Prostate cancer is the most frequently diagnosed cancer in men and a leading cause of cancer death. More than 218,000 cases will occur in 2007, and prostate cancer will lead to more than 27,000 deaths.1 Although the 5-year survival rate is excellent for localized stages (100%), the relative survival rapidly decreases to 33% when prostate cancer metastasizes.1 Cancer cells from prostate primary tumors commonly colonize bones, and postmortem analyses show that 65 to 75% of patients with advanced disease have bone metastases.2 Scientific knowledge of bone metastasis pathophysiology has increased in recent years, but effective therapy for this devastating complication of cancer remains suboptimal. In this issue of The American Journal of Pathology, Buijs and colleagues3 shed more light into pathophysiology of bone metastases. More importantly, they apply this knowledge to develop novel therapy for bone metastases using bone morphogenetic protein 7 (BMP7).
In the bone microenvironment, tumor cells interact with bone cells to disrupt normal bone remodeling, causing abnormal new bone formation or bone destruction, characteristic of osteoblastic and osteolytic metastases, respectively (Figure 1). This imbalance increases patient morbidity from pathological fractures, intractable bone pain, spinal cord compression, and hypercalcemia. As noted by Stephen Paget in the 19th century, bone osteotropism of cancers, such as prostate, have been attributed to the characteristics of these cells as seeds to survive and grow in the fertile soil of the bone microenvironment.4 Mineralized bone matrix is a major storehouse of growth factors, such as transforming growth factor-β (TGF-β), which is released and activated by tumor stimulation of osteoclastic bone resorption. These cancer cells, stimulated by TGF-β, secrete more osteolytic factors such as parathyroid hormone-related protein (PTHrP), interleukin (IL)-6, and IL-11 that can in turn further stimulate osteoclastic resorption and increase more TGF-β release from bone. TGF-β plays a central role in this feed-forward stimulation of osteoclastic bone resorption, referred to as the vicious cycle of bone metastasis.5,6,7 The TGF-β signaling pathway in tumor cells represents a promising therapeutic target; different modalities to block TGF-β signaling are under investigation in mice and in humans. Buijs and colleagues3 demonstrate that, in prostate cancer cells, BMP7, also known as osteogenic protein-1 (OP-1), is an antagonist of the TGF-β pathway and can inhibit osteolytic metastases attributable to prostate cancer.
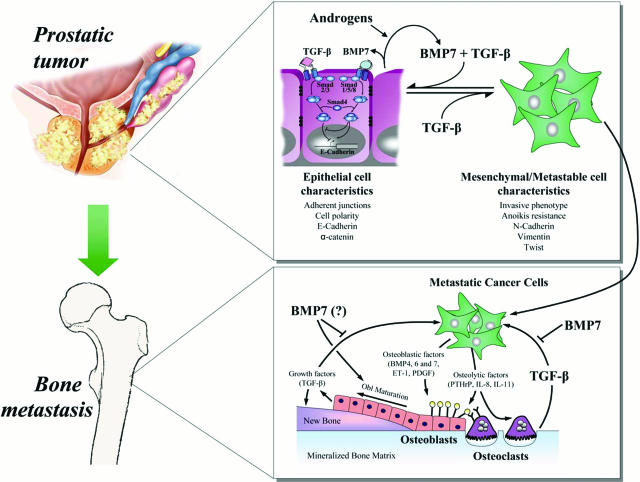
Figure 1.
Role of BMP7 in prostate cancer progression and in bone metastases. Top: In the prostate gland, androgens stimulate the production of BMP7 by normal epithelial cells. Through Smad1/5/8, specifics of BMP signaling, BMP7 represses EMT via different mechanisms including the regulation of E-cadherin expression. BMP7, combined with TGF-β, which signals through Smad2/3, further increases E-cadherin expression and then more efficiently prevents EMT and cancer progression. In the later stage of the disease, BMP7 ceases to act on cancer cells, potentially because of the loss of androgen-induced production of BMP7 after anti-androgen therapy. TGF-β alone represses E-cadherin expression and favors EMT. Cancer cells then acquire a metastatic potential (metastable cells) and can disseminate through the bloodstream to other organs, including bones. Bottom: At sites of bone metastases, cancer cells produce osteolytic factors such as PTHrP, IL-8, or IL-11 that increase bone resorption by directly acting on osteoclasts and their precursors or via the osteoblasts through production of RANK ligand. Growth factors released from resorbed mineralized bone matrix, such as TGF-β, promote tumor growth in bone by enhancing the production of tumor factors that stimulate bone resorption and bone formation. Cancer cells can also induce osteoblast maturation and new bone formation by production of endothelin-1 (ET-1); platelet-derived growth factor (PDGF); and BMP4, -6, or -7. Mature osteoblasts secrete growth factors such as TGF-β that may fuel the progression of bone metastases. BMP7, used as a therapeutic agent, can inhibit TGF-β signaling and progression of this vicious cycle of tumor growth of bone metastases.
