Abstract
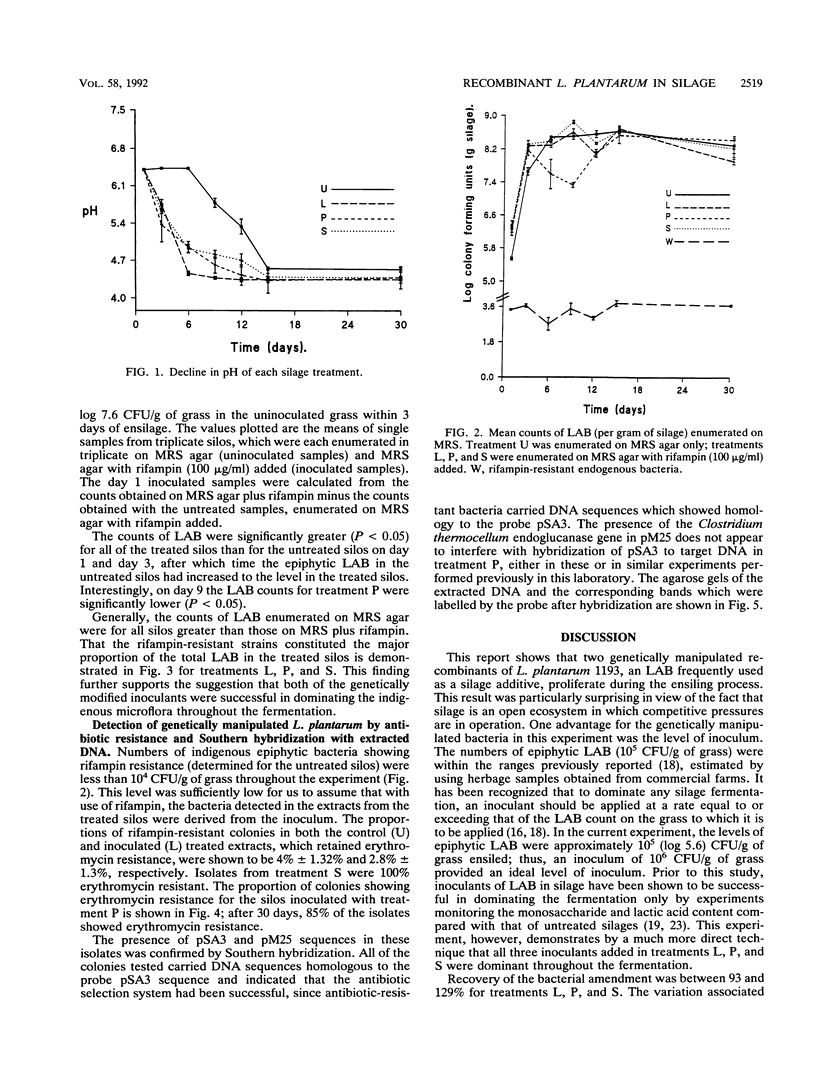
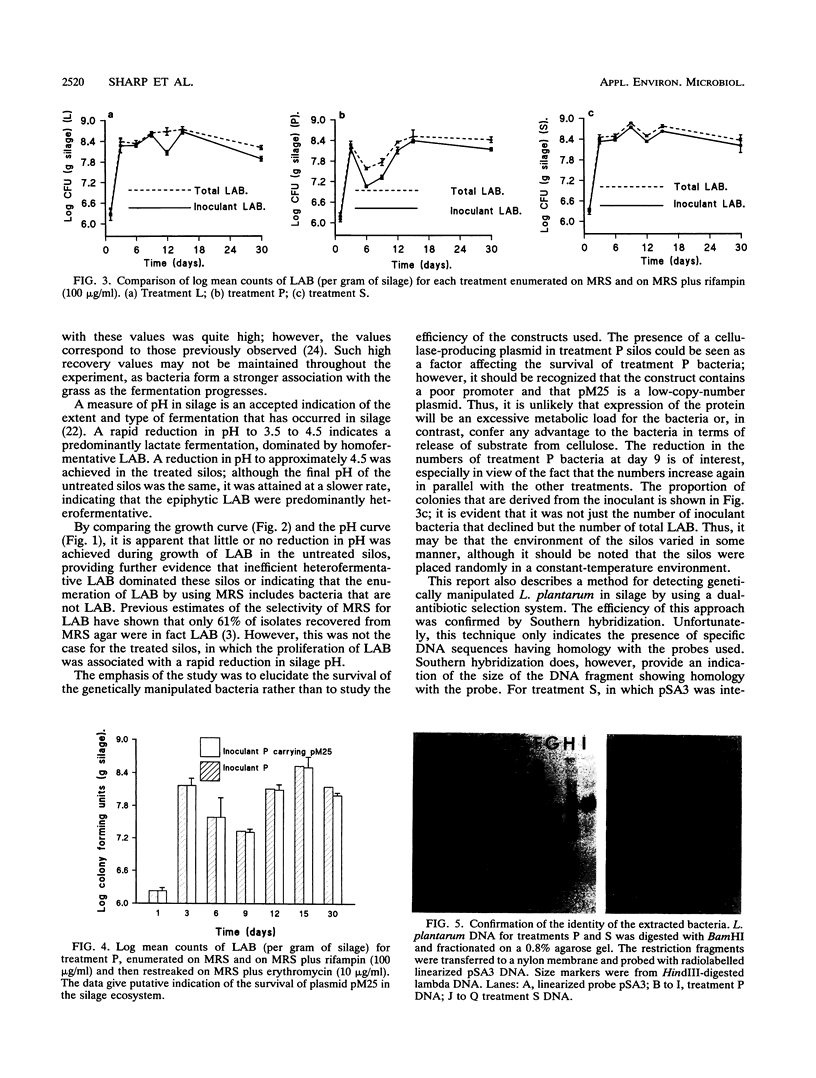
The growth and persistence of two genetically manipulated forms of Lactobacillus plantarum NCDO (National Collection of Dairy Organisms) 1193 have been monitored in grass silage. Both recombinants contained pSA3, a shuttle vector for gram-positive organisms that encodes erythromycin resistance. In one of the recombinants, pSA3 was integrated onto the chromosome, whereas in the other, a pSA3 derivative designated pM25, which contains a Clostridium thermocellum cellulase gene cloned into pSA3, was maintained as an extrachromosomal element. This extrachromosomal element is a plasmid. Rifampin-resistant mutants were selected for the recombinants and the parent strain. When applied to minisilos at a rate of 106 CFU/g of grass, both the recombinants and the parent strain proliferated to dominate the epiphytic microflora and induced an increase in the decline in pH compared with that of the noninoculated silos. The presence of extra genetic material did not appear to disadvantage the bacterium in comparison with the parent strain. The selective recovery of both strains by using rifampin and erythromycin was confirmed by Southern hybridization. Interestingly, the free plasmid (pM25) appeared more stable in silage than was expected from studies in MRS broth. The plasmid was retained by 85% of the rifampin-resistant L. plantarum colonies isolated from a day 30 silo. These data answer an important question by showing that genetically manipulated recombinants of L. plantarum can proliferate and compete with epiphytic lactic acid bacteria in silage.