Abstract
Objective
To determine the effect of new, persistent or reverted ischaemic ECG abnormalities at ages 50 and 70 years on the risk of subsequent cardiovascular disease.
Design, setting and participants
A prospective community‐based observational cohort of 50‐year‐old men in Sweden, followed for 32 years. 2322 men of age 50 years participated in 1970–3, and 1221 subjects were re‐examined at the age of 70 years.
Main outcome measures
Myocardial infarction (MI), cardiovascular mortality and overall mortality.
Results
At 50 years of age, after adjusting for established conventional risk factors, T wave abnormalities, ST segment depression, major Q/QS pattern and ECG‐left ventricular hypertrophy were all found to be independent risk factors for the main outcome measures during the 32 years of follow‐up. When ECG variables were re‐measured at 70 years of age, they were still found to be independent risk factors for the mortality outcomes, but lost in significance for prediction of MI. Regarding mortality, it was twice as dangerous to have persistent T wave abnormalities (HR 4.63; 95% CI 2.18 to 9.83) or ST segment depression (HR 5.66; 95% CI 1.77 to 18.1), as with new T wave abnormalities (HR 2.20; 95% CI 1.48 to 3.29) or ST segment depression (HR 2.55; 95% CI 1.74 to 3.75), developing between ages 50 and 70 years. The addition of “ECG indicating ischaemia” significantly increased the predictive power of the Framingham score (p<0.001).
Conclusions
It is worthwhile to obtain serial ECGs for proper risk assessment, since persistent ST‐T abnormalities carried twice as high a risk for future mortality compared with new or reverted abnormalities.
The existing literature has consistently shown that the resting ECG carries important independent prognostic information for future cardiac events. ECG indicating ischaemia,1 abnormal Q/QS patterns2 and ST‐T abnormalities1,3,4,5,6 have been consistently associated with an increased risk of cardiovascular disease (CVD) events, with most studies reporting a doubled relative risk. Also, ECG‐left ventricular hypertrophy (ECG‐LVH), usually based on a voltage criteria accompanied by ST segment depression or characteristic “strain” pattern, has been associated with an increased risk for CVD morbidity and mortality.7 De Bacquer et al1 have shown that the prognostic value of major ECG findings for CVD and coronary artery disease is more powerful than established conventional risk factors.
Many pathological ECG findings, such as ST‐T abnormalities3 and abnormal Q/QS patterns,8 may be transient. One year after a Q wave myocardial infarction (MI), the Q wave persisted in 73% of cases,8 and those with a persistent Q wave had worse prognosis, compared with subjects in whom ECG normalised. Regression of ECG‐LVH has been associated with a lower risk for future cardiovascular events.9 On the contrary, the risk of death over a 29‐year follow‐up period was higher for subjects in whom repeated ECG examination 5 years after initial investigation showed persistent minor ST‐T abnormalities, than for those in whom ECG had normalised.3 There are not many longitudinal studies with long follow‐up and repeated ECG examinations: one is the Manitoba study10 with 30 years of follow‐up and with ECG examinations every 3–5 years. However, information on the prognostic significance of persistent versus reverted ECG findings from longitudinal studies, with serial ECG tracings, is still limited.
Our hypothesis was that ECG‐LVH and “ECG indicating ischaemia” on resting ECG, including Q/QS patterns, left bundle branch block (LBBB) and ST‐T abnormalities, may be sensitive indicators of cardiac damage, and that subjects with persistent ECG abnormalities would have a higher risk for future CVD, independently of established conventional risk factors, compared with subjects developing ECG abnormalities de novo or those in whom ECG abnormalities normalised over time.
Using the Uppsala Longitudinal Study of Adult Men, our primary objective was to determine how persistent versus new or reverted ECG ischaemic abnormalities at age 70 years affected the risk of fatal and non‐fatal MI, CVD mortality, and all‐cause mortality.
Methods
Study sample
In 1970–3, all 50‐year‐old men, born in 1920–4 and residing in Uppsala, Sweden, were invited to a health survey aimed at identifying the risk factors for cardiovascular disease.11 Of the invited subjects, 2322 (82%) participated. Twenty years later, eligible participants were invited for re‐examination at age 70 years. Of these, 1221 (73%) participated. During the first 20‐year period, 422 had died and 219 had moved out of the region. Of the 1681 subjects invited to the 70‐year survey, 1221 participated. ECGs are available for 1139 of the subjects participating at the age of 70 years. All subjects gave informed consent, and the Ethics Committee, Faculty of Medicine, Uppsala University, Uppsala, Sweden, approved the study. The censored date was 31 December 2003. Excluded subjects were the six subjects with pacemaker at age 70 years, and, for assessment of the association of ECG findings with subsequent fatal or non‐fatal MI, subjects hospitalised due to MI before the 50‐year examination (n = 7).
Electrocardiography
During the surveys at age 50 and 70 years, a 12‐lead resting ECG was recorded. The following ECG abnormalities were coded according to the Minnesota Code (MC) published in 196012: major Q/QS pattern (1.1), minor Q/QS pattern (1.2 and 1.3), ST segment depression (4.1–4.2), T wave abnormality (5.1–5.3), LBBB (7.1), right bundle branch block (7.2), atrioventricular (AV)‐block 1 (6.3), atrial fibrillation/flutter (8.3) and high R‐amplitude (3.1/3.3). In this study, ECG‐LVH was based on high R‐wave amplitude (3.1/3.3), accompanied by ST segment depression (4.1–4.2). The variable ECG indicating ischaemia included codes 1.1–1.3, 4.1–4.2, 5.1–5.3 and 7.1. ECGs were classified as normal in the absence of MC 1.1–1.3, 3.1/3.3, 4.1–4.2, 5.1–5.3, 6.3, 7.1, 7.2 and 8.3.
