Abstract
Background
Whole‐body water immersion leads to a significant shift of blood from the periphery to the intrathoracic circulation, followed by an increase in central venous pressure and heart volume. In patients with severely reduced left ventricular function, this hydrostatically induced volume shift might overstrain the cardiovascular adaptive mechanisms and lead to cardiac decompensation.
Aim
To assess the haemodynamic response to water immersion, gymnastics and swimming in patients with chronic heart failure (CHF).
Methods
10 patients with compensated CHF (62.9 (6.3) years, ejection fraction 31.5% (4.1%), peak oxygen consumption (V̇o2) 19.4 (2.8) ml/kg/min), 10 patients with coronary artery disease (CAD) but preserved left ventricular function (57.2 (5.6) years, ejection fraction 63.9% (5.5%), peak V̇o2 28 (6.3) ml/kg/min), and 10 healthy controls (32.8 (7.2) years, peak V̇o2 45.6 (6) ml/kg/min) were examined. Haemodynamic response to thermoneutral (32°C) water immersion and exercise was measured using a non‐invasive foreign gas rebreathing method during stepwise water immersion, water gymnastics and swimming.
Results
Water immersion up to the chest increased cardiac index by 19% in controls, by 21% in patients with CAD and by 16% in patients with CHF. Although some patients with CHF showed a decrease of stroke volume during immersion, all subjects were able to increase cardiac index (by 87% in healthy subjects, by 77% in patients with CAD and by 53% in patients with CHF). V̇o2 during swimming was 9.7 (3.3) ml/kg/min in patients with CHF, 12.4 (3.5) ml/kg/min in patients with CAD and 13.9 (4) ml/kg/min in controls.
Conclusions
Patients with severely reduced left ventricular function but stable clinical conditions and a minimal peak V̇o2 of at least 15 ml/kg/min during a symptom‐limited exercise stress test tolerate water immersion and swimming in thermoneutral water well. Although cardiac index and V̇o2 are lower than in patients with CAD with preserved left ventricular function and controls, these patients are able to increase cardiac index adequately during water immersion and swimming.
Exercise in thermoneutral water has a long tradition in rehabilitative training institutions and has been used for many years in patients with coronary artery disease (CAD). Exercises to improve mobility, strength and cardiovascular fitness can easily be performed in water.
Whole‐body head‐up immersion leads to a significant shift of blood into the intrathoracic circulation, followed by an increase in central venous pressure, heart volume and cardiac output.1,2,3 Because this hydrostatically induced volume shift might overstrain the cardiovascular adaptive mechanisms in patients with heart failure and lead to left ventricular decompensation, recent guidelines state that patients with diastolic and systolic dysfunction should refrain from swimming.4
On the other hand, exposure to thermoneutral water leads to a number of physiological responses, which may be beneficial in patients with heart failure. Both systemic and pulmonary vascular resistance have been shown to decrease during bathing in warm water in these patients,5 and an improvement in the ventilation/perfusion ratio of the lungs6 may increase oxygen consumption (V̇o2). Water immersion leads to a reduction of renin, angiotensin II and aldosterone activity whereas increased release of atrial natriuretic peptide elicits natriuresis.7,8
The aim of this study was to evaluate cardiovascular adaptations in patients with stable chronic heart failure (CHF) during stepwise water immersion, water gymnastics and swimming, and to compare the results with those in patients with coronary artery disease (CAD) with preserved left ventricular function and in healthy controls. We hypothesised that patients with stable CHF and a functional class A or B (peak V̇o2 during a symptom‐limited cardiopulmonary exercise test >14 ml/kg/min), according to the Weber classification,9 are able to increase stroke volume and cardiac index during water immersion and to tolerate water gymnastics and swimming without symptoms of pulmonary congestion.
