Abstract
Social gliding motility of Myxococcus xanthus requires polar type IV pili. Tgl mutants lack pili and lack social motility. However, both defects can be rescued phenotypically, but not genotypically, when tgl+ donor and tgl− recipient cells make physical contact with each other. What is the cellular and molecular basis of this transfer of phenotype, which is called stimulation? Stimulation does not occur in liquid nor in soft (0.5%) agar; however, on a more firm surface (1.0% agar) cells stimulate each other efficiently. Microscopy revealed that cells placed on 1.0% agar readily became aligned whereas they remained poorly aligned on 0.5% agar. It was observed, moreover, that stimulation is greatly reduced when donor and recipient cells lack the ability to move because of mutations in any of several different motility genes. Microscopy showed that motile cells became highly aligned, and can even align nonmotile cells. Using a stimulation assay based on the assembly of pili, a strong correlation was found between conditions that promote cell alignment and the speed or extent of stimulation. Because pili are assembled only at the end(s) of a cell, and a parallel alignment of cells in contact brings them end-to-end, we would suggest that end-to-end contacts are important for stimulation of pilus assembly.
Myxococcus xanthus has two systems called adventurous (A) and social (S) motility that control its gliding motility and swarming behavior (1). Mutations in A- or S-motility genes inactivate the corresponding system, but cells are still motile by means of their other system. However, no A−S− double mutant can move more than a cell’s length nor swarm. A-motility is characterized by the movements of well-isolated single cells and groups or rafts of cells. In contrast, S-motility requires close cell proximity, and isolated cells do not move (2). A-motility also allows cells to move on firm agar surfaces (1.0%), whereas S-motile cells move better on soft agar, such as 0.5% (3).
The A-motility system involves the products of more than 37 genes (4). The S-motility system includes 12 known genes and may contain two or three times that number (4, 5). S-motility absolutely depends on polar type IV pili and, in fact, nearly all the known genes are involved in regulating pilus assembly or function (6–9). Mutations in pilus genes do not change A-motility. In addition to the A- and S-motility genes there is the mgl locus that encodes a Ras-like protein and is required for both motility systems, because mgl mutants have neither A- nor S-motility and do not swarm (4). The frequency of reversal of gliding direction is controlled by the frizzy system (10). The frz genes are homologous to the enteric bacterial chemotaxis genes for swimming, though M. xanthus is nonflagellated and cannot swim.
Mutations in the S-motility gene, tgl, result in the loss of pili and loss of S-motility (1, 6). Among the pili-lacking mutants, when tgl− cells are mixed with tgl+ donor cells, S-motility and piliation of the tgl− recipient cells are transiently restored. This process, called stimulation, does not involve a factor that can diffuse from cell to cell; instead physical contact between the donor and recipient cells is necessary. Stimulation is transient and only occurs phenotypically, because the offspring of stimulated cells remain S− and lack pili (6). Recently, the tgl gene product has been characterized, and it appears to be exposed to the periplasm (11, 12). Five groups of mutants in the A-motility system (cglB, C, D, E, F), which lack A-motility, can be stimulated for A-motility by donor cells that have the corresponding wild-type alleles cglB+, cglC+, etc. (13, 14). Stimulation may play a general role in the swarming of M. xanthus because it is found in both motility systems.
Hodgkin and Kaiser previously reported that stimulation in general does not occur in liquid, but only on a solid surface (6, 13, 14); we show here that this applies to Tgl stimulation. Restriction to surfaces suggests that the collisions between cells that occur in solution lack some quality necessary for stimulation. Given that M. xanthus cells are asymmetric (they are 10 times longer than they are wide) and pili are polarly localized, might orientation between donor and recipient cells be relevant to stimulation? Aligned cell–cell interactions in M. xanthus has been shown to be important for C signaling in fruiting body development (15). Here we show a strong correlation between conditions that promote cell alignment and the efficiency of Tgl stimulation.
MATERIALS AND METHODS
M. xanthus Strains and Media.
Strains DK1217 (aglB1), DK6204 (ΔmglBA), DK10405 (Δtgl∷Tcr; tetracycline resistance), DK10407 (ΔpilA∷Tcr), and DK10410 (ΔpilA) have been described previously (1, 11, 16, 17). The following strains were constructed by generalