Abstract
To assess the usefulness of oncostatin M (osm) gene therapy in liver regeneration, we examined whether the introduction of OSM cDNA enhances the regeneration of livers damaged by dimethylnitrosamine (DMN) in rats. Repeated injection of OSM cDNA enclosed in hemagglutinating virus of Japan envelope into the spleen resulted in the exclusive expression of OSM protein in Kupffer cells of the liver, which was accompanied by increases in body weight, liver weight, and serum albumin levels and the reduction of serum liver injury parameters (bilirubin, aspartate aminotransferase, and alanine aminotransferase) and a serum fibrosis parameter (hyaluronic acid). Histological examination showed that osm gene therapy reduced centrilobular necrosis and inflammatory cell infiltration and augmented hepatocyte proliferation. The apoptosis of hepatocytes and fibrosis were suppressed by osm gene therapy. Time-course studies on osm gene therapy before or after DMN treatment showed that this therapy was effective not only in enhancing regeneration of hepatocytes damaged by DMN but in preventing hepatic cytotoxicity caused by subsequent treatment with DMN. These results indicate that OSM is a key mediator for proliferation and anti-apoptosis of hepatocytes and suggest that osm gene therapy is useful, as preventive and curative means, for the treatment of patients with liver damage.
The liver has a remarkable ability to respond to injuries inflicted by various causes, such as partial hepatectomy, toxic exposure, and virus infection.1,2 Hepatocytes, which are liver parenchymal cells and normally in the quiescent G0 phase, re-enter the cell cycle after injury to restore its mass, architecture, and function. In this process, a number of growth factors and cytokines have been reported to be involved.3,4,5,6,7,8,9 For example, hepatocyte growth factor (HGF) functions as a potent mitogen for hepatocytes,10 and the administration of HGF has been shown to ameliorate hepatic injury in animal models of fulminant hepatic failure.11,12,13 It has also been shown that the introduction of hgf gene into rat cirrhotic livers using liposome with the hemagglutinating virus of Japan (HVJ) inhibits fibrogenesis and hepatocyte apoptosis, leading to the complete resolution of fibrosis and improvement of survival rate.14
Oncostatin M (OSM) is a member of the interleukin (IL)-6 cytokine family that includes IL-6, IL-11, leukemia inhibitory factor, ciliary neurotrophic factor, cardiotrophin-1, and novel neutrophin-1/B-cell-stimulating factor-3.15,16,17,18 Mouse OSM receptor is composed of the gp130 subunit, common to all of the IL-6 family cytokines, and an OSM-specific subunit (hereafter called OSM-specific receptor; OSM-R).19 Binding of OSM to its receptor complex activates Janus tyrosine kinases (Jak1, Jak2, and Tyk2), which in turn activates downstream signaling pathways, including SHP-2 tyrosine phosphatase and signal transducer and activator of transcription protein 3 (STAT3). Recently, OSM has been shown to induce maturation of mouse hepatocytes derived from embryonic day 14.5 liver.20 In addition, Nakamura and colleagues21 have shown that OSM-R knockout (OSM-R−/−) mice exhibit delayed hepatocyte proliferation, persistent liver necrosis, and increased tissue destruction after CCl4 treatment. They have also shown that the administration of OSM reduces CCl4-induced acute liver failure in wild-type mice.21 These results suggest that OSM, like HGF, plays an important role in liver regeneration.
Recently, we have succeeded in isolating rat OSM cDNA.22 In this study, we examined whether the introduction of this OSM cDNA enhances liver regeneration and suppresses fibrogenesis in rats administrated with dimethylnitrosamine (DMN). Repeated injection of HVJ envelope complex23 with rat OSM cDNA (hereafter called HVJ-OSM) into the spleen reduced centrilobular necrosis and inflammatory cell infiltration, induced hepatocyte proliferation, and suppressed hepatocyte apoptosis. Serum liver injury parameters, such as bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT), and a fibrosis parameter, hyaluronic acid, were improved. In addition, osm gene therapy was effective in both protection against hepatic cytotoxicity caused by subsequent treatment with DMN and enhancement of regeneration of hepatocytes damaged by DMN. These results support a crucial role of OSM in liver regeneration and suggest that osm gene therapy is useful, as preventive and curative means, for the treatment of patients with liver damage.
Materials and Methods
Rats and Treatments
Male Sprague-Dawley rats of 4 weeks of age were purchased from Japan SLC (Hamamatsu, Shizuoka, Japan). To examine the effect of OSM cDNA introduction on the physiological condition of rats, we injected HVJ-OSM, prepared from pEF-BOS with rat OSM cDNA22 by HVJ Envelope Vector kit (GenomONE-Neo; Ishihara Industry Corp., Osaka, Japan) according to the manufacturer’s protocol, or the HVJ envelope complex without rat OSM cDNA (hereafter called HVJ-Vector) into rats (n = 12) weekly for 3 weeks. On the next day of the last injection, rats were sacrificed, blood was taken from the right ventricle of hearts, and sera were prepared and stored at −80°C until analysis. Livers were promptly removed to determine the weight and examine histology. Albumin, total bilirubin (T-Bil), direct bilirubin (D-Bil), AST, ALT, and hyaluronic acid in the sera were measured by standard laboratory techniques. Other rats were then divided into two groups. One group (n = 6) received osm gene therapy that was performed as follows. DMN (Sigma-Aldrich Co., St. Louis, MO) was given intraperitoneally at 15 mg/kg body weight for 3 consecutive days per week for 3 weeks to induce liver damage and fibrosis. On day 4 of each week, HVJ-OSM was injected into the spleen. The other group (n = 6) that served as control was treated with DMN similarly for 3 consecutive days per week for 3 weeks, and on day 4 of each week, HVJ-Vector was injected into the spleen. On day 5 of the last week, rats were sacrificed. For time-course studies on osm gene therapy before DMN treatment, HVJ-OSM or HVJ-Vector was injected into the spleens of rats (n = 18), and 24 hours later, DMN was given intraperitoneally at 21 mg/kg body weight. For time-course studies on osm gene therapy after DMN treatment, DMN was given intraperitoneally at 21 mg/kg body weight, and HVJ-OSM or HVJ-Vector was injected into the spleen of rats (n = 18) 24 hours later. In both experiments, rats were sacrificed every day. All surgical procedures on rats were done under pentobarbital sodium anesthesia. All experimental procedures were approved by the Animal Care Committee of Hyogo College of Medicine and performed in accordance with the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Science.
Histological Examination
The removed liver was fixed with methacarn (methanol/chloroform/glacial acetic acid, 6:3:1) or 10% formalin neutral buffer solution, embedded in paraffin, and cut into 5-μm-thick sections. For OSM immunostaining, sections were incubated with goat anti-mouse OSM antibody (R&D Systems, Minneapolis, MN) and then sequentially with horseradish peroxidase-conjugated donkey anti-goat IgG antibody (Santa Cruz Biotechnology, Santa Cruz, CA), biotinyl tyramine (DAKO, Glostrup, Denmark), and horseradish peroxidase-conjugated streptavidin (DAKO). For Ki-67 immunostaining, sections were heated in 10 mmol/L sodium citrate buffer (pH 6.0) at 95°C for 40 minutes to facilitate antigen retrieval. The sections were incubated with rabbit polyclonal antibody against human Ki-67 nuclear antigen (Novocastra Laboratories Ltd., Benton Lane, UK) and then with Histofine Simple Stain MAX-PO(R) (Nichirei Corporation, Tokyo, Japan). Immunoreacted cells for OSM and Ki-67 were visualized with Simple Stain DAB solution (Nichirei). The sections were lightly counterstained with hematoxylin. Total and Ki-67-positive hepatocytes were counted in 10 portal fields selected randomly in each specimen. Apoptotic hepatocytes were visualized by terminal dUTP nick-end labeling (TUNEL) staining using an apoptosis in situ detection kit (Wako Pure Chemical Industries, Ltd., Osaka, Japan). Apoptotic hepatocytes were counted in 10 central vein areas selected randomly in each specimen.
Confocal Laser-Scanning Microscope
For double staining for OSM and ED2, the sections were incubated with goat anti-mouse OSM antibody and sequentially with horseradish peroxidase-conjugated donkey anti-goat IgG antibody, biotinyl tyramine, and fluorescein isothiocyanate-conjugated streptavidin. After washing, the sections were incubated with mouse anti-rat ED2 antibody (Serotec Ltd., Oxford, UK) and then with R-phycoerythrin-conjugated goat anti-mouse IgG (DAKO) and covered with mounting medium for fluorescence with 4′,6-diamidino-2-phenylindole (Vectashield; Vector Laboratories, Inc., Burlingame, CA). Co-localization of fluorescein isothiocyanate and R-phycoerythrin was analyzed with a confocal laser-scanning microscope (LSM510; Carl Zeiss Jena GmbH, Jena, Germany).
Statistical Analysis
Statistical analysis was performed by unpaired, two-tailed Student’s t-test. A P value <0.05 was considered significant.
Results
Introduction of HVJ-OSM into the Spleen and Expression of OSM at the Protein Level in the Liver
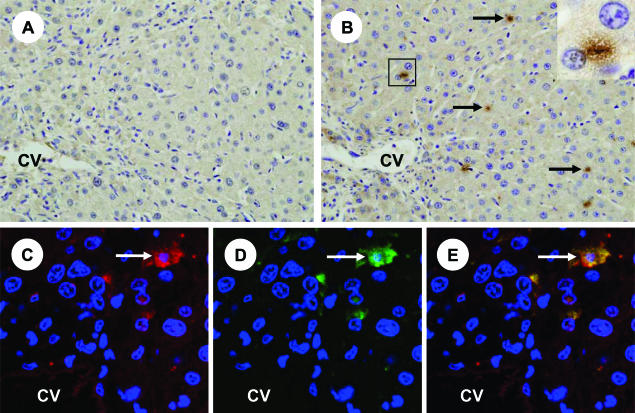
We injected rat OSM cDNA in HVJ envelope (HVJ-OSM) into the spleens of rats weekly for 3 weeks (Figure 1) and analyzed the liver immunohistochemically. The results showed that OSM protein was exclusively expressed in small cells lining the sinusoid of the liver, whereas OSM-positive cells were hardly detectable in the liver of rats injected with HVJ-Vector (Figure 2, A and B). Because the location of OSM protein-expressing cells appeared to correspond to that of Kupffer cells, we performed immunohistochemical double staining of ED2 and OSM. Kupffer cells identified by ED2 expression were found to be positive for OSM protein (Figure 2, C–E), indicating that injection of OSM cDNA into the spleen resulted in expression at the protein level exclusively in Kupffer cells of the liver.
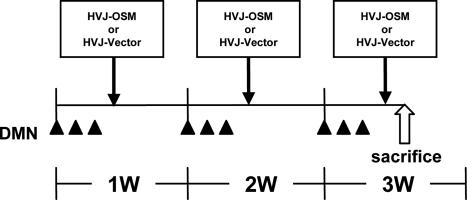
Figure 1.
Schedule of DMN administration and HVJ-OSM or HVJ-Vector injection. Rats were given DMN intraperitoneally at 15 mg/kg body weight for 3 consecutive days per week for 3 weeks. On day 4 of each week, HVJ-OSM or HVJ-Vector was injected into the spleen. On day 5 of the last week, rats were sacrificed for analysis. w, week.
Figure 2.
Expression of OSM in the liver of a rat injected with HVJ-OSM. A: Immunostaining of OSM in the liver of a rat injected with HVJ-Vector. B: Immunostaining of OSM in the liver of a rat injected with HVJ-OSM. Arrows indicate representative OSM-expressing cells. Inset in B is a higher magnification of the squared region, and shows an OSM-expressing cell. C: Fluorescein for ED2, which is a marker of Kupffer cells (red). D: Fluorescein for OSM (green). E: Merged confocal image of C and D. Coexpression of ED2 and OSM is shown as yellow. White arrows indicate representative coexpression of ED2 and OSM. C to E are photographs of the same section, taken by confocal laser-scanning microscope. CV, central vein. Original magnifications: ×200 (A and B); ×600 (inset).
Repeated Injection of HVJ-OSM Attenuates Liver Damage
First, we examined whether the introduction of OSM cDNA weekly for 3 weeks affects the physiological condition of rats. There is no significant difference between rats injected with HVJ-OSM and with HVJ-Vector in the body weight, liver weight, serum albumin level, and serum liver injury parameters (T-Bil, D-Bil, AST, and ALT values), and a serum fibrosis parameter (hyaluronic acid value), and no obvious liver damage in both these rats (Supplemental Figure S1 available at http://ajp.amjpathol.org). Histology of livers showed no significant difference between rats injected with HVJ-OSM and with HVJ-Vector (Supplemental Figure S2 available at http://ajp.amjpathol.org). These results indicated that the introduction of OSM cDNA does not affect the physiological conditions of rats.