Methods
Patients
We examined 30 male subjects: 10 patients with stable CHF, 10 patients with CAD and preserved left ventricular function, and 10 healthy controls (table 1). Patients with heart failure included eight patients with ischaemic heart disease and two patients with idiopathic dilated cardiomyopathy. All patients were taking β‐blockers, whereas the controls were taking no drugs. Patients had to be swimmers and in a stable clinical condition. They were informed about the study procedure and written informed consent was obtained from them. The study protocol was reviewed and accepted by the local ethical committee.
Table 1 Characteristics of the study population.
| Characteristic | CHF (n = 10) | CAD (n = 10) | Healthy controls (n = 10) | p Value | ||
|---|---|---|---|---|---|---|
| CHF vs CAD | CHF vs healthy | CAD vs healthy | ||||
| Age (years) | 62.9 (6.3) | 57.2 (5.6) | 32.8 (7.2) | 0.063 | <0.001 | <0.001 |
| BMI (kg/m2) | 27.2 (3.4) | 26.4 (3.1) | 22.5 (2.1) | 0.739 | 0.002 | 0.003 |
| Peak V̇o2 (ml/kg/min) | 19.4 (2.8) | 28.0 (6.3) | 45.6 (6.0) | 0.020 | <0.001 | 0.003 |
| V̇o2 VT (ml/kg/min) | 13.5 (3.2) | 19.6 (6.2) | 26.4 (6.7) | 0.106 | <0.001 | 0.106 |
| Power output (W) | 124.8 (28.9) | 175.3 (43.3) | 289.1 (78.4) | 0.007 | <0.001 | <0.001 |
| ΔV̇o2/Δwatt | 8.1 (1.5) | 9.2 (0.4) | 10.0 (0.6) | 0.183 | 0.018 | 0.143 |
| Ejection fraction (%) | 31.5 (4.1) | 63.9 (5.5) | — | <0.001 | — | — |
| BNP (pg/ml) | 163 (98) | — | — | — | — | — |
BMI, body mass index; BNP, brain natriuretic peptide; CAD, coronary artery disease; CHF, chronic heart failure; V̇o2, oxygen consumption; V̇o2 VT, oxygen consumption at ventilatory threshold.
All values are mean (SD).
Experimental setting
The study was performed in the swimming pool of the physiotherapeutic facilities at the University Hospital of Bern, Bern, Switzerland. The water temperature was 32°C throughout the study.
Haemodynamic measurements were performed with an inert gas rebreathing method using an infrared photoacoustic gas analyser (Innocor, Innovision A/S, Odense, Denmark). The patient breathes a gas mixture containing two physiologically inert compounds in a closed rebreathing assembly, one being soluble in blood (N2O, 0.5%) and the other being insoluble in blood (SF6, 0.1%). When the blood‐soluble gas comes in contact with the blood in the lung capillaries, it is dissolved and washed out by the blood perfusing the lungs. In the absence of pulmonary shunts (defined as an arterial saturation >98%), the pulmonary blood flow is proportional to the rate of washout of the blood‐soluble compound, measured continuously by a gas analyser. The blood‐insoluble compound is used to determine the lung volume, which is also required in the equation used to calculate cardiac output from the measured washout curve of the blood‐soluble compound.10 Previous validations of the foreign gas rebreathing method showed that the method gives accurate measurements of cardiac output at both rest and exercise.10,11
Rebreathing was performed over 15 s with a gas volume of 3 litres. Heart rate, blood pressure and oxygen saturation of haemoglobin were measured simultaneously, and stroke volume, cardiac index and systemic vascular resistance were calculated.
As β‐blocker treatment is recommended by current guidelines in patients with CHF and CAD, and to allow for an optimal comparability of the haemodynamic measures, we only included patients who were taking β‐blocker treatment. The healthy subjects, however, were not treated.
Study protocol
An initial measurement was taken at rest at the border of the pool with the patient standing outside the water. Thereafter, the patient walked into the water step by step on a staircase. Additional measurements were taken at immersion to pelvis and chest, followed by the “jumping‐jack” exercise for 30 s and a 60 s swim along the edges of the pool. Haemodyamic measurements were taken three times at every stage and mean values were calculated.
Statistical analysis
Data analysis was performed using SPSS V.12.0 for Windows software. All data are expressed as mean (SD). Mean comparisons were effectuated using non‐parametric (Wilcoxon and Mann–Whitney U) tests. For bivariate correlation analysis, Pearson's correlation coefficient was calculated. A value of p<0.05 was considered significant.
Results
Heart rate
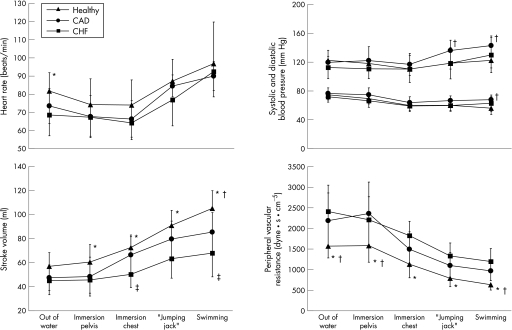
During stepwise water immersion up to the pelvis and chest, heart rate decreased from 69 (11) to 64 (9) beats/min in patients with CHF, from 74 (10) to 66 (10) beats/min in patients with CAD with preserved ventricular function, and from 82 (10) to 74 (14) beats/min in controls (fig 1). During swimming, heart rate increased to 93 (14) beats/min, 93 (16) beats/min and 97 (23) beats/min in patients with CHF, patients with CAD and controls, respectively. Compared with controls and patients with CAD, the decrease in heart rate during water immersion was proportionally lower in patients with CHF, whereas the rate response during water gymnastics and swimming paralleled the other two groups.
Figure 1 Haemodynamic adaptation to water immersion, water gymnastics and swimming in patients with stable chronic heart failure (CHF), patients with coronary artery disease (CAD) with preserved left ventricular systolic function and healthy controls (Healthy). *p<0.05 (healthy vs CHF); †p<0.05 (healthy vs CAD); ‡p<0.05 (CAD vs CHF).
Stroke volume
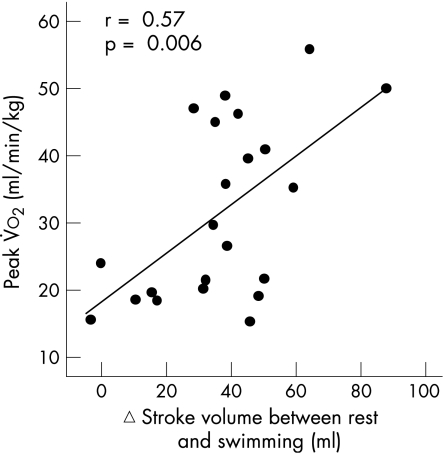
Water immersion up to the chest led to an increase in stroke volume of 30% in normal subjects and of 41% in the patients with CAD (fig 1). In patients with CHF, stroke volume increased by 17%. During swimming, stroke volume was increased further by 47% in controls, by 30% in patients with CAD and by 35% in patients with CHF. Changes in stroke volume showed a positive correlation with peak V̇o2 achieved during a maximal cardiopulmonary exercise test (r = 0.57; p = 0.006; fig 2).
Figure 2 Relationship between peak oxygen uptake V̇o2 during maximal cardiopulmonary exercise test and change (▵) in stroke volume between rest and swimming.
Blood pressure
Systolic blood pressure decreased from 122 (11) to 110 (9) mm Hg during water immersion in controls, from 119 (17) to 117 (15) mm Hg in patients with CAD, and from 113 (15) to 110 (19) mm Hg in patients with CHF. Diastolic pressure decreased from 75 (7) to 60 (7) mm Hg, from 77 (8) to 64 (8) mm Hg, and from 72 (8) to 59 (8) mm Hg, respectively, in controls, in patients with CAD and in patients with CHF. During swimming, systolic blood pressure rose up to 130 (24), 143 (13) and 122 (10) mm Hg in patients with CHF, CAD and controls, respectively, whereas diastolic blood pressure revealed only minor changes (63 (11), 68 (4) and 56 (9) mm Hg, respectively (fig 1).
Peripheral vascular resistance
Peripheral resistance, which was highest in patients with CHF, decreased in all study groups during water immersion (by 21% in patients with CHF, by 30% in patients with CAD and by 28% in controls). During swimming, vascular resistance decreased further by 33%, 33% and 43%, respectively (fig 1).
Cardiac index
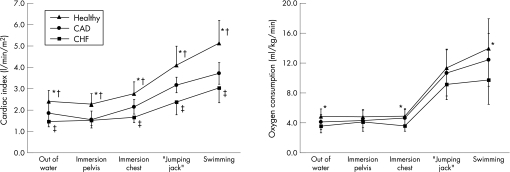
Water immersion up to the chest increased cardiac index by 19% in controls, by 21% in patients with CAD and by 16% in patients with CHF (fig 3). During exercise, cardiac index increased further by 87% in controls, by 77% in patients with CAD and by 53% in patients with CHF. Figure 4 shows the individual responses of the patients with heart failure. Although cardiac index decreased in two patients during water immersion up to the chest, all patients were able to increase cardiac output during swimming.
Figure 3 Adaptation of cardiac index and oxygen consumption to water immersion, water gymnastics and swimming in patients with stable chronic heart failure (CHF), in patients with coronary artery disease (CAD) with preserved left ventricular systolic function and in healthy controls (Healthy). *p<0.05 (healthy vs CHF); †p<0.05 (healthy vs CAD); ‡p<0.05 (CAD vs CHF).
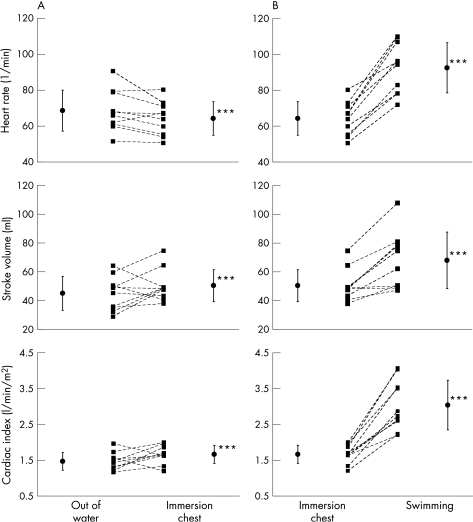
Figure 4 Individual haemodynamic response to water immersion (A) and swimming (B) in patients with heart failure (n = 10). ***p<0.001.
Oxygen uptake
V̇o2 was unaffected by water immersion. Water gymnastics required a V̇O2 of 11.4 (2.5) ml/kg/min in controls. Patients with CAD and CHF achieved a V̇o2 of 10.7 (3.1) and 9.2 (2.1) ml/kg/min, respectively. During swimming, V̇o2 rose to 9.7 (3.3) ml/kg/min in patients with CHF, to 12.4 (3.5) ml/kg/min in patients with CAD, and to 13.9 (4) ml/kg/min in controls (fig 3).
Discussion
During head‐out water immersion, intrathoracic blood volume increases. The reason for this volume shift from the periphery to the central organs is an increase of venous return as a consequence of the effect of hydrostatic pressure on the capacitance vessels, supported by a diminution of gravity dependency of the lower body.
To compensate for decreased myocardial contractility and to maintain an adequate cardiac output in patients with heart failure, the left ventricular end‐diastolic volume increases according to the Frank–Starling mechanism. Furthermore, increased diastolic distensibility leads to increased end‐diastolic volume tolerance in order to avoid end‐diastolic pressure rise, which could lead to pulmonary oedema. Volume shifts, as they occur during water immersion, might potentially overstrain these compensatory mechanisms and as a consequence lead to a decrease in stroke volume, a further rise in the end‐diastolic pressure and the occurrence of pulmonary congestion.
In our study, the cardiac response to water immersion up to the chest was characterised by a decrease in heart rate, an increase in stroke volume, no change in systolic blood pressure but a slight decrease in diastolic blood pressure and a reduction in peripheral vascular resistance (fig 1).
The observed response is the result of the following reflex mechanisms, all interfering with each other: a raise in atrial pressure due to the increase in venous return would lead to an increase in heart rate to evacuate the higher circulating volume (Bainbridge reflex). On the other hand, the rise in right atrial pressure and the increase in the circulating volume also initiates the Frank–Starling mechanism, which increases stroke volume and systolic blood pressure. The rise in blood pressure on his part activates the arterial baroreceptor control system located in the wall of the internal carotid arteries, the carotid sinus and the aortic arch. This leads finally, through excitation of the vagal centre, to a decrease in heart rate and venous and arteriolar tone over‐ruling, therefore the Bainbridge reflex.
The patients with heart failure were characterised by the lowest heart rate, stroke volume and blood pressure but the highest peripheral vascular resistance of the three groups. The picture of the haemodynamic response, nevertheless, was similar in all three groups, which means that also in the population with heart failure, the reflex mechanisms were still intact. Despite their severely reduced left ventricular function with a peak V̇o2 between 15.4 and 24.1 ml/kg/min, they tolerated water immersion, gymnastics and swimming clinically well. Although stroke volume decreased in 4/10 patients and cardiac index decreased in 2/10 patients during water immersion, all patients were able to increase cardiac index during exercise (fig 4).
After a 60 s swim in a pool at 32°C, we found a mean V̇o2 of 9.7 ml/kg/min in patients with heart failure, 12.4 ml/kg/min in patients with CAD with preserved left ventricular function and 13.9 ml/kg/min in controls (fig 3). The differences in V̇o2 between controls and patients with cardiac problems, for the same physical activity, might result from a slightly different swimming speed and technique or, more likely, from differences in the ΔV̇o2/Δwatt relationship during exercise (CHF, 8.1 (1.5); CAD, 9.2 (0.4); healthy, 10.0 (0.6); table 1), reflecting a reduced central haemodynamic response to exercise in patients with impaired left ventricular ejection fraction and/or condition after myocardial infarction.
Left ventricular function at rest is a poor predictor of exercise capacity.12,13 When it comes to recommendations for water sports in patients with heart failure, parameters of exercise capacity rather than echocardiographic measures seem therefore to be of particular importance. Water aerobics, water callisthenics and swimming correspond to a metabolic equivalent (MET) intensity level of 4 or a V̇o2 of 14 ml/kg/min (1 MET = 3.5 ml/kg/min).14 The oxygen uptake at the anaerobic threshold in our study was 13.5 (3.2), 19.6 (6.2) and 26.4 (6.7) ml/kg/min for the patients with CHF, CAD and controls, respectively. This shows that swimming in the tested conditions (with a mean V̇o2 of 9.7 (3.3), 12.4 (3.5) and 13.9 (4.0) ml/kg/min in the patients with CHF, CAD and controls, respectively) corresponds to an intensity level below the anaerobic threshold, even in patients with heart failure.
As V̇o2 is proportional to stroke volume, peak V̇o2 can be used to appreciate the behaviour of the stroke volume during water immersion and swimming (fig 4). Thus, in a stable clinical condition, a minimal peak V̇o2 of at least 15 ml/kg/min and an anaerobic threshold >10 ml/kg/min characterises patients who can participate in water sports in the described conditions without the risk of cardiac decompensation. In patients with a peak V̇o2 of <15 ml/kg/min or a ΔV̇o2/Δwatt relationship clearly <8, however, caution has to be raised.