The TGF-β superfamily encompasses BMPs and TGF-β. TGF-β1, TGF-β2, and TGF-β3 isoforms have a high homology, possess similar biological functions, and bind the type I (also known as ALK5) and II TGF-β receptors.8 The serine/threonine kinase activity of the heterotetrameric receptor complex phosphorylates the receptor-associated Smad2 and Smad3, which when associated with the Co-Smad, Smad4, and co-activators or co-repressors control targeted gene expression to regulate cell growth, differentiation, and migration.8 TGF-β receptors can also activate Smad-independent signaling in cells through mitogen activated protein kinase kinase and other pathways. TGF-β has a complex role in malignancy. On one hand, it is a tumor suppressor and inhibits growth of epithelial cells. On the other hand, TGF-β promotes invasion and metastasis in transformed cells. Thus, it is considered to be both tumor suppressor and inducer in the early and late stage of the pathology, respectively.8 BMPs activate a combination of type I receptors (type IA and IB BMP receptors and type IA activin receptor) and type II receptors (type II BMP receptor, and type II and IIB activin receptors). The serine/threonine activity of these BMP receptors activates Smad1, Smad5, and Smad8, which associate with Smad4 and transcription factors to participate in gene transcription regulation. The core Smad complexes, based on their composition, bind different DNA motifs: the Smad-binding elements (SBE) are targeted by TGF-β and BMP pathways, whereas the CAGA motives and the BMP-response elements are specific of TGF-β and BMP pathways, respectively. Using a CAGA-luciferase construct, Buijs and colleagues3 showed that BMP7 inhibits the luciferase activity induced by TGF-β and therefore the TGF-β pathway in prostate cancer cells in vitro. The effects of BMP7 were tested in immunodeficient mice inoculated with the highly aggressive PC-3M-Pro4 human prostate cancer cells expressing the firefly luciferase directly into the tibia or into the left cardiac ventricle to induce bone metastases detectable by bioluminescence. In cancer cells in vivo, BMP7 treatment triggered BMP pathway activity as shown by the nuclear accumulation of phosphorylated Smad1 on sections of bone metastases. This was associated with decreased tumor burden. The effect of BMP7 was bone-specific because it had no effect on orthotopic prostate cancer tumors. Thus, BMP7 could be used as a TGF-β antagonist in the treatment of osteolytic metastases from prostate cancer. These data are consistent with those derived from breast cancer and melanoma bone metastases in which inhibition of TGF-β signaling by either small-molecule inhibitors or overexpression of the inhibitory Smad7 reduced bone metastases.9,10,11 Whether BMP7 inhibits TGF-β signaling in vivo as it does in vitro is not conclusively demonstrated by Buijs and colleagues.3 This would require demonstration that BMP7 reduced luciferase activity of PC-3M-Pro4 prostate cancer cells that stably express a TGF-β-specific, CAGA-controlled luciferase vector in vivo.
