Abstract
Congestive heart failure (CHF) has emerged as a major worldwide epidemic and its main causes seem to be the aging of the population and the survival of patients with post-myocardial infarction. Cardiomyocyte dropout (necrosis and apoptosis) plays a critical role in the progress of CHF; thus treatment of CHF by exogenous cell implantation will be a promising medical approach. In the acute phase of cardiac damage cardiac stem cells (CSCs) within the heart divide symmetrically and/or asymmetrically in response to the change of heart homeostasis, and at the same time homing of bone marrow stem cells (BMCs) to injured area is thought to occur, which not only reconstitutes CSC population to normal levels but also repairs the heart by differentiation into cardiac tissue. So far, basic studies by using potential sources such as BMCs and CSCs to treat animal CHF have shown improved ventricular remodelling and heart function. Recently, however, a few of randomized, double-blind, placebo-controlled clinical trials demonstrated mixed results in heart failure with BMC therapy during acute myocardial infarction.
Keywords: Congestive heart failure, Acute myocardial infarction, Myocardial regeneration, Cell therapy
INTRODUCTION
Congestive heart failure (CHF) has emerged as a major worldwide epidemic. Its incidence and prevalence has continued to increase with the aging of the population (Hunt, 2005) and the improved treatment of acute myocardial infarction (AMI) which leads to a longer post-myocardial infarction survival (Menasche, 2003). Evidences have shown that patients with ischemic cardiomyopathy may have a worse prognosis than patients with nonischemic cardiomyopathy such as hypertensive and idiopathic dilated cardiomyopathy (Bart et al., 1997).
Irrespective of the etiology, failing heart is characterized by cardiac hypertrophy, insufficient vascularization and cardiomyocyte loss (Anversa et al., 1993; Fortuno et al., 2001). Cumulative evidences indicated that the normal heart is not a terminally differentiated organ (Anversa and Nadal-Ginard, 2002; Beltrami et al., 2001), and a fraction of cardiomyocytes may be able to reenter the cell-cycle and that limited regeneration can occur though recruitment of resident and/or circulating stem cells (Nadal-Ginard et al., 2003). For example, the human heart replaces completely its myocyte population ≈18 times during the course of life, independently of cardiac diseases (Anversa et al., 2006). Recent literature also showed that primitive cells with properties of stem cells are present in the myocardium; they dwell in stem cell niches in the adult heart. These cardiac stem cells (CSCs) divide symmetrically and/or asymmetrically in response to the change of heart homeostasis, but asymmetric division predominates and the replicating CSCs give rise to one daughter stem cell and one daughter committed cell. By this mechanism of growth kinetics, the pool of primitive CSCs is preserved, and a myocyte progeny is generated together with endothelial and smooth muscle cells (SMCs) (Urbanek et al., 2006).
Following myocardial damage, homing of bone marrow stem cells (BMCs) such as endothelial progenitor cells (EPCs) (Leone et al., 2005) and multipotent adult progenitor cells (Jiang et al., 2002a) to injured area is thought to occur; the stimulus may be stem cell factor (SCF) and/or granulocyte colony-stimulating factor (G-CSF) and other cytokines released from the injured region (Lapidot and Petit, 2002). Thus, although CSC populations are acutely depleted they are reconstituted to normal levels by both self-proliferation and selective homing of BMCs (Fazel et al., 2006; Mouquet et al., 2005). The above forms the cardiomyocyte regenerating pool (Fig.1). New cycling cells may constantly enter this pool through the differentiation of the stem-like cells, whereas other myocytes continuously exit this pool when they become terminally differentiated, losing their capacity to reenter the cell cycle (Urbanek et al., 2005). Ischemic heart disease and cardiac failure increase the rate of flux through this transient pool (Anversa and Nadal-Ginard, 2002), however, because of insufficient CSC recruitment caused by ischemia stress (Linke et al., 2005; Urbanek et al., 2005), and/or functional decline in circulating and BMCs resulted from aging and environmental risk factors (Rauscher et al., 2003), this natural regenerative process is believed to be inadequate in repairing major injuries with massive cell lost such as AMI and CHF.
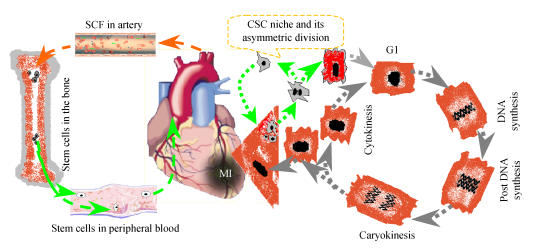
Fig. 1.
The cardiomyocyte regenerating pool
The regeneration of cardiomyocytes comes from mitosis of young myocytes and differentiation of resident CSCs. They go through G1 phase, DNA synthesis, post DNA synthesis, caryokinesis and cytokinesis. However, in pathological conditions such as myocardial infarction (MI), the injured myocardium may enhance not only differentiating CSCs into myocytes but secreting cytokines such as stem cell factor (SCF) into circulation and mobilizing bone marrow stem cells into peripheral blood and then driving them seeding the injured myocardium and differentiating into new myocytes
Replacement and regeneration of functional cardiac muscle after an ischemic insult could be achieved by either stimulating proliferation of endogenous mature cardiomyocytes and resident CSCs or by implanting exogenous donor-derived cells. Thus, new approaches, based on harnessing and expanding the self-renewal potential of the myocardium and enhancing the recruitment and mobilization of exogenous donor-derived cells, should improve the chances to produce true myocardial regeneration that will maintain or restore cardiac function, and delay the time or reduce the need for cardiac transplantation.
POTENTIAL SOURCES
To restore function to nonviable infarcted myocardium or to replace the lost heart muscle cells during pathological or aging process, optimal cell approaches should generate contractile cells that integrate both functionally and structurally into the surrounding viable myocardium and develop a network of capillaries and larger size blood vessels for supply of oxygen and nutrients to the injured region. The newly formed cardiomyocytes must integrate precisely into the existing myocardial wall to augment contractile function of the residual myocardium in a synchronized manner and avoid alterations in the electrical conduction and syncytial contraction of the heart, potentially resulting in life-threatening consequences.
Embryonic stem cells (ESCs)
ESCs are very conducive to cell implantation therapy, mainly because they are pluripotent. These unique pluripotent cell lines can be propagated in the undifferentiated state in culture and coaxed to differentiate into cell derivatives of all three germ layers, including cardiomyocytes (Capi and Gepstein, 2006). A defining characteristic of ESCs is their ability to give rise to teratoma upon implantation, an obviously unacceptable outcome within an injured heart (Ott et al., 2007; Thomson et al., 1998). The human ESCs can differentiate in vitro into cardiomyocytes having electrophysiologic, structural, and contractile properties characteristic of neonatal myocytes (Kehat et al., 2001), and even displaying atrial and ventricular subtypes (Rosenstrauch et al., 2005). ESCs will be the most promising sources of donor cells for cardiac regeneration therapy, however, the future work for cardial regeneration must be dependent on whether differentiated ESCs to cardiogenic lineage are fully induced, which matches the basic instinct of CSCs, otherwise this kind of cell implantation remains to be settled.
Fetal and neonatal cardiomyocytes
These cells are obtained from fetal and neonatal hearts, respectively, and then grown in tissue cultures. In animal models of myocardial infarction, the cells have been successfully engrafted through epicardial injection into ischemic myocardium (Scorsin et al., 1997) and shown to connect with host cells through intercalated discs containing desmosomes and gap junctions (Zhang et al., 2001). However, these exciting results are offset by the strong immunogenicity and small-scale production of engrafted cells and, above all, by the significant ethical issues raised by their use (Menasche, 2003).
