buffered saline containing 4% paraformaldehyde, overnight cryoprotection in 30% sucrose solution, embedding in OCT compound, freezing to −80°C, and sectioning on a Leica cryotome.
For histological analysis, brain, spinal cord, and sciatic nerve were placed overnight at 4°C in cacodylate-buffered fixative containing 25% paraformaldehyde, 25% glutaraldehyde, and 0.05% picric acid. After extensive washing in 0.1 M sodium cacodylate (pH 7.3), tissues were postfixed in cacodylate-buffered 1% OsO4 for 2 h, dehydrated in an ascending ethanol series, and embedded in Epon resin. For light microscopy, semithin sections (1-μm thickness) were stained with para-phenylene-diamine (PPD) (7) and viewed under a Leica microscope. For electron microscopy, ultrathin sections (50-nm thickness) were stained with uranyl acetate and lead citrate and examined with a Zeiss EM902 electron microscope (10). Determination of g ratios (the numerical ratio between the diameter of the axon proper and the outer diameter of the myelinated fiber) was performed using Biovision software (Soft Imaging System GmbH) by digital tracing around the inner and outer myelinated fiber circumferences, as previously described (27). No fewer than 160 myelinated axons per genotype and age were measured. Data were displayed as a scatter plot against axon diameter or processed using a binning analysis, with axons placed in subgroups of different g ratio ranges (0.05 increments).
In situ hybridization, immunohistochemistry, and TUNEL assays.
Twenty-micrometer-thick cryosections of genotyped, age-matched mouse embryos or tissues were used for in situ hybridization with digoxigenin-labeled antisense riboprobes specific for MBP and proteolipid protein (PLP) as described previously (23).
Alternatively, 10-μm cryosections were used for immunohistochemistry with the following primary antibodies: anti-PCNA mouse monoclonal (1:100 dilution; Roche Biochemicals), anti-NeuN mouse monoclonal (1:500 dilution; Chemicon), anti-MBP mouse monoclonal (1:500 dilution; Chemicon), anti-GFP mouse monoclonal (1:100 dilution; Roche), anti-GFP rabbit antiserum (1:500 dilution; Molecular Probes), anti-glial fibrillary acidic protein (anti-GFAP) rabbit antiserum (1:300 dilution; Neomarkers), anti-Iba-1 rabbit antiserum (1:250 dilution; WAKO), anti-Olig2 rabbit antiserum (1:50,000 dilution; gift of D. Rowitch, University of California—San Francisco), anti-Sox10 guinea pig antiserum (1:1,000 dilution) (13), anti-Sox11 guinea pig antiserum (1:1,000 dilution), anti-Sox4 guinea pig antiserum (1:1,500 dilution), and anti-Ki67 rabbit monoclonal (1:500 dilution, NeoMarkers). Secondary antibodies conjugated to Cy2, Cy3, and Alexa 488 immunofluorescent dyes (Dianova and Invitrogen) were used for detection. Terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling (TUNEL) assays were performed according to the manufacturer's protocol (Chemicon). Samples were analyzed and documented using a Leica inverted microscope (DMIRB) equipped with a cooled MicroMax charge-coupled device camera (Princeton Instruments, Trenton, NJ).
Quantitative RT-PCR analysis.
For quantitative reverse transcription-PCR (RT-PCR) analysis, total RNA was extracted from brain and spinal cord at postnatal day 7 and postnatal day 14 and in the adult using TRIzol reagent (Invitrogen). Two micrograms of each RNA sample was reverse transcribed into cDNA for 1 h at 37°C, using 200 U of Moloney murine leukemia virus reverse transcriptase (NEB) and 100 pmol of oligo(dT) primer under standard conditions. From each cDNA, 2 μl was amplified with primer pairs for Sox4 (5′-TCGTGAACTGCAATCGACTG-3′ and 5′-CGCGTTGTTGGTCTGTTGTA-3′) or β-actin (5′-CCTGGGCATGGAGTCCTG-3′ and 5′-GGAGCAATGATCTTGATCTTC-3′). Primers for Sox4 recognize transcripts from the endogenous Sox4 gene as well as from the Sox4 transgene. PCRs were performed on a Roche Lightcycler according to the manufacturer′s instructions in a 10-μl reaction mixture containing 10% dimethyl sulfoxide and 0.5 μM of each primer at an annealing temperature of 56°C.
RESULTS
Analysis of oligodendrocyte development in Sox11-deficient mice.
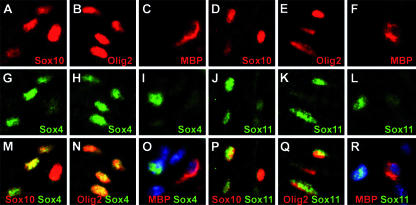
Expression of Sox4 and Sox11 in the oligodendrocyte lineage has so far been analyzed in cell culture (12). In the embryonic spinal cord, expression of both group C Sox proteins is not restricted to the oligodendendrocyte lineage (5). At 18.5 dpc, many cells of the spinal cord expressed the two Sox proteins. Amounts varied significantly between cells but were usually highest in neuronal cells of the mantle zone. However, both Sox proteins were also expressed in the marginal zone (i.e., the future white matter region), which is devoid of neurons (Fig. 1). At 18.5 dpc, the marginal zone contains many oligodendrocyte precursors and a few differentiating oligodendrocytes. Many marginal zone cells that were positive for Sox4 also expressed Sox10 or Olig2 (Fig. 1A, B, G, H, M, and N), indicating that these cells belong to the oligodendrocyte lineage. Similar results were also obtained for Sox11 (Fig. 1D, E, J, K, P, and Q). In contrast to this significant overlap with oligodendrocyte markers, MBP-expressing cells of the marginal zone lacked Sox4 or Sox11 expression (Fig. 1C, F, I, L, O, and R). As MBP selectively labels the fraction of differentiating oligodendrocytes, we conclude that Sox4 and Sox11 are expressed in oligodendrocyte precursors but are downregulated as these cells undergo terminal differentiation. Oligodendrocytic expression of Sox4 and Sox11 in the spinal cord and in cell culture thus follows a similar pattern.
FIG. 1.