The revised MC published in 1982,13 which requires that 4.1–4.3 should be accompanied by a 5 code, was not followed, and mutually exclusive groups were not created upon coding.
Interobserver agreement on classification was assessed on a subsample (n = 100) of ECGs, and the percentage agreement for coding was ⩾92%, for all variables.
Covariates
The investigations have been described extensively elsewhere (http://www.pubcare.uu.se/ULSAM/).11 Blood pressure was measured in the recumbent position after a 10 min rest with a mercury manometer. Diabetes mellitus was defined as fasting plasma glucose ⩾7.0 mmol/l14 and/or pharmacological treatment for diabetes mellitus. The Framingham score was determined15 and used to represent the conventional risk factors. The Framingham score was selected since it is a score that is internationally well recognised, it also includes diabetes mellitus as a risk factor and can be used up to the age of 70 years.
Follow‐up and outcomes
All men were followed for first occurrence of fatal and non‐fatal MI, mortality due to CVD, and all‐cause mortality due to any change, using the Swedish cause‐of‐death registry and the hospital discharge registry data. MI was defined according to the International Classification of Disease 9th revision as code 410 or according to the 10th revision as code I21 and CVD was defined as code 390–459 or I00–I99, respectively. The two different baselines considered were: the examination dates of the 50‐year and the 70‐year survey. The total follow‐up period was 32 years (censored date 31 December 2003) after the 50‐year survey. After the 70‐year‐survey, the total follow‐up period was 12 years. Results from a 12‐year follow‐up period after the 50‐year survey were also calculated.
Statistical analyses
The statistical analyses were carried out using Stata V 8.0. Associations between ECG abnormalities at age 50 and 70 years and the three different outcomes, were analysed by Cox's proportional hazard regression model. Two sets of Cox proportional hazards were performed with baseline at age 50 years and at age 70 years, respectively, using all follow‐up time available from each baseline, 32 and 12 years, respectively. A third set of analyses with 12 years of follow‐up from baseline at age 50 years was also performed. All results were adjusted for age at entry, as customary. However, the age distribution around the average age at each baseline was very narrow, thus results were very similar even without age adjustment. The Framingham score15 was included in the multivariate Cox regression analyses, together with the different ECG abnormalities, to determine whether the ECG abnormalities were associated with the three outcome measures, independently of the major cardiovascular risk factors. To exclude the influence of Q wave MI, when evaluating ST‐T abnormalities and ECG‐LVH, the major Q/QS pattern (1.1) was included as a confounding variable in the model. LBBB was excluded from all analyses, since it is an independent predictor of mortality.16 Participants with LBBB were few (5 and 18 subjects from the 50‐ and 70‐year examinations, respectively), and also in our study the presence of LBBB was a predictor of CVD mortality. In univariate analyses, at age 50 years, LBBB was a predictor of CVD mortality (HR 4.32, 95% CI 1.62 to 11.6), and at 70 years, LBBB was a predictor of overall mortality (HR 2.71, 95% CI 1.44 to 5.09), CVD mortality (HR 4.21, 95% CI 1.97 to 9.02), and fatal and non‐fatal MI (HR 4.16, 95% CI 1.69 to 10.2).
The estimated relative risks that were calculated referred to the presence or absence of the particular ECG abnormality under consideration: however, for ECG indicating ischaemia, analyses were also carried out in relation to normal ECG. The total number of ECGs was reduced from 2322 ECGs to 1940, when excluding 382 ECGs that did not fulfil the criteria for normal ECG. For the 70‐year examination, the total number of ECGs were reduced from 1139 to 798, when excluding 341 ECGs that did not fulfil the criteria for normal ECG. Results comparing the presence of a particular ECG abnormality with either absence of the same ECG abnormality or a normal ECG gave very similar results.
Analyses on persistent ECG abnormalities included subjects in whom the abnormality had been present both at age 50 and at age 70 years. Analyses on new abnormalities excluded subjects who already had the abnormality being studied at age 50 years. For analyses on reverted ECG, the subjects included were those in whom the ECG abnormality had been present at age 50 years, but had reverted at 70 years. Inspection of log–log survival curves for each outcome confirmed the assumption of proportional hazards.
In order to investigate the added value of the resting ECG, we tested the hypothesis that at age 70 years, Framingham score and ECG indicating ischaemia predicted CVD mortality better than the Framingham score alone, by comparing areas under the receiver‐operating characteristic (ROC) curves (C‐statistics).
Results
Baseline characteristics of conventional cardiovascular risk factors
Table 1 presents the baseline characteristics of conventional cardiovascular risk factors at 50‐ and 70‐year baseline investigations. The mean (SD) Framingham score at age 50 and 70 years was 7.0 (1.7) and 10.1 (1.6), respectively.
Table 1 Baseline characteristics of conventional cardiovascular risk factors at 50‐ and 70‐year baseline investigations.
| Characteristics | Mean (SD) at 50 years | n | Mean (SD) at 70 years | n |
|---|---|---|---|---|
| Age (years) | 49.6 (0.6) | 2322 | 71.0 (0.6) | 1221 |
| SBP supine (mm Hg) | 133.1 (18.1) | 2321 | 146.8 (18.5) | 1216 |
| Fasting glucose (mmol/l)* | 5.5 (1.0) | 2314 | 5.8 (1.5) | 1219 |
| Serum total cholesterol (mmol/l) | 6.9 (1.3) | 2322 | 5.8 (1.0) | 1220 |
| HDL cholesterol (mmol/l) | 1.4 (0.4) | 1880 | 1.3 (0.3) | 1218 |
| LDL cholesterol (mmol/l) | 5.3 (1.3) | 1880 | 3.9 (0.9) | 1214 |
| Body mass index (kg/m2) | 25.0 (3.2) | 2322 | 26.3 (3.4) | 1215 |
| Prevalence of smoking (%) | 51.1 | 2322 | 20.8 | 1139 |
HDL, high‐density lipoprotein; LDL, low‐density lipoprotein; SBP, systolic blood pressure.