Skeletal myoblasts (SMs)
Histologically, SMs are committed progenitors of skeletal muscle cells. Normally, they reside under the basement membrane of skeletal muscle fibers, where they remain quiescent until recruited for repair or regeneration of damaged muscle tissue. Studies in rats and human being confirmed that implanted SMs could repopulate scar tissue, resulting in ventricular wall thickening, elevated left ventricular ejection fraction (LVEF), and improved contractility (Murry et al., 1996; Taylor et al., 1998). However, as shown by fluorescent studies, the engrafted cells also developed a peculiar phenotype of hyperexcitable myotubes with contractile activity that was completely unaffected by neighboring muscle cells (Leobon et al., 2003). Another investigation showed that SM-derived myocytes did not necessarily integrate into the surrounding host myocardium, as evidenced by the normal expression in vitro versus downregulated expression in vivo of 2 key proteins involved in electromechanical cell integration N-cadherin and connexin-43 (Reinecke et al., 2000). Thus, intensive studies aimed at improving cooperation between myoblasts and cardiomyocytes in response to synchronous electrophysiological stimuli, including studies using conditioned gene transfer technologies, are necessary.
Bone marrow stem cells (BMCs)
Adult BMCs mainly consist of hemopoietic stem cells, EPCs and mesenchymal stem cells (MSCs); they can be directly enriched from the bone marrow elements or indirectly from the peripheral circulation after mobilization with the cytokine treatment; thus there are no ethical and immunological complications associated with the use of cardiomyocytes derived from fetal tissues or ESCs. Whether derived from peripheral blood or bone marrow, BMCs are readily accessible and are already part of the current clinical arsenal for management of hematological diseases so that BMCs have been used in recent attempts at myocardial regeneration and neoangiogenesis (Jackson et al., 2001; Kamihata et al., 2001).
Among the multipotent cells found in bone marrow, MSCs seem to be an exciting source for cell therapy, for which can differentiate into nerve cells, skeletal muscle cells, and vascular endothelial cells (Jiang et al., 2002b), and into cells with cardiomyocyte features in vitro, either through exposure to 5-azacytidine (Makino et al., 1999) or coculture with cardiomyocytes (Rangappa et al., 2003). Furthermore, these cells are positive for β-myosin heavy chain, cardiac troponin T, α-cardiac actin and desmin after differentiation and they exhibit Ca2+ transients, several types of action potentials (Li et al., 2007), and respond to α and β adrenergic stimulation with an increase in contractility, phosphorylation of ERK1/2 and an increase in cAMP.
Cardiac stem cells (CSCs)
A distinct population of CSCs has been isolated from adult rat heart. These small, primitive cells are negative for blood lineage markers (Lin−) and positive for c-kit (c-kit+). They are self-renewing, clonogenic, and multipotent, with the ability to generate cardiomyocytes, SMCs, and endothelial cells in vitro and in vivo (Barile et al., 2007; Leri et al., 2005). When injected into border zone 5 h after infarction in adult rats, these CSCs induced the formation of myocardium, composed of cardiomyocytes, capillaries, and arterioles in the infarcted area (Beltrami et al., 2003). When delivered in a clinically relevant manner—intracoronary injection, CSCs traverse the vessel barrier, regenerate infarcted myocardium, and improve cardiac function (Dawn et al., 2005). In the nonmyocyte fraction of mouse heart, a resident population of CSCs characterized by expression of stem cell antigen 1 (Sca-1), but lacking expression of blood lineage markers and c-kit, has been reported. Although these cells do not spontaneously differentiate in vitro, when they are exposed to 5-azacytidine, a small fraction of the population acquires biochemical evidence and functional evidence of cardiac myocyte differentiation. When injected intravenously into mice subjected to myocardial ischemia—reperfusion, these Sca-1+ cells were able to home to the injured tissue and differentiate into cardiomyocytes (Oh et al., 2003).
SP cells are side population stem cells in the heart; they have the ability to efflux the dye, Hoechst 33342, by a multi-drug-like transporter, and do not express myosin heavy chain and rapidly decrease from 4 d after birth. SP cells can acquire an adult cardiomyocyte phenotype when placed in a permissive environment. Among cardiac SP cells, the greatest potential for cardiomyogenic differentiation is restricted to cells that express Sca-1 but not CD31 (Pfister et al., 2005).
Isl1 cells are another group of resident CSCs which are characterized by the expression of the antigen ‘islet 1’ (Isl1). Isl1 is a gene initially iterated in early embryonic mesodermal cells that were clearly committed to a cardiogenic lineage, and Isl1+ cells express the homeobox gene islet-1 and these Isl1+ cells develop into cells with a mature cardiac phenotype including intact Ca2+-cycling and the generation of action potentials (Laugwitz et al., 2005).
Cardiosphere cells are isolated from myocyte depleted fractions of murine and human atria and ventricles that form spheres when cultured in ‘cardiosphere growing medium’ (Messina et al., 2004). They express c-kit and are self-renewing, clonogenic, and multipotent, with the ability to spontaneously differentiate into cardiomyocytes, SMCs, and endothelial cells. When injected into the myocardium in severe combined immunodeficient disease mice immediately after myocardial infarction, they acquired phenotypic markers of cardiac cells such as lamin A/C, a human myocyte marker, and connexin-43. Cardiosphere-derived cells are cardiogenic in vitro; they promote cardiac regeneration and improve heart function in a mouse infarct model, which provides motivation for further development for therapeutic applications in patients (Smith et al., 2007).
Uncommitted cardiac progenitor cells (UPCs) isolated from neonatal and adult rat ventricles have been characterized by expression of the ESC marker stage-specific embryonic antigen 1 (SSEA-1) (Ott et al., 2007). In the neonatal heart, small clusters of SSEA-1+ cells were dispersed among mature cardiomyocytes throughout the heart. In the adult heart, SSEA-1+ cells remained as single cells in the right and left ventricles and as clusters in the right ventricular outflow tract. When highly enriched populations of suspended UPCs were isolated and expanded over a cardiac-derived mesenchymal feeder layer, temporal maturation of the original cells, from an uncommitted SSEA-1+ or SSEA-1+/octamer-4+ (oct-4+) state to a mesodermally committed flk-1+ state, then to an flk-1+ and Sca-1+ state and then to a cardiac committed Nkx2.5+, GATA-4+, or Isl1+ state, was observed. Ultimately, cells differentiated into mature cardiomyocytes, endothelial cells, and SMCs. When adenovirally green fluorescence protein-labelled, SSEA-1+ UPCs were injected into the failing rat heart, they generated mature cardiomyocytes and vasculature and led to substantial functional improvement and attenuation of left ventricular remodelling, but no evidence of teratoma was seen (Ott et al., 2007).
DELIVERY METHODS
Transvascular strategies
Transvascular strategies are especially suited for the prevention of recently infarct-induced heart failure when chemoattractants and cell adhesion molecules are highly expressed.
Intracoronary vessel infusion
Intracoronary application delivers a maximum concentration of cells homogeneously to the site of injury during first passage. Unselected BMCs (Ge et al., 2006; Wollert et al., 2004), circulating blood-derived progenitor cells (Erbs et al., 2005), and MSCs (Chen et al., 2004) have been delivered via the intracoronary artery route in patients with AMI and ischemic cardiomyopathy in which cells were delivered through the central lumen of an over-the-wire balloon catheter during transient balloon inflations to maximize the contact time of the cells with the microcirculation of the infarct-related artery.
The intracoronary venous route, the retrograde coronary perfusion, is another important delivery method for stem cell therapy (Giordano, 2003; Murad-Netto et al., 2004). The coronary venous system was cannulated with a novel, investigational, double-balloon catheter. This device comprises 2 concentric catheters; a proximal, outer catheter bearing a large balloon, and a distal, inner catheter bearing a smaller balloon; the distal balloon is used to minimize washout of delivered cells into the systemic circulation via distal, low-resistance veno-venous anastomoses (Hou et al., 2005).
Intravenous injection
Intravenous injection is the simplest and least invasive method of administration of stem cells and obviates the need for cardiac surgery or cardiac catheterization (Kocher et al., 2001). In this mode of delivery, the optimal dose of stem cells is of particular importance. However, because of the long circulation time, cells could be lost by extraction toward noncardiac organs and fail to home to the area of infarct (Hofmann et al., 2005).
In the acute phase of cardiac damage, BMCs could also be mobilized with G-CSF and SCF injections (Kawada et al., 2004; Orlic et al., 2001a). This kind of approach decreased mortality, infarct size, cavity dilatation, and diastolic stress in a murine infarct model (Orlic et al., 2001a). In addition, there were improvements in LVEF and hemodynamics due to the formation of new cardiomyocytes with arterioles and capillaries (Zohlnhöfer et al., 2007).
Direct injection in the ventricular wall
Direct injection is the preferred route for cell delivery in patients with CHF or when cell homing signals are expressed at low levels in the heart (scar tissue).
Transendocardial injection is using an injection needle catheter advanced across the aortic valve and positioned against the endocardial surface; cells can be directly injected into the left ventricular wall (Amado et al., 2005). Electromechanical mapping of the endocardial surface can be used to delineate viable, ischemic, and scarred myocardium before cell injections, which is the best method so far to deliver cells in CHF patients (Perin et al., 2003).
Percutaneous transcoronary-venous transplantation was performed using the TransAccess catheter system under fluoroscopic and intravascular ultrasound guidance (Siminiak et al., 2005). Working through the coronary sinus, a specialized catheter system was easily delivered to the anterior interventricular coronary vein. The composite catheter system (TransAccess) incorporates a phased-array ultrasound tip for guidance and a sheathed, extendable nitinol needle for transvascular myocardial access. A microinfusion (IntraLume) catheter was advanced through the needle, deep into remote myocardium, and the cells suspension was injected (Thompson et al., 2003).
Transepicardial injection has been performed as an adjunct to a surgical procedure such as coronary artery bypassing grafting (Archundia et al., 2005; Patel et al., 2005), which during opening heart surgery allows for a direct visualization of the myocardium and a targeted application of cells to scarred areas and/or the border zone of an infarct scar.
POSSIBLE MECHANISMS
Differentiation of CSCs to cardiogenic cell lineages
Lin−/c-kit+ cells, with the properties of CSCs, exist in the rat heart. They are self-renewing, clonogenic, and multipotent, giving rise to myocytes, smooth muscle, and endothelial cells; when injected into an ischemic heart, these cells or their clonal progeny reconstitute well-differentiated myocardium, formed by blood-carrying new vessels and myocytes with the characteristics of young cells (Beltrami et al., 2003). In the adult mice heart CSCs expressing Sca-1 exist and given intravenously after ischemia-reperfusion, Sca-1+ cells home to injured myocardium. By using a Cre/Lox donor/recipient pair, differentiation was shown to occur roughly equally, with and without fusion to host cells (Oh et al., 2003).
Rat CSCs delivered intravascularly traverse the vessel barrier, regenerate infarcted myocardium, and improve cardiac function (Dawn et al., 2005). Activation of resident CSCs by hepatocyte growth factor and insulin-like growth factor 1 in the damaged dog heart can promote a significant restoration of dead tissue, which is paralleled by a progressive improvement in cardiac function (Linke et al., 2005).
Unsettled transdifferentiation of BMCs to cardiomyocytes
In 2001, an exciting publication (Orlic et al., 2001b) showed that locally delivered BMCs could generate de novo myocardium, ameliorating the outcome of coronary artery disease. However, in 2004, two independent studies seriously challenged the above findings (Balsam et al., 2004; Murry et al., 2004). Both sets of authors used state-of-the-art genetic tools to examine whether BMCs transplanted into damaged hearts could take on the role of heart muscle cells and improve heart function. In both studies BMCs showed little evidence of becoming cardiac muscle cells after they were transplanted into damaged mouse hearts. Instead, these stem cells developed into different blood-cell types, despite being in the heart.
More recently, a group of investigators (Dai et al., 2005) demonstrated that allogeneic MSCs could survive in infarcted myocardium as long as 6 months and express markers that suggested muscle and endothelium phenotypes. Another group of investigators (Kajstura et al., 2005) obtained c-kit+ BMCs from male transgenic mice expressing enhanced green fluorescence protein (EGFP). EGFP and the Y-chromosome were used as markers of the progeny of the transplanted cells in the recipient heart. By these approaches, they demonstrated that BMCs, when properly administrated in the infarcted heart, efficiently differentiated into cardiomyocytes and coronary vessels with no detectable differentiation into hematopoietic lineages. Within the infarct, in 10 d, nearly 4.5 million biochemically and morphologically differentiated myocytes together with coronary arterioles and capillary structures were generated.
In contrast to the unsettled transdifferentiation of BMCs, other phenomena, such as cell fusion and cell integration, are well-recognized; this may benefit a little bit of ventricular remodelling and improve cardiac performance (Nygren et al., 2004). Cell fusion occurs with host nuclei penetrating the implanted cells, whereas cell integration refers to the ability of certain cell types such as SMs, to integrate into the cardiac syncytium without transdifferentiating into the myocardium.
Neovascularization
Neovascularization is an important process, as increasing perfusion to infarcted myocardium may decrease ventricular dilatation and improve cardiac function by rescuing hibernating myocardium, decreasing apoptosis of hypertrophied cardiomyocytes, and helping restore injured matrix. Intravenously administrated EPCs into rats after myocardial infarction were shown to migrate into the infarcted region within 48 h, differentiate into endothelial cells, induce neovascularization, limit apoptosis of hypertrophied cardiomyocytes in the peri-infarct rim, reduce progressive collagen deposition and scar formation, and prevent late ventricular remodelling (Kocher et al., 2001). Several experiments on various animal models showed that implantation of BMCs or MSCs after myocardial infarction increased capillary density together with an improvement of contractile function (Kobayashi et al., 2000; Krause et al., 2007; Silva et al., 2005; Tomita et al., 2002).
Para- and auto-secretion
Another potential mechanism of the observed beneficial effects is the possibility that transplanted cells can release various chemokines and cytokines which not only stimulate proliferation and/or differentiation of CSCs, but exert cytoprotective functions, thus, affect myocardial remodelling and function (Mazhari and Hare, 2007).
Local delivery of cytokines and growth factors are responsible for ischemic cell survival and endogenous stem cell mobilization and homing (Rafii and Lyden, 2003; Rajnoch et al., 2001). Recent literature showed that myocardial medium for hypoxic preconditioning can induce MSCs differentiation into myocardial-like cells (Xie et al., 2006), and genetically modified MSCs overexpressing the Akt1 gene release paracrine factor(s) that exert cytoprotective effects on cardiomyocytes exposed to hypoxia, and notably, myocardial protection can also be achieved in vivo by Akt concentrated medium injection after acute infarction (Gnecchi et al., 2005). Thus, a substantial portion of the salutary effects of Akt modified MSCs is attributable to protection of ischemic myocardium instead of de novo regeneration by cardiomyogenic differentiation of the donor cells.
Others
There is strong evidence that some of the beneficial effects of cell transplantation were due to a reorganization of the extracellular matrix of both the injured heart region and the normal myocardium to prevent CHF (Murtuza et al., 2004). Disruption of the matrix network impairs support for heart cells, leading to ventricular dilatation. Transplanted cells may reorganize, secrete and incorporate new matrix elements into deficient areas. Restoration of the extracellular matrix could therefore improve structural support for host heart cells, limiting infarct expansion and improving regional function (Fedak et al., 2003).
CLINICAL TRIALS
So far, basic studies to treat animal CHF with cell implantation have shown improved ventricular remodelling and heart function (Fuchs et al., 2001; Li et al., 1996; Miyahara et al., 2006; Nagaya et al., 2005; Ohnishi et al., 2007; Tang et al., 2005; Wei et al., 2006; Yamada et al., 2007; Zimmermann et al., 2006). However, a few of randomized, double-blind, placebo-controlled clinical trials demonstrated mixed results in heart failure with BMC therapy during AMI (Adler and Maddox, 2007; Metharom et al., 2007; Rosenzweig, 2006).
In the treatment of CHF with BMC transfer, a very small controlled trial by Perin et al.(2003) enrolled 21 patients (first 14 patients, treatment; last 7 patients, control) with end-stage ischemic heart disease. BMCs were injected by NOGA Myostar injection catheter. Electromechanical mapping was used to identify viable myocardium for treatment. At 2 months, there was a significant reduction in total reversible defect and improvement in global left ventricular function within the treatment group and between the treatment and control groups on quantitative electrocardiogram-gated single-photon-emission computed tomography analysis. At 4 months, there was improvement in LVEF from a baseline and a reduction in end-systolic volume in the treated patients. Electromechanical mapping revealed significant mechanical improvement of the injected segments (P<0.0005) at 4 months after treatment.
Another trial by Assmus et al.(2006) investigated the effects of cell transplantation in patients with healed myocardial infarction. They randomly assigned, in a controlled crossover study, 75 patients with stable ischemic heart disease who had had a myocardial infarction at least 3 months previously to receive either no cell infusion (23 patients) or infusion of circulating progenitor cells (24 patients) or BMCs (28 patients) into the patent coronary artery supplying the most dyskinetic left ventricular area. Results showed that intracoronary infusion of progenitor cells is safe and feasible in patients with healed myocardial infarction; transplantation of BMCs is associated with moderate but significant improvement in LVEF after 3 months.
In the treatment of AMI and delaying the progress of heart failure, the BOOST study (Wollert et al., 2004) involved 60 patients, and treatment (autologous BMCs versus control) was administered approximately 5 d after a heart attack. At 6 months, there was a mild increase in LVEF in the stem cell group, from about 49% to about 53%. Transfer of BMCs enhanced left ventricular systolic function primarily in myocardial segments adjacent to the infarcted area. Cell transfer did not increase the risk of adverse clinical events, in-stent restenosis, or proarrhythmic effects. However, the relative improvement in LVEF after infusion of BMCs at 6 months, as compared with no infusion, was no longer significant at 18 months, suggesting that the main effect was an acceleration of recovery (Meyer et al., 2006).
The REPAIR-AMI investigator group (Schachinger et al., 2006) randomly assigned patients with AMI to receive an intracoronary infusion of progenitor cells derived from bone marrow (n=101) or placebo medium (n=98) into the infarct artery 3 to 7 d after successful reperfusion therapy. At 4 months, the absolute improvement in LVEF was significantly greater in the BMC group than in the placebo group [(5.5±7.3)% vs (3.0±6.5)%; P=0.01]. Patients with a baseline LVEF at or below the median value of 48.9% derived the most benefit (absolute improvement in LVEF, 5.0%; 95% confidence interval, 2.0 to 8.1). At 1 year, intracoronary infusion of BMCs was associated with a reduction in the prespecified combined clinical endpoint of death, recurrence of myocardial infarction, and any revascularization procedure (P=0.01).
At the same time Janssens et al.(2006) did a small randomized, double-blind, placebo-controlled study on 67 patients from whom the bone marrow was harvested 1 d after successful percutaneuos coronary intervention for ST-elevation myocardial infarction, patients were assigned to receive optimum medical treatment and infusion of placebo (n=34) or BMCs (n=33). The endpoint was the increase in LVEF and the secondary endpoints were changes in infarct size and regional left ventricular function at 4 months’ follow-up, all assessed by magnetic resonance imaging. However, they did not find a significant improvement in LVEF at 6 months in the BMC group, as compared with the control group; compared with placebo infusion, BMC transfer was associated with a significant reduction in myocardial infarct size and a better recovery of regional systolic function. Thus, they concluded that intracoronary transfer of autologous BMCs within 24 h of optimum reperfusion therapy did not augment recovery of global left ventricular function after myocardial infarction, but could favorably affect infarct remodelling. Another small clinical trial by Lunde et al.(2006) enrolled patients with ST-elevation myocardial infarction of the anterior wall treated with percutaneuos coronary intervention. Patients were randomly assigned to the group that underwent intracoronary injection of autologous mononuclear BMCs (n=50) or to the control group (n=47). Left ventricular function was assessed with the use of quantitative electrocardiogram-gated single-photon-emission computed tomography and echocardiography at baseline and magnetic resonance imaging 2 to 3 weeks after the infarction. These procedures were repeated 6 months after the infarction. The two groups did not differ significantly in changes in LVEF and in left ventricular end-diastolic volume or infarct size and had similar rates of adverse events.
The current rationale for cell therapy after AMI and CHF is derived from the premise that the capacity of the adult myocardium for repair after severe injury is inadequate. Such regenerative inadequacy occurs despite demonstrable mobilization of endogenous progenitor cells into the circulation (Fazel et al., 2006; Leone et al., 2005). The clinical investigators enriched stem cells from bone marrow or bloodstream and reinfused into heart vessels in order to expand the intrinsic repair ability of human. Indeed, circulating endothelial progenitor cells prepared ex vivo and readministered in vivo induced vasculogenesis and angiogenesis and improved cardiac recovery after myocardial infarction in both animals and human (Asahara et al., 1997; Kalka et al., 2000). However, animal models have questioned the ability of BMCs to effectively generate cardiomyocytes (Balsam et al., 2004; Murry et al., 2004). Furthermore, clinical evidences have suggested that after intracoronary delivery only 1.3% to 2.6% of infused BMCs are retained in the heart (Hofmann et al., 2005). Functional benefits may also be mediated through paracrine secretion of growth factors or cytokines, which could indirectly promote survival of cardiomyocytes, mobilization of endogenous progenitor cells, or neovascularization. The effects of cell therapy seemed to be restricted to the infarct-related territory, for which current therapeutic strategies might be adequate for smaller infarcts (Janssens et al., 2006; Meyer et al., 2006), leave very small room for cell therapy. Thus, application of cell therapy to improve the outcome and myocardial recovery in patients with large myocardial infarcts, who experience a moderate reduction in LVEF (<40%) despite optimum conventional therapy, warrants continued research in this field (Metharom et al., 2007). Several other possible explanations for the lack of myocardial repair by BMCs exist, including insufficient progenitor recruitment and mobilization for a sufficient time period, functional decline in circulating progenitors resulted from aging and environmental risk factors (Rauscher et al., 2003), the timing of cell administration, and microvascular obstruction in severely ischemic heart (Anversa and Sonnenblick, 1990).
Fortunately, the current ascertained source of cardiogenic lineage differentiation is adult CSCs which can be acquired from endomyocardial biopsy specimens and provide motivation for further development for therapeutic applications in patients (Smith et al., 2007). Another way of getting donor cells may be through somatic cell nuclear transfer (SCNT). This technique, also known as ‘therapeutic cloning’, involves the transfer of the nucleus of a somatic cell into an enucleated donor oocyte. Recently, SCNT has led to the successful derivation of human ESCs that were genetically identical to the somatic nuclear donor cells (Hwang et al., 2004). In theory, cells acquired from SCNT can be directed to develop into heart muscle cells. Because the vast majority of genetic information is contained in the nucleus, these cells would essentially be genetically identical to the patient with the failing heart. When these heart muscle cells were transplanted back into the patient, there would likely be no rejection and no need to expose the patient to immunosuppressants.
CONCLUSION
It was ascertained that CSCs isolated from animals or human being are committed to cardiogenic lineage and have the ability to generate cardiomyocytes, SMCs, and endothelial cells in vitro and in vivo. Implanted SMs could repopulate scar tissue, resulting in ventricular wall thickening, elevated LVEF, and improved contractility despite an increased risk of ventricular arrhythmias. These may be overcome by using conditioned gene transfer technologies. Another source to get door cells for the treatment of heart failure is from human ESCs by SCNT, this application will avoid the growth of teratoma upon implantation. Though the ability of BMCs to effectively generate cardiomyocytes is questioned, functional benefits may be mediated through paracrine secretion of growth factors or cytokines. In the acute phase of myocardial damage BMCs are mobilized and seed the host myocardium reconstituting CSCs to normal levels. Thus, to elucidate clearly which subcomponents of BMCs are responsible for differentiation of cardiogenic lineage is warranted and then prepare ex vivo and readminister in vivo to enhance the intrinsic repair ability. Clinically, it is worth conducting randomized with placebo controlled trials on cell therapy of CHF with adult CSCs which can be acquired from endomyocardial biopsy specimens.
Footnotes
Project (No. 20060400200) supported by the Postdoctoral Science Foundation, China
References
- 1.Adler ED, Maddox TM. Cell therapy for cardiac disease: where do we go from here? Nat Clin Pract Cardiovasc Med. 2007;4(1):2–3. doi: 10.1038/ncpcardio0729. [DOI] [PubMed] [Google Scholar]
- 2.Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, Cattaneo S, Durand DJ, Fitton T, Kuang JQ, Stewart G, et al. Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA. 2005;102(32):11474–11479. doi: 10.1073/pnas.0504388102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anversa P, Sonnenblick EH. Ischemic cardiomyopathy: pathophysiologic mechanisms. Prog Cardiovasc Dis. 1990;33(1):49–70. doi: 10.1016/0033-0620(90)90039-5. [DOI] [PubMed] [Google Scholar]
- 4.Anversa P, Nadal-Ginard B. Myocyte renewal and ventricular remodelling. Nature. 2002;415(6868):240–243. doi: 10.1038/415240a. [DOI] [PubMed] [Google Scholar]
- 5.Anversa P, Li P, Zhang X, Olivetti G, Capasso JM. Ischaemic myocardial injury and ventricular remodelling. Cardiovasc Res. 1993;27(2):145–157. doi: 10.1093/cvr/27.2.145. [DOI] [PubMed] [Google Scholar]
- 6.Anversa P, Kajstura J, Leri A, Bolli R. Life and death of cardiac stem cells: a paradigm shift in cardiac biology. Circulation. 2006;113(11):1451–1463. doi: 10.1161/CIRCULATIONAHA.105.595181. [DOI] [PubMed] [Google Scholar]
- 7.Archundia A, Aceves JL, Lopez-Hernandez M, Alvarado M, Rodriguez E, Diaz Quiroz G, Paez A, Rojas FM, Montano LF. Direct cardiac injection of G-CSF mobilized bone-marrow stem-cells improves ventricular function in old myocardial infarction. Life Sci. 2005;78(3):279–283. doi: 10.1016/j.lfs.2005.04.080. [DOI] [PubMed] [Google Scholar]
- 8.Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, Witzenbichler B, Schatteman G, Isner JM. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275(5302):964–967. doi: 10.1126/science.275.5302.964. [DOI] [PubMed] [Google Scholar]
- 9.Assmus B, Honold J, Schachinger V, Britten MB, Fischer-Rasokat U, Lehmann R, Teupe C, Pistorius K, Martin H, Abolmaali ND, et al. Transcoronary transplantation of progenitor cells after myocardial infarction. N Engl J Med. 2006;355(12):1222–1232. doi: 10.1056/NEJMoa051779. [DOI] [PubMed] [Google Scholar]
- 10.Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC. Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature. 2004;428(6983):668–673. doi: 10.1038/nature02460. [DOI] [PubMed] [Google Scholar]
- 11.Barile L, Chimenti I, Gaetani R, Forte E, Miraldi F, Frati G, Messina E, Giacomello A. Cardiac stem cells: isolation, expansion and experimental use for myocardial regeneration. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl. 1):S9–S14. doi: 10.1038/ncpcardio0738. [DOI] [PubMed] [Google Scholar]
- 12.Bart BA, Shaw LK, McCants CBJr, Fortin DF, Lee KL, Califf RM, O′Connor CM. Clinical determinants of mortality in patients with angiographically diagnosed ischemic or nonischemic cardiomyopathy. J Am Coll Cardiol. 1997;30(4):1002–1008. doi: 10.1016/S0735-1097(97)00235-0. [DOI] [PubMed] [Google Scholar]
- 13.Beltrami AP, Urbanek K, Kajstura J, Yan SM, Finato N, Bussani R, Nadal-Ginard B, Silvestri F, Leri A, Beltrami CA, et al. Evidence that human cardiac myocytes divide after myocardial infarction. N Engl J Med. 2001;344(23):1750–1757. doi: 10.1056/NEJM200106073442303. [DOI] [PubMed] [Google Scholar]
- 14.Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, Kasahara H, Rota M, Musso E, Urbanek K, et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell. 2003;114(6):763–776. doi: 10.1016/S0092-8674(03)00687-1. [DOI] [PubMed] [Google Scholar]
- 15.Capi O, Gepstein L. Myocardial regeneration strategies using human embryonic stem cell-derived cardiomyocytes. J Control Release. 2006;116(2):211–218. doi: 10.1016/j.jconrel.2006.06.027. [DOI] [PubMed] [Google Scholar]
- 16.Chen SL, Fang WW, Ye F, Liu YH, Qian J, Shan SJ, Zhang JJ, Chunhua RZ, Liao LM, Lin S, et al. Effect on left ventricular function of intracoronary transplantation of autologous bone marrow mesenchymal stem cell in patients with acute myocardial infarction. Am J Cardiol. 2004;94(1):92–95. doi: 10.1016/j.amjcard.2004.03.034. [DOI] [PubMed] [Google Scholar]
- 17.Dai W, Hale SL, Martin BJ, Kuang JQ, Dow JS, Wold LE, Kloner RA. Allogeneic mesenchymal stem cell transplantation in postinfarcted rat myocardium: short- and long-term effects. Circulation. 2005;112(2):214–223. doi: 10.1161/CIRCULATIONAHA.104.527937. [DOI] [PubMed] [Google Scholar]
- 18.Dawn B, Stein AB, Urbanek K, Rota M, Whang B, Rastaldo R, Torella D, Tang XL, Rezazadeh A, Kajstura J, et al. Cardiac stem cells delivered intravascularly traverse the vessel barrier, regenerate infarcted myocardium, and improve cardiac function. Proc Natl Acad Sci USA. 2005;102(10):3766–3771. doi: 10.1073/pnas.0405957102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Erbs S, Linke A, Adams V, Lenk K, Thiele H, Diederich KW, Emmrich F, Kluge R, Kendziorra K, Sabri O, et al. Transplantation of blood-derived progenitor cells after recanalization of chronic coronary artery occlusion: first randomized and placebo-controlled study. Circ Res. 2005;97(8):756–762. doi: 10.1161/01.RES.0000185811.71306.8b. [DOI] [PubMed] [Google Scholar]
- 20.Fazel S, Cimini M, Chen L, Li S, Angoulvant D, Fedak P, Verma S, Weisel RD, Keating A, Li RK. Cardioprotective c-kit+ cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines. J Clin Invest. 2006;116(7):1865–1877. doi: 10.1172/JCI27019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fedak PW, Weisel RD, Verma S, Mickle DA, Li RK. Restoration and regeneration of failing myocardium with cell transplantation and tissue engineering. Semin Thorac Cardiovasc Surg. 2003;15(3):277–286. doi: 10.1016/S1043-0679(03)70007-0. [DOI] [PubMed] [Google Scholar]
- 22.Fortuno MA, Ravassa S, Fortuno A, Zalba G, Diez J. Cardiomyocyte apoptotic cell death in arterial hypertension: mechanisms and potential management. Hypertension. 2001;38(6):1406–1412. doi: 10.1161/hy1201.099615. [DOI] [PubMed] [Google Scholar]
- 23.Fuchs S, Baffour R, Zhou YF, Shou M, Pierre A, Tio FO, Weissman NJ, Leon MB, Epstein SE, Kornowski R. Transendocardial delivery of autologous bone marrow enhances collateral perfusion and regional function in pigs with chronic experimental myocardial ischemia. J Am Coll Cardiol. 2001;37(6):1726–1732. doi: 10.1016/S0735-1097(01)01200-1. [DOI] [PubMed] [Google Scholar]
- 24.Ge J, Li Y, Qian J, Shi J, Wang Q, Niu Y, Fan B, Liu X, Zhang S, Sun A, et al. Efficacy of emergent transcatheter transplantation of stem cells for treatment of acute myocardial infarction (TCT-STAMI) Heart. 2006;92(12):1764–1767. doi: 10.1136/hrt.2005.085431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Giordano FJ. Retrograde coronary perfusion: a superior route to deliver therapeutics to the heart? J Am Coll Cardiol. 2003;42(6):1129–1131. doi: 10.1016/S0735-1097(03)00903-3. [DOI] [PubMed] [Google Scholar]