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bates E. E., Gilbert H. J., Hazlewood G. P., Huckle J., Laurie J. I., Mann S. P. Expression of a Clostridium thermocellum endoglucanase gene in Lactobacillus plantarum. Appl Environ Microbiol. 1989 Aug;55(8):2095–2097. doi: 10.1128/aem.55.8.2095-2097.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Gibson E. M., Chace N. M., London S. B., London J. Transfer of plasmid-mediated antibiotic resistance from streptococci to lactobacilli. J Bacteriol. 1979 Jan;137(1):614–619. doi: 10.1128/jb.137.1.614-619.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers J. A., Sanchez D., Elwell L. P., Falkow S. Simple agarose gel electrophoretic method for the identification and characterization of plasmid deoxyribonucleic acid. J Bacteriol. 1976 Sep;127(3):1529–1537. doi: 10.1128/jb.127.3.1529-1537.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheirlinck T., Mahillon J., Joos H., Dhaese P., Michiels F. Integration and expression of alpha-amylase and endoglucanase genes in the Lactobacillus plantarum chromosome. Appl Environ Microbiol. 1989 Sep;55(9):2130–2137. doi: 10.1128/aem.55.9.2130-2137.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West C. A., Warner P. J. Plasmid profiles and transfer of plasmid-encoded antibiotic resistance in Lactobacillus plantarum. Appl Environ Microbiol. 1985 Nov;50(5):1319–1321. doi: 10.1128/aem.50.5.1319-1321.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]