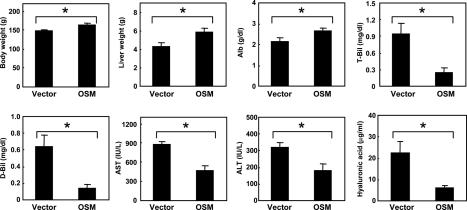
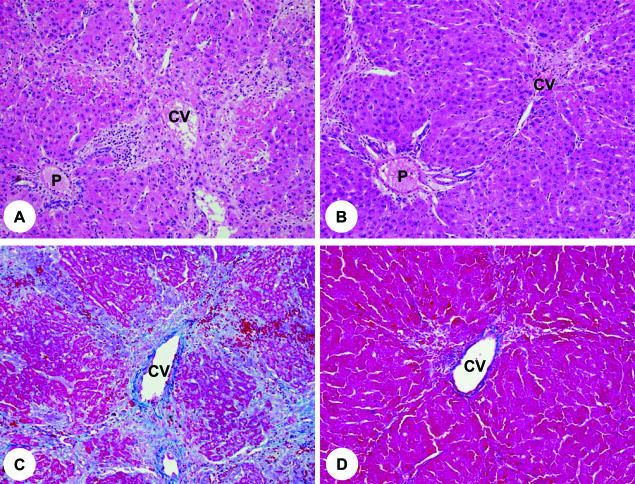
Next, we examined the effect of OSM cDNA introduction on rats damaged by DMN. Rats injected with HVJ-OSM and HVJ-Vector weighed 164 ± 6 and 149 ± 3 g, respectively, indicating that the OSM introduction leads to the improvement of the general condition of DMN-damaged rats (Figure 3). We then analyzed the pathological state of livers by examining the liver weight, serum albumin levels, and serum liver injury parameters, ie, T-Bil, D-Bil, AST, and ALT values. Liver weights and serum albumin values of rats injected with HVJ-OSM were significantly higher than those of rats injected with HVJ-Vector, and all serum liver injury parameters were significantly lower (Figure 3). We also analyzed a serum fibrosis parameter, hyaluronic acid value, and found that hyaluronic acid value of rats injected with HVJ-OSM was significantly lower than that of rats injected with HVJ-Vector (Figure 3). These results showed that the fibrogenesis of rats injected with HVJ-OSM was less than those injected with HVJ-Vector. In addition, we examined histology of the liver. In the liver of rats injected with HVJ-Vector, centrilobular necrosis and numerous inflammatory cells were observed (Figure 4A). On the other hand, rats injected with HVJ-OSM showed much less centrilobular necrosis and a smaller number of inflammatory cells (Figure 4B). The development of liver fibrosis was inhibited by HVJ-OSM injection (Figure 4, C and D). Taken together, these results indicate that the repeated injection of HVJ-OSM attenuates DMN-induced liver damage and stimulates regeneration.
Figure 3.
Effects of OSM on body weight, liver weight, serum albumin levels, serum liver injury parameters, and serum, a fibrosis parameter in rats with DMN-induced liver damage. Vector: Rats injected with HVJ-Vector. OSM: Rats injected with HVJ-OSM. Alb, albumin. Data represent the mean of six rats. Bars are standard errors. *P < 0.05, significant difference by t-tests.
Figure 4.
Histology of livers of rats injected with HVJ-Vector or HVJ-OSM. A and C: Liver sections of a rat injected with HVJ-Vector. B and D: Liver sections of a rat injected with HVJ-OSM. Sections of A and B were stained with H&E, and those of C and D with Azan. P, portal vein; CV, central vein. Original magnifications: ×90 (A and B); ×100 (C and D).
Effects of HVJ-OSM Transfection on Proliferation and Apoptosis of Hepatocytes
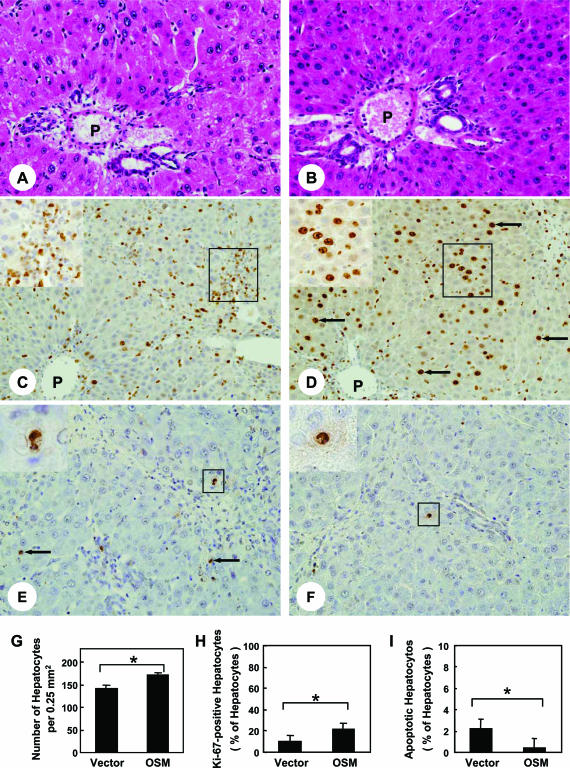
To evaluate the effects of HVJ-OSM injection on proliferative activity of hepatocytes, we examined the density of hepatocytes and the percentage of hepatocytes expressing Ki-67. Histological examination showed that rats injected with HVJ-OSM contained a much larger number of hepatocytes around the portal field than those injected with HVJ-Vector (Figure 5, A, B, and G). Immunohistochemical examination of Ki-67 showed that Ki-67-expressing cells were mainly hepatocytes in rats injected with HVJ-OSM, whereas they were inflammation cells in rats injected with HVJ-Vector (Figure 5, C and D). The proportion of Ki-67-positive hepatocytes to total hepatocytes in portal field was significantly higher in rats injected with HVJ-OSM than those injected with HVJ-Vector (Figure 5H). These results indicate that repeated injection of HVJ-OSM augments hepatocyte proliferation.
Figure 5.