- 26.Gnecchi M, He H, Liang OD, Melo LG, Morello F, Mu H, Noiseux N, Zhang L, Pratt RE, Ingwall JS, et al. Paracrine action accounts for marked protection of ischemic heart by Akt-modified mesenchymal stem cells. Nat Med. 2005;11(4):367–368. doi: 10.1038/nm0405-367. [DOI] [PubMed] [Google Scholar]
- 27.Hofmann M, Wollert KC, Meyer GP, Menke A, Arseniev L, Hertenstein B, Ganser A, Knapp WH, Drexler H. Monitoring of bone marrow cell homing into the infarcted human myocardium. Circulation. 2005;111(17):2198–2202. doi: 10.1161/01.CIR.0000163546.27639.AA. [DOI] [PubMed] [Google Scholar]
- 28.Hou D, Youssef EA, Brinton TJ, Zhang P, Rogers P, Price ET, Yeung AC, Johnstone BH, Yock PG, March KL. Radiolabeled cell distribution after intramyocardial, intracoronary, and interstitial retrograde coronary venous delivery: implications for current clinical trials. Circulation. 2005;112(9 Suppl.):I150–I156. doi: 10.1161/CIRCULATIONAHA.104.526749. [DOI] [PubMed] [Google Scholar]
- 29.Hunt SA. ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure) J Am Coll Cardiol. 2005;46(6):e1–e82. doi: 10.1016/j.jacc.2005.08.022. [DOI] [PubMed] [Google Scholar]
- 30.Hwang WS, Ryu YJ, Park JH, Park ES, Lee EG, Koo JM, Jeon HY, Lee BC, Kang SK, Kim SJ, et al. Evidence of a pluripotent human embryonic stem cell line derived from a cloned blastocyst. Science. 2004;303(5664):1669–1674. doi: 10.1126/science.1094515. [DOI] [PubMed] [Google Scholar]
- 31.Jackson KA, Majka SM, Wang H, Pocius J, Hartley CJ, Majesky MW, Entman ML, Michael LH, Hirschi KK, Goodell MA. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest. 2001;107(11):1395–1402. doi: 10.1172/JCI12150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Janssens S, Dubois C, Bogaert J, Theunissen K, Deroose C, Desmet W, Kalantzi M, Herbots L, Sinnaeve P, Dens J, et al. Autologous bone marrow-derived stem-cell transfer in patients with ST-segment elevation myocardial infarction: double-blind, randomised controlled trial. Lancet. 2006;367(9505):113–121. doi: 10.1016/S0140-6736(05)67861-0. [DOI] [PubMed] [Google Scholar]
- 33.Jiang Y, Vaessen B, Lenvik T, Blackstad M, Reyes M, Verfaillie CM. Multipotent progenitor cells can be isolated from postnatal murine bone marrow, muscle, and brain. Exp Hematol. 2002;30(8):896–904. doi: 10.1016/S0301-472X(02)00869-X. [DOI] [PubMed] [Google Scholar]
- 34.Jiang Y, Jahagirdar BN, Reinhardt RL, Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund T, Blackstad M, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41–49. doi: 10.1038/nature00870. [DOI] [PubMed] [Google Scholar]
- 35.Kajstura J, Rota M, Whang B, Cascapera S, Hosoda T, Bearzi C, Nurzynska D, Kasahara H, Zias E, Bonafe M, et al. Bone marrow cells differentiate in cardiac cell lineages after infarction independently of cell fusion. Circ Res. 2005;96(1):127–137. doi: 10.1161/01.RES.0000151843.79801.60. [DOI] [PubMed] [Google Scholar]
- 36.Kalka C, Masuda H, Takahashi T, Kalka-Moll WM, Silver M, Kearney M, Li T, Isner JM, Asahara T. Transplantation of ex vivo expanded endothelial progenitor cells for therapeutic neovascularization. Proc Natl Acad Sci USA. 2000;97(7):3422–3427. doi: 10.1073/pnas.070046397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kamihata H, Matsubara H, Nishiue T, Fujiyama S, Tsutsumi Y, Ozono R, Masaki H, Mori Y, Iba O, Tateishi E, et al. Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation. 2001;104(9):1046–1052. doi: 10.1161/hc3501.093817. [DOI] [PubMed] [Google Scholar]
- 38.Kawada H, Fujita J, Kinjo K, Matsuzaki Y, Tsuma M, Miyatake H, Muguruma Y, Tsuboi K, Itabashi Y, Ikeda Y, et al. Nonhematopoietic mesenchymal stem cells can be mobilized and differentiate into cardiomyocytes after myocardial infarction. Blood. 2004;104(12):3581–3587. doi: 10.1182/blood-2004-04-1488. [DOI] [PubMed] [Google Scholar]
- 39.Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, Livne E, Binah O, Itskovitz-Eldor J, Gepstein L. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest. 2001;108(3):407–414. doi: 10.1172/JCI200112131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kobayashi T, Hamano K, Li TS, Katoh T, Kobayashi S, Matsuzaki M, Esato K. Enhancement of angiogenesis by the implantation of self bone marrow cells in a rat ischemic heart model. J Surg Res. 2000;89(2):189–195. doi: 10.1006/jsre.2000.5828. [DOI] [PubMed] [Google Scholar]
- 41.Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J, Homma S, Edwards NM, Itescu S. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med. 2001;7(4):430–436. doi: 10.1038/86498. [DOI] [PubMed] [Google Scholar]
- 42.Krause U, Harter C, Seckinger A, Wolf D, Reinhard A, Bea F, Dengler T, Hardt S, Ho A, Katus HA, et al. Intravenous delivery of autologous mesenchymal stem cells limits infarct size and improves left ventricular function in the infarcted porcine heart. Stem Cells Dev. 2007;16(1):31–37. doi: 10.1089/scd.2006.0089. [DOI] [PubMed] [Google Scholar]
- 43.Lapidot T, Petit I. Current understanding of stem cell mobilization: the roles of chemokines, proteolytic enzymes, adhesion molecules, cytokines, and stromal cells. Exp Hematol. 2002;30(9):973–981. doi: 10.1016/S0301-472X(02)00883-4. [DOI] [PubMed] [Google Scholar]
- 44.Laugwitz KL, Moretti A, Lam J, Gruber P, Chen Y, Woodard S, Lin LZ, Cai CL, Lu MM, Reth M, et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature. 2005;433(7026):647–753. doi: 10.1038/nature03215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Leobon B, Garcin I, Menasche P, Vilquin JT, Audinat E, Charpak S. Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc Natl Acad Sci USA. 2003;100(13):7808–7811. doi: 10.1073/pnas.1232447100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Leone AM, Rutella S, Bonanno G, Abbate A, Rebuzzi AG, Giovannini S, Lombardi M, Galiuto L, Liuzzo G, Andreotti F, et al. Mobilization of bone marrow-derived stem cells after myocardial infarction and left ventricular function. Eur Heart J. 2005;26(12):1196–1204. doi: 10.1093/eurheartj/ehi164. [DOI] [PubMed] [Google Scholar]
- 47.Leri A, Kajstura J, Anversa P. Cardiac stem cells and mechanisms of myocardial regeneration. Physiol Rev. 2005;85(4):1373–1416. doi: 10.1152/physrev.00013.2005. [DOI] [PubMed] [Google Scholar]
- 48.Li RK, Jia ZQ, Weisel RD, Mickle DA, Zhang J, Mohabeer MK, Rao V, Ivanov J. Cardiomyocyte transplantation improves heart function. Ann Thorac Surg. 1996;62(3):654–660. doi: 10.1016/S0003-4975(96)00389-X. (Discussion in p.660-661) [DOI] [PubMed] [Google Scholar]
- 49.Li X, Yu X, Lin Q, Deng C, Shan Z, Yang M, Lin S. Bone marrow mesenchymal stem cells differentiate into functional cardiac phenotypes by cardiac microenvironment. J Mol Cell Cardiol. 2007;42(2):295–303. doi: 10.1016/j.yjmcc.2006.07.002. [DOI] [PubMed] [Google Scholar]