The development of bone metastases also depends on primary tumor expression of factors such as chemokine receptor CXCR4, integrin αVβ3, and MMPs that facilitate the journey of cancer cells from the primary site to bone.6 Detachment of tumor cells from the primary site is considered a first step of the metastatic cascade and controversially considered to reproduce epithelial-to-mesenchymal transition (EMT). EMT occurs in embryonic development during the migration and differentiation of cells away from the neural crest to form bone, smooth muscle cells, peripheral neurons and glia, and melanocytes. Epithelial cells undergoing EMT lose their polarity and cell-to-cell contacts, undergo cytoskeleton remodeling, and acquire mesenchymal and migration phenotypes that can be related to the transition from localized to invasive tumor (Figure 1). This progression is associated with the decreased expression of epithelial markers, such as E-cadherin, and the increase of mesenchymal markers, such as N-cadherin or vimentin. By signaling through the Smad pathway, TGF-β is a well-known inducer of EMT. TGF-β can increase the expression of N-cadherin in mammary epithelial cells12 and repress the expression of E-cadherin in mammary and renal epithelial cells.12,13 Conversely, in the same renal epithelial cells, BMP7 enhanced E-cadherin expression and antagonized TGF-β repression through a Smad-dependent mechanism.13 This phenomenon seems to be analogous to the BMP7-inhibition of the TGF-β pathway in prostate cancer cells. Buijs and colleagues3 show that BMP7 induced E-cadherin promoter activity in prostate cancer cells, and its expression in prostate cancer cells in vitro was correlated with the E-cadherin ratio and inversely correlated with aggressiveness. Consequently, prostate cancer cells had a decreased BMP7 expression compared with normal epithelial prostatic cells when the expression of BMP7 was compared in matched normal epithelial and malignant prostate cancer cells from patients using laser microdissection or immunohistochemistry methods.3 Although E-cadherin expression in prostate cancer is inversely correlated with the Gleason score, BMP7 expression in prostate cells did not reach a significant inverse correlation with the Gleason score because of statistical limitation. These findings describe a model whereby BMP7 expressed in normal epithelial cells induces E-cadherin expression and contributes to maintenance of an epithelial state, whereas during cancer progression BMP7 expression is lost and E-cadherin expression is decreased leading to EMT and metastasis (Figure 1). In such a model, BMP7 treatment of the primary tumor would prevent metastasis development. This effect could be further potentiated in prostate cancer cells by an environment rich in TGF-β because the combination of BMP7 and TGF-β induced a higher increase of E-cadherin and decrease of vimentin expression than each growth factor alone (Figure 1).3 This up-regulation, however, was not detected in renal epithelial cells13 and may be cell-specific for reasons that remain to be explained. Buijs and colleagues3 did not notice any effect of BMP7 on the local spreading of orthotopically implanted PC-3M-Pro4 cells, but such an inhibition may occur during micrometastatic deposit in the bone marrow in prostate and breast cancer bone metastases.
Despite the significance of these findings, limitations of the models prevent complete extrapolation to the human situation. First, the models (intracardiac inoculation or direct injection into bone) do not reproduce the entire metastatic process, and orthotopic inoculation of PC-3M-Pro4 does not lead to distant metastases. Second, the cell lines used in vivo, derived from PC-3, do not express androgen receptor or respond to androgens. Finally, PC-3 and its derivatives cause osteolytic bone metastases, unlike the osteoblastic lesions that occur in men with prostate cancer.
To study the relative role of BMP7 inhibition of EMT in metastases to distant sites of bone and other organs requires an animal model in which tumor cells metastasize from the primary site to bone. That said, there are very few and limited models that recapitulate the events of the entire metastatic cascade attributable to prostate or breast cancer. Inoculation of the 4T1 murine breast cancer cells into the mammary gland results in formation of a primary tumor that disseminates to the lung, the adrenal glands, and to bone. Using this model, Muraoka and colleagues14 showed that inhibition of TGF-β signaling with a soluble type II receptor significantly reduced metastases. However, these investigators did not study EMT or bone metastases; such studies would be highly relevant, given the findings of Buijs and colleagues3 reported here. Another relevant model that recapitulates the metastatic cascade and shows enhanced metastasis with increased TGF-β signaling uses a genetically engineered expression of an oncogenic version of the Neu receptor tyrosine kinase (c-Neu) under the control of the mouse mammary tumor virus (MMTV) in mouse mammary gland. Here, mammary tumors spontaneously develop and later metastasize to lung.15 Overexpression of a constitutively active form of the TGF-β type I receptor [either MTV/TβRI(AAD) or MMTV-TβRI(S223/225)] increased metastasis formation,15,16 whereas impairment of the TGF-β pathway in mammary cells with a dominant-negative form of the TGF-β type II receptor lacking its kinase cytoplasmic domain [MMTV/TβRII(ΔCyt)] decreased the frequency of metastasis to the lungs.15 Analysis of endogenous expression of BMP7 and EMT markers, as well as testing the effects of a BMP7 treatment could answer questions about BMP7 antagonism of TGF-β-induced EMT and metastasis. However, because bone metastases do not develop in this model, it would not be useful to study such events in the bone microenvironment.
BMP7 expression in prostate cells is androgen-dependent because orchidectomy decreased BMP7 expression whereas testosterone and dihydrotestosterone increased it.17,18 Further, Buijs and colleagues3 show that BMP7 expression is reduced in prostate cancer cell lines that lack androgen receptor. These observations raise several questions relative to androgen regulation of BMP7: i) Is BMP7 expression correlated with androgen independence? ii) Could the evolution of prostate cancer to metastatic dissemination after hormonal therapy be linked to the loss of androgen-induced BMP7 expression and loss of epithelial phenotype maintenance? iii) How does androgen deprivation therapy affect this process? iv) Are there bone-specific implications?