Effects of OSM on proliferation and apoptosis of hepatocytes in rats with DMN-induced liver damage. A, C, and E: Liver sections of a rat injected with HVJ-Vector. B, D, and F: Liver sections of a rat injected with HVJ-OSM. Sections of A and B were stained with H&E, those of C and D were immunostained with Ki-67 antibody, and those of E and F were subjected to TUNEL staining. Arrows in D and E indicate representative Ki-67-positive and apoptotic hepatocytes, respectively. The inset in C is a high magnification of the squared region and shows inflammatory cells expressing Ki-67 antigen. The inset in D is a high magnification of the squared region and shows hepatocytes expressing Ki-67 antigen. Each inset in E and F is a high magnification of the squared region and shows apoptotic hepatocytes. P, portal vein. G and H show the number of total hepatocytes per portal field and the proportion of Ki-67-positive hepatocytes to total hepatocytes, respectively. I shows the proportion of TUNEL-positive hepatocytes to total hepatocytes. Data represent the mean of six rats. Bars are standard errors. *P < 0.05, significant difference by t-tests. Vector: Rats injected with HVJ-Vector. OSM: Rats injected with HVJ-OSM. Original magnifications: ×150 (A and B); ×100 (C and D); ×170 (E and F); ×160 (C and D, insets); ×520 (E and F, insets).
Because DMN is known to induce apoptosis as well as necrosis of hepatocytes,24,25 we examined whether repeated injection of HVJ-OSM prevents apoptosis of hepatocytes. The proportion of TUNEL-positive hepatocytes to total hepatocytes in central vein area (0.25 mm2) of HVJ-OSM-injected livers was 0.4 ± 0.1, which was much less than that of HVJ-Vector-injected livers, 2.2 ± 0.2 (Figure 5, E, F, and I). These results indicate that repeated injection of HVJ-OSM prevents apoptosis of hepatocytes.
Time-Course Studies on osm Gene Therapy before or after DMN Treatment
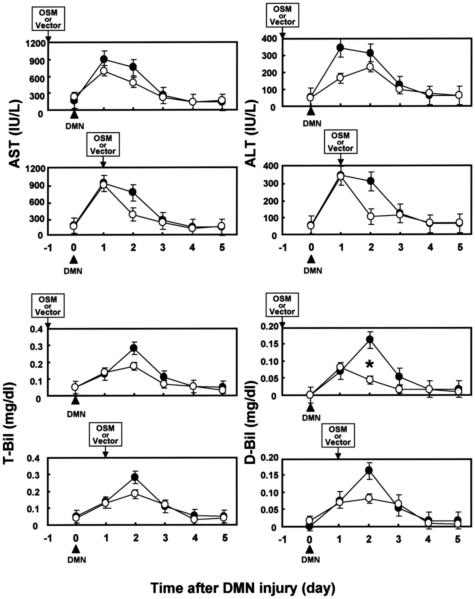
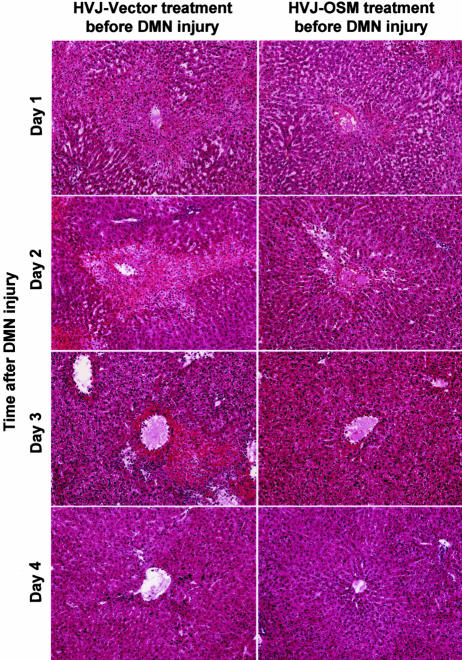
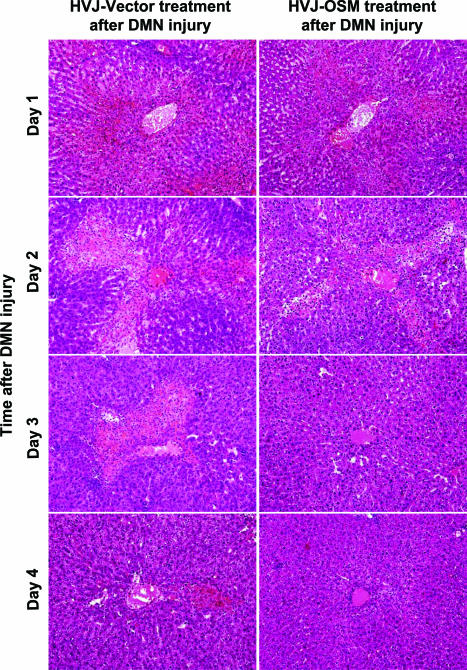
To clarify the biological role of OSM on livers damaged by DMN, we performed time-course studies of the effect of OSM cDNA introduction before or after DMN treatment. In the case of OSM pretreatment, AST and ALT values of rats injected with HVJ-OSM were lower than those of rats injected with HVJ-Vector on days 1 and 2 after DMN treatment. T-Bil and D-Bil values of rats injected with HVJ-OSM were also lower than those of rats injected with HVJ-Vector on day 2 (Figure 6). Centrilobular necrosis caused by subsequent treatment with DMN was much less in rats pretreated with HVJ-OSM than those pretreated with HVJ-Vector (Figure 7). These results indicate that OSM protects livers from cytotoxicity caused by DMN. In the case of posttreatment, serum liver injury parameters (AST, ALT, T-Bil, and D-Bil values) were improved in rats injected with HVJ-OSM, as compared with those injected with HVJ-Vector, on day 1 after HVJ-OSM introduction (Figure 6). Histological study showed that centrilobular necrosis induced by DMN disappeared more rapidly in rats injected with HVJ-OSM than in rats injected with HVJ-Vector (Figure 8). These results indicate that OSM promotes liver regeneration. Thus, OSM is able to protect livers from cytotoxicity caused by DMN and also accelerate regeneration of liver damaged by DMN.
Figure 6.
Time-course studies of the effect of OSM treatment before or after DMN injury on serum liver injury parameters in rats with DMN-induced liver damage. (•): Rats injected with HVJ-Vector. (○): Rats injected with HVJ-OSM. Data represent the mean of three rats. Bars are standard errors. *P < 0.05, significant difference by t-tests.
Figure 7.
Time-course studies on the liver histology of rats injected with HVJ-Vector or HVJ-OSM before DMN injury. Sections were stained with H&E. Original magnifications, ×40.