*Data given are plasma concentrations (blood‐glucose concentrations at 50 years of age multiplied by a conversion factor of 1.11 to be comparable to plasma, according to IFCC recommendations.17
Regression of ECG abnormalities
At the 70‐year examination, 329 (29%) had new ECG indicating ischaemia, 40 (3.5%) had persistent ECG indicating ischaemia, while 11 (1%) had lost the ischaemic pattern observed at previous examination (fig 1).
Figure 1 Prevalence of ECG indicating ischaemia at 50‐ and 70‐year investigations. Only those participants with ECG available at both investigations are included.
ECGs from the 50‐year examination
The incidence of fatal and non‐fatal MI, CVD mortality and all‐cause mortality were higher for subjects with pathological ECG findings at the 50‐year examination than for those with normal ECG (tables 2 and 3). Even though the prevalence of ECG abnormalities was low, T wave abnormalities, ST segment depression, major Q/QS pattern, ECG‐LVH and ECG indicating ischaemia had a long‐term prognostic value for subsequent CVD mortality and all‐cause mortality, independently of Framingham score and major Q/QS patterns (tables 2 and 3). Also, at age 50 years, ECG indicating ischaemia had a long‐term prognostic value for subsequent fatal and non‐fatal MI (HR 2.27, 95% CI 1.72 to 3.00). Even though there were only eight subjects with major Q/QS pattern at the 50‐year examination, these ECG findings were the most strongly associated with subsequent CVD events (table 2).
Table 2 CVD mortality and all‐cause mortality in relation to ECG findings at 50 years (adjusted for age at baseline) during a follow‐up period of 32 years.
| Age 50 years (32 years of follow‐up) | Deaths due to CVD (n = 533) | All‐cause deaths (n = 1150) | ||||||
|---|---|---|---|---|---|---|---|---|
| Inc rates | RR (95% CI) | Adjusted RR (95% CI) | Inc rates | RR (95% CI) | Adjusted RR (95% CI) | |||
| T, n = 136 (5.9%) | 23.1 | 3.27 (2.54 to 4.21)* | 2.75 (2.11 to 3.59)*† | 32.4 | 2.03 (1.65 to 2.49)* | 1.79 (1.44 to 2.22)*† | ||
| ST, n = 53 (2.3%) | 31.9 | 4.22 (2.93 to 6.07)* | 2.98 (2.00 to 4.43)*† | 41.9 | 2.62 (1.92 to 3.57)* | 2.07 (1.48 to 2.85)*† | ||
| Isolated T, n = 98 (3.9%) | 18.5 | 2.61 (1.89 to 3.61)* | 2.26 (1.62 to 3.17)*† | 27.5 | 1.69 (1.30 to 2.20)* | 1.55 (1.18 to 2.03)†‡ | ||
| Q, n = 8 (0.3%) | 65.4 | 12.69 (5.99 to 26.9)* | 13.77 (6.50 to 29.2)*§ | 74.8 | 6.95 (3.46 to 13.97)* | 7.32 (3.64 to 14.71)*§ | ||
| LVH, n = 27 (1.2%) | 35.2 | 4.71 (2.90 to 7.64)* | 3.06 (1.79 to 5.23)*† | 47.6 | 2.95 (1.95 to 4.45)* | 2.19 (1.40 to 3.41)†‡ | ||
| Ischaemia vs non‐ischaemia, n = 157 (6.8%) | 21.9 | 3.11 (2.45 to 3.97)* | 2.77 (2.15 to 3.55)*§ | 31.6 | 1.98 (1.63 to 2.41)* | 1.85 (1.51 to 2.26)*§ | ||
| Ischaemia vs normal n = 92 (4.7%) | 23.3 | 3.29 (2.41 to 4.49)* | 2.95 (2.13 to 4.08)*§ | 42.9 | 1.89 (1.46 to 2.45)* | 1.78 (1.36 to 2.33)*§ | ||
| Normal ECG, n = 2061 (88.8%) | 7.8 | 0.47 (0.38 to 0.58)* | 0.48 (0.39 to 0.60)*§ | 17.9 | 0.66 (0.56 to 0.78)* | 0.67 (0.57 to 0.79)*§ | ||
CVD, cardiovascular disease; Inc rates, incidence/mortality rate for 1000 patient‐years of follow‐up; Ischaemia, ECG indicating ischaemia (1.1–1.3, 4.1–4.2, 5.1–5.3, 7.1); Isolated T, subjects with abnormal T waves but normal ST segment; LVH, left ventricular hypertrophy (ECG‐LVH (3.1/3.3 and 4.1–4.2)); MI, myocardial infarction; Normal, normal ECG (absence of 1.1–1.3, 3.1/3.3, 4.1–4.2, 5.1–5.3, 6.3, 7.1, 7.2 and 8.3); Q, major Q/QS pattern (1.1); ST, ST segment depression (4.1–4.2); T, negative T wave (5.1–5.3).
RR refers to the presence and absence of each particular ECG abnormality.
RR for normal ECG refers to the ratio between normal ECG (defined as absence of 1.1–1.3, 3.1/3.3, 4.1–4.2, 5.1–5.3, 6.3, 7.1, 7.2 or 8.3) and abnormal ECG.
*p<0.001.
†Multivariate Cox regression with adjustment for Framingham score and major Q/QS pattern (1.1). Subjects with left bundle branch block (LBBB) were dropped from multivariate analyses.
‡p<0.01.
§Multivariate Cox regression with adjustment for Framingham score. Subjects with LBBB were dropped from multivariate analyses.
Table 3 Cardiovascular disease mortality and all‐cause mortality in relation to ECG findings at age 50 and 70 years (adjusted for age at baseline).
| Age 50 years (12 years of follow‐up) | Deaths CVD, n = 80 | All‐cause deaths, n = 182 | ||||
|---|---|---|---|---|---|---|
| Inc rates | RR (95% CI) | Adjusted RR (95% CI) | Inc rates | RR (95% CI) | Adjusted RR (95% CI) | |
| T, n = 136 (5.9%) | 13.4 | 5.75 (3.47 to 9.55)* | 4.40 (2.54 to 7.62)* † | 17.4 | 2.87 (1.90 to 4.35)* | 2.32 (1.48 to 3.64)†§ |
| ST, n = 53 (2.3%) | 13.0 | 10.2 (5.63 to 18.5)* | 6.56 (3.34 to 12.9)*† | 16.9 | 5.37 (3.25 to 8.84)* | 3.93 (2.26 to 6.83)*† |
| Isolated T, n = 98 (3.9%) | 6.9 | 2.90 (1.33 to 6.35)‡ | 2.21 (0.95 to 5.17)† | 9.8 | 1.61 (0.85 to 3.05) | 1.37 (0.70 to 2.71)† |
| Q, n = 8 (0.3%) | 28.0 | 15.5 (4.86 to 49.6)* | 16.5 (5.12 to 53.2)*§ | 37.4 | 8.58 (3.17 to 23.2)* | 8.78 (3.24 to 23.8)*§ |
| LVH, n = 27 (1.2%) | 18.6 | 14.3 (7.16 to 28.7)* | 8.70 (4.01 to 18.8)*† | 22.8 | 7.22 (3.92 to 13.3)* | 5.05 (2.60 to 9.84)*† |
| Ischaemia vs non‐ischaemia n = 157 (6.8%) | 5.97 | 5.28 (3.21 to 8.68)* | 4.40 (2.62 to 7.39)*§ | 8.2 | 2.81 (1.89 to 4.18)* | 2.54 (1.69 to 3.82)*§ |
| Ischaemia vs normal n = 92 (4.7%) | 5.3 | 5.35 (2.76 to 10.4)* | 4.48 (2.22 to 9.03)*§ | 7.7 | 2.67 (1.59 to 4.50)* | 2.45 (1.43 to 4.20)‡§ |
| Normal ECG n = 2061 (88.8%) | 1.0 | 0.30 (0.19 to 0.49)* | 0.33 (0.21 to 0.55)*§ | 2.7 | 0.53 (0.37 to 0.77)‡ | 0.56 (0.38 to 0.82)‡§ |
| Age 70 (12 years of follow‐up) | Deaths CVD, n = 139 | All‐cause deaths, n = 309 | ||||
|---|---|---|---|---|---|---|
| Inc rates | RR (95% CI) | Adjusted RR (95% CI) | Inc rates | RR (95% CI) | Adjusted RR (95% CI) | |
| T, n = 182 (16.1%) | 27.5 | 2.75 (1.93 to 3.93)* | 2.34 (1.61 to 3.39)*† | 37.3 | 1.40 (1.06 to 1.86)¶ | 1.28 (0.96 to 1.71)† |
| ST, n = 163 (14.4%) | 27.3 | 2.64 (1.83 to 3.82)* | 2.65 (1.83 to 3.84)*† | 40.3 | 1.54 (1.16 to 2.04)‡ | 1.53 (1.15 to 2.03)†‡ |
| Isolated T, n = 106 (11%) | 23.3 | 3.07 (1.93 to 4.88)* | 2.73 (1.69 to 4.43)*† | 42.9 | 1.43 (1.00 to 2.06) | 1.33 (0.92 to 1.94)† |
| Q, n = 104 (9.2%) | 36.2 | 3.25 (2.19 to 4.81)* | 3.29 (2.21 to 4.88)* § | 51.6 | 1.95 (1.43 to 2.66)* | 1.95 (1.43 to 2.69)*§ |
| LVH, n = 73 (6.4%) | 24.4 | 2.07 (1.23 to 3.50)‡ | 2.31 (1.36 to 3.92)†‡ | 33.5 | 1.21 (0.78 to 1.87) | 1.27 (0.82 to 1.96)† |
| Ischaemia vs non‐ischaemia, n = 351 (31%) | 24.2 | 3.21 (2.29 to 4.50)* | 2.91 (2.06 to 4.11)*§ | 37.7 | 1.56 (1.24 to 1.96)* | 1.45 (1.15 to 1.84) ‡§ |
| Ischaemia vs normal, n = 227 (28.5%) | 24.1 | 3.94 (2.58 to 6.00)* | 3.63 (2.35 to 5.61)*§ | 43.5 | 1.81 (1.37 to 2.39)* | 1.69 (1.27 to 2.25)*§ |
| Normal ECG, n = 633 (55.6%) | 7.6 | 0.36 0.25 to 0.55)* | 0.38 (0.27 to 0.54)*§ | 23.5 | 0.63 (0.50 to 0.78)* | 0.65 (0.52 to 0.81)*§ |
CVD, cardiovascular disease; Inc rates, Incidence/mortality rate for 1000 patient‐years of follow‐up; Ischaemia, ECG indicating ischaemia (1.1–1.3, 4.1–4.2, 5.1–5.3, 7.1); Isolated T, subjects with abnormal T waves but normal ST‐segment LVH, left ventricular hypertrophy (ECG‐LVH (3.1/3.3 and 4.1–4.2)); MI, myocardial infarction; Normal, normal ECG (absence of 1.1–1.3, 3.1/3.3, 4.1–4.2, 5.1–5.3, 6.3, 7.1, 7.2 and 8.3); Q, major Q/QS pattern (1.1); ST, ST segment depression (4.1–4.2); T, negative T wave (5.1–5.3).
RR refers to the presence and absence of each particular ECG abnormality.
RR for normal ECG refers to the ratio between normal ECG (defined as absence of 1.1–1.3, 3.1/3.3, 4.1–4.2, 5.1–5.3, 6.3, 7.1, 7.2 or 8.3) and abnormal ECG.
Follow‐up period for both age 50 and age 70 years is 12 years.
*p<0.001.
†Multivariate Cox regression with adjustment for Framingham score and major Q/QS pattern (1.1). Subjects with left bundle branch block (LBBB) were dropped from multivariate analyses.
‡p<0.01.
§Multivariate Cox regression with adjustment for Framingham score. Subjects with LBBB were dropped from multivariate analyses.
¶p<0.05.
ECGs from the 70‐year examination
Subjects with pathological ECG findings at the 70‐year examination had higher CVD mortality and all‐cause mortality, even after adjusting for the Framingham score and major Q/QS patterns (1.1; tables 2 and 3). When ECGs were re‐measured at age 70 years, they lost in significance for prediction of MI. At age 70 years, ECG indicating ischaemia did not predict fatal and non‐fatal MI (HR 0.92, (95% CI 0.52 to 1.62). The only ECG variables significantly associated with future MI were persistent T wave abnormalities and persistent ECG indicating ischaemia.
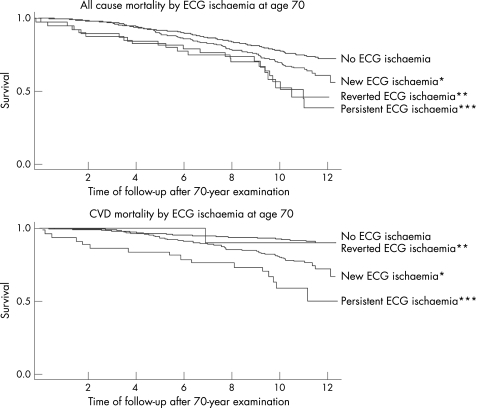
In general, persistent T wave abnormalities (HR 4.63, 95% CI 2.18 to 9.83), ST segment depression (HR 5.66, 95% CI 1.77 to 18.1) or ECG indicating ischaemia (HR 5.19, 95% CI 2.70 to 9.97) had twice the risk for developing future CVD mortality or all‐cause mortality compared with new T wave abnormalities (HR 2.20, 95% CI 1.48 to 3.29), ST segment depression (HR 2.55, 95% CI 1.74 to 3.75) or ECG indicating ischaemia (HR 2.72, 95% CI 1.90 to 3.90). Regression of the ECG abnormality improved the prognosis (table 4 and fig 2). Due to the low power for analysis of persistent and reverted ECG‐LVH and major Q/QS patterns, we did not pursue analyses of these variables further.
Table 4 RRs for myocardial infarction, cardiovascular disease (CVD) and all‐cause mortality by new, persistent and reverted T wave abnormality (5.1–5.3), ST‐segment depression (4.1–4.2) and ECG indicating ischaemia at baseline at 70‐year examination.
| ECG variable | CVD mortality | All‐cause mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Inc rates | RR | 95% CI | p Value | Inc rates | RR | 95% CI | p Value | |
| T wave abnormality | ||||||||
| Unadjusted* (n = 1139) | ||||||||
| New (n = 161) | 25.3 | 2.68 | 1.81 to 3.92 | <0.001 | 34.8 | 1.34 | 0.99 to 1.82 | 0.06 |
| Persistent (n = 21) | 46.5 | 5.30 | 2.56 to 11.0 | <0.001 | 58.1 | 2.30 | 1.22 to 4.33 | 0.01 |
| Reverted (n = 22) | 38.7 | 4.55 | 2.10 to 9.85 | <0.001 | 60.8 | 2.44 | 1.33 to 4.46 | 0.004 |
| Adjusted† (n = 1139) | ||||||||
| New (n = 161) | 2.20 | 1.48 to 3.29 | <0.001 | 1.21 | 0.89 to 1.65 | 0.2 | ||
| Persistent (n = 21) | 4.63 | 2.18 to 9.83 | <0.001 | 2.17 | 1.14 to 4.11 | 0.02 | ||
| Reverted (n = 22) | 2.40 | 0.85 to 6.78 | 0.1 | 1.49 | 0.69 to 3.22 | 0.3 | ||
| ST segment depression | ||||||||
| Unadjusted* (n = 1139) | ||||||||
| New (n = 157) | 26.6 | 2.56 | 1.76 to 3.75 | <0.001 | 39.6 | 1.52 | 1.14 to 2.03 | 0.005 |
| Persistent (n = 6) | 62.5 | 5.80 | 1.83 to 18.3 | 0.003 | 62.5 | 2.36 | 0.76 to 7.37 | 0.1 |
| Reverted (n = 8) | 30.3 | 2.77 | 0.68 to 11.2 | 0.2 | 60.6 | 2.23 | 0.83 to 5.98 | 0.1 |
| Adjusted† (n = 1139) | ||||||||
| New (n = 157) | 2.55 | 1.74 to 3.75 | <0.001 | 1.51 | 1.13 to 2.02 | 0.006 | ||
| Persistent (n = 6) | 5.66 | 1.77 to 18.1 | 0.003 | 2.38 | 0.76 to 7.46 | 0.1 | ||
| Reverted (n = 8) | 1.74 | 0.24 to 12.5 | 0.6 | 1.34 | 0.33 to 5.39 | 0.7 | ||
| ECG indicating ischaemia | ||||||||
| Unadjusted* (n = 1139) | ||||||||
| New (n = 329) | 21.9 | 2.90 | 2.04 to 4.14 | <0.001 | 35.5 | 1.47 | 1.16 to 1.87 | 0.002 |
| Persistent (n = 40) | 45.6 | 6.21 | 3.51 to 11.0 | <0.001 | 57.8 | 2.43 | 1.51 to 3.90 | <0.001 |
| Reverted (n = 11) | 9.7 | 1.40 | 0.19 to 10.1 | 0.7 | 50.3 | 1.64 | 0.72 to 3.70 | 0.2 |
| Adjusted ‡ (n = 1139) | ||||||||
| New (n = 329) | 2.72 | 1.90 to 3.90 | <0.001 | 1.41 | 1.10 to 1.80 | 0.006 | ||