Oliogdendroglial expression of Sox4 and Sox11. Antibodies directed against Sox4 (G, H, and I) and Sox11 (J, K, and L) (both in green) were combined with antibodies directed against Sox10 (A and D), Olig2 (B and E) and MBP (C and F) (all in red) in coimmunohistochemistry (M, N, O, P, Q, and R) on transverse sections of wild-type spinal cords from 18.5-dpc embryos. Images were taken from the marginal zone. Coexpression of Sox4 or Sox11 and oligodendroglial markers is visible as a yellow signal in panels M, N, O, P, Q, and R. Nuclei were additionally visualized by DAPI (4′,6′-diamidino-2-phenylindole) staining and are shown in blue in some panels (O and R).
Neither Sox4-deficient nor Sox11-deficient embryos exhibit overt signs of abnormal CNS development until the time of their death. However, development of the oligodendrocyte lineage has not been analyzed in detail. Taking into account that Sox4-deficient mice die already at 13.5 dpc (15), a time when oligodendrocyte development has just started, we concentrated our analysis on Sox11-deficient mice.
When spinal cords of Sox11-deficient embryos were analyzed at 15.5 dpc or 18.5 dpc using Olig2 or Sox10 as two independent markers of the oligodendrocyte lineage, cells were clearly present and exhibited a distribution throughout the spinal cord that resembled the wild type (Fig. 2A and B [data not shown]). Quantification furthermore revealed that overall numbers of oligodendroglial lineage cells were not significantly different between Sox11-deficient embryos and wild-type littermates at 18.5 dpc, the latest time at which an analysis could be carried out because of the perinatal lethality of mutant mice (Fig. 2E [data not shown]).
FIG. 2.
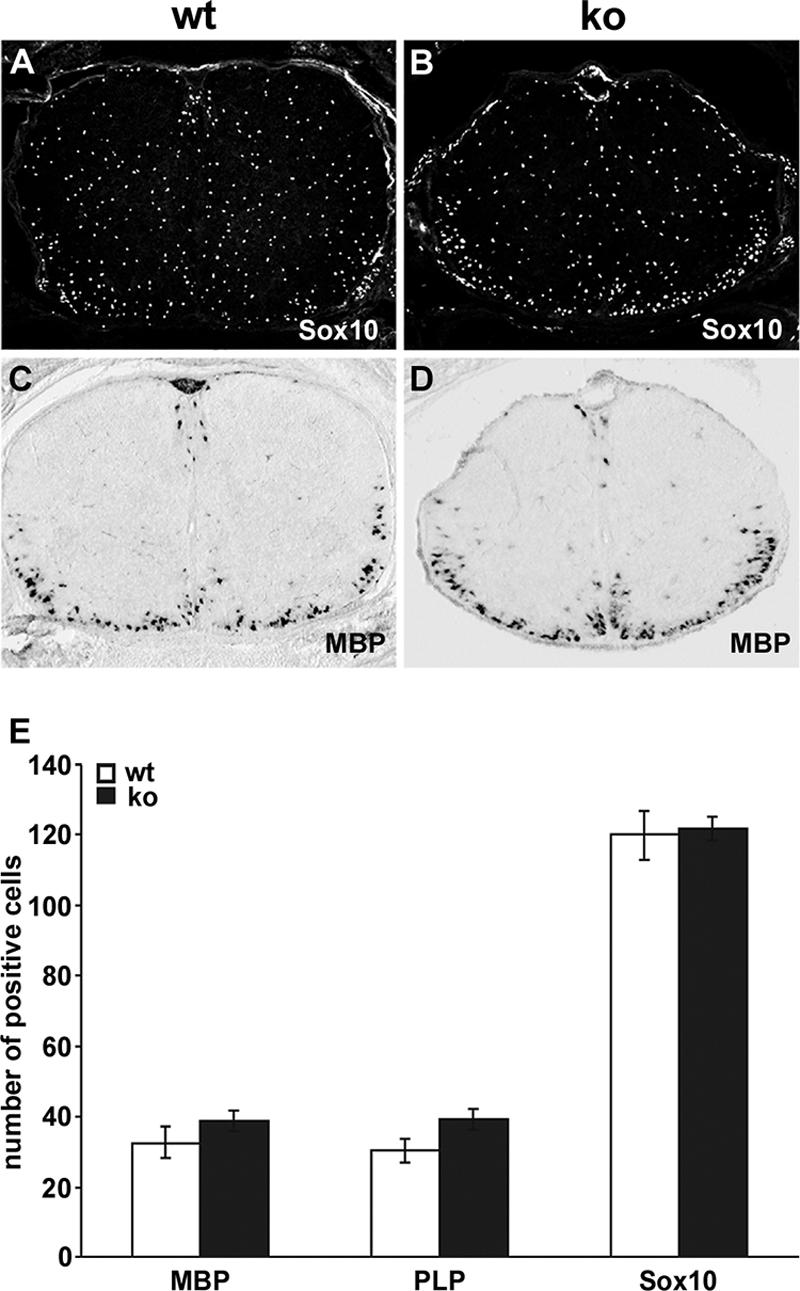
Oligodendroglial development in the embryonic spinal cord of Sox11-deficient mice. Immunohistochemistry with antibodies specific for the oligodendrocyte lineage marker Sox10 (A and B) and in situ hybridizations with an antisense riboprobe specific for MBP (C and D) were performed on transverse sections of wild-type (wt [A and C]) and Sox11lacZ/lacZ (ko [B and D]) spinal cords from 18.5-dpc embryos. (E) The numbers of MBP-positive, PLP-positive, and Sox10-positive cells were determined at 18.5 dpc in wild-type (wt) and Sox11lacZ/lacZ knockout (ko) spinal cord halves, as indicated below the bars. At least 15 separate 20-μm sections from the forelimb region of two independent embryos were counted for each genotype. Data are presented as means ± standard deviations. Differences from the wild type were not statistically significant for any marker, as determined by Student's t test.
At 18.5 dpc, the majority of cells in the oligodendrocyte lineage are in the oligodendrocyte precursor stage, with terminal differentiation and myelin gene expression just commencing in the future white matter region of the spinal cord. To analyze whether terminal differentiation starts on time in the Sox11-deficient embryos, we performed in situ hybridizations with probes directed against transcripts from the myelin genes MBP and PLP (Fig. 2C and D [data not shown]). The numbers of MBP- and PLP-expressing oligodendrocytes in the Sox11-deficient and wild-type spinal cords were comparable, suggesting that terminal differentiation of oligodendrocytes had started on schedule in the absence of Sox11 (Fig. 2E).