Figure 8.
Time-course studies on the liver histology of rats injected with HVJ-Vector or HVJ-OSM after DMN injury. Sections were stained with H&E. Original magnifications, ×40.
Discussion
OSM was originally characterized by its ability to inhibit the proliferation of tumor cells,17,18,26 but since then it has been shown to be involved in inflammation, hematopoiesis, embryonic development, and tissue remodeling.17,18,27 In addition, OSM has recently been shown to play an important role in liver development and regeneration.17,18,20,21,22,28 In this study, we examined whether the introduction of rat OSM cDNA enhances the regeneration of rat livers damaged by DMN. Repeated injection of HVJ-OSM into the spleen resulted in increases in body weight, liver weight, and serum albumin levels and the reduction of serum parameters of liver injury and fibrosis. Moreover, histologically, osm gene therapy reduced centrilobular necrosis and inflammatory cell infiltration, promoted hepatocyte proliferation, and suppressed hepatocyte apoptosis. The fibrosis identified by Azan stain was also attenuated by osm gene therapy. These results indicate that OSM is a key mediator for proliferation and anti-apoptosis of hepatocytes and suggest that osm gene therapy is useful for the treatment of patients with liver damage.
Time-course studies on the effect of HVJ-OSM treatment on subsequent liver injury caused by DMN showed that serum liver injury parameters (T-Bil, D-Bil, AST, and ALT values) of rats pretreated with HVJ-OSM were lower than those of rats pretreated with HVJ-Vector. Centrilobular necrosis caused by subsequent treatment with DMN was also much less in rats pretreated with HVJ-OSM than those pretreated with HVJ-Vector. Time-course studies on HVJ-OSM treatment after DMN injury showed that serum AST, ALT, T-Bil, and D-Bil values were improved in rats injected with HVJ-OSM after HVJ-OSM introduction. Centrilobular necrosis induced by DMN was also shown to disappear more rapidly in rats injected with HVJ-OSM than in rats injected with HVJ-Vector. These results indicate that OSM is effective in combating DMN-caused liver injury by administrating either before or after DMN treatment, suggest that osm gene therapy is useful, as preventive and curative means, for the treatment of liver injury.
In this study, we directly injected HVJ-OSM into the spleen of rats with DMN-damaged livers. HVJ-OSM injected into the spleen is expected to reach the liver, because the blood of spleen flows into the portal vein through the splenic hilus. The HVJ envelope is ∼280 nm in diameter,23 whereas the diameter of the fenestrae of sinusoidal endothelial cells, which separate sinusoidal blood from the space of Disse, is ∼175 nm in the periportal area,29 indicating that HVJ-OSM hardly go through the fenestrae of sinusoidal endothelial cells. It is, therefore, likely that HVJ-OSM may be retained in Kupffer cells, which are present within the sinusoid. Ogushi and colleagues30 reported that when mice were injected with fluorescein isothiocyanate-labeled oligodeoxynucleotides encapsulated HVJ liposome into the spleen, more than 95% of fluorescein isothiocyanate-labeled oligodeoxynucleotides were transferred into Kupffer cells and only 1% into endothelial cells of the liver. These findings supported the present results that OSM protein was found exclusively in Kupffer cells after injection of OSM cDNA into the spleen.
The role of OSM in inflammatory responses is complex, performing in either a proinflammatory or an anti-inflammatory manner. In proinflammatory action, OSM increases P-selectin expression, induces granulocyte-macrophage colony-stimulating factor (GM-CSF), granulocyte colony-stimulating factor, and IL-6 secretion in endothelial cells in vitro, and causes an inflammatory response when injected subcutaneously into mice.31,32,33,34 On the other hand, in anti-inflammatory action, OSM induces tissue inhibitor of metalloproteinase-1 (TIMP-1)35 and down-regulates IL-1-induced proinflammatory mediators, such as IL-8, GM-CSF, and RANTES, in human fibroblasts.36 Transfection of mouse osm gene in adenoviral vector has also been shown to induce acute-phase proteins and TIMP-1 expression in mice.37 The present study showed that repeated injection of HVJ-OSM into the spleen of rats with DMN-induced liver damage significantly reduced centrilobular necrosis and the infiltration of inflammatory cells into the damaged sites of livers. Consistent with these findings, Sanchez and colleagues38 have reported that adenoviral transfer of osm gene suppresses dextran-sodium sulfate-induced colitis in mice by reducing macrophage infiltration and apoptosis. These results support the role of OSM as an anti-inflammatory cytokine.
Recently, OSM-R−/− mice have been shown to decrease in proliferating cell nuclear antigen-positive hepatocytes after CCl4-induced liver damage.21 It has also been shown that restoration of liver mass after 70% hepatectomy is delayed in OSM-R−/− mice.21 In this study, we found that the number of total hepatocytes and the proportion of Ki-67-positive hepatocytes to total hepatocytes of rats injected with HVJ-OSM were significantly higher than those injected with HVJ-Vector. These results suggest that OSM-R-mediated signaling is required for the proliferative response of hepatocytes in damaged liver. In human adipose tissue-derived mesenchymal stem cells, two separate signaling pathways, MEK/ERK and JAK3/STAT1, have been shown to be independently involved in the OSM-stimulated proliferation.39 It is therefore possible that these two separate signaling pathways participate in OSM-stimulated proliferation of hepatocytes in the liver regeneration, resulting in a striking hepatocyte growth. Recently, Cohen and colleagues40 reported a specific up-regulation of HGF synthesis by OSM, most likely through the MAPK pathway, in human lung fibroblasts, suggesting that OSM participates in lung repair through HGF production. Because HGF is an important growth factor for hepatocytes, the up-regulation of HGF synthesis by OSM may be involved in the proliferation of hepatocytes in the liver regeneration. The AP-1 transcription factor c-Jun is a key regulator of hepatocyte proliferation. Mice lacking c-Jun display impaired liver regeneration after partial hepatectomy. Liver regeneration is regulated by c-Jun/AP-1 through a pathway involving p53, p21, and the stress kinase p38α.41 These molecules may be also involved in OSM-stimulated liver regeneration. However, the mechanism by which OSM prevents liver injury and enhances liver regeneration remains to be clarified.