Most patients with prostate cancer metastases to bone have osteoblastic lesions on radiographs. However, osteoclastic bone resorption is clearly evident and contributes to pathophysiology of bone metastasis in this setting. The bone resorption marker N-telopeptide is increased in prostate cancer patients with bone metastases and is a stronger predictor of death than prostate-specific antigen,19 and bisphosphonates improve skeletal morbidity in patients with osteoblastic disease.20 The model used by Buijs and colleagues,3 PC-3M-Pro4, a subclone of PC-3, is similar to MDA-MB-231 breast cancer or B16F10 melanoma in that it causes osteolytic bone metastases in mice. Although this model lacks the osteoblastic component evident in humans with prostate cancer bone metastases, important information can be gleaned regarding the role of BMP7 in osteolytic prostate cancer bone metastases. Nonetheless, the effect of BMP7 on bone metastases should be evaluated in known osteoblastic models such as those induced by ZR-75-1 breast cancer21 and LuCap 23.1 or C4-2B prostate cancer. As the authors suggest, BMP7 could trigger different effects with different bone metastasis phenotypes. Depending on the cell type, BMP7 can induce EMT as assessed by E-cadherin expression in the parental prostate cancer cell line PC-3 or have no effect in other prostate cancer cells.22 BMPs can also protect some prostate cancer cells (LnCap, C4-2B) from apoptosis22 or induce the apoptosis of myeloma cells, which are known to induce osteolysis.23 In other bone metastasis models, BMPs have been shown to induce bone metastases.24,25,26 Noggin inhibits BMP signaling by binding BMP ligands, and when Noggin was overexpressed in PC-3 or LAPC-9 prostate cancer cells, there was a decrease of osteolytic and osteoblastic lesions, respectively.25,26 This positive effect of BMPs in the osteoblastic process could be related to an increase of BMP7 expression at sites of osteoblastic prostate cancer metastases.27 BMPs induce the differentiation of cells from the osteoblast lineage and enhance osteoblast activity; BMP7-knockout mice have a smaller skeleton and decreased bone mineralization.28 Thus, BMP7 at sites of bone metastases could potentially promote the osteoblastic reaction and enhance osteoblastic bone metastasis. This distinction would be important to confirm in animal models before proceeding to clinical trials.
Overall, Buijs and colleague3 provide a role for BMP7 expression in the maintenance of epithelial behavior in the prostate (Figure 1). In normal epithelial cells, BMP7 and TGF-β concur to induce the expression of E-cadherin, repress vimentin synthesis, and prevent EMT. In later stage, because of loss of expression of BMP7 by cancer cells and/or resistance of these cells to the BMP7 secreted by surrounding normal epithelial cells, malignant cells lose the expression of E-cadherin under the influence of TGF-β, which then induces EMT and leads to metastatic spreading. This model re-emphasizes the double-edged-sword properties and the complexity of TGF-β and members of the TGF-β superfamily in cancer biology. It also provides a rationale for further investigation of BMP7 in the prevention of osteolytic bone metastases attributable to prostate cancer. This effect is bone-specific, and whether it is dependent on the bone metastasis phenotype remains to be studied. Buijs and colleagues3 offer interesting and clinically relevant perspectives for the therapeutic use of BMP7, but they also raise numerous questions regarding its role at the different stages of prostate cancer that exemplify the promises but also the versatility of the members of the TGF-β superfamily.
Footnotes
Address reprint requests to Theresa A. Guise, Division of Endocrinology and Metabolism, Department of Internal Medicine, University of Virginia, PO Box 801419, Charlottesville, VA 22908-1419. E-mail: tag4n@virginia.edu.
See related article on page 1047
Supported by the National Institutes of Health (grants CA69158, DK067333, and DK065837 to T.A.G.), the Department of Defense (grants PC061185 to P.G.J.F. and PC040341 to T.A.G.), the Prostate Cancer Foundation, The Mellon Institute, and the Gerald D. Aurbach Endowment of the University of Virginia.
This commentary relates to Buijs et al, Am J Pathol 2007, 171:1047–1057, published in this issue.
