Abstract
Objective
To present our experience with three cases of rectoprostatic fistula with special emphasis on diagnosis and conservative management.
Patients and Methods
Three middle-aged men presented to us differently. All had spontaneous rectoprostatic fistulas. Biopsy showed tuberculosis though three consecutive urine samples for acid-fast bacilli were negative. None of the patients were immunocompromised. Their upper tracts were normal and all had a past history of pulmonary tuberculosis. They were started on antitubercular drugs and urinary diversion with or without faecal diversion.
Results
All fistulae healed completely within 6 weeks of starting antitubercular treatment. One patient healed with bladder neck stenosis that required bladder neck incision. Voiding was normal on 1-year follow-up.
Conclusion
Spontaneous tubercular rectoprostatic fistulae are rare. There should be a strong clinical suspicion in endemic areas. Prostatic biopsy proves the diagnosis. Conservative management with antitubercular drugs and urinary diversion with or without faecal diversion has a high success rate and should be the first line of treatment even if urine is negative for acid-fast bacilli.
Keywords: Tuberculosis, Rectoprostatic fistula, Rectourethral fistula, Genitourinary fistula
For obvious anatomical reasons, direct communication between the rectum and prostatic urethra has been confined to males. Rectourethral fistula is a rare condition.1 Literature on this subject is scanty and has been provided by surgeons interested in either rectal neoplasm or imperforate anus and by urologists involved in misfortunes of prostatectomy. Strangely, inflammatory disease of the intestine like Crohn's rarely (0.3%) cause rectourethral fistula.2 Tubercular rectoprostatic fistulae are very uncommon. Literature reveals 6 cases and none in the last 15 years. We recently treated three such cases with diversion and antitubercular drugs. All the fistulae healed and patients are voiding normally.
Patients and Methods
Since August 2003, three patients with spontaneous tubercular rectoprostatic fistula were diagnosed and managed conservatively at our institution with complete healing in all the patients. The cases are discussed individually below.
Case 1
A 40-year-old man presented with a 3-month history of lower abdominal pain and severe burning while passing urine. He also had had increased frequency (12–15 times in the day and 4–5 times at night) for 2 months and one episode of haematuria with clots. One month previously, he had experienced fever with chills and rigor and a week prior to visiting the clinic had started passing small amounts of urine per anum. The past history was noteworthy in that he had been treated for pulmonary tuberculosis 25 years previously. He had also undergone surgery for a perianal fistula 10 years ago.
Systemic examination was unremarkable. Rectal examination revealed a perianal scar at 2 ‘o’-clock on the anal verge and a firm irregular tender mass 6 cm from the anal verge. Mucosa was free with no induration. The prostate could not be made out separately from the mass.
Urine microscopy confirmed numerous leukocytes and few RBCs while culture grew significant colonies of Escherichia coli sensitive to routine drugs. Three consecutive samples of urine for AFB smear were negative. Plain skiagram and ultrasound abdomen were normal. The prostate on ultrasound showed an hypoechoic mass 25 × 15 cm at bladder base extending into the right seminal vesicle. Micturating cystourethrogram showed a 1.5 cm oblique fistulous tract connecting the rectum and the prostatic urethra. CT of abdomen and pelvis confirmed the above and showed thickening of anterior rectal and posterior bladder wall with inflammatory changes noted in the periprostatic tissues (Fig. 1). Both kidneys were normal and there was no ureteric dilatation. There were multiple para-aortic, aorto-caval and right para-iliac lymph nodes (largest 1.4 × 0.9 cm). Cystoscopy under anaesthesia showed whitish nodules in the anterior urethra. The prostatic urethra was inflamed and rigid. A 1.5-cm oblique fistulous tract was seen extending posterosuperiorly from prostatic urethra just proximal to veru with inflamed edges. The distal extent of the tract could not be seen. The edges of the fistula were inflamed and friable. No fistulous opening could be palpated rectally. Biopsy of the fistulous opening in the prostate showed granulomatous inflammation consistent with tuberculous but no AFB were seen. He was put on suprapubic catheter (SPC) drainage and antitubercular treatment was started. The fistula healed in a month after which the SPC was removed. He is voiding normally at 1-year follow-up.
Figure 1.

CT pelvis of case 1 showing extravasation of contrast from the prostatic urethra tracking posteriorly into the rectum.
Case 2
A 64-year-old man was admitted to his local hospital with a history of dribbling urine, high-grade fever and severe dysuria for the last month. He developed acute urinary retention 3 days later in the hospital for which he was catheterised and ∼2-l of urine drained immediately. He gave a history of severe rectal bleeding for which a sigmoidoscopy was done that confirmed a rectal ulcer. The rest of the colon was normal. He was referred to us on a urethral catheter. He had past history of treatment for multi-drug resistant pulmonary tuberculosis over 20 years.
Examination was unremarkable except for a small 7-mm rectal ulcer felt on the anterior wall overlying the prostate. The prostate was small, firm and nodular.
Urine microscopy showed pyuria and microscopic haematuria; repeated cultures grew a mixture of microorganisms. Total PSA was 2.8 ng/ml and free PSA was 0.28 ng/ml. Ultrasound showed normal upper tracts and the prostate was heterogeneous in echotexture. Since the DRE was abnormal and the ultrasound showed an heterogeneous echotexture, TRUS biopsy was done; this was inconclusive and the clinical picture suggested bladder outflow obstruction. A TURP was performed. Cystoscopy was normal and TURP was uneventful. After catheter removal, he had urinary leak per rectum and pneumaturia. A diverting sigmoid colostomy and SPC was done.
TUR biopsy of the prostate showed necrotising granulomatous inflammation consistent with tuberculosis. He was started on antitubercular drugs. On his first visit after 6 weeks, the fistula had healed. An MCU confirmed this (Fig. 2), the SPC was removed and he was voiding without any complaints. The colostomy was closed after 6 weeks and he was well at 1-year follow-up.
Figure 2.
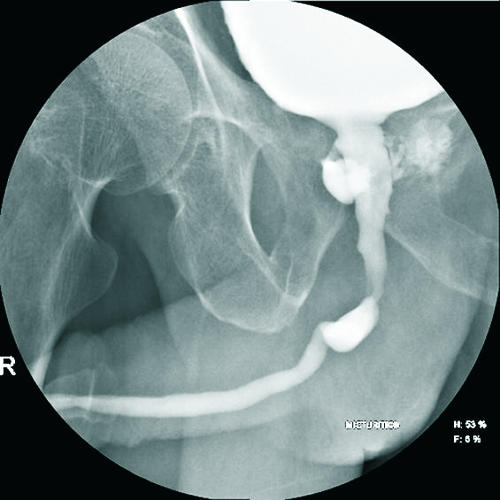
Post-treatment MCU of case 2 showing residual focal pooling of contrast in prostatic cavity with a healed rectoprostatic fistula.
Case 3
A 52-year-old labourer presented to the emergency department with history of moderate fever with chills and rigors, dysuria, frequency and cough with productive sputum. As urine microscopy showed pyuria, he was referred to the urology department after starting antibiotics. On examination, he was also found to have purulent discharge per urethra, a palpable bladder, bilateral epididymoorchitis, a boggy prostate on rectal examination and perianal abscess. Suprapubic catheterisation with incision and drainage of perianal abscess was done. The perianal wound continued to discharge purulent material and did not heal. One month later, he complained of passage of urine per rectum following blockage of the SPC. MCU showed fistulous communication with the distal rectum (Fig. 3). Ultrasound showed enlarged prostate with an hypoechoic region 4.7 × 4.5 × 3.7 cm within the prostate. Upper tracts were normal. Cystoscopy showed 4–5 fistulous openings on either side of the verumontanum. There was no stricture, bladder neck and bladder were normal. A diagnosis of rectoprostatic fistula and high anal fistula was made. He was advised further evaluation but was lost to follow-up for 6 months. A diverting colostomy for the high anal fistula and rectoprostatic fistula was planned. On pre-operative evaluation, he was found to have fibrocavitatory lesions in both lung apices and sputum AFB was positive. The ESR was 100 mm/h and serum creatinine was 0.6 mg%. Diverting colostomy was deferred and he was started on antitubercular therapy with 4 drugs. Six weeks after treatment with antitubercular drugs, a diverting colostomy was done. The prostato-rectal fistula healed clinically and was confirmed with an MCU (Fig. 4). The SPC was clamped after which he voided with a poor flow. Urodynamic study confirmed bladder outflow obstruction. Cystoscopy showed a cavity in prostatic urethra and a narrowed bladder neck. A bladder neck incision was done. He is voiding normally without any leak. He is awaiting a colostomy closure.
Figure 3.

RGU of case 3 showing extravasation of contrast into the rectum.
Figure 4.

Post-treatment MCU of case 3 showing a healed rectoprostatic fistula and a small residual prostatic biopsy.