| Persistent (n = 40) | 5.19 | 2.70 to 9.97 | <0.001 | 1.97 | 1.14 to 3.40 | 0.02 | ||
| Reverted (n = 11) | 1.30 | 0.18 to 9.48 | 0.8 | 1.58 | 0.70 to 3.58 | 0.3 | ||
Inc rates, incidence/mortality rate for 1000 patient‐years of follow‐up.
*Adjusted for age at entry.
†Multivariate Cox regression with adjustment for age at entry, Framingham score and major Q/QS pattern (1.1). Subjects with left bundle branch block (LBBB) were dropped from multivariate analyses.
‡Multivariate Cox regression with adjustment for age at entry and Framingham score. Subjects with LBBB were dropped from multivariate analyses.
Figure 2 Kaplan–Meier plots of the effects of persistent, new and reverted ECG indicating ischaemia at age 70 years for all‐cause mortality and mortality due to cardiovascular disease (CVD) during 12 years of follow‐up. (Note: There was only one ECG with reverted ischaemia for CVD mortality; p value not possible to calculate). *p value <0.01 compared with “no ECG ischaemia”. **For overall mortality: p value <0.01 compared with “no ECG ischaemia”. ***p value <0.01 compared with “no ECG ischaemia”.
ROC curves for CVD mortality
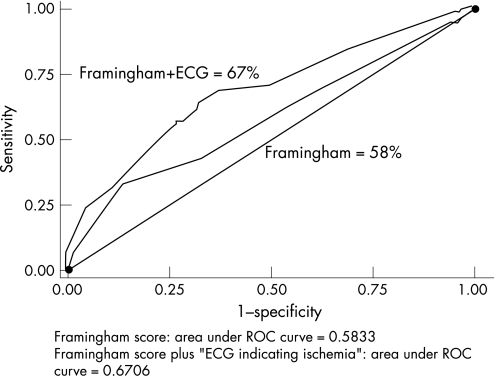
Comparison of area under ROC curves for (1) Framingham score alone and (2) Framingham score and ECG indicating ischaemia, for prediction of CVD mortality, over the 12 years after the 70‐year examination, showed that the addition of ECG indicating ischaemia significantly increased the predictive power of the Framingham score (areas under ROC curves: 0.58 vs 0.67; p<0.001; fig 3).
Figure 3 Receiver‐operating characteristic curves showing the performance of Framingham score alone, and Framingham score and ECG indicating ischaemia, in predicting CVD mortality over 12 years of follow‐up, after the 70‐year examination. p for difference between curves <0.001.
Discussion
The present study confirmed the hypothesis that it was more dangerous to have persistent T wave abnormalities, ST segment depression or ECG indicating ischaemia compared with new or reverted abnormalities at age 70 years. At age 50 years, also after adjusting for conventional risk factors, T wave abnormalities, ST segment depression, major Q/QS pattern, ECG‐LVH and ECG indicating ischaemia were all independent risk factors for first MI event, CVD mortality and all cause mortality. These ECG variables, were also independent risk factors for CVD and all‐cause mortality at age 70 years, but lost in significance regarding MI.
Study strengths and limitations
Our study has not taken into consideration the effect of treatment of cardiovascular risk factors over time, even though it is reasonable to assume that regression of ECG abnormalities is the result of different pharmaceutical and non‐pharmaceutical interventions, aimed at reducing the impact of cardiovascular risk factors.
An obvious limitation is the lack of women in the study. However, studies have found that the predictive value of ECG findings are comparable between men and women.1 There was no validation of the outcome detection system. However, combining data from the Cause of Death Registry and In‐Patient Registry in Sweden has been shown to be an efficient, validated alternative to revised hospital discharge notes and death certificates.18,19
Strengths of the Uppsala Longitudinal Study of Adult Men study include its large study population, repeated investigations, its long follow‐up period, low number of subjects lost to follow‐up and its longitudinal design. Since explanatory variables change over time, a shorter re‐examination interval between the ages of 50 years and 70 years would have been interesting from a clinical point of view. However, such examination with ECGs did not take place. Other studies with long follow‐up and repeated ECG examination include the Chicago Western Electric Study,3 the Framingham Heart Study8 and the Manitoba Study,10 but none of these studies have compared the prognostic value of persistent versus reverted major ischaemic ECG abnormalities within the same cohort.