References
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007;57:43–66. doi: 10.3322/canjclin.57.1.43. [DOI] [PubMed] [Google Scholar]
- Coleman RE. Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer Treat Rev. 2001;27:165–176. doi: 10.1053/ctrv.2000.0210. [DOI] [PubMed] [Google Scholar]
- Buijs JT, Rentsch CA, van der Horst G, van Overveld PGM, Schwaninger R, Henriquez NV, Papapoulos SE, Pelger RCM, Vukicevic S, Cecchini MG, Löwik CWGM, van der Pluijm G. BMP7, a putative regulator of epithelial homeostasis in the human prostate, is a potent inhibitor of prostate cancer bone metastasis in vivo. Am J Pathol. 2007;171:1047–1057. doi: 10.2353/ajpath.2007.070168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- Kang Y, He W, Tulley S, Gupta GP, Serganova I, Chen CR, Manova-Todorova K, Blasberg R, Gerald WL, Massague J. Breast cancer bone metastasis mediated by the Smad tumor suppressor pathway. Proc Natl Acad Sci USA. 2005;102:13909–13914. doi: 10.1073/pnas.0506517102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang Y, Siegel PM, Shu W, Drobnjak M, Kakonen SM, Cordon-Cardo C, Guise TA, Massague J. A multigenic program mediating breast cancer metastasis to bone. Cancer Cell. 2003;3:537–549. doi: 10.1016/s1535-6108(03)00132-6. [DOI] [PubMed] [Google Scholar]
- Yin JJ, Selander K, Chirgwin JM, Dallas M, Grubbs BG, Wieser R, Massague J, Mundy GR, Guise TA. TGF-β signaling blockade inhibits PTHrP secretion by breast cancer cells and bone metastases development. J Clin Invest. 1999;103:197–206. doi: 10.1172/JCI3523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massagué J, Gomis RR. The logic of TGFβ signaling. FEBS Lett. 2006;580:2811–2820. doi: 10.1016/j.febslet.2006.04.033. [DOI] [PubMed] [Google Scholar]
- Bandyopadhyay A, Agyin JK, Wang L, Tang Y, Lei X, Story BM, Cornell JE, Pollock BH, Mundy GR, Sun LZ. Inhibition of pulmonary and skeletal metastasis by a transforming growth factor-β type I receptor kinase inhibitor. Cancer Res. 2006;66:6714–6721. doi: 10.1158/0008-5472.CAN-05-3565. [DOI] [PubMed] [Google Scholar]
- Javelaud D, Mohammad KS, McKenna CR, Fournier P, Luciani F, Niewolna M, Andre J, Delmas V, Larue L, Guise TA, Mauviel A. Stable overexpression of Smad7 in human melanoma cells impairs bone metastasis. Cancer Res. 2007;67:2317–2324. doi: 10.1158/0008-5472.CAN-06-3950. [DOI] [PubMed] [Google Scholar]
- Stebbins EG, Mohammad KS, Niewolna M, McKenna CR, Mison AP, Schimmoller F, Murphy A, Chakravarty S, Dugar S, Higgins LS, Wong DH, Guise TA. SD-208, a small molecule inhibitor of transforming growth factor-β receptor I kinase reduces breast cancer metastases to bone and improves survival in a mouse model. J Bone Miner Res. 2005;20:S55. [Google Scholar]
- Deckers M, van Dinther M, Buijs J, Que I, Lowik C, van der Pluijm G, ten Dijke P. The tumor suppressor Smad4 is required for transforming growth factor β-induced epithelial to mesenchymal transition and bone metastasis of breast cancer cells. Cancer Res. 2006;66:2202–2209. doi: 10.1158/0008-5472.CAN-05-3560. [DOI] [PubMed] [Google Scholar]
- Zeisberg M, Hanai J, Sugimoto H, Mammoto T, Charytan D, Strutz F, Kalluri R. BMP-7 counteracts TGF-β1-induced epithelial-to-mesenchymal transition and reverses chronic renal injury. Nat Med. 2003;9:964–968. doi: 10.1038/nm888. [DOI] [PubMed] [Google Scholar]