In addition, we found that osm gene therapy reduced apoptosis of hepatocytes. A number of studies have shown that STAT3 activated by IL-6 exhibits anti-apoptotic effects through the induction of Bcl-2, Bcl-xL, and FLICE inhibitor protein, which in turn inhibits FLICE and caspase-3 in hepatocytes.42,43 Adenovirus-mediated expression of an active form of STAT3 has also been shown to induce the expression of redox-associated protein redox factor-1 and reduce Fas-mediated apoptosis in the liver.44 Because OSM is known to activate STAT3,17,18 it is possible that OSM may reduce DMN-induced apoptosis of hepatocytes through STAT3 activation.
DMN induces liver fibrosis in a highly reproducible manner, first inducing a central hemorrhagic necrosis followed by the formation of septa and establishing micronodular cirrhosis after 3 weeks of treatment.45 Biological and ultrastructural studies suggest that inflammatory mechanisms are involved in DMN-induced liver fibrosis.45,46,47,48 The present study showed that osm gene therapy could suppress liver fibrosis induced by DMN. Because OSM may function as an anti-inflammatory cytokine, as mentioned above, there is a possibility that the production of inflammatory cytokines involved in fibrosis is suppressed by osm gene therapy. Alternatively, the tissue repair by fibrosis is not indispensable because OSM induces the proliferation of hepatocytes to fill the space of centrilobular necrosis caused by DMN.
The response to partial hepatectomy is impaired in IL-6-deficient mice.8 The studies using liver-specific conditional knockout mice have also shown that activation of STAT3 is required for liver regeneration.49 These findings demonstrate that IL-6 and a downstream mediator of IL-6 signaling pathway, STAT3, are very important molecules for liver regeneration. OSM, like IL-6, activates Jak1, Jak2, and Tyk2 and the activated Jaks in turn activate STAT3.17,18 Recently, Nakamura and colleagues21 have shown that liver regeneration is impaired in OSM-R−/− mice as well as IL-6−/− mice. They have also shown that OSM expression after CCl4 exposure is greatly decreased in IL-6−/− mice, whereas IL-6 expression after CCl4 exposure is not altered in OSMR−/− mice. Moreover, OSM administration in IL-6−/− mice has been shown to induce phosphorylation of STAT3 with the normal kinetics. These results suggest that OSM is a downstream mediator of IL-6 in liver regeneration. Therefore, it is likely that osm gene therapy may be more direct and efficient than the therapy using il-6 gene in the treatment of patients with liver damage.
OSM-R is hardly expressed in hepatocytes in mouse and human livers.21,50 This may reflect very low levels of proliferation of hepatocytes in normal liver. On the other hand, the expression of OSM-R has been shown to rapidly increase in hepatocytes after liver injury.21 Therefore, it is proposed that, in osm gene therapy, a large number of OSM-R in injured hepatocytes interacts with OSM generated from the introduced gene to inhibit hepatocyte apoptosis and promote hepatocyte proliferation, leading to active liver regeneration.
Supplementary Material
Acknowledgments
We thank Ms. Michiko Kakihana, Ms. Mio Kawasumi, and Mr. Hirotsugu Kubo, Hyogo College of Medicine, for their technical assistance.
Footnotes
Address reprint requests to Tohru Tsujimura, M.D., Ph.D., Department of Pathology, Hyogo College of Medicine, 1, Mukogawa, Nishinomiya, Hyogo 663-8501, Japan. E-mail: tohru@hyo-med.ac.jp.
Supported in part by the Ministry of Education, Science, Sports, Culture, and Technology of Japan (grants-in-aid for scientific research and Hitec Research Center grant) and Hyogo College of Medicine (grants-in-aid for promotion of core research projects, graduate students, and researchers).
Supplemental material for this article can be found on http://ajp. amjpathol.org.
References
- Fausto N, Laird AD, Webber EM. Liver regeneration. 2. Role of growth factors and cytokines in hepatic regeneration. FASEB J. 1995;9:1527–1536. doi: 10.1096/fasebj.9.15.8529831. [DOI] [PubMed] [Google Scholar]
- Michalopoulos GK, DeFrances MC. Liver regeneration. Science. 1997;276:60–66. doi: 10.1126/science.276.5309.60. [DOI] [PubMed] [Google Scholar]
- Lindroos PM, Zarnegar R, Michalopoulos GK. Hepatocyte growth factor (hepatopoietin A) rapidly increases in plasma before DNA synthesis and liver regeneration stimulated by partial hepatectomy and carbon tetrachloride administration. Hepatology. 1991;13:743–750. [PubMed] [Google Scholar]
- Rubin RA, O’Keefe EJ, Earp HS. Alteration of epidermal growth factor-dependent phosphorylation during rat liver regeneration. Proc Natl Acad Sci USA. 1982;79:776–780. doi: 10.1073/pnas.79.3.776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webber EM, FitzGerald MJ, Brown PI, Bartlett MH, Fausto N. Transforming growth factor-alpha expression during liver regeneration after partial hepatectomy and toxic injury, and potential interactions between transforming growth factor-alpha and hepatocyte growth factor. Hepatology. 1993;18:1422–1431. [PubMed] [Google Scholar]
- Yamada Y, Fausto N. Deficient liver regeneration after carbon tetrachloride injury in mice lacking type 1 but not type 2 tumor necrosis factor receptor. Am J Pathol. 1998;152:1577–1589. [PMC free article] [PubMed] [Google Scholar]
- Webber EM, Bruix J, Pierce RH, Fausto N. Tumor necrosis factor primes hepatocytes for DNA replication in the rat. Hepatology. 1998;28:1226–1234. doi: 10.1002/hep.510280509. [DOI] [PubMed] [Google Scholar]