To what extent parameters of cardiopulmonary exercise obtained on land can be compared with exercise in water has been investigated in various studies.2,15,16 In controls, the central shift of blood volume with head‐out water immersion results in a higher stroke volume at rest and during graded intensities of exercise, compared with values on land in the same posture and at the same metabolic rate.15 Despite an increased stroke volume in water, there is no proportional decrease in heart rate, at least at submaximal exercise levels, and therefore cardiac output is higher in water. Similarly, mean right atrial and pulmonary arterial pressures were found to remain increased, indicating that preload remains increased during graded exercise up to maximal effort in water.2
In patients with cardiac problems, Hanna et al16 compared the effect of increased preload during head‐out water immersion on exercise response in men with healed myocardial infarction without signs of congestive heart failure and with an exercise capacity of at least 5 MET. At rest, cardiac output and stroke volume increased during water immersion, whereas heart rate did not change. During exercise, contrary to the studies conducted on healthy individuals,3,15 these patients did not show a shift of the cardiac output–V̇o2 curve to the left, which means that cardiac output was not increased in water compared with land‐based exercise. This difference has been explained by a lack of further increase of the stroke volume from rest to exercise in the patient group. This might have been the consequence of a more intense adaptation using the Frank–Starling mechanism to maintain cardiac output during land‐based exercise in patients after myocardial infarction and a rapid exhaustion of this compensatory mechanism in water. Furthermore, controls and patients after myocardial infarction showed a different heart rate response to exercise. In healthy people, heart rate response to exercise on land and in water are similar up to a work load of 40% of peak V̇o2, but becomes lower at higher work loads in water. By contrast, subjects with a healed myocardial infarction had the same average heart rate in water and at land at all work loads examined up to 75% of peak V̇o2. It can be concluded that in controls, the haemodynamic response to exercise in water is mainly determined by mechanisms including preload and stroke volume, whereas in patients with a reduced ejection fraction, cardiac output is regulated predominantly by changes in heart rate.
In practice, two important points can be deduced from this fact. (1) Chronotropic response during an exercise stress test is a key parameter in the evaluation of water immersion tolerance in patients with heart failure. In our cohort, mean heart rate increased by 24 beats/min. By generalising these data, one can state that an increase in heart rate during submaximal exercise (eg, up to the anaerobic threshold) of this dimension would be a prerequisite for safe water immersion. (2) The data from Sheldahl et al15 and Christie et al2 show that healthy subjects have a similar heart rate response to exercise on land and in water up to 60% of peak V̇o2 but a somewhat lower heart rate in water at higher work loads with a difference of approximately 10 beats/min. In patients after a myocardial infarction no difference in heart rate response to exercise on land and at all work loads examined in water (up to 75% of peak V̇o2) was found.16 Therefore, there is no need for adaptation of the training heart rate for exercise on land or in water at intensities <60% of peak V̇o2, especially in patients with reduced left ventricular function.
Distention of the peripheral vessels by thermoneutral water results in several salutary effects, which may be particularly beneficial for patients with heart failure: systemic vascular resistance decreases, whereas arginine, vasopressin, renin and norepinephrine are suppressed.17 Activation of cardiac mechanoreceptors leads to reflex adjustments of water and electrolyte excretions from the kidney,18 a mechanism that is preserved in patients with heart failure. Gabrielsen et al8 also showed that intravascular and central blood volume expansion in compensated heart failure suppresses the activity of the renin–angiotensin–aldosterone system, increases the release of atrial natriuretic peptide and elicits a natriuresis, which is enhanced when angiotensin II and aldosterone concentrations are suppressed by ACE inhibitor treatment. It can be concluded that water immersion elicits a number of physiological reactions that are similar to those achieved by modern pharmacological treatment. While the impact on the renin–angiotensin–aldosterone system is of rather short duration, patients with difficulties in regulating their fluid status could particularly profit from the enhancement of natriuresis and therefore be ideal candidates for regular treatment in water.