Discussion
Tubercular involvement of the prostate gland is known to present as granulomatous prostatitis. The exact incidence is unknown at present, but is reportedly low. In India, a survey of 126 patients who underwent fine needle aspiration cytology for suspicion of malignancy revealed tuberculosis in 3%.1 Despite the prevalence of urogenital tuberculosis in the non-industrialised world, rectoprostatic fistula caused by tuberculosis is extremely rare. A possible explanation for this rarity is that the fascia between the prostate and rectum acts as a barrier.
All three cases presented differently. Case 1 presented with a rectoprostatic fistula and cystoscopic findings; biopsy of the prostate confirmed tuberculosis. Case 2 presented with a rectal ulcer and abnormal DRE with normal PSA. A TUR biopsy was done as TRUS-guided needle biopsy was inconclusive; this confirmed the diagnosis. Case 3 presented with a prostatic abscess. The untreated prostatic abscess drained through the rectum and led to the rectoprostatic fistula. There was indirect evidence of tuberculosis as the fistula persisted for about a year till antitubercular treatment was given to treat active pulmonary tuberculosis. The cystoscopic findings of multiple openings of sinus near the verumontanum are characteristic of prostatic TB as described by Veenema and Lattimer.3 The lesion healed with bladder neck scarring requiring a bladder neck incision as reported elsewhere.4
All the patients were ELISA negative for HIV infection. There was no evidence of kidney, ureter or bladder involvement and the urine was negative for AFB (3 samples). None had evidence of upper tract involvement. They were voiding normally at a 1-year follow-up.
With regard to treatment, different methods have been employed in cases reported in the literature. Three cases in which M. tuberculosis was found in urine or pus had spontaneous closure of fistula after antitubercular chemotherapy.4–6 Another case underwent a successful pull-through operation.7 One case6 received no treatment whereas another8 required a combined abdominoperineal approach with omental interposition and no faecal or urinary diversion. All three patients were treated with antitubercular drugs and urinary diversion with or without faecal diversion. This resulted in successful closure of all the fistulae in spite of the fact that they did not have M. tuberculosis in the urine as suggested in the literature.4–6 We feel that a simple SPC and faecal diversion with antitubercular drugs should be the first line of treatment in patients with spontaneous tubercular rectoprostatic fistula.
Conclusions
Spontaneous tubercular rectoprostatic fistulae are a rare complication of prostatic tuberculosis. There is no renal, ureteric or bladder involvement. The fistulae open adjacent to the verumontanum in the prostatic urethra. Urine for acidfast bacilli may be negative and prostatic biopsy proves the diagnosis. Conservative management with antitubercular drugs and urinary diversion with or without faecal diversion has a high success rate and should be the first line of treatment even if urine is negative for acid-fast bacilli.
References
- 1.Mondal A, Ghosh E, Ghose A. The role of transrectal fine needle aspiration cytology in the diagnosis of prostatic nodules suspicious of malignancy – a study of 126 cases. Ind J Pathol Microbiol. 1990;33:23. [PubMed] [Google Scholar]
- 2.Stamler JS, Bauer JJ, Janonitz HD. Rectourethroperineal fistula in Crohn's disease. Am J Gastroenterol. 1985;80:111–2. [PubMed] [Google Scholar]
- 3.Veenema RJ, Lattimer JK. Genital tuberculosis in the male: clinical pathology and effect on fertility. J Urol. 1957;78:65–77. doi: 10.1016/S0022-5347(17)66400-0. [DOI] [PubMed] [Google Scholar]
- 4.Culp OS, Calhoo HW. A variety of rectourethral fistulas: experience with twenty cases. J Urol. 1964;91:560–71. doi: 10.1016/S0022-5347(17)64171-5. [DOI] [PubMed] [Google Scholar]
- 5.Symes JM, Blandy JP. Tuberculosis of the male urethra. Br J Urol. 1973;45:432–6. doi: 10.1111/j.1464-410x.1973.tb12184.x. [DOI] [PubMed] [Google Scholar]
- 6.Suton S, Shiraishi Y, Sasaki T. Tuberculous rectourethral fistula: a case report of two cases. Med J Aomor. 1974;20:151. [Google Scholar]
- 7.Takai S, et al. Experience of three cases of rectourethral fistula. Jpn J Urol. 1962;53:483. [Google Scholar]
- 8.Toshikazu O, Akimi O, Masahisa W. Tuberculous rectourethral fistula. Urology. 1988;31:424–6. doi: 10.1016/0090-4295(88)90741-8. [DOI] [PubMed] [Google Scholar]