- Cressman DE, Greenbaum LE, DeAngelis RA, Ciliberto G, Furth EE, Poli V, Taub R. Liver failure and defective hepatocyte regeneration in interleukin-6-deficient mice. Science. 1996;274:1379–1383. doi: 10.1126/science.274.5291.1379. [DOI] [PubMed] [Google Scholar]
- Kovalovich K, DeAngelis RA, Li W, Furth EE, Ciliberto G, Taub R. Increased toxin-induced liver injury and fibrosis in interleukin-6-deficient mice. Hepatology. 2000;31:149–159. doi: 10.1002/hep.510310123. [DOI] [PubMed] [Google Scholar]
- Gohda E, Tsubouchi H, Nakayama H, Hirono S, Sakiyama O, Takahashi K, Miyazaki H, Hashimoto S, Daikuhara Y. Purification and partial characterization of hepatocyte growth factor from plasma of a patient with fulminant hepatic failure. J Clin Invest. 1988;81:414–419. doi: 10.1172/JCI113334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okano J, Shiota G, Kawasaki H. Protective action of hepatocyte growth factor for acute liver injury caused by d-galactosamine in transgenic mice. Hepatology. 1997;26:1241–1249. doi: 10.1053/jhep.1997.v26.pm0009362368. [DOI] [PubMed] [Google Scholar]
- Kosai K, Matsumoto K, Funakoshi H, Nakamura T. Hepatocyte growth factor prevents endotoxin-induced lethal hepatic failure in mice. Hepatology. 1999;30:151–159. doi: 10.1002/hep.510300102. [DOI] [PubMed] [Google Scholar]
- Matsuda Y, Matsumoto K, Yamada A, Ichida T, Asakura H, Komoriya Y, Nishiyama E, Nakamura T. Preventive and therapeutic effects in rats of hepatocyte growth factor infusion on liver fibrosis/cirrhosis. Hepatology. 1997;26:81–89. doi: 10.1053/jhep.1997.v26.pm0009214455. [DOI] [PubMed] [Google Scholar]
- Ueki T, Kaneda Y, Tsutsui H, Nakanishi K, Sawa Y, Morishita R, Matsumoto K, Nakamura T, Takahashi H, Okamoto E, Fujimoto J. Hepatocyte growth factor gene therapy of liver cirrhosis in rats. Nat Med. 1999;5:226–230. doi: 10.1038/5593. [DOI] [PubMed] [Google Scholar]
- Gómez-Lechón MJ. Oncostatin M: signal transduction and biological activity. Life Sci. 1999;65:2019–2030. doi: 10.1016/s0024-3205(99)00296-9. [DOI] [PubMed] [Google Scholar]
- Taga T, Kishimoto T. Gp130 and the interleukin-6 family of cytokines. Annu Rev Immunol. 1997;15:797–819. doi: 10.1146/annurev.immunol.15.1.797. [DOI] [PubMed] [Google Scholar]
- Miyajima A, Kinoshita T, Tanaka M, Kamiya A, Mukouyama Y, Hara T. Role of Oncostatin M in hematopoiesis and liver development. Cytokine Growth Factor Rev. 2000;11:177–183. doi: 10.1016/s1359-6101(00)00003-4. [DOI] [PubMed] [Google Scholar]
- Tanaka M, Miyajima A. Oncostatin M, a multifunctional cytokine. Rev Physiol Biochem Pharmacol. 2003;149:39–52. doi: 10.1007/s10254-003-0013-1. [DOI] [PubMed] [Google Scholar]
- Tanaka M, Hara T, Copeland NG, Gilbert DJ, Jenkins NA, Miyajima A. Reconstitution of the functional mouse oncostatin M (OSM) receptor: molecular cloning of the mouse OSM receptor β subunit. Blood. 1999;93:804–815. [PubMed] [Google Scholar]
- Kamiya A, Kinoshita T, Ito Y, Matsui T, Morikawa Y, Senba E, Nakashima K, Taga T, Yoshida K, Kishimoto T, Miyajima A. Fetal liver development requires a paracrine action of oncostatin M through the gp130 signal transducer. EMBO J. 1999;18:2127–2136. doi: 10.1093/emboj/18.8.2127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura K, Nonaka H, Saito H, Tanaka M, Miyajima A. Hepatocyte proliferation and tissue remodeling is impaired after liver injury in oncostatin M receptor knockout mice. Hepatology. 2004;39:635–644. doi: 10.1002/hep.20086. [DOI] [PubMed] [Google Scholar]
- Okaya A, Kitanaka J, Kitanaka N, Satake M, Kim Y, Terada K, Sugiyama T, Takemura M, Fujimoto J, Terada N, Miyajima A, Tsujimura T. Oncostatin M inhibits proliferation of rat oval cells, OC15-5, inducing differentiation into hepatocytes. Am J Pathol. 2005;166:709–719. doi: 10.1016/S0002-9440(10)62292-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaneda Y, Tabata Y. Non-viral vectors for cancer therapy. Cancer Sci. 2006;97:348–354. doi: 10.1111/j.1349-7006.2006.00189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pritchard DJ, Butler WH. Apoptosis—the mechanism of cell death in dimethylnitrosamine-induced hepatotoxicity. J Pathol. 1989;158:253–260. doi: 10.1002/path.1711580314. [DOI] [PubMed] [Google Scholar]
- Horn TL, Bhattacharjee A, Schook LB, Rutherford MS. Altered hepatic mRNA expression of apoptotic genes during dimethylnitrosamine exposure. Toxicol Sci. 2000;57:240–249. doi: 10.1093/toxsci/57.2.240. [DOI] [PubMed] [Google Scholar]
- Zarling JM, Shoyab M, Marquardt H, Hanson MB, Lioubin MN, Todaro GJ. Oncostatin M: a growth regulator produced by differentiated histiocytic lymphoma cells. Proc Natl Acad Sci USA. 1986;83:9739–9743. doi: 10.1073/pnas.83.24.9739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace PM, MacMaster JF, Rouleau KA, Brown TJ, Loy JK, Donaldson KL, Wahl AF. Regulation of inflammatory responses by oncostatin M. J Immunol. 1999;162:5547–5555. [PubMed] [Google Scholar]
- Kinoshita T, Sekiguchi T, Xu MJ, Ito Y, Kamiya A, Tsuji K, Nakahata T, Miyajima A. Hepatic differentiation induced by oncostatin M attenuates fetal liver hematopoiesis. Proc Natl Acad Sci USA. 1999;96:7265–7270. doi: 10.1073/pnas.96.13.7265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisse E, De Zanger RB, Charels K, Van Der Smissen P, McCuskey RS. The liver sieve: considerations concerning the structure and function of endothelial fenestrae, the sinusoidal wall and the space of Disse. Hepatology. 1985;5:683–692. doi: 10.1002/hep.1840050427. [DOI] [PubMed] [Google Scholar]