Recently, the effect of 8 weeks of hydrotherapy in 25 elderly (72 (6) years), stable patients with CHF has been reported.19 These patients with a New York Heart Association (NYHA) functional class II to III and with an exercise capacity that was markedly lower than in our patient group (V̇o2 14.3 (2.7) vs 18.8 ml/kg/min) were randomised either to 8 weeks of hydrotherapy with three weekly sessions of 45 min (n = 15) or to a control group (n = 10). Hydrotherapy yielded improvements in exercise capacity and was well tolerated without any adverse events, suggesting that such a treatment can be safely offered to patients with CHF. These results are now supported by our haemodynamic studies and should therefore stimulate a paradigm shift in the recommendations of water gymnastics or swimming in thermoneutral water in patients with stable CHF and an NYHA functional class I to II.
Study limitations
The study has been effectuated in a controlled indoor setting, and in thermoneutral water. Therefore the results cannot be translated into activities in different environmental conditions. For example, for swimming at lower temperatures, in rivers, lakes or the sea, a higher exercise capacity would be required. Such activity has been shown to correspond to an intensity level of 6 METs or a V̇o2 of 21 ml/kg/min.14
We did not study patients with CHF with severely impaired exercise capacity and a V̇o2 capacity <14 ml/kg/min, which is the reason why we cannot draw any conclusion about patients in NYHA class III or IV. In addition, the fact that two of our patients showed a decrease in cardiac output during water immersion (although during exercise, cardiac output increased) should remind us to advise such patients with appropriate caution.
The absolute values of cardiac index measured at rest in this study seem to be rather low. However, it has to be kept in mind that these measures have been effectuated in a standing position, which could account for some differences of cardiac index values known from the literature, in general measured in supine position. Whereas validation studies have proved reliable compared with invasive techniques to determine cardiac output,10,11 some limitations have to be mentioned. Uneven distribution between ventilation, lung tissue volume, alveolar volume and pulmonary blood flow, as they are observed in more severe forms of lung disease, may cause errors.20 Thus the presence of pulmonary disease might present one obstacle to the use of the rebreathing method.
Conclusion
Stable clinical conditions and a minimal peak V̇o2 of at least 15 ml/kg/min with an anaerobic threshold >10 ml/kg/min during a symptom‐limited exercise stress test characterise patients with severely reduced ejection fraction who can safely participate in water sports in thermoneutral water. Although cardiac index and V̇o2 are lower in these patients compared with patients with CAD and preserved left ventricular function and controls, such patients are able to adequately increase cardiac index during water gymnastics and swimming in thermoneutral water.
In general practice, the ability of a patient to exercise safely on a cycle ergometer with a workload of 70 W (corresponding to 4 METs) or 110 W (corresponding to 6 METs) is a good indicator that water gymnastics and swimming in thermoneutral water are safe even in the presence of stable CHF. However, an inadequate chronotropic response to exercise might denote patients with impaired tolerance to water sports, given the fact that in patients with a reduced ejection fraction, cardiac output during water immersion and swimming is regulated predominantly by the heart rate.
Abbreviations
CAD - coronary artery disease
CHF - chronic heart failure
MET - metabolic equivalent
NYHA - New York Heart Association
V̇o2 - oxygen consumption
Footnotes
Competing interests: None declared.