Generation of mice that overexpress SoxC proteins in differentiating oligodendrocytes.
Because Sox11 function in the oligodendrocyte lineage might have been masked by coexpressed and functionally redundant Sox4 in Sox11-deficient spinal cords (5), we switched from a loss-of-function to a gain-of-function approach by generating mice in which either Sox4 or Sox11 was ectopically expressed in differentiating oligodendrocytes. To achieve this goal, we placed the corresponding open reading frames under the control of a 3.1-kb fragment from the MBP promoter (Fig. 3A). This fragment has previously been shown to direct transgene expression specifically to differentiating oligodendrocytes during the peak of myelination in the first postnatal weeks (9). It has also been shown that the activity of this regulatory element is strongly reduced during later postnatal times, so that expression of our transgene was expected to be transient. To better follow transgene expression, we also included an EGFP cassette in the construct which was linked via an IRES to the Sox open reading frame and should allow detection of transgene expression by autofluorescence. Additionally, we placed the construct between H19 insulators to prevent potential influences on transgene expression from regulatory regions at the integration site (Fig. 3A).
Progeny from two female founder mice expressed the MBP-Sox4 transgene, whereas none expressed the MBP-Sox11 transgene. The presence of the H19 insulators in our construct caused transgene expression to be shut off when passaged through the male germ line, possibly through imprinting effects. We thus propagated the transgene through the female germ line. All experiments shown in this article were performed with animals derived from a single MBP-Sox4 transgenic founder that carried 11 copies of the transgene and transmitted the transgene faithfully and with high frequency to its offspring (Fig. 3B). Progeny of the second MBP-Sox4 transgenic founder exhibited a qualitatively similar, slightly more severe phenotype (data not shown).
Beginning at postnatal week 2, transgenic animals started to exhibit coarse, generalized action tremors very reminiscent of those described for dysmyelinating mouse mutants. Additionally, attacks with a collection of dyskinetic behaviors were observed, including listing to one side, exaggerated flexion or extension of the trunk, and tonic extension or retraction of the limbs. The severity of the phenotype increased until the end of the fourth postnatal week. Thereafter, the mice gradually recovered so that transgenic littermates could no longer be easily identified in the cage at 2 months of age. None of these symptoms was observed in mice with strong oligodendrocytic YFP expression (see below). Taking into account that YFP and EGFP are 98% identical, the phenotype of our MBP-Sox4 transgenic mice is unlikely to result from expression of the EGFP cassette but rather is a bona fide effect of ectopic Sox4 expression.
CNS expression of the MBP-Sox4 transgene during early postnatal development.
Total Sox4 expression was analyzed in transgenic and wild-type mice during postnatal development by quantitative RT-PCR on brain and spinal cord RNA. At postnatal day 7, we noted a moderate two- to threefold-higher Sox4 expression in the transgenic progeny (Fig. 3C). This moderate increase in fact corresponds to a strong transgene expression in oligodendroglial cells, if the widespread and strong expression of the endogenous Sox4 gene in many cells at this time is taken into account (5). Seven days later, expression of the Sox4 gene was almost completely extinguished in the CNS (12), whereas the MBP-Sox4 transgene continued to be expressed. As a consequence, brain and spinal cord expression of Sox4 in transgenic animals reached seven- to eightfold the amount of Sox4 in the wild type at postnatal day 14 (Fig. 3C). In contrast, Sox4 transcript levels in brain and spinal cord were comparable between adult transgenic and wild-type mice, suggesting that transgene expression was downregulated after the first postnatal weeks when the 3.1-kb MBP promoter fragment has been reported to lose its activity (9).
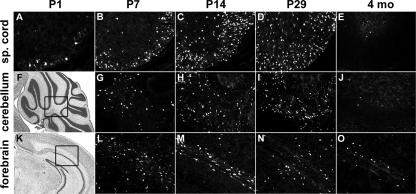
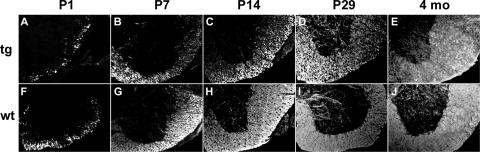
We also followed expression of the MBP-Sox4 transgene by autofluorescence and immunohistochemistry against EGFP. In the spinal cord, EGFP was detected already 1 day after birth in a few select cells in the future white matter region (Fig. 4A). The number of these cells was strongly increased on postnatal day 7 (Fig. 4B). At postnatal day 14, EGFP-labeled cells were also apparent in the gray matter (Fig. 4C). Both the timing with which EGFP-labeled cells appear and their location were thus indicative of a selective expression of the MBP-Sox4 transgene in differentiating oligodendrocytes of the spinal cord. Many differentiating oligodendrocytes still showed the presence of EGFP at postnatal day 29 (Fig. 4D), whereas virtually no EGFP was present in the adult spinal cord (Fig. 4E).
FIG. 4.
Postnatal CNS expression of the MBP-Sox4 transgene. Autofluorescence or immunohistochemistry against EGFP was used to determine transgene expression in spinal cord (sp. cord [A to E]), cerebellum (G to J), and corpus callosum (L to O) at postnatal days 1 (A), 7 (B, G, and L), 14 (C, H, and M), and 29 (D, I, and N) and at 4 months (E, J, and O). The areas depicted in panels G to J and L to O are boxed in the hematoxylin-eosin-stained overviews of cerebellum (F) and forebrain (K), respectively.
A comparable pattern of transgene expression was also observed in other CNS regions with high numbers of differentiating oligodendrocytes, including the white matter region of the cerebellum (Fig. 4G to J) and the corpus callosum (Fig. 4L to O). Compared to the spinal cord, oligodendrocyte differentiation in these areas is slightly delayed. In agreement, EGFP-positive signals were usually not detected in corpus callosum or cerebellum at postnatal day 1 but were clearly present at postnatal day 7 (Fig. 4G and L [data not shown]). Again, we observed a strong decrease of the EGFP signal after the fourth postnatal week such that EGFP-positive cells were mostly missing from the adult cerebellum and severely reduced in the corpus callosum (compare Fig. 4I and N to J and O).