- Ogushi I, Iimuro Y, Seki E, Son G, Hirano T, Hada T, Tsutsui H, Nakanishi K, Morishita R, Kaneda Y, Fujimoto J. Nuclear factor κB decoy oligodeoxynucleotides prevent endotoxin-induced fetal liver failure in a murine model. Hepatology. 2003;38:335–344. doi: 10.1053/jhep.2003.50298. [DOI] [PubMed] [Google Scholar]
- Brown TJ, Liu J, Brashem-Stein C, Shoyab M. Regulation of granulocyte colony-stimulating factor and granulocyte-macrophage colony-stimulating factor expression by oncostatin M. Blood. 1993;82:33–37. [PubMed] [Google Scholar]
- Brown TJ, Rowe JM, Liu JW, Shoyab M. Regulation of IL-6 expression by oncostatin M. J Immunol. 1991;147:2175–2180. [PubMed] [Google Scholar]
- Yao L, Pan J, Setiadi H, Patel KD, McEver RP. Interleukin 4 or oncostatin M induces a prolonged increase in P-selectin mRNA and protein in human endothelial cells. J Exp Med. 1996;184:81–92. doi: 10.1084/jem.184.1.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modur V, Feldhaus MJ, Weyrich AS, Jicha DL, Prescott SM, Zimmerman GA, McIntyre TM. Oncostatin M is a proinflammatory mediator. In vivo effects correlate with endothelial cell expression of inflammatory cytokines and adhesion molecules. J Clin Invest. 1997;100:158–168. doi: 10.1172/JCI119508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards CD, Shoyab M, Brown TJ, Gauldie J. Selective regulation of metalloproteinase inhibitor (TIMP-1) by oncostatin M in fibroblasts in culture. J Immunol. 1993;150:5596–5603. [PubMed] [Google Scholar]
- Richards CD, Langdon C, Botelho F, Brown TJ, Agro A. Oncostatin M inhibits IL-1-induced expression of IL-8 and granulocyte-macrophage colony-stimulating factor by synovial and lung fibroblasts. J Immunol. 1996;156:343–349. [PubMed] [Google Scholar]
- Kerr C, Langdon C, Graham F, Gauldie J, Hara T, Richards CD. Adenovirus vector expressing mouse oncostatin M induces acute-phase proteins and TIMP-1 expression in vivo in mice. J Interferon Cytokine Res. 1999;19:1195–1205. doi: 10.1089/107999099313145. [DOI] [PubMed] [Google Scholar]
- Sanchez AL, Langdon CM, Akhtar M, Lu J, Richards CD, Bercik P, McKay DM. Adenoviral transfer of the murine oncostatin M gene suppresses dextran-sodium sulfate-induced colitis. J Interferon Cytokine Res. 2003;23:193–201. doi: 10.1089/107999003765027393. [DOI] [PubMed] [Google Scholar]
- Song HY, Jeon ES, Jung JS, Kim JH. Oncostatin M induces proliferation of human adipose tissue-derived mesenchymal stem cells. Int J Biochem Cell Biol. 2005;37:2357–2365. doi: 10.1016/j.biocel.2005.05.007. [DOI] [PubMed] [Google Scholar]
- Cohen M, Marchand-Adam S, Lecon-Malas V, Marchal-Somme J, Boutten A, Durand G, Crestani B, Dehoux M. HGF synthesis in human lung fibroblasts is regulated by oncostatin M. Am J Physiol. 2006;290:L1097–L1103. doi: 10.1152/ajplung.00166.2005. [DOI] [PubMed] [Google Scholar]
- Stepniak E, Ricci R, Eferl R, Sumara G, Sumara I, Rath M, Hui L, Wagner EF. c-Jun/AP-1 controls liver regeneration by repressing p53/p21 and p38 MAPK activity. Genes Dev. 2006;20:2306–2314. doi: 10.1101/gad.390506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukada T, Hibi M, Yamanaka Y, Takahashi-Tezuka M, Fujitani Y, Yamaguchi T, Nakajima K, Hirano T. Two signals are necessary for cell proliferation induced by a cytokine receptor gp130: involvement of STAT3 in anti-apoptosis. Immunity. 1996;5:449–460. doi: 10.1016/s1074-7613(00)80501-4. [DOI] [PubMed] [Google Scholar]
- Kovalovich K, Li W, DeAngelis R, Greenbaum LE, Ciliberto G, Taub R. Interleukin-6 protects against Fas-mediated death by establishing a critical level of anti-apoptotic hepatic proteins FLIP, Bcl-2, and Bcl-xL. J Biol Chem. 2001;276:26605–26613. doi: 10.1074/jbc.M100740200. [DOI] [PubMed] [Google Scholar]
- Haga S, Terui K, Zhang HQ, Enosawa S, Ogawa W, Inoue H, Okuyama T, Takeda K, Akira S, Ogino T, Irani K, Ozaki M. Stat3 protects against Fas-induced liver injury by redox-dependent and -independent mechanisms. J Clin Invest. 2003;112:989–998. doi: 10.1172/JCI17970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jézéquel AM, Mancini R, Rinaldesi ML, Macarri G, Venturini C, Orlandi F. A morphological study of the early stages of hepatic fibrosis induced by low doses of dimethylnitrosamine in the rat. J Hepatol. 1987;5:174–181. doi: 10.1016/s0168-8278(87)80570-6. [DOI] [PubMed] [Google Scholar]
- Jézéquel AM, Mancini R, Rinaldesi ML, Ballardini G, Fallani M, Bianchi F, Orlandi F. Dimethylnitrosamine-induced cirrhosis. Evidence for an immunological mechanism. J Hepatol. 1989;8:42–52. doi: 10.1016/0168-8278(89)90160-8. [DOI] [PubMed] [Google Scholar]
- Myers MJ, Schook LB. Immunotoxicity of nitrosamines. Smialowicz RJ, Holsapple MP, editors. Boca Raton: CRC Press,; Experimental Immunotoxicology. 1996:pp 351–366. [Google Scholar]
- Jin YL, Enzan H, Kuroda N, Hayashi Y, Toi M, Miyazaki E, Hamauzu T, Hiroi M, Guo LM, Shen ZS, Saibara T. Vascularization in tissue remodeling after rat hepatic necrosis induced by dimethylnitrosamine. Med Mol Morphol. 2006;39:33–43. doi: 10.1007/s00795-005-0306-3. [DOI] [PubMed] [Google Scholar]
- Li W, Liang X, Kellendonk C, Poli V, Taub R. STAT3 contributes to the mitogenic response of hepatocytes during liver regeneration. J Biol Chem. 2002;277:28411–28417. doi: 10.1074/jbc.M202807200. [DOI] [PubMed] [Google Scholar]
- Znoyko I, Sohara N, Spicer SS, Trojanowska M, Reuben A. Expression of oncostatin M and its receptors in normal and cirrhotic human liver. J Hepatol. 2005;43:893–900. doi: 10.1016/j.jhep.2005.04.020. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.