References
- 1.Arborelius M, Jr, Ballidin U I, Lilja B.et al Hemodynamic changes in man during immersion with the head above water. Aerosp Med 197243592–598. [PubMed] [Google Scholar]
- 2.Christie J L, Sheldahl L M, Tristani F E.et al Cardiovascular regulation during head‐out water immersion exercise. J Appl Physiol 199069657–664. [DOI] [PubMed] [Google Scholar]
- 3.Risch W D, Koubenec H J, Beckmann U.et al The effect of graded immersion on heart volume, central venous pressure, pulmonary blood distribution, and heart rate in man. Pflugers Arch 1978374115–118. [DOI] [PubMed] [Google Scholar]
- 4.Giauzzi P, Tavazzi L, Meyer K.et al Recommendations for exercise training in chronic heart failure patients. Eur Heart J 200122125–135. [DOI] [PubMed] [Google Scholar]
- 5.Tei C, Horikiri Y, Park J C.et al Acute hemodynamic improvement by thermal vasodilation in congestive heart failure. Circulation 1995912582–2590. [DOI] [PubMed] [Google Scholar]
- 6.Arborelius M, Jr, Balldin U I, Lila B.et al Regional lung function in man during immersion with the head above water. Aerosp Med 197243701–707. [PubMed] [Google Scholar]
- 7.Gauer O H. Recent advances in the physiology of whole body immersion. Acta Astronaut 1975231–39. [DOI] [PubMed] [Google Scholar]
- 8.Gabrielsen A, Bie P, Holstein‐Rathlou N H.et al Neuroendocrine and renal effects of intravascular volume expansion in compensated heart failure. Am J Physiol Regul Integr Comp Physiol 2001281R459–R467. [DOI] [PubMed] [Google Scholar]
- 9.Weber K T, Janicki J S. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol 19855522A–31A. [DOI] [PubMed] [Google Scholar]
- 10.Gabrielsen A, Videbaek R, Schou M.et al Non‐invasive measurement of cardiac output in heart failure patients using a new foreign gas rebreathing technique. Clin Sci (Lond) 2002102247–252. [PubMed] [Google Scholar]
- 11.Agostoni P, Cattadori G, Apostolo A.et al Noninvasive measurement of cardiac output during exercise by inert gas rebreathing technique: a new tool for heart failure evaluation. J Am Coll Cardiol 2005461779–1781. [DOI] [PubMed] [Google Scholar]
- 12.Franciosa J A, Ziesche S, Wilen M. Functional capacity of patients with chronic left ventricular failure. Relationship of bicycle exercise performance to clinical and hemodynamic characterization. Am J Med 197967460–466. [DOI] [PubMed] [Google Scholar]
- 13.Cohen‐Solal A, Logeart D, Guiti C.et al Cardiac and peripheral responses to exercise in patients with chronic heart failure. Eur Heart J 199920931–945. [DOI] [PubMed] [Google Scholar]
- 14.Ainsworth B E, Haskell W L, Whitt M C.et al Compendium of physical activities: an update of activity codes and MET intensities. Med Sci Sports Exerc 200032(Suppl)S498–S504. [DOI] [PubMed] [Google Scholar]
- 15.Sheldahl L M, Tristani F E, Clifford P S.et al Effect of head‐out water immersion on cardiorespiratory response to dynamic exercise. J Am Coll Cardiol 1987101254–1258. [DOI] [PubMed] [Google Scholar]
- 16.Hanna R D, Sheldahl L M, Tristani F E. Effect of enhanced preload with head‐out water immersion on exercise response in men with healed myocardial infarction. Am J Cardiol 1993711041–1044. [DOI] [PubMed] [Google Scholar]
- 17.Gabrielsen A, Sorensen V B, Pump B.et al Cardiovascular and neuroendocrine responses to water immersion in compensated heart failure. Am J Physiol Heart Circ Physiol 2000279H1931–H1940. [DOI] [PubMed] [Google Scholar]
- 18.Epstein M. Renal effects of head‐out water immersion in humans: a 15‐year update. Physiol Rev 199272563–621. [DOI] [PubMed] [Google Scholar]
- 19.Cider A, Schaufelberger M, Sunnerhagen K S.et al Hydrotherapy—a new approach to improve function in the older patient with chronic heart failure. Eur J Heart Fail 20035527–535. [DOI] [PubMed] [Google Scholar]
- 20.Petrini M F, Peterson B T, Hyde R W. Lung tissue volume and blood flow by rebreathing theory. J Appl Physiol 197844795–802. [DOI] [PubMed] [Google Scholar]