In contrast to the widespread occurrence of EGFP-positive cells throughout the CNS, we failed to detect EGFP in the peripheral nervous system. The sciatic nerve, in particular, was completely devoid of EGFP (data not shown), arguing that transgene expression was restricted to the CNS. This was expected as the enhancer that drives MBP expression in Schwann cells has previously been mapped to a region outside the MBP promoter fragment used in the transgenic construct (6).
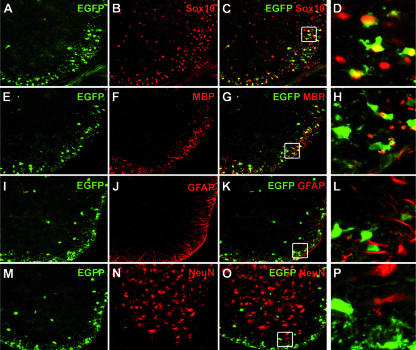
Coimmunohistochemistry on spinal cord and corpus callosum confirmed that EGFP-positive cells also expressed the oligodendrocyte lineage marker Sox10 (Fig. 5A to D; also see Fig. S1A to D in the supplemental material). The relative percentage of cells double labeled for Sox10 and EGFP among all Sox10-positive cells furthermore varied with age and region analyzed, as expected for selective expression of the MBP-Sox4 transgene in differentiating oligodendrocytes (data not shown). In agreement, there was also a significant overlap between EGFP and MBP expression during the first postnatal week (Fig. 5E to H; also see Fig. S1E to H in the supplemental material). Coexpression was, however, not observed in those cells that expressed high levels of EGFP or MBP. In contrast, there was no significant overlap between EGFP and the astrocyte marker GFAP (Fig. 5I to L; also see Fig. S1I to L in the supplemental material) or the panneuronal marker NeuN (Fig. 5M to P; also see Fig. S1M to P in the supplemental material) at any of the time points analyzed. In summary, these data not only prove that the MBP-Sox4 transgene is selectively expressed in the oligodendrocyte lineage but also show that its expression is restricted to differentiating oligodendrocytes during the peak time of postnatal myelination before being extinguished at later postnatal stages.
FIG. 5.
Cell-type-specific expression of the MBP-Sox4 transgene in the spinal cord. Antibodies directed against EGFP (in green) (A, E, I, and M) were combined with antibodies directed against the oligodendroglial marker Sox10 (B), the marker for differentiating oligodendrocytes, MBP (F), the astrocyte marker GFAP (J), and the panneuronal marker NeuN (N) (all in red) in coimmunohistochemistry (C, D, G, H, K, L, O, and P) on sections of transgenic spinal cord at postnatal day 3 (A to H) and postnatal day 7 (I to P). Coexpression of EGFP and cell-type-specific marker is visible as a yellow signal in panels C, D, G, H, K, L, O, and P. Panels D, H, L, and P represent magnifications of the boxed area in panels C, G, K, and O.
Reduced and delayed myelin gene expression in the spinal cord of MBP-Sox4 transgenic mice.
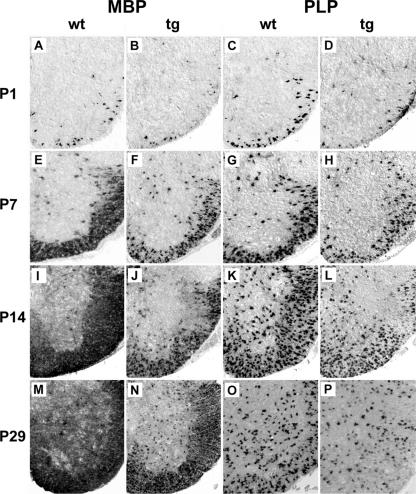
Expression of myelin genes was next examined in MBP-Sox4 transgenic animals. Using in situ hybridization with antisense probes directed against MBP and PLP, we followed myelin gene expression in the early postnatal spinal cord. When compared to the wild type, we observed a strong reduction in MBP- and PLP-positive cells in the white matter region of transgenic spinal cords already at postnatal day 1 (compare Fig. 6A and C to B and D). Although MBP- and PLP-expressing cells increased steadily in the MBP-Sox4 transgenic spinal cord during the next 4 weeks (Fig. 6F, H, J, L, N, and P), their numbers remained lower in the transgenic animal than in the wild type (Fig. 6E, G, I, K, M, and O). At postnatal day 29, the difference between wild-type and transgenic spinal cord was less dramatic than at postnatal day 7 or 14, arguing that myelin gene expression in the transgenic spinal cord increases at these later stages.
FIG. 6.
Occurrence of myelin gene transcripts in MBP-Sox4 transgenic animals. In situ hybridization was performed with antisense riboprobes directed against MBP (A, B, E, F, I, J, M, and N) and PLP (C, D, G, H, K, L, O, and P) on spinal cord sections from wild-type (wt) animals (A, C, E, G, I, K, M, and O) and MBP-Sox4 transgenic (tg) littermates (B, D, F, H, J, L, N, and P) at postnatal days 1 (A to D), 7 (E to H), 14 (I to L), and 29 (M to P).
Very similar results were also obtained in immunohistochemistry using antibodies directed against MBP. At postnatal day 1, there were fewer MBP-positive cells in the transgenic spinal cord than in the wild type (Fig. 7A and F). At later stages of postnatal development, single MBP-expressing cells could no longer be resolved. Nevertheless, the MBP-stained white matter region was narrower in the transgenic spinal cord than in the wild type at both postnatal days 7 and 14 and the MBP signal appeared less intense and less uniform (compare Fig. 7B and C to G and H). At postnatal day 29, this difference was already less pronounced and was no longer obvious in the adult (Fig. 7D, E, I, and J). We conclude that the reduced myelin gene expression in the MBP-Sox4 transgenic animal also translates into a reduction of myelin proteins.
FIG. 7.
Occurrence of MBP in spinal cords of MBP-Sox4 transgenic animals. Immunohistochemistry with antibodies directed against MBP was performed on spinal cord sections from MBP-Sox4 transgenic (tg) animals (A to E) and wild-type (wt) littermates (F to J) at postnatal days 1 (A and F), 7 (B and G), 14 (C and H), and 29 (D and I) and at 4 months (E and J).
