Abstract
Generation of effector cytotoxic T lymphocytes (CTLs) is a process tightly governed by regulatory helper T (Th) cells. The nature of cellular interactions as well as the precise role of distinct Th cell subsets involved in efficient CTL activation remains elusive. Employing in vitro cultures for primary induction of human, peptide-specific CTL, a strict requirement for Th cells and linkage of epitopes for helper and CTLs on the surface of antigen presenting cells was found, suggesting a three cell type cluster as minimal immune regulatory entity. Cognate and antigen-driven interactions of T cells were neither essential nor sufficient to override the need for linked epitopes. Within the three cell type cluster complex, keyhole limpit hemocyanin or tetanus toxoid-reactive Th cells promoted generation of MAGE-3- or HIV-gag-specific CTL. Both type 1 and type 2 Th cells were recruited and induced by CTL. Interleukin 2 and interferon γ were essential in early stages, and interleukin 4 was utilized in later stages, of CTL maturation. Synergistic effects of CD45RA+ and CD45RO+ Th cells were found. The data reported here suggest a critical link between the innate and adaptive immune system in the initiation process of cytolytic immune responses and offers the basis for efficient vaccine strategies.
CD8+ precursor cytotoxic T lymphocytes (CTLs) mature toward lytic effector cells along a multistep inductive process that is composed of a variety of regulatory events. In addition to recognition of specific antigen complexed with major histocompatibility complex (MHC) class I molecules by clonotypic T cell receptors, activation of effector functions often requires costimulation that is delivered through accessory and adhesion molecules as well as by receptors for lymphokines (1). CD4+ and MHC class II restricted helper T (Th) cells in many cases regulate CTL effector responses by providing stimulatory or inhibitory signals (2).
Despite the biological importance, the exact nature of cellular interactions, requirements for antigen organization and presentation, the precise role of distinct Th cell subsets, and the influence of antagonistically acting secretory products and surface molecules involved in helper and cytotoxic T cell collaboration is not well understood.
In mice, T cells do not express MHC class II antigens. Based on this finding it has been proposed that the presentation of antigen for Th cells is delivered by MHC class II restriction molecules on the surface of antigen presenting cells (APCs). Linkage of epitopes for helper and cytolytic T lymphocytes on one APC to approximate interacting T cells has been demonstrated in the murine system (3, 4).
In humans, immunoregulation differs fundamentally from murine systems by the capability of T lymphocytes to express MHC class I and II molecules upon activation, suggesting regulatory functions of these structures in antigen-driven T cellular interactions and thus providing the possibility for direct helper and cytolytic T cell collaboration without the participation of additional cells.
Immunoregulation of the cellular immune response against a defined epitope is a highly complex process and involves the exchange of signals between helper and cytotoxic T lymphocytes. Regulatory Th cells are not a homogeneous population and comprise subpopulations exerting antagonistic functions defined by differential expression of surface molecules such as CD45 isoforms and patterns of lymphokine secretion.
T cells expressing the high molecular weight form of CD45(RA) are considered “naive type” cells that after encounter of antigen may switch to the CD45RO “memory” phenotype. Although functional differences of CD45RO+ and CD45RA+ Th cells have been demonstrated (5) little is known about their respective roles in primary CTL induction. After activation, Th cells differentiate into subpopulations that can be defined by the production of distinct cytokine patterns. Type 1 Th cells (Th1), producing interleukin 2 (IL-2), tumor necrosis factor β, and interferon γ (IFN-γ), and type 2 Th cells (Th2), secreting IL-4, IL-5, and IL-10, have been reported to execute antagonistic immunoregulatory properties supporting cellular or humoral immune responses, respectively (6).
Designing vaccination strategies for efficient induction of cytolytic immune responses in humans requires a fundamental knowledge of the immune regulatory properties of the Th cells. Employing in vitro cultures, we show that generation of specific CTLs takes place in cell clusters composed of cytotoxic and several Th cell subsets collaborating on the surface of one APC. Coordination of T cellular interactions appear to be organized by linkage of antigens on the APC.
MATERIALS AND METHODS
Cells.
Cells were prepared using previously described methods and reagents (7). Briefly, HLA-A2 positive human peripheral blood mononuclear cells were isolated from healthy individuals by Ficoll/Hypaque (Pharmacia) density gradient centrifugation and E-rosetting using 5% sheep erythrocyte (Microbiologic Associates, Bethesda). Purification of CD8, CD4, CD4/CD45RA+ and CD4/CD45RO+ T cells and monocytes was accomplished by negative selection using goat anti-mouse IgG-conjugated magnetic beads (PerSeptive Diagnostics, Cambridge, MA) and mAbs reactive with CD3, CD4, CD8, CD14, CD45RO, CD45RA, MHC class II, κ, and λ. CD8 and CD4 cells thus obtained were >95% CD8+ and CD4+, respectively, as determined by flow cytometry analysis. CD4/CD45RA and CD4/CD45RO populations were >90% positive for CD45RA or CD45RO and monocytes were >85% reactive with anti-CD14 mAb, containing <3% contaminating B or T lymphocytes.
The human antigen-processing-defective lymphoblast hybrid T2 (147×CEM.T2) was purchased from the American Type Culture Collection and used as target cells for HLA-A2-restricted lysis (8). Due to a deletion of genes for peptide transporter subunits TAP 1 and TAP 2 within the MHC class II region, T2 cells are unable to present endogenously processed peptides. Exogenous peptides, however, can be complexed with HLA-A2 or B5 restriction molecules and be expressed on the cell surface.
Antigens/Peptides.
HIV-gag77–85 (Ser-Leu-Tyr-Asn-Thr-Val-Ala-Thr-Leu) (9) and MAGE-3271–279 (Phe-Leu-Trp-Gly-Pro-Arg-Ala-Leu-Val) (10) peptides were prepared by the Molecular Biology Core Facility, Dana–Farber Cancer Institute, according to published sequences on an automated peptide synthesizer (Applied Biosystems model 433A) using fluorenylmethoxycarbonyl (Fmoc) chemistry and purified by HPLC. The peptides carrying HLA-A2 motifs were used as epitopes for CTLs.
Keyhole limpet hemocyanin (KLH; Sigma), pigeon cytochrome C (Sigma), and tetanus toxoid (TT; Calbiochem) were utilized as source for helper epitopes. The proteins contain a variety of Th cell epitopes that can be presented on most MHC class II restriction molecules (11).
In Vitro Cultures and Assays.
Cultures were set up in α-MEM (minimal essential medium without nucleosides; Life Technologies, Gaithersburg, MD) supplemented with penicillin (100 units/ml), streptomycin (100 μg/ml), and glutamine (2 mM). Ten percent heat inactivated fetal serum albumin (North American Biologicals, Miami) or human AB serum (North American Biologicals) were used in cultures designed for cytotoxicity analysis or flow cytometry analysis, respectively.
Cultures were conducted using modified conditions according to previously published methods (12). If not otherwise noted, purified monocytes were pulsed with the indicated antigens at concentrations of 50 μg/ml for proteins and 20 μg/ml for peptides in low-protein hybridoma medium (Life Technologies) for 3 h and washed two times with PBS. Cells of the indicated types and numbers were mixed in 200 μl cultures in 96-well flat bottom plates and incubated for the indicated times in duplicate, after which 51Cr-release assays, flow cytometry analyses, and ELISA techniques were performed.
Specific lysis was determined by addition of 5000 51Cr-labeled T2 target cells to the cultured cells. 51Cr-labeled T2 cells were pulsed for 2 h with 20 μg/ml MAGE-3 or HIV-gag peptides in 0.5 ml hybridoma medium before adding to the cultures. Spontaneous release and maximum release was determined by incubation of 51Cr-labeled T2 cells in medium or detergents, respectively. Percent specific lysis was assessed after 6-h incubation at 37°C as 100 × (experimental release − spontaneous release)/(maximum release − spontaneous release). Standard deviations were <15% of shown specific lysis values. Antibodies when required were added to the cultures at a concentration of 10 μg/ml. In some experiments, cultures were set up with 5 units/ml recombinant (r) IL-2 (Collaborative Biomedical Products, Bedford, MA) or 500 units/ml rIL-4 (Sigma).
Lymphokine analysis for IL-2, IL-4, and IFN-γ [BioSource International (Camarillo, CA) or Tago Immunologicals] were carried out using ELISA techniques according to manufacturers instructions and correlation to the cell population producing the respective lymphokine was accomplished by detection of intracellular lymphokines and phenotyping using flow cytometry analysis as described (13). Briefly, after amplification of intracellular lymphokine contents by restimulation of cultured cells with 50 ng/ml phorbol 12-myristate 13-acetate, 500 ng/ml ionomycin (Calbiochem), and 10 μmol brefeldin A (Sigma) for 3 h, cells were washed in 2% BSA/PBS (Sigma) and stained with phycoerythrin-conjugated CD45R0 (Sigma) and Cy5-conjugated anti-CD8 (Coulter Immunology) for 30 min at 4°C. After washing, cells were fixed with 2% formaldehyde/PBS for 20 min at room temperature. After washing, the cells were permeabilized in 50 μl 0.5% saponin, 2% BSA/PBS, and fluorescein isothiocyanate (Pierce)-labeled antibodies reactive with human IL-2, IL-4, IFN-γ (BioSource), and ECD-conjugated anti-CD4 (Coulter Immunology) was added. In some experiments, phycoerythrin-conjugated IL-4-specific Ab were used (PharMingen). After staining for 30 min at room temperature, cells were washed twice with 0. 5% saponin and 2% BSA/PBS. The samples were analyzed using a Epics C flow cytometer (Coulter Electronics).
Specificity of antilymphokine staining was controlled using fluorescein isothiocyanate-conjugated irrelevant Abs. Less than 2% staining was observed by previous blocking with unlabeled lymphokine-specific Abs or excess of rIL-2 and rIL-4 or if saponin was excluded.
RESULTS
Induction of Cytolytic T Lymphocytes by Th Cells Requires Organized Antigens.
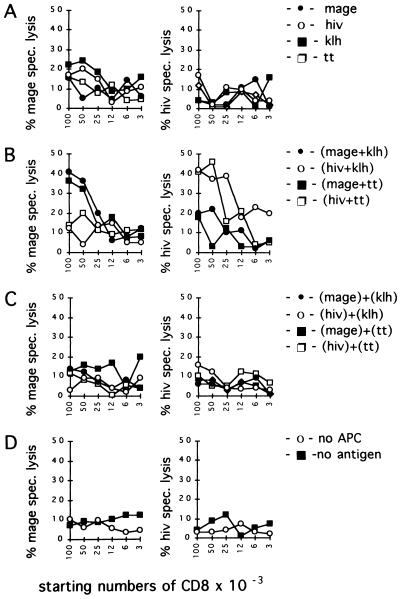
To investigate the role of Th cells in the generation of human, peptide-specific CTLs, we developed induction cultures in vitro. HLA-A2 positive peripheral blood mononuclear cells were separated to provide CD8+ T cells as a source for CTL precursors, CD4+ T cells as a source for Th cells, and monocytes, which were used as APCs. After pulsing with helper antigens KLH or TT or with HLA-A2 motifs carrying HIV-gag- and MAGE-3-derived CTL epitopes or with both helper and CTL antigens, monocytes were cocultivated with the T cell populations. Induction of peptide-specific cytotoxicity was determined after 9 days by the capability of the cultured cells to lyse T2 target cells, loaded with MAGE-3 or HIV-gag peptides, respectively.
In Fig. 1 we demonstrate generation of peptide-specific cytolytic activity in cultures, where helper and CTL epitopes have been coexpressed on the surface of one APC (Fig. 1B). Induction of cytotoxicity was negligible when monocytes presented helper or killer epitopes alone (Fig. 1A), no antigens (Fig. 1D), or in assays, where helper and CTL epitopes were expressed separately on different APCs (Fig. 1C) while identical concentrations of antigens and cell numbers were maintained.
Figure 1.
Th cell-dependent induction of CTLs. The indicated numbers of CTLs were coincubated with 0.5 × 105 Th cells and 0.3 × 105 monocytes for 9 days, after which 5000 51Cr-labeled T2 target cells, pulsed with MAGE (Left) or HIV-gag peptides (Right), were added and cytolytic activity was assessed as described. Monocytes were pulsed before employing in cultures with the CTL epitopes MAGE-3 or HIV-gag or with the helper antigens KLH and TT alone (A), combinations of helper and killer epitopes coexpressed by one APC (B), combinations of helper and killer epitopes but presented on different APCs (C), or no antigen (D).
KLH- as well as TT-specific Th cells have been shown to be capable of supporting CTL activation that were specific for MAGE-3 or HIV-gag peptides, respectively. Specificity of cytolytic responses was confirmed by demonstrating that induced effector T lymphocytes lysed T2 target cells pulsed with the peptide against which they have been raised, but not T2 cells pulsed with different epitopes, thus excluding nonspecific or MHC class II restricted killing, because the MHC class II region of T2 target cells is deleted.
No induction of cytolytic activity was observed in control experiments, where Th cells or CTLs were omitted (data not presented). In addition, mAb reactive with CD8 or MHC class I, but not mAb directed against CD4 or MHC class II molecules given with peptide pulsed T2 targets, inhibited their destruction, indicating MHC class I restricted killing and CD8+ T cells as lytic active components in this experimental system (data not presented). HLA-A2 restriction was confirmed by assaying peptide coated Jurkat cells (HLA-A9, A25, B7, B41), which were treated identically to T2 cells but were not lysed (data not presented).
A Three Cell Type Cluster Comprising APCs, Helper, and CTLs Constitute the Minimal Immune Regulatory Entity.
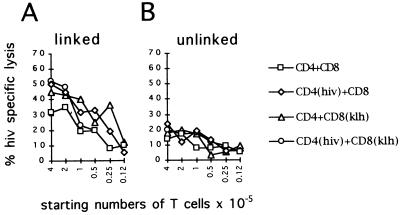
To test whether APCs are essential for productive T–T cell collaborations or if the strict requirements for epitope linkage on APCs as demonstrated above may be overridden by antigen driven T–T interactions, we examined the ability of antigen loaded helper and CTLs to generate cytolytic activity.
Th cells were pulsed with HIV-gag CTL epitopes and CTLs were loaded with KLH, the antigen for Th cells. APCs, expressing linked (Fig. 2A) or unlinked (Fig. 2B) helper and CTL epitopes were cocultivated for 9 days with the T cells as indicated, and lysis of HIV-gag peptide loaded T2 target cells was assayed as described.
Figure 2.
Linked epitopes on APCs are required for productive T cellular collaboration. Th cells were pulsed with HIV-gag, and CTLs were pulsed with KLH or not treated. The T cells were mixed at 1:1 ratios in starting numbers as indicated and coincubated with monocytes, expressing HIV-gag and KLH linked on one population (A) or separately pulsed with individual antigens and mixed (B). Cytolytic activity was determined by addition of 5000 51Cr-labeled and HIV-gag pulsed T2 cells as described.
As shown in Fig. 2A, induction of peptide-specific CTL was possible under conditions where APCs coexpressed both helper and CTL epitopes. Expression of killer epitopes on Th cells or helper epitopes on CTLs amplified elicited cytotoxicity. No generation of cytolytic activity was observed in situations where the antigens for the T cell populations were not linked on the APC (Fig. 2B), even under circumstances where Th cells expressed killer epitopes and CTLs presented helper antigens in the appropriate MHC context. Thus antigen-driven interactions between the T cell populations were not sufficient to promote generation of effector CTLs when specific antigen for helper and killer cells were separated on different APCs.
The data presented point to a three cell type cluster, composed of APCs, helper, and cytotoxic T cells as minimal immune regulatory unit required for Th cell-dependent induction of CTLs.
Differential Effects of CD45RA+ and CD45RO+ Helper T Cell Subpopulations on Induction of CTLs.
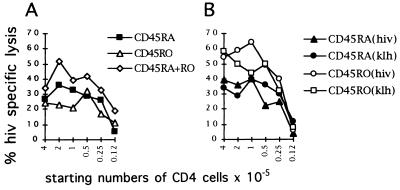
To further define the induction process, we tested the functions and activation requirements of distinct T helper cell subsets. Accordingly, we separated CD45RO+ and CD45RA+ Th cell populations and assayed their capacity to promote CTL generation. Cultures were set up as described above using APCs, coexpressing KLH as helper antigen and HIV-gag CTL epitopes.
As shown in Fig. 3A, both Th cell subsets were found capable to induce HIV-gag specific CTLs. The greater ability of CD45RA+ Th cells for CTL induction compared with CD45RO+ Th cells confirms previously published observations reporting CD45RA+, but not CD45RO+ Th cells, as the responding population to KLH (11). The best CTL-mediated cytolytic responses, however, were found when both CD45RA and CD45RO Th cells were mixed and added to the cultures, indicating synergistic and additive functions of the distinct Th cell subsets for CTL generation. The ability to support CTL induction was greatly enhanced if CD45RO, but not CD45RA, Th cells were pulsed with helper or killer epitopes (Fig. 3B), indicating differential requirements for optimal activation and induction of helper functions for the distinct Th cell subsets.
Figure 3.
Requirements for antigen organization for CD45RO+ and CD45RA+ Th cells. The indicated numbers of CD45RO+ and CD45RA+ Th cells were pulsed with no antigen (A) or with HIV-gag or KLH (B) and coincubated with 1 × 105 CTLs and 0.3 × 105 monocytes, coexpressing HIV-gag and KLH antigens. After 9 days, cytolytic activity was assessed as described using HIV-gag pulsed T2 target cells.
Enhancement of HIV-gag-specific cytolytic responses was also observed in cases, where CD45RO+ Th cells were pulsed with pigeon cytochrome C helper epitopes that were not presented on the APCs but could be recognized by other Th (data not shown).
Immunoregulation by Th Cell-Derived Lymphokines.
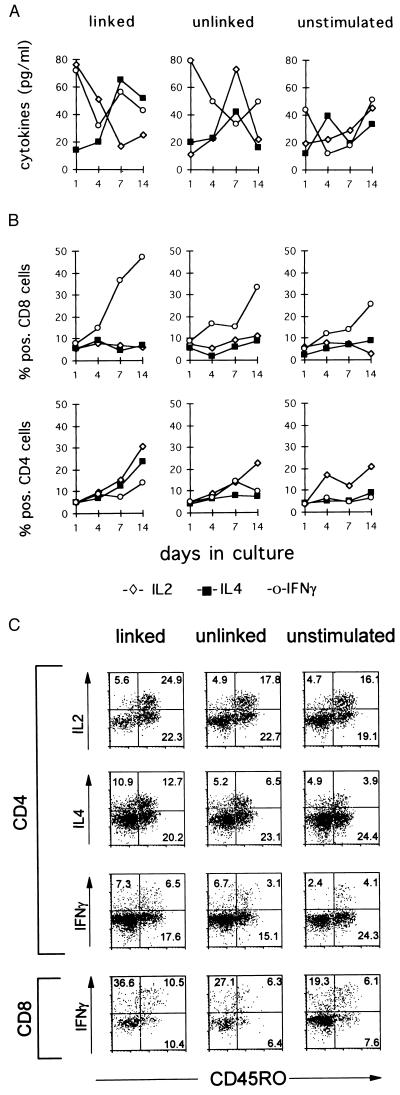
Immunoregulation is mediated by soluble and membrane-expressed molecules, that in concert with antigen-specific signals are required for efficient CTL induction. Accordingly, we next set out to determine lymphokines that are produced in the context of the three cell type cluster complex described above. Because type I lymphokines, represented by IL-2 and IFN-γ and type II lymphokines represented predominantly by IL-4 may exert antagonistic functions, we investigated their role in primary induction of peptide-specific CTLs. Supernatants from cultures, which have been set up with helper and CTLs and APCs, coexpressing KLH as helper and MAGE-3 as CTL epitopes, with APC expressing both antigens but separated on different cells and unpulsed monocytes were assayed for IL-2, IL-4, and IFN-γ content at indicated time points by ELISA.
In cultures, where antigens for Th cells and CTLs were linked on the surface of one APC, maximal IL-2 expression was found as early as 1 day after start of the cultures (Fig. 4A). In cultures containing APCs expressing unlinked helper and cytotoxic antigens, comparable levels of secreted IL-2 were observed that peaked on day 7. In cultures with linked epitopes, IL-4 was found at double the concentration compared with cultures set up with unlinked or no antigens, while no significant differences were observed for IFN-γ production. In cultures expressing either linked or unlinked antigens, IFN-γ concentrations peaked at day 1, while peak IL-4 concentrations were reached at day 7.
Figure 4.
Lymphokine production by CTL and Th cell subsets. Cultures were set up using 1 × 105 Th, 1 × 105 CTLs, and 0.3 × 105 APCs, which have been pulsed with KLH and MAGE-3 antigens (Left, linked), with KLH or MAGE-3 and mixed after pulsing (Center, unlinked), or used without pulsing with antigens (Right, unstimulated). (A) At the indicated time points, supernatants of the cultures were assayed for IL-2 (⋄), IL-4 (▪), and IFN-γ (○) content using ELISA techniques. (B) Detection of intracellular lymphokines was accomplished by flow cytometry analysis. Cells were stained for CD4, CD8, and CD45RO expression and subsequent staining for intracellular lymphokines. (C) Data are shown for day 14.
To define the cell populations responsible for production of the respective cytokines, we performed flow cytometry analysis for intracellular detection of lymphokines on single cell level. Cultured cells were phenotyped after indicated time points using CD4, CD8, and CD45RO reactive mAbs, and detection of intracellular lymphokine production was assessed as described.
Both helper and cytotoxic T lymphocytes were capable of lymphokine production in cultures in vitro. A higher percentage of Th cells expressing IL-2, IL-4, and IFN-γ were detected in culture that were set up with APCs, coexpressing epitopes for CTLs and Th cells compared with cultures where the antigens have not been linked or where the antigens have been omitted as shown in Fig. 4B. CTLs were found to be the major source for IFN-γ, while Th cells produced both IL-2 and IL-4, which were found preferentially in the CD45RO+ Th cell subpopulation (Fig. 4C). After 2 weeks in culture, the ratios of IL-4 to IFN-γ-producing Th cells was assessed as 1.7 in the linked system, 1.2 using unlinked epitopes, and 1.35 in the absence of any antigen. Simultaneous staining for IFN-γ and IL-4 using fluorescein isothiocyanate- and phycoerythrin-labeled antibodies revealed <3% double positive lymphocytes (data not shown).
The interaction of the T cell populations was found to be interdependent, because Th cells induce CTLs for cytotoxic reactions as well as for IFN-γ production and CTLs in turn bias Th cells from an early type I-dominated lymphokine production to a regulatory situation where type I as well as type II lymphokines were produced. It should be noted again that in the experimental set up applied to this cultures, CD45RA, more than CD45RO+, helper cells supported generation of effector CTLs.
Because the distinct CD45 isoforms are highly regulated upon T cell activation, conversion of Th cells from the CD45RA to the CD45RO phenotype followed by subsequent expression of lymphokines may be one explanation for the results observed. To address this possibility, we separated Th cells into CD45RO+ and CD45RA+ cells and assayed the distinct populations separately for lymphokine production.
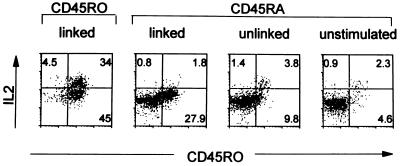
As illustrated in Fig. 5, originally CD45RA+ Th cells, although converting into the CD45RO phenotype in cultures with linked epitopes, produced significant lower amounts of IL-2 compared with identically treated originally CD45RO+ Th cells. The results reported here point to two fundamentally distinct populations of Th cells.
Figure 5.
Functional difference between CD45RO and CD45RA Th cells. A total of 105 CTLs, 105 CD45RO, or CD45RA+ Th cells were cultivated with 0.3 × 105 APCs, coexpressing KLH and MAGE-3 antigens on one cell (linked), with KLH and MAGE-3 presented on different cells (unlinked) or presenting no antigens (unstimulated). After 10 days, cells were stained for CD4, CD8, and CD45RO, and detection of intracellular lymphokines was assessed as described.
Dynamic Functions of Immunoregulatory Lymphokines.
To test the functional roles of type I and type II lymphokines in the process of regulating effector CTL generation, we added rIL-2 and rIL-4 and mAb reactive with IL-2, IL-4, IL-7, IL-12, and IFN-γ to cultures initially containing helper and cytotoxic T cells and APCs coexpressing KLH and MAGE-3 antigens.
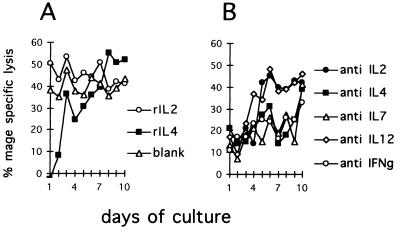
As shown in Fig. 6A, addition of IL-2 in the first days of cultures slightly amplify induced cytotoxicity. In contrast, IL-4 was found to inhibit completely Th cell-dependent generation of effector CTLs if added at the beginning of cultures. However, stimulating effects were found for rIL-4 if added after 7 days of culture. The stimulating potency of IL-4 added during the latter period of the culture incubation resulted in higher cytotoxicity then that observed after addition of rIL-2.
Figure 6.
Role of lymphokines in Th cell-dependent induction of CTLs. Culture were set up using 105 Th, 105 CTLs, and 0.3 × 105 APCs, coexpressing KLH and MAGE-3 antigens. At indicated time points, 5 units/ml rIL-2 or 500 units/ml rIL-4 (A) or neutralizing antibodies directed against IL-2, IL-4, IL-7, IL-12, and IFN-γ at 10 μg/ml (B) were added. After 11 days, 5000 51Cr-labeled and MAGE-3 pulsed T2 target cells were added, and cytolytic activity of the cultured cells was assessed as described.
To test if the lymphokines are essential for the process of cytotoxic T cell induction, we blocked IL-2, IL-4, IL-7, IFN-γ, and APC-derived IL-12 with neutralizing-specific mAb. As shown in Fig. 6B, mAb directed against all lymphokines assayed here inhibited generation of cytotoxicity. However, Abs reactive with IL-2 or IL-12 after 5 days of culture showed no effect, indicating the importance of these lymphokines during the early stages of helper and cytotoxic T cell collaboration. In contrast, IL-4, IFN-γ, and IL-7-specific reagents maintained their inhibiting functions throughout 9 days, pointing toward a long-lasting T–T interaction involving multiple regulatory steps.
DISCUSSION
We have investigated the role of antigen organization in the process of effector CTL generation by Th cell subsets in humans. The data presented in this report demonstrate cluster formation of helper and CTLs and one APC as a prerequisite for collaboration resulting in specific cytolytic responses. To establish productive interactions, antigens for helper and cytotoxic T cells have to be presented by the same APCs. CTLs presenting antigens to Th cells or Th cells expressing CTL epitopes in the proper HLA context are not sufficient to initiate cytolytic immune responses, nor is induction of CTLs possible in cases, where both T cell populations recognize their respective antigens presented by T cell partners and on two separate APCs. Importantly, the antigens for the respective T cell partner need not be related or derived from the same molecule. The regulatory and effector arms of the cellular immune system thus have to be connected by coexpression of the respective antigens on one APC pointing to a cluster of at least three different cell types as the fundamental immunoregulatory entity in humans. Clusters of APCs, surrounded by 10–100 T cells, is a common feature in the T cell compartment of lymph nodes (14) and provides cytological evidence for a mechanism of cytotoxic T cell induction whereby APCs control T cell reactivity by controlling epitope expression to T cells (15).
Within the three cell type cluster, two mechanisms of T cellular communication are possible; both explain the need for the epitope linkage observed. Helper and cytotoxic T cells may communicate via inducible functions of the APCs, which upon activation by helper or cytotoxic T cells may enhance subsequent T cell binding and stimulation or interaction of the T cell subsets requires simultaneous antigen recognition and membrane interactions between T cells and APCs.
APC-derived IL-12 is an essential factor in the experimental system used, because mAb directed against this lymphokine inhibit generation of CTLs. IL-12 production by APCs has been shown to be induced by IFN-γ (16), which is produced predominantly by CD8+ CTLs in the system used here. IL-12 in turn induces IFN-γ production and activates Th cells for IL-2 expression (17–19). Because expression of MHC class I and class II and adhesion molecules are highly regulated by IFN-γ and IL-2-derived signals, the contribution of CTLs for Th cell induction and vice versa has a mechanistic basis provided they interact with the same APCs expressing antigens for helper and cytotoxic T cells.
To dissect immunoregulatory signals occurring within the three cell type cluster, we analyzed lymphokine production of the interacting cells. IL-2, IL-4, IL-7, IFN-γ, and IL-12 are demonstrated to be nonredundant factors for CTL induction. In concordance with reports by other authors, we demonstrate CD45RO+ Th cells as the major source for IL-2 and IL-4 (5, 13), which both have been reported to be essential for CTL maturation (20). Interestingly, we observed IL-2 more than IL-4 production restricted to the CD45RO+ Th population and no significant difference for the Th subsets in IFN-γ expression. IL-2 is required early in the initiation process of CTL induction. In contrast, IL-4 strongly inhibits generation of cytolytic activity if applied at early stages of differentiation, but is essential and exerts stimulatory effects for CTL generation if given several days after initiation of the cultures. These findings may reflect altered reception of immunoregulatory signals by CTLs along their maturation toward effector cells as reported from other laboratories (21) and provide strong evidence for a multistep process of CTL maturation (22) governed by lymphokine-producing Th cells .
Although IL-4 and IFN-γ are expressed in a reciprocally exclusive manner at the single cell level, polarization of lymphokine patterns at the gross level as reported for immune responses against certain microorganisms (23) are not found. In contrast, CTLs bias Th cells for IL-2 and IL-4 production within the clusters. Th cells in turn support IFN-γ production by CTLs, pointing to mutual interaction and interdependent activation of collaborating helper and cytotoxic T cells. Interdependence of helper and cytotoxic T cells may contribute to the strict requirement for linked epitopes on the APCs.
The experiments described here provide evidence that the lymphokines examined are not inhibitory or stimulatory per se, but are required at defined stages of CTL generation. We interpret our findings that the fundamental function of this cluster is to coordinate a variety of T cellular interactions.
In the system applied, CD45RA+ are better than CD45RO+ Th cells at supporting generation of effector CTLs, confirming previously published results that CD45RA is the predominantly responding population to the non-recall antigen KLH used in these experiments (11). By modulating cellular interactions of the distinct helper subsets within the three cell type cluster, we show that the capacity to induce CTLs was greatly enhanced if CD45RO+ but not CD45RA+ Th cells are pulsed with helper or killer epitopes. This result indicates that CD45RO cells indeed respond specifically to KLH, but that they appear to require additional signals for efficient activation.
Because the best cytolytic responses are observed when both CD45RO+ and CD45RA+ cells are present, we conclude that primary CTL induction is regulated by synergistic and additive functions of the respective Th subsets similar to previously published results in mixed lymphocyte reactions (24). Considering the requirement for epitope linkage for both Th cell subsets, more than one Th cell may cluster with CTLs and APCs.
Interestingly, although expressing the CD45RO phenotype after activation (25), originally CD45RA+ Th cells fail to produce lymphokines to the same extent as was found for originally CD45RO+ cells. These findings support reports that propose that the CD45RO+ and CD45RA+ Th subsets are functionally distinct populations that, despite interconversion of CD45 isoforms, maintain discrete features (5, 26). The exact functions of CD45RA+ Th cells remains unclear. As yet unidentified surface molecules and lymphokines may contribute to the greater ability of this population to support CTL generation compared with CD45RO+ Th cells. mAbs directed against likely candidate molecules such as CD27, CD28, or CD70 (27), which have been shown to execute crucial functions in direct T–T cellular interactions, failed to block CTL generation at concentrations of 10 μg/ml.
CTLs are major effector cells responsible for clearance of intracellular pathogens and are involved in immune responses against tumors. Accordingly, various trials have been initiated to improve the efficacy of CTL induction directed against defined epitopes such as virus-derived peptides or tumor antigens. Given the importance of multistep induction and varying signal requirements for efficient effector CTL generation, recruitment of Th cells could be more rewarding than replacing Th cell functions by a single stimulatory molecule (28, 29).
Acknowledgments
We would like to thank H. Levine and J. F. Daley for expert technical assistance and Dr. J. S. Duke-Cohan for helpful discussions. This work was supported by grants from the National Institutes of Health (AI 12069) and a fellowship from the Deutsche Forschungsgemeinschaft (G.S.).
Footnotes
.
Abbreviations: KLH, keyhole limpet hemocyanin; IL, interleukin; r, recombinant; TT, tetanus toxoid; MHC, major histocompatibility complex; Th cell, helper T cell; CTL, cytotoxic T lymphocyte; APC, antigen presenting cell; IFN-γ, interferon γ
References
- 1.Weiss A, Littman D R. Cell. 1994;76:263–274. doi: 10.1016/0092-8674(94)90334-4. [DOI] [PubMed] [Google Scholar]
- 2.Bretscher P. Immunol Today. 1992;13:74–76. doi: 10.1016/0167-5699(92)90138-W. [DOI] [PubMed] [Google Scholar]
- 3.Mitchison N A. Eur J Immunol. 1990;20:699–704. doi: 10.1002/eji.1830200335. [DOI] [PubMed] [Google Scholar]
- 4.Stuhler G, Walden P. Eur J Immunol. 1993;23:2279–2286. doi: 10.1002/eji.1830230934. [DOI] [PubMed] [Google Scholar]
- 5.Ferrer J M, Plaza A, Kreisler M, Diaz-Espada F. Cell Immunol. 1992;141:10–20. doi: 10.1016/0008-8749(92)90123-7. [DOI] [PubMed] [Google Scholar]
- 6.Paul W E, Seder R A. Cell. 1994;76:241–251. doi: 10.1016/0092-8674(94)90332-8. [DOI] [PubMed] [Google Scholar]
- 7.Kobata T, Agematsu K, Kameoka J, Schlossman S F, Morimoto C. J Immunol. 1994;153:5422–5432. [PubMed] [Google Scholar]
- 8.Salter R D, Cresswell P. EMBO J. 1986;5:943–949. doi: 10.1002/j.1460-2075.1986.tb04307.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tsomides T J, Aldovini A, Johnson R P, Walker B D, Young R A, Eisen H N. J Exp Med. 1994;180:1283–1293. doi: 10.1084/jem.180.4.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van der Bruggen P, Bastin J, Gajewski T, Coulie P G, Boel P, De Smet C, Traversari C, Townsend A, Boon T. Eur J Immunol. 1994;24:3038–3043. doi: 10.1002/eji.1830241218. [DOI] [PubMed] [Google Scholar]
- 11.Plebanski M, Saunders M, Burtles S S, Crowe S, Hooper D C. Immunology. 1992;75:86–91. [PMC free article] [PubMed] [Google Scholar]
- 12.Macatonia S E, Patterson S, Knight S C. Immunology. 1991;74:399–406. [PMC free article] [PubMed] [Google Scholar]
- 13.Elson L H, Nutman T B, Metcalf D D, Prussin C. J Immunol. 1995;154:4294–4301. [PubMed] [Google Scholar]
- 14.Breel M, Mobius R E, Kraal G. Eur J Immunol. 1987;17:1555–1559. doi: 10.1002/eji.1830171105. [DOI] [PubMed] [Google Scholar]
- 15.Brunner M C, Mitchison N A, Schneider S C. Folia Biol (Prague) 1994;40:359–369. [PubMed] [Google Scholar]
- 16.Ma X, Chow J M, Gri G, Carra G, Gerosa F, Wolf S F, Dzialo R, Trinchieri G. J Exp Med. 1996;183:147–157. doi: 10.1084/jem.183.1.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gerosa F, Paganin C, Peritt D, Paiola F, Scupoli M T, Aste-Amezaga M, Frank I, Trinchieri G. J Exp Med. 1996;183:2559–2569. doi: 10.1084/jem.183.6.2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Peng X, Kasran A, Warmerdam P A, de Boer M, Ceuppens J L. Eur J Immunol. 1996;26:1621–1627. doi: 10.1002/eji.1830260732. [DOI] [PubMed] [Google Scholar]
- 19.Heufler C, Koch F, Stanzl U, Topar G, Wysocka M, Trinchieri G, Enk A, Steinman R, Romani N, Schuler G. Eur J Immunol. 1996;26:659–668. doi: 10.1002/eji.1830260323. [DOI] [PubMed] [Google Scholar]
- 20.Widmer M B, Grabstein K H. Nature (London) 1987;326:795–798. doi: 10.1038/326795a0. [DOI] [PubMed] [Google Scholar]
- 21.Widmer M B, Acres R B, Sassenfeld H M, Grabstein K H. J Exp Med. 1987;166:1447–1455. doi: 10.1084/jem.166.5.1447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bachmann M F, Sebzda E, Kündig T M, Shahinian A, Speiser D E, Mak T W, Ohashi P S. Eur J Immunol. 1996;26:2017–2022. doi: 10.1002/eji.1830260908. [DOI] [PubMed] [Google Scholar]
- 23.Yamamura M, Uyemura K, Deans R J, Weinberg K, Rea T H, Bloom B R, Modlin R L. Science. 1991;254:277–279. doi: 10.1126/science.254.5029.277. [DOI] [PubMed] [Google Scholar]
- 24.Akbar A N, Salmon M, Ivory K, Taki S, Pilling D, Janossy G. Eur J Immunol. 1991;21:2517–2522. doi: 10.1002/eji.1830211031. [DOI] [PubMed] [Google Scholar]
- 25.Rothstein D M, Yamda A, Schlossman S F, Morimoto C. J Immunol. 1991;146:1175–1183. [PubMed] [Google Scholar]
- 26.Rothstein D M, Sohen S, Daley J F, Schlossman S F, Morimoto C. Cell Immunol. 1990;129:449–467. doi: 10.1016/0008-8749(90)90220-l. [DOI] [PubMed] [Google Scholar]
- 27.Agematsu K, Kobata T, Sugita K, Hirose T, Schlossman S F, Morimoto C. J Immunol. 1995;154:3627–3635. [PubMed] [Google Scholar]
- 28.Ostrand-Rosenberg S, Thakur A, Clements V. J Immunol. 1990;144:4068–4071. [PubMed] [Google Scholar]
- 29.Stuhler G, Walden P. Cancer Immunol Immunother. 1994;39:342–345. doi: 10.1007/BF01519989. [DOI] [PMC free article] [PubMed] [Google Scholar]