- 50.Linke A, Muller P, Nurzynska D, Casarsa C, Torella D, Nascimbene A, Castaldo C, Cascapera S, Bohm M, Quaini F, et al. Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function. Proc Natl Acad Sci USA. 2005;102(25):8966–8971. doi: 10.1073/pnas.0502678102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lunde K, Solheim S, Aakhus S, Arnesen H, Abdelnoor M, Egeland T, Endresen K, Ilebekk A, Mangschau A, Fjeld JG, et al. Intracoronary injection of mononuclear bone marrow cells in acute myocardial infarction. N Engl J Med. 2006;355(12):1199–1209. doi: 10.1056/NEJMoa055706. [DOI] [PubMed] [Google Scholar]
- 52.Makino S, Fukuda K, Miyoshi S, Konishi F, Kodama H, Pan J, Sano M, Takahashi T, Hori S, Abe H, et al. Cardiomyocytes can be generated from marrow stromal cells in vitro. J Clin Invest. 1999;103(5):697–705. doi: 10.1172/JCI5298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mazhari R, Hare JM. Mechanisms of action of mesenchymal stem cells in cardiac repair: potential influences on the cardiac stem cell niche. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl. 1):S21–S26. doi: 10.1038/ncpcardio0770. [DOI] [PubMed] [Google Scholar]
- 54.Menasche P. Cell transplantation in myocardium. Ann Thorac Surg. 2003;75(6 Suppl.):S20–S28. doi: 10.1016/S0003-4975(03)00462-4. [DOI] [PubMed] [Google Scholar]
- 55.Messina E, de Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MV, Coletta M, et al. Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res. 2004;95(9):911–921. doi: 10.1161/01.RES.0000147315.71699.51. [DOI] [PubMed] [Google Scholar]
- 56.Metharom P, Doyle B, Caplice NM. Clinical trials in stem cell therapy: pitfalls and lessons for the future. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl. 1):S96–S99. doi: 10.1038/ncpcardio0730. [DOI] [PubMed] [Google Scholar]
- 57.Meyer GP, Wollert KC, Lotz J, Steffens J, Lippolt P, Fichtner S, Hecker H, Schaefer A, Arseniev L, Hertenstein B, et al. Intracoronary bone marrow cell transfer after myocardial infarction: eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrOw transfer to enhance ST-elevation infarct regeneration) trial. Circulation. 2006;113(10):1287–1294. doi: 10.1161/CIRCULATIONAHA.105.575118. [DOI] [PubMed] [Google Scholar]
- 58.Miyahara Y, Nagaya N, Kataoka M, Yanagawa B, Tanaka K, Hao H, Ishino K, Ishida H, Shimizu T, Kangawa K, et al. Monolayered mesenchymal stem cells repair scarred myocardium after myocardial infarction. Nat Med. 2006;12(4):459–465. doi: 10.1038/nm1391. [DOI] [PubMed] [Google Scholar]
- 59.Mouquet F, Pfister O, Jain M, Oikonomopoulos A, Ngoy S, Summer R, Fine A, Liao R. Restoration of cardiac progenitor cells after myocardial infarction by self-proliferation and selective homing of bone marrow-derived stem cells. Circ Res. 2005;97(11):1090–1092. doi: 10.1161/01.RES.0000194330.66545.f5. [DOI] [PubMed] [Google Scholar]
- 60.Murad-Netto S, Moura R, Romeo LJ, Manoel Neto A, Duarte N, Barreto F, Jensen A, Vina RF, Vraslovik F, Oberdan A, et al. Stem cell therapy with retrograde coronary perfusion in acute myocardial infarction. A new technique. Arq Bras Cardiol. 2004;83(4):352–354. 349–351. doi: 10.1590/S0066-782X2004001600010. [DOI] [PubMed] [Google Scholar]
- 61.Murry CE, Wiseman RW, Schwartz SM, Hauschka SD. Skeletal myoblast transplantation for repair of myocardial necrosis. J Clin Invest. 1996;98(11):2512–2523. doi: 10.1172/JCI119070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Murry CE, Soonpaa MH, Reinecke H, Nakajima H, Nakajima HO, Rubart M, Pasumarthi KB, Virag JI, Bartelmez SH, Poppa V, et al. Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature. 2004;428(6983):664–668. doi: 10.1038/nature02446. [DOI] [PubMed] [Google Scholar]
- 63.Murtuza B, Suzuki K, Bou-Gharios G, Beauchamp JR, Smolenski RT, Partridge TA, Yacoub MH. Transplantation of skeletal myoblasts secreting an IL-1 inhibitor modulates adverse remodeling in infarcted murine myocardium. Proc Natl Acad Sci USA. 2004;101(12):4216–4221. doi: 10.1073/pnas.0306205101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nadal-Ginard B, Kajstura J, Leri A, Anversa P. Myocyte death, growth, and regeneration in cardiac hypertrophy and failure. Circ Res. 2003;92(2):139–150. doi: 10.1161/01.RES.0000053618.86362.DF. [DOI] [PubMed] [Google Scholar]
- 65.Nagaya N, Kangawa K, Itoh T, Iwase T, Murakami S, Miyahara Y, Fujii T, Uematsu M, Ohgushi H, Yamagishi M, et al. Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation. 2005;112(8):1128–1135. doi: 10.1161/CIRCULATIONAHA.104.500447. [DOI] [PubMed] [Google Scholar]
- 66.Nygren JM, Jovinge S, Breitbach M, Sawen P, Roll W, Hescheler J, Taneera J, Fleischmann BK, Jacobsen SE. Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nat Med. 2004;10(5):494–501. doi: 10.1038/nm1040. [DOI] [PubMed] [Google Scholar]
- 67.Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y, Pocius J, Michael LH, Behringer RR, Garry DJ, et al. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci USA. 2003;100(21):12313–12318. doi: 10.1073/pnas.2132126100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ohnishi S, Yanagawa B, Tanaka K, Miyahara Y, Obata H, Kataoka M, Kodama M, Ishibashi-Ueda H, Kangawa K, Kitamura S, et al. Transplantation of mesenchymal stem cells attenuates myocardial injury and dysfunction in a rat model of acute myocarditis. J Mol Cell Cardiol. 2007;42(1):88–97. doi: 10.1016/j.yjmcc.2006.10.003. [DOI] [PubMed] [Google Scholar]
- 69.Orlic D, Kajstura J, Chimenti S, Limana F, Jakoniuk I, Quaini F, Nadal-Ginard B, Bodine DM, Leri A, Anversa P. Mobilized bone marrow cells repair the infarcted heart, improving function and survival. Proc Natl Acad Sci USA. 2001;98(18):10344–10349. doi: 10.1073/pnas.181177898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, et al. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410(6829):701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 71.Ott HC, Matthiesen TS, Brechtken J, Grindle S, Goh SK, Nelson W, Taylor DA. The adult human heart as a source for stem cells: repair strategies with embryonic-like progenitor cells. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl. 1):S27–S39. doi: 10.1038/ncpcardio0771. [DOI] [PubMed] [Google Scholar]
- 72.Patel AN, Geffner L, Vina RF, Saslavsky J, Urschel HCJr, Kormos R, Benetti F. Surgical treatment for congestive heart failure with autologous adult stem cell transplantation: a prospective randomized study. J Thorac Cardiovasc Surg. 2005;130(6):1631–1638. doi: 10.1016/j.jtcvs.2005.07.056. [DOI] [PubMed] [Google Scholar]
- 73.Perin EC, Dohmann HF, Borojevic R, Silva SA, Sousa AL, Mesquita CT, Rossi MI, Carvalho AC, Dutra HS, Dohmann HJ, et al. Transendocardial, autologous bone marrow cell transplantation for severe, chronic ischemic heart failure. Circulation. 2003;107(18):2294–2302. doi: 10.1161/01.CIR.0000070596.30552.8B. [DOI] [PubMed] [Google Scholar]
- 74.Pfister O, Mouquet F, Jain M, Summer R, Helmes M, Fine A, Colucci WS, Liao R. CD31− but Not CD31+ cardiac side population cells exhibit functional cardiomyogenic differentiation. Circ Res. 2005;97(1):52–61. doi: 10.1161/01.RES.0000173297.53793.fa. [DOI] [PubMed] [Google Scholar]