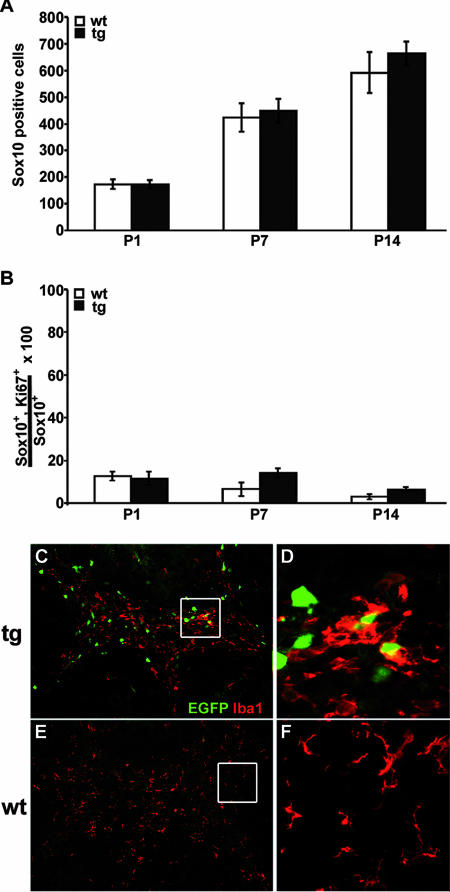
Despite this reduction in myelin gene transcripts and myelin proteins, the numbers of Sox10-positive cells in MBP-Sox4 transgenic and wild-type spinal cords were very similar at postnatal days 1, 7, and 14 (Fig. 8A). As Sox10 labels all cells of the oligodendrocyte lineage, dramatic alterations in the overall number of oligodendrocytes are unlikely to be responsible for the reduced myelin expression. Rather, fewer oligodendrocytes appear to produce myelin at any given time.
FIG. 8.
Oligodendroglial and microglial parameters in the CNS of MBP-Sox4 transgenic animals. (A) The number of Sox10-positive cells was determined in spinal cord halves from wild-type (wt) and MBP-Sox4 transgenic (tg) animals at postnatal days 1, 7, and 14, as indicated below the bars. (B) The percentage of Sox10-positive cells that also express the proliferation marker Ki67 was determined in wild-type and transgenic spinal cords at postnatal days 1, 7, and 14. (C to F) Immunohistochemistry with antibodies directed against the microglial marker Iba-1 was performed on cerebellar sections from MBP-Sox4 transgenic (C and D) and wild-type (E and F) mice at postnatal day 7 and is shown together with EGFP autofluorescence at low (C and E) and high (D and F) magnification.
Using Ki67 or PCNA as a marker for proliferating cells, we only detected modest increases in proliferating oligodendrocytes at postnatal days 7 and 14 (Fig. 8B [data not shown]). EGFP-expressing cells in the MBP-Sox4 transgenic animals did not colabel with either Ki67 or PCNA, indicating that transgene-expressing oligodendrocytes are postmitotic, as expected. Although microglia in white matter regions of MBP-Sox4 transgenic animals had a morphology characteristic of activated microglia and were often associated with EGFP-expressing oligodendrocytes (compare Fig. 8C and D to E and F), rates of apoptosis were not significantly altered, as assessed by TUNEL staining or by the number of caspase-3-positive cells (data not shown). Oligodendroglial cells expressing the MBP-Sox4 transgene thus appear to persist in the postnatal spinal cord in a postmitotic but premyelinating or early myelinating state.
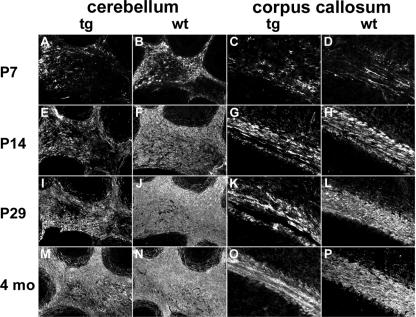
Reduced and delayed myelin gene expression in corpus callosum and cerebellum of MBP-Sox4 transgenic mice.
The reduction in myelin proteins was also evident in other CNS regions such as the white matter tract of the cerebellum or the corpus callosum. Compared to the spinal cord and in agreement with a later onset of myelin gene expression in these areas, differences in the amount and occurrence of MBP were more pronounced at postnatal day 14 than at postnatal day 7 (compare Fig. 9B, D, F, and H to A, C, E, and G) and were still clearly detectable at postnatal day 29 or at 4 months (compare Fig. 9J, L, N, and P to I, K, M, and O). In the transgenic corpus callosum, single MBP-expressing cells could, for instance, still be distinguished at postnatal day 29 (Fig. 9K). At that time, the corpus callosum already appeared as a homogenously stained white matter tract in the wild type (Fig. 9L). Even in the adult transgenic brain, the corpus callosum remained thinner and stained less intensely for MBP than in the wild type (Fig. 9O and P). At no time did we detect similar reductions in myelin gene expression in spinal cord, cerebellum, or corpus callosum of mice with oligodendrocytic YFP expression. Instead, postnatal myelination occurred in a manner comparable to that in the wild type (see Fig. S2 in the supplemental material [data not shown]), confirming that ectopic expression of an autofluorescent protein in oligodendrocytes does not interfere with proper differentiation and myelination.
FIG. 9.
Occurrence of MBP in the brains of MBP-Sox4 transgenic animals. Immunohistochemistry with antibodies directed against MBP was performed on cerebellar sections (A, B, E, F, I, J, M, and N) and sections of the corpus callosum (C, D, G, H, K, L, O, and P) from MBP-Sox4 transgenic (tg) animals (A, C, E, G, I, K, M, and O) and wild-type (wt) littermates (B, D, F, H, J, L, N, and P) at postnatal days 7 (A to D), 14 (E to H), and 29 (I to L) and at 4 months (M to P).
Structural myelin abnormalities in MBP-Sox4 transgenic mice.
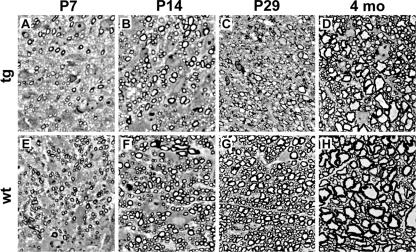
Reduced myelin gene expression also correlated with structural abnormalities in myelin. Spinal cords from MBP-Sox4 transgenic mice revealed a strong reduction in the number and thickness of myelin figures in their white matter at postnatal days 7, 14, and 29 (compare Fig. 10A, B, and C to E, F, and G). Although myelin sheaths were still thinner in adult transgenic animals, the overall number and size distribution of myelinated axons began to resemble those in the wild type (Fig. 10D and H), confirming that with a loss of transgene expression, myelination recovered. In accord with a lack of transgene expression in Schwann cells, myelination was normal in the sciatic nerve (data not shown).