The Framingham score was used to adjust for confounders and to investigate the added value of the resting ECG in predicting CVD mortality. The use of the score developed for Europeans by the Systemic COronary Risk Evaluation project20 could be an alternative to the Framingham score, since it has been validated in a European population and the end point is more in accordance with our analysis. However, the Systemic COronary Risk Evaluation risk score does not include diabetes mellitus, and its validation beyond the age of 70 years has not been published. For CVD mortality, our major outcome, adjustment for the individual risk factors included in the Framingham score, gave essentially the same results as using the Framingham score.
Risk associated with ECG indicating ischaemia
In agreement with other studies, ECG indicating ischaemia was independently associated with CVD mortality. However, it has not been shown previously that, in older people, a persistent ECG indicating ischaemia has a fivefold increased risk of CVD mortality, in comparison with a twofold increased risk associated with a newly diagnosed ECG indicating ischaemia.
Risk associated with ST‐T abnormalities
Consistently with other studies, ST‐T abnormalities were related to CVD events in both middle‐aged and older men. If persistent, they were twice as dangerous as new ECG abnormalities, and regression improved the prognosis.
Lancellotti et al21 are among the few other authors who have addressed the long‐term prognostic significance of persistent versus transient negative T waves after acute MI. They have shown that persistent negative T waves have a poorer long‐term prognosis than patients with either early or late T wave normalisation. The evolutionary changes of negative T waves after acute MI are a reflection of the myocardial state, with delayed T wave normalisation occurring mainly in patients with viable but jeopardised or hibernating myocardium.22
Risk associated with Q/QS patterns
In our study, the ECG pattern classified as MC 1.1 was selected to represent major Q/QS patterns, since 1.1 patterns are more often associated with necropsy evidence of cardiac infarction in older people compared with 1.2 or 1.3 patterns.23 Major Q/QS patterns increased with age, but, even though they predicted CVD mortality and all‐cause mortality, they did not predict MI at the age of 70 years, suggesting that in older people, previous Q/QS patterns may be less important for the development of the clinical MI.
The long‐term prognostic significance of persistent versus transient abnormal Q/QS patterns after acute MI is still controversial. Due to the low number of persistent Q/QS patterns at the age of 70 years (n = 1) in our cohort, we could not address this question.
Risk associated with LVH
Left ventricular mass may reflect cumulative life‐long exposure to cardiovascular insults from hypertension, obesity or from coronary artery disease, and, thereby, LVH assessment may provide a better estimate of the extent of cardiac end‐organ damage than is provided by casual measurement of conventional risk factors.24 However, even though specificity is high, the ECG is a poor screening test for echocardiographic LVH, since it identifies only a small fraction of subjects with echocardiographic hypertrophy.25 Despite low sensitivity, ECG‐LVH, as defined in our study, was associated with CVD mortality, which is consistent with other studies.26
The role of resting ECG and future perspectives
Different authors have questioned the prognostic and screening role of the resting ECG, especially in apparently healthy subjects.6,27 The presence of ST‐T abnormalities occurs not only with myocardial ischaemia and infarction but also with cardiomyopathy, pulmonary embolism, electrolyte abnormalities and drugs such as digitalis.28 The low prevalence of ECG abnormalities age <50 years, as well as the transient and reversible aspect of many ECG findings, has probably contributed to the ambiguous position that the resting ECG has had in the global assessment of cardiovascular risk. Our results show that repeated examination of ECG after the age of 50 years contributes to the identification of high‐risk groups. Due to its strong prognostic value for CVD events and all‐cause mortality, the resting ECG deserves a stronger role in cardiovascular risk assessment. Also, serial resting ECG, by identifying the reversibility of ECG abnormalities, could have a role in following the success of therapeutic intervention directed at myocardial ischaemia or established conventional risk factors, even though that has to be confirmed in future studies.
Conclusion
It is worthwhile obtaining serial ECGs for proper risk assessment, since persistent T wave abnormalities and ST segment depression carry twice as high a risk for future mortality as new or reverted abnormalities, and since inclusion of ECG indicating ischaemia significantly increased the predictive power of the Framingham score.
Acknowledgements
This work was funded by the Medical Faculty at Uppsala University, Royal Society of Science, Swedish Council for Planning and Co‐ordination of Research, Swedish National Association against Heart and Lung Disease, and Swedish Medical Research Council.
Abbreviations
CVD - cardiovascular disease
LBBB - left bundle branch block
LVH - left ventricular hypertrophy
MC - Minnesota Code
MI - myocardial infarction
ROC - receiver‐operating characteristics
Footnotes
Competing interests: AstraZeneca R&D, Sweden, also employs CSM and LL. AstraZeneca R&D, Sweden, had no role in the study design; in the collection, analysis and interpretation of data; in the writing of the manuscript; nor in the decision to submit the manuscript for publication.