- 75.Rafii S, Lyden D. Therapeutic stem and progenitor cell transplantation for organ vascularization and regeneration. Nat Med. 2003;9(6):702–712. doi: 10.1038/nm0603702. [DOI] [PubMed] [Google Scholar]
- 76.Rajnoch C, Chachques JC, Berrebi A, Bruneval P, Benoit MO, Carpentier A. Cellular therapy reverses myocardial dysfunction. J Thorac Cardiovasc Surg. 2001;121(5):871–878. doi: 10.1067/mtc.2001.112937. [DOI] [PubMed] [Google Scholar]
- 77.Rangappa S, Entwistle JW, Wechsler AS, Kresh JY. Cardiomyocyte-mediated contact programs human mesenchymal stem cells to express cardiogenic phenotype. J Thorac Cardiovasc Surg. 2003;126(1):124–132. doi: 10.1016/S0022-5223(03)00074-6. [DOI] [PubMed] [Google Scholar]
- 78.Rauscher FM, Goldschmidt-Clermont PJ, Davis BH, Wang T, Gregg D, Ramaswami P, Pippen AM, Annex BH, Dong C, Taylor DA. Aging, progenitor cell exhaustion, and atherosclerosis. Circulation. 2003;108(4):457–463. doi: 10.1161/01.CIR.0000082924.75945.48. [DOI] [PubMed] [Google Scholar]
- 79.Reinecke H, MacDonald GH, Hauschka SD, Murry CE. Electromechanical coupling between skeletal and cardiac muscle. Implications for infarct repair. J Cell Biol. 2000;149(3):731–740. doi: 10.1083/jcb.149.3.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Rosenstrauch D, Poglajen G, Zidar N, Gregoric ID. Stem celltherapy for ischemic heart failure. Tex Heart Inst J. 2005;32(3):339–347. [PMC free article] [PubMed] [Google Scholar]
- 81.Rosenzweig A. Cardiac cell therapy-mixed results from mixed cells. N Engl J Med. 2006;355(12):1274–1277. doi: 10.1056/NEJMe068172. [DOI] [PubMed] [Google Scholar]
- 82.Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H, Yu J, Corti R, Mathey DG, Hamm CW, et al. Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med. 2006;355(12):1210–1221. doi: 10.1056/NEJMoa060186. [DOI] [PubMed] [Google Scholar]
- 83.Scorsin M, Hagege AA, Marotte F, Mirochnik N, Copin H, Barnoux M, Sabri A, Samuel JL, Rappaport L, Menasche P. Does transplantation of cardiomyocytes improve function of infarcted myocardium? Circulation. 1997;96(9 Suppl.):II188–II193. [PubMed] [Google Scholar]
- 84.Silva GV, Litovsky S, Assad JA, Sousa AL, Martin BJ, Vela D, Coulter SC, Lin J, Ober J, Vaughn WK, et al. Mesenchymal stem cells differentiate into an endothelial phenotype, enhance vascular density, and improve heart function in a canine chronic ischemia model. Circulation. 2005;111(2):150–156. doi: 10.1161/01.CIR.0000151812.86142.45. [DOI] [PubMed] [Google Scholar]
- 85.Siminiak T, Fiszer D, Jerzykowska O, Grygielska B, Rozwadowska N, Kalmucki P, Kurpisz M. Percutaneous trans-coronary-venous transplantation of autologous skeletal myoblasts in the treatment of post-infarction myocardial contractility impairment: the POZNAN trial. Eur Heart J. 2005;26(12):1188–1195. doi: 10.1093/eurheartj/ehi159. [DOI] [PubMed] [Google Scholar]
- 86.Smith RR, Barile L, Cho HC, Leppo MK, Hare JM, Messina E, Giacomello A, Abraham MR, Marban E. Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation. 2007;115(7):896–908. doi: 10.1161/CIRCULATIONAHA.106.655209. [DOI] [PubMed] [Google Scholar]
- 87.Tang YL, Zhao Q, Qin X, Shen L, Cheng L, Ge J, Phillips MI. Paracrine action enhances the effects of autologous mesenchymal stem cell transplantation on vascular regeneration in rat model of myocardial infarction. Ann Thorac Surg. 2005;80(1):229–236. doi: 10.1016/j.athoracsur.2005.02.072. (Discussion in p.236-237) [DOI] [PubMed] [Google Scholar]
- 88.Taylor DA, Atkins BZ, Hungspreugs P, Jones TR, Reedy MC, Hutcheson KA, Glower DD, Kraus WE. Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med. 1998;4(8):929–933. doi: 10.1038/nm0898-929. [DOI] [PubMed] [Google Scholar]
- 89.Thompson CA, Nasseri BA, Makower J, Houser S, McGarry M, Lamson T, Pomerantseva I, Chang JY, Gold HK, Vacanti JP, et al. Percutaneous transvenous cellular cardiomyoplasty. A novel nonsurgical approach for myocardial cell transplantation. J Am Coll Cardiol. 2003;41(11):1964–1971. doi: 10.1016/S07351097(03)00397-8. [DOI] [PubMed] [Google Scholar]
- 90.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 91.Tomita S, Mickle DA, Weisel RD, Jia ZQ, Tumiati LC, Allidina Y, Liu P, Li RK. Improved heart function with myogenesis and angiogenesis after autologous porcine bone marrow stromal cell transplantation. J Thorac Cardiovasc Surg. 2002;123(6):1132–1140. doi: 10.1067/mtc.2002.120716. [DOI] [PubMed] [Google Scholar]
- 92.Urbanek K, Rota M, Cascapera S, Bearzi C, Nascimbene A, de Angelis A, Hosoda T, Chimenti S, Baker M, Limana F, et al. Cardiac stem cells possess growth factor-receptor systems that after activation regenerate the infarcted myocardium, improving ventricular function and long-term survival. Circ Res. 2005;97(7):663–673. doi: 10.1161/01.RES.0000183733.53101.11. [DOI] [PubMed] [Google Scholar]
- 93.Urbanek K, Cesselli D, Rota M, Nascimbene A, de Angelis A, Hosoda T, Bearzi C, Boni A, Bolli R, Kajstura J, et al. Stem cell niches in the adult mouse heart. Proc Natl Acad Sci USA. 2006;103(24):9226–9231. doi: 10.1073/pnas.0600635103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wei HJ, Chen SC, Chang Y, Hwang SM, Lin WW, Lai PH, Chiang HK, Hsu LF, Yang HH, Sung HW. Porous acellular bovine pericardia seeded with mesenchymal stem cells as a patch to repair a myocardial defect in a syngeneic rat model. Biomaterials. 2006;27(31):5409–5419. doi: 10.1016/j.biomaterials.2006.06.022. [DOI] [PubMed] [Google Scholar]
- 95.Wollert KC, Meyer GP, Lotz J, Ringes-Lichtenberg S, Lippolt P, Breidenbach C, Fichtner S, Korte T, Hornig B, Messinger D, et al. Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet. 2004;364(9429):141–148. doi: 10.1016/S01406736(04)16626-9. [DOI] [PubMed] [Google Scholar]
- 96.Xie XJ, Wang JA, Cao J, Zhang X. Differentiation of bone marrow mesenchymal stem cells induced by myocardial medium under hypoxic conditions. Acta Pharmacol Sin. 2006;27(9):1153–1158. doi: 10.1111/j.17457254.2006.00436.x. [DOI] [PubMed] [Google Scholar]
- 97.Yamada Y, Yokoyama S, Wang XD, Fukuda N, Takakura N. Cardiac stem cells in brown adipose tissue express CD133 and induce bone marrow nonhematopoietic cells to differentiate into cardiomyocytes. Stem Cells. 2007;25(5):1326–1333. doi: 10.1634/stemcells.2006-0588. [DOI] [PubMed] [Google Scholar]
- 98.Zhang M, Methot D, Poppa V, Fujio Y, Walsh K, Murry CE. Cardiomyocyte grafting for cardiac repair: graft cell death and anti-death strategies. J Mol Cell Cardiol. 2001;33(5):907–921. doi: 10.1006/jmcc.2001.1367. [DOI] [PubMed] [Google Scholar]
- 99.Zimmermann WH, Melnychenko I, Wasmeier G, Didie M, Naito H, Nixdorff U, Hess A, Budinsky L, Brune K, Michaelis B, et al. Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nat Med. 2006;12(4):452–458. doi: 10.1038/nm1394. [DOI] [PubMed] [Google Scholar]
- 100.Zohlnhöfer D, Kastrati A, Schomig A. Stem cell mobilization by granulocyte-colony-stimulating factor in acute myocardial infarction: lessons from the REVIVAL-2 trial. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl. 1):S106–S109. doi: 10.1038/ncpcardio0745. [DOI] [PubMed] [Google Scholar]