FIG. 10.
Hypomyelination in the postnatal spinal cords of MBP-Sox4 transgenic mice. Shown is PPD staining of myelin sheaths in transverse sections of spinal cords from MBP-Sox4 transgenic (tg) animals (A to D) and wild-type (wt) littermates (E to H) at postnatal days 7 (A and E), 14 (B and F), and 29 (C and G) and at 4 months (D and H).
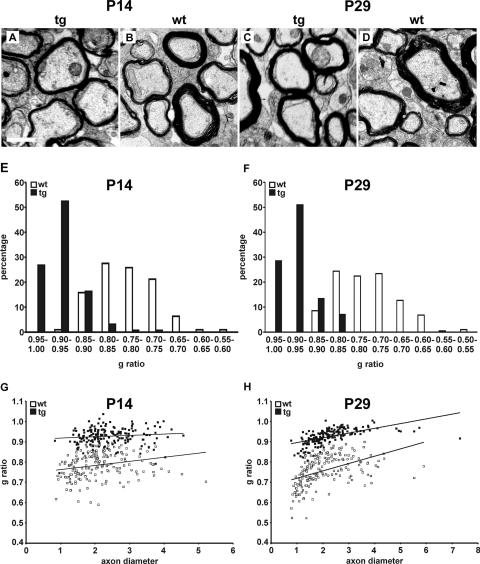
Reduced myelin thickness was particularly evident from electron microscopic studies of the white matter of transgenic spinal cords at postnatal days 14 and 29, while compaction and membrane periodicity were mostly normal (Fig. 11A to D [data not shown]). When g ratios as the numerical ratio between the diameter of the axon proper and the outer diameter of the myelinated fiber were determined at postnatal days 14 and 29, differences between MBP-Sox4 transgenic mice and wild-type littermates were immediately obvious. The average g ratio was shifted from 0.79 (± 0.06) at postnatal day 14 and 0.76 (± 0.07) at postnatal day 29 in the wild type to 0.93 (± 0.04) in the transgenic animal, indicating that myelin was generally thinner. At both ages analyzed, axons with high g ratios were preferentially observed in the MBP-Sox4 transgenic mice, whereas axons with low g ratios and therefore thick myelin sheaths were almost exclusively found in wild-type animals (Fig. 11E to H). Reduced myelin thickness was furthermore observed for all axons independent of their caliber size (Fig. 11G and H).
FIG. 11.
Myelin ultrastructure in MBP-Sox4 transgenic mice. Shown are representative ultrathin sections from 14- and 29-day-old MBP-Sox4 transgenic (tg) mice (A and C) and wild-type (wt) littermates (B and D). The scale bar is 1 μm. From these sections, g ratios were determined at both postnatal days 14 (E and G) and 29 (F and H). Data are presented by showing the relative distribution of g ratios (E and F) and by showing the g ratio of each fiber as a function of the respective axon diameter in a scatter blot (G and H). The scatter blots show that small and large axons are similarly affected by hypomyelination.
DISCUSSION
We had previously shown in primary cultures and now confirmed in situ that the two related transcription factors Sox4 and Sox11 are expressed in the oligodendrocyte lineage until cells undergo terminal differentiation (12). Such an expression pattern is compatible with a function of these transcription factors in oligodendrocyte precursors.
Analysis of oligodendrocyte development in the Sox11-deficient spinal cord revealed that a normal complement of oligodendrocyte precursors had formed by the time of birth and that the percentage of oligodendrocytes that had started to terminally differentiate and transcribe myelin genes was comparable to that in the wild type. These results argue that oligodendrocyte development can proceed in the absence of Sox11. One possibility is that Sox11 does not influence oligodendrocyte development. Alternatively, Sox11 effects on oligodendrocyte development may be compensated for by the continued presence of the closely related Sox4 in cells of the oligodendrocyte lineage (5). Such functional redundancy between other closely related Sox proteins has been shown for a number of different developmental processes, including oligodendrocyte development (3, 4, 17, 21, 24, 25).
A role for Sox4 and Sox11 in the oligodendrocyte lineage will thus require concomitant deletion of both factors. Taking the many early developmental defects into account that have already been associated with the single mutants (15, 18), as well as our inability to generate compound heterozygous mice, the necessary tissues will only be obtained using conditional mutagenesis and highly cell-specific Cre recombinases. Additionally, it deserves to be noted that both Sox4 and Sox11 are closely related to a third group C protein. From the very few data that are available for Sox12, a significant overlap of expression with Sox4 and Sox11 is obvious (11). In the case in which Sox12 is also expressed in the oligodendrocyte lineage, even the Sox4 Sox11 double mutant may not be informative.
Therefore, we used overexpression as an alternative approach to study the role of group C Sox proteins in the oligodendrocyte lineage. Despite the use of analogous strategies in the construction of Sox4 and Sox11 transgenic constructs and a similar number of pronucleus injections, we only obtained Sox4-overexpressing transgenic animals. For the Sox11 transgene, 4 males and 2 females with genomic integration were obtained from pronucleus injections at the time of weaning. Their progeny did not exhibit significant transgene expression, as judged by EGFP autofluorescence or signs of dysmyelination. Because the presence of H19 insulators in our construct would have shut off transgene expression after passage through the male germ line, only the two females could have transmitted an expressed Sox11 transgene. Taking the low number of female founders into account, their failure to do so is not sufficient to identify negative selection against ectopic Sox11 expression as the underlying cause of our unsuccessful attempts.
To overexpress group C proteins in the oligodendrocyte lineage, we used 3.1 kb of the MBP upstream region. MBP expression in oligodendrocytes and Schwann cells has previously been reported to be controlled by the activity of multiple enhancers that function in a time- and region-specific pattern (8). The 3.1-kb fragment used in our study does not contain those regulatory elements that drive MBP expression in Schwann cells of the peripheral nervous system (6), nor does it maintain MBP expression in the adult central nervous system (9). Rather it contains regulatory elements for efficient upregulation of MBP expression during the peak of myelination. Expression of our Sox4 transgene mimics this expected pattern. There was no expression of the transgene and thus no effect of transgene expression throughout the peripheral nervous system. Rather, the transgene was selectively turned on in the central nervous system when oligodendrocytes started to undergo terminal differentiation. It was extinguished again after the first postnatal weeks despite the fact that myelination was not complete in the transgenic animal at this time. This implies that the activity of the CNS-specific MBP enhancers in the 3.1-kb fragment is not regulated so much cell intrisically by the achieved myelination status of oligodendrocytes but rather by changing extrinsic signals of the CNS environment.
As a consequence of transgene activity, Sox4 expression is prolonged in the oligodendrocyte lineage. Whereas Sox4 is normally turned off when oligodendrocytes become postmitotic and undergo terminal differentiation, it is now maintained past this point. The result of this prolonged Sox4 expression is a severe hypomyelination with all of its typical signs, including a “shiverer” phenotype, reduced levels of myelin gene expression, and thinner myelin sheaths during the early postnatal period. The phenotype only recovers once the transgene is turned off. Interestingly, hypomyelination did not go along with increased rates of apoptosis, and proliferation rates in the oligodendrocyte lineage were only mildly increased. Considering additionally that the MBP-Sox4 transgene was only rarely expressed in proliferating oligodendroglial cells, Sox4 expression in postmitotic oligodendrocytes appears incompatible with their terminal differentiation. Instead, postmitotic oligodendrocytes become transiently arrested in a premyelinating or early myelinating state until the transgene is switched off.
Our study therefore reveals an antidifferentiation effect of Sox4 and thus argues that Sox4 normally prevents precocious terminal differentiation of oligodendrocyte precursors. If functional redundancy is assumed, Sox11 could have similar effects. Interestingly, human gliomas exhibit strong Sox11 expression in accord with the highly dedifferentiated nature of these cells (26).
Recently, the two group D Sox proteins Sox5 and Sox6 have also been reported to counteract precocious oligodendrocyte differentiation (24). In the case of Sox5 and Sox6, both proteins were shown to bind to Sox binding sites in myelin gene promoters and exert their function by directly interfering with the ability of Sox10 to activate myelin genes. In contrast to Sox5 and Sox6, the group C Sox proteins do not interfere with Sox10-dependent upregulation of myelin genes in transient transfections (data not shown), nor did we find any evidence for a direct repression of the 3.1-kb MBP upstream region by group C Sox proteins in reporter gene assays. Therefore, we assume that group C proteins prevent oligodendrocyte maturation by a mechanism that is distinct from that of group D Sox proteins. This failure to detect a direct negative effect of Sox4 on expression from the 3.1-kb MBP upstream region may also explain why the MBP-Sox4 transgene can be expressed in differentiating oligodendrocytes and at the same time inhibit endogenous MBP gene expression. Very likely, the transgene's effect on endogenous MBP gene expression is indirect and not mediated by the 3.1-kb MBP promoter sequence present in the transgene.
A recent study addressed the impact of group C Sox protein overexpression on neurogenesis in the neural tube of the early chicken embryo (1). This study revealed that both Sox4 and Sox11 precociously induce panneuronal traits in immature neuronal precursors. Whereas we prolonged Sox gene expression in glial precursors, Bergsland and colleagues (1) induced Sox gene expression prematurely in neuronal precursors. Thus, it cannot be excluded that the results of both studies are differentially influenced by their respective experimental setups. Nevertheless, it is intriguing that group C Sox proteins appear to have opposing effects on neuronal versus glial differentiation, promoting one and repressing the other. In this context, it will be particularly interesting to analyze oligodendrocyte differentiation in the absence of group C Sox proteins once compound mutant mice are available.
Supplementary Material
Acknowledgments
We thank A. Garratt and H. Wende for help with g ratio determinations and A. Fischer for expert technical assistance. H. Clevers is acknowledged for providing mice with a Sox4 null allele, F. Constantini for providing mice with a Rosa26stop-YFP allele, A. Roach for the MBP promoter, and V. Lefebvre for the mouse Sox4 plasmid.
This work was supported by a grant from the Wilhelm-Sander Stiftung to M.W. (2002.048.1) and a grant from the Deutsche Forschungsgemeinschaft to E.S. (So251/3-1).
Footnotes
Published ahead of print on 21 May 2007.
Supplemental material for this article may be found at http://mcb.asm.org/.