- Muraoka RS, Dumont N, Ritter CA, Dugger TC, Brantley DM, Chen J, Easterly E, Roebuck LR, Ryan S, Gotwals PJ, Koteliansky V, Arteaga CL. Blockade of TGF-β inhibits mammary tumor cell viability, migration, and metastases. J Clin Invest. 2002;109:1551–1559. doi: 10.1172/JCI15234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muraoka RS, Koh Y, Roebuck LR, Sanders ME, Brantley-Sieders D, Gorska AE, Moses HL, Arteaga CL. Increased malignancy of Neu-induced mammary tumors overexpressing active transforming growth factor beta1. Mol Cell Biol. 2003;23:8691–8703. doi: 10.1128/MCB.23.23.8691-8703.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel PM, Shu W, Cardiff RD, Muller WJ, Massague J. Transforming growth factor beta signaling impairs Neu-induced mammary tumorigenesis while promoting pulmonary metastasis. Proc Natl Acad Sci USA. 2003;100:8430–8435. doi: 10.1073/pnas.0932636100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masuda H, Fukabori Y, Nakano K, Shimizu N, Yamanaka H. Expression of bone morphogenetic protein-7 (BMP-7) in human prostate. Prostate. 2004;59:101–106. doi: 10.1002/pros.20030. [DOI] [PubMed] [Google Scholar]
- Thomas R, Anderson WA, Raman V, Reddi AH. Androgen-dependent gene expression of bone morphogenetic protein 7 in mouse prostate. Prostate. 1998;37:236–245. doi: 10.1002/(sici)1097-0045(19981201)37:4<236::aid-pros5>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- Coleman RE, Major P, Lipton A, Brown JE, Lee K-A, Smith M, Saad F, Zheng M, Hei YJ, Seaman J, Cook R. Predictive value of bone resorption and formation markers in cancer patients with bone metastases receiving the bisphosphonate zoledronic acid. J Clin Oncol. 2005;23:4925–4935. doi: 10.1200/JCO.2005.06.091. [DOI] [PubMed] [Google Scholar]
- Saad F, Gleason DM, Murray R, Tchekmedyian S, Venner P, Lacombe L, Chin JL, Vinholes JJ, Goas JA, Chen B. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458–1468. doi: 10.1093/jnci/94.19.1458. [DOI] [PubMed] [Google Scholar]
- Yin JJ, Mohammad KS, Kakonen SM, Harris S, Wu-Wong JR, Wessale JL, Padley RJ, Garrett IR, Chirgwin JM, Guise TA. A causal role for endothelin-1 in the pathogenesis of osteoblastic bone metastases. Proc Natl Acad Sci USA. 2003;100:10954–10959. doi: 10.1073/pnas.1830978100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S, Zhong C, Frenkel B, Reddi AH, Roy-Burman P. Diverse biological effect and Smad signaling of bone morphogenetic protein 7 in prostate tumor cells. Cancer Res. 2005;65:5769–5777. doi: 10.1158/0008-5472.CAN-05-0289. [DOI] [PubMed] [Google Scholar]
- Ro TB, Holt RU, Brenne AT, Hjorth-Hansen H, Waage A, Hjertner O, Sundan A, Borset M. Bone morphogenetic protein-5, -6 and -7 inhibit growth and induce apoptosis in human myeloma cells. Oncogene. 2004;23:3024–3032. doi: 10.1038/sj.onc.1207386. [DOI] [PubMed] [Google Scholar]
- Dai J, Keller J, Zhang J, Lu Y, Yao Z, Keller ET. Bone morphogenetic protein-6 promotes osteoblastic prostate cancer bone metastases through a dual mechanism. Cancer Res. 2005;65:8274–8285. doi: 10.1158/0008-5472.CAN-05-1891. [DOI] [PubMed] [Google Scholar]
- Feeley BT, Gamradt SC, Hsu WK, Liu N, Krenek L, Robbins P, Huard J, Lieberman JR. Influence of BMPs on the formation of osteoblastic lesions in metastatic prostate cancer. J Bone Miner Res. 2005;20:2189–2199. doi: 10.1359/JBMR.050802. [DOI] [PubMed] [Google Scholar]
- Feeley BT, Krenek L, Liu N, Hsu WK, Gamradt SC, Schwarz EM, Huard J, Lieberman JR. Overexpression of noggin inhibits BMP-mediated growth of osteolytic prostate cancer lesions. Bone. 2006;38:154–166. doi: 10.1016/j.bone.2005.07.015. [DOI] [PubMed] [Google Scholar]
- Masuda H, Fukabori Y, Nakano K, Takezawa Y, Suzuki TC, Yamanaka H. Increased expression of bone morphogenetic protein-7 in bone metastatic prostate cancer. Prostate. 2003;54:268–274. doi: 10.1002/pros.10193. [DOI] [PubMed] [Google Scholar]
- Jena N, Martin-Seisdedos C, McCue P, Croce CM. BMP7 null mutation in mice: developmental defects in skeleton, kidney, and eye. Exp Cell Res. 1997;230:28–37. doi: 10.1006/excr.1996.3411. [DOI] [PubMed] [Google Scholar]