References
- 1.De Bacquer D, De Backer G, Kornitzer M.et al Prognostic value of ECG findings for total, cardiovascular disease, and coronary heart disease death in men and women. Heart 199880570–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Caird F I, Campbell A, Jackson T F. Significance of abnormalities of electrocardiogram in old people. Br Heart J 1974361012–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Daviglus M L, Liao Y, Greenland P.et al Association of nonspecific minor ST‐T abnormalities with cardiovascular mortality: the Chicago Western Electric Study. JAMA 1999281530–536. [DOI] [PubMed] [Google Scholar]
- 4.Ostor E. Electrocardiographic findings and their association with mortality in the Copenhagen City Heart Study. Eur Heart J 19812317–328. [DOI] [PubMed] [Google Scholar]
- 5.Knutsen R. The predictive value of resting electrocardiograms for 12‐year incidence of coronary heart disease in the Honolulu heart program. J Clin Epidemiol 198841293–302. [DOI] [PubMed] [Google Scholar]
- 6.Ashley E A, Raxwal V K, Froelicher V F. The prevalence and prognostic significance of electrocardiographic abnormalities. Curr Probl Cardiol 2000251–72. [DOI] [PubMed] [Google Scholar]
- 7.Larsen C T, Dahlin J, Blackburn H.et al Prevalence and prognosis of electrocardiographic left ventricular hypertrophy, ST segment depression and negative T‐wave; the Copenhagen City Heart Study. Eur Heart J 200223268–270. [DOI] [PubMed] [Google Scholar]
- 8.Wong N D, Levy D, Kannel W B. Prognostic significance of the electrocardiogram after Q wave myocardial infarction. The Framingham Study. Circulation 199081780–789. [DOI] [PubMed] [Google Scholar]
- 9.Okin P M, Devereux R B, Jern S.et al Regression of electrocardiographic left ventricular hypertrophy during antihypertensive treatment and the prediction of major cardiovascular events. JAMA 20042922343–2349. [DOI] [PubMed] [Google Scholar]
- 10.Rabkin S, Mathewson F, Tate R. The electrocardiogram in apparently healthy men and the risk of sudden death. Br Heart J 198247546–552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hedstrand H. A study of middle‐aged men with particular reference to risk factors for cardiovascular disease. Ups J Med Sci Suppl 1975191–61. [PubMed] [Google Scholar]
- 12.Blackburn H, Keys A, Simonson E.et al The electrocardiogram in population studies. A classification system. Circulation 1960211160–1175. [DOI] [PubMed] [Google Scholar]
- 13.Prineas R, Crow R, Blackburn H.The Minnesota Code manual of electrocardiographic findings, standards and procedures for measuring and classification. Bristol, UK: John Wright, 1982
- 14.Alberti K G, Zimmet P Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabetes Med 199815539–553. [DOI] [PubMed] [Google Scholar]
- 15.Wilson P W, D'Agostino R B, Levy D.et al Prediction of coronary heart disease using risk factor categories. Circulation 1998971837–1847. [DOI] [PubMed] [Google Scholar]
- 16.Freedman R, Alderman E, Sheffield L.et al Bundle branch block in patients with chronic coronary artery disease: angiographic correlates and prognostic significance. J Am Coll Cardiol 19871073–80. [DOI] [PubMed] [Google Scholar]
- 17.D' Orazio P, Burnett R W, Fogh‐Andersen N.et al Approved IFCC recommendation on reporting results for blood glucose (abbreviated). Clin Chem 2005511573–1576. [DOI] [PubMed] [Google Scholar]
- 18.Merlo J, Lindblad U, Pessah‐Rasmussen H.et al Comparison of different procedures to identify probable cases of myocardial infarction and stroke in two Swedish prospective cohort studies using local and national routine registers. Eur J Epidemiol 200016235–243. [DOI] [PubMed] [Google Scholar]
- 19.Lindblad U, Rastam L, Ranstam J.et al Validity of register data on acute myocardial infarction and acute stroke: the Skaraborg Hypertension Project. Scand J Soc Med 1993213–9. [DOI] [PubMed] [Google Scholar]
- 20.Conroy R M, Pyorala K, Fitzgerald A P.et al Estimation of ten‐year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J 200324987–1003. [DOI] [PubMed] [Google Scholar]
- 21.Lancellotti P, Gerard P L, Kulbertus H E.et al Persistent negative T waves in the infarct‐related leads as an independent predictor of poor long‐term prognosis after acute myocardial infarction. Am J Cardiol 200290833–837. [DOI] [PubMed] [Google Scholar]
- 22.Pierard L A, Lancellotti P. Determinants of persistent negative T waves and early versus late T wave normalisation after acute myocardial infarction. Heart 2005911008–1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kurihara H, Kuramoto K, Terasawa F.et al Reliability of abnormal Q and QS patterns classified by the Minnesota code for the diagnosis of myocardial infarction in aged people. Jpn Heart J 19678514–521. [DOI] [PubMed] [Google Scholar]
- 24.Levy D, Garrison R J, Savage D D.et al Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med 19903221561–1566. [DOI] [PubMed] [Google Scholar]
- 25.Levy D, Labib S B, Anderson K M.et al Determinants of sensitivity and specificity of electrocardiographic criteria for left ventricular hypertrophy. Circulation 199081815–820. [DOI] [PubMed] [Google Scholar]
- 26.Larsen C T, Blackburn H, Bruun N E.et al The ischemic electrocardiogram: a harbinger for ischemic heart disease independent of the blood pressure level. The Copenhagen City Heart Study. Eur J Epidemiol 200520301–309. [DOI] [PubMed] [Google Scholar]
- 27.Sox H C, Jr, Garber A M, Littenberg B. The resting electrocardiogram as a screening test. A clinical analysis. Ann Intern Med 1989111489–502. [DOI] [PubMed] [Google Scholar]
- 28.Fisch C, Braunwald Electrocardiography. In: ed. Heart disease, a textbook of cardiovascular medicine, 5th edn Chapter 4, Philadelphia, PA: Saunders 1977108–152.