REFERENCES
- 1.Bergsland, M., M. Werme, M. Malewicz, T. Perlmann, and J. Muhr. 2006. The establishment of neuronal properties is controlled by Sox4 and Sox11. Genes Dev. 20:3475-3486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bondurand, N., M. Girard, V. Pingault, N. Lemort, O. Dubourg, and M. Goossens. 2001. Human Connexin 32, a gap junction protein altered in the X-linked form of Charcot-Marie-Tooth disease, is directly regulated by the transcription factor SOX10. Hum. Mol. Genet. 10:2783-2795. [DOI] [PubMed] [Google Scholar]
- 3.Bylund, M., E. Andersson, B. G. Novitch, and J. Muhr. 2003. Vertebrate neurogenesis is counteracted by Sox1-3 activity. Nat. Neurosci. 6:1162-1168. [DOI] [PubMed] [Google Scholar]
- 4.Chaboissier, M.-C., A. Kobayashi, V. I. P. Vidal, S. Lützkendorf, H. J. G. van de Kant, M. Wegner, D. G. de Rooij, R. R. Behringer, and A. Schedl. 2004. Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development 131:1891-1901. [DOI] [PubMed] [Google Scholar]
- 5.Cheung, M., M. Abu-Elmagd, H. Clevers, and P. J. Scotting. 2000. Roles of Sox4 in central nervous system development. Mol. Brain Res. 79:180-191. [DOI] [PubMed] [Google Scholar]
- 6.Denarier, E., R. Forghani, H. F. Farhadi, S. Dib, N. Dionne, H. C. Friedman, P. Lepage, T. J. Hudson, R. Drouin, and A. Peterson. 2005. Functional organization of a Schwann cell enhancer. J. Neurosci. 25:11210-11217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Estable-Puig, J. F., W. C. Bauer, and J. M. Blumberg. 1965. Technical note: paraphenylendiamine staining of osmium-fixed plastic embedded tissue for light and phase microscopy. J. Neuropathol. Exp. Neurol. 24:531-536. [Google Scholar]
- 8.Farhadi, H. F., P. Lepage, R. Forghani, H. C. Friedman, W. Orfali, L. Jasmin, W. Miller, T. J. Hudson, and A. C. Peterson. 2003. A combinatorial network of evolutionarily conserved myelin basic protein regulatory sequences confers distinct glial-specific phenotypes. J. Neurosci. 23:10214-10223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Foran, D. R., and A. C. Peterson. 1992. Myelin acquisition in the central nervous system of the mouse revealed by an MBP-LacZ transgene. J. Neurosci. 12:4890-4897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Friedrich, R. P., B. Schlierf, E. R. Tamm, M. R. Bösl, and M. Wegner. 2005. The class III POU domain protein Brn-1 can fully replace the related Oct-6 during Schwann cell development and myelination. Mol. Cell. Biol. 25:1821-1829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jay, P., I. Sahly, C. Goze, S. Taviaux, F. Poulat, G. Couly, M. Abitbol, and P. Berta. 1997. Sox22 is a new member of the Sox gene family, mainly expressed in human nervous tissue. Hum. Mol. Genet. 6:1069-1077. [DOI] [PubMed] [Google Scholar]
- 12.Kuhlbrodt, K., B. Herbarth, E. Sock, J. Enderich, I. Hermans-Borgmeyer, and M. Wegner. 1998. Cooperative function of POU proteins and Sox proteins in glial cells. J. Biol. Chem. 273:16050-16057. [DOI] [PubMed] [Google Scholar]
- 13.Maka, M., C. C. Stolt, and M. Wegner. 2005. Identification of Sox8 as a modifier gene in a mouse model of Hirschsprung disease reveals underlying molecular defect. Dev. Biol. 277:155-169. [DOI] [PubMed] [Google Scholar]
- 14.Matsuoka, T., P. E. Ahlberg, N. Kessaris, P. Iannarelli, U. Dennehy, W. D. Richardson, A. P. McMahon, and G. Koentges. 2005. Neural crest origins of the neck and shoulder. Nature 436:347-355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schilham, M. W., M. A. Oosterwegel, P. Moerer, J. Ya, P. A. J. Deboer, M. Vandewetering, S. Verbeek, W. H. Lamers, A. M. Kruisbeek, A. Cumano, and H. Clevers. 1996. Defects in cardiac outflow tract formation and pro-B-lymphocyte expansion in mice lacking Sox-4. Nature 380:711-714. [DOI] [PubMed] [Google Scholar]
- 16.Schlierf, B., T. Werner, G. Glaser, and M. Wegner. 2006. Expression of Connexin47 in oligodendrocytes is regulated by the Sox10 transcription factor. J. Mol. Biol. 361:11-21. [DOI] [PubMed] [Google Scholar]
- 17.Smits, P., P. Li, J. Mandel, Z. Zhang, J. M. Deng, R. R. Behringer, B. de Crombrugghe, and V. Lefebvre. 2001. The transcription factors L-Sox5 and Sox6 are essential for cartilage formation. Dev. Cell 1:277-290. [DOI] [PubMed] [Google Scholar]
- 18.Sock, E., S. D. Rettig, J. Enderich, M. R. Bösl, E. R. Tamm, and M. Wegner. 2004. Gene targeting reveals a widespread role for the high-mobility-group transcription factor Sox11 in tissue remodeling. Mol. Cell. Biol. 24:6635-6644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sohn, J., J. Natale, L. J. Chew, S. Belachew, Y. Cheng, A. Aguirre, J. Lytle, B. Nait-Oumesmar, C. Kerninon, M. Kanai-Azuma, Y. Kanai, and V. Gallo. 2006. Identification of Sox17 as a transcription factor that regulates oligodendrocyte development. J. Neurosci. 26:9722-9735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Srinivas, S., T. Watanabe, C. S. Lin, C. M. William, Y. Tanabe, T. M. Jessell, and F. Costantini. 2001. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev. Biol. 1:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stolt, C. C., P. Lommes, R. P. Friedrich, and M. Wegner. 2004. Transcription factors Sox8 and Sox10 perform non-equivalent roles during oligodendrocyte development despite functional redundancy. Development 131:2349-2358. [DOI] [PubMed] [Google Scholar]
- 22.Stolt, C. C., P. Lommes, E. Sock, M.-C. Chaboissier, A. Schedl, and M. Wegner. 2003. The Sox9 transcription factor determines glial fate choice in the developing spinal cord. Genes Dev. 17:1677-1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Stolt, C. C., S. Rehberg, M. Ader, P. Lommes, D. Riethmacher, M. Schachner, U. Bartsch, and M. Wegner. 2002. Terminal differentiation of myelin-forming oligodendrocytes depends on the transcription factor Sox10. Genes Dev. 16:165-170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stolt, C. C., A. Schlierf, P. Lommes, S. Hillgärtner, T. Werner, T. Kosian, E. Sock, N. Kessaris, W. D. Richardson, V. Lefebvre, and M. Wegner. 2006. SoxD proteins influence multiple stages of oligodendrocyte development and modulate SoxE protein function. Dev. Cell 11:697-710. [DOI] [PubMed] [Google Scholar]
- 25.Stolt, C. C., S. Schmitt, P. Lommes, E. Sock, and M. Wegner. 2005. Impact of transcription factor Sox8 on oligodendrocyte specification in the mouse embryonic spinal cord. Dev. Biol. 281:323-331. [DOI] [PubMed] [Google Scholar]
- 26.Weigle, B., R. Ebner, A. Temme, S. Schwind, M. Schmitz, A. Kiessling, M. A. Rieger, G. Schackert, H. K. Schackert, and E. P. Rieber. 2005. Highly specific overexpression of the transcription factor SOX11 in human malignant gliomas. Oncol. Rep. 13:139-144. [PubMed] [Google Scholar]
- 27.Willem, M., A. N. Garratt, B. Novak, M. Citron, S. Kaufmann, A. Rittger, B. DeStrooper, P. Saftig, C. Birchmeier, and C. Haass. 2006. Control of peripheral nerve myelination by the beta-secretase BACE1. Science 314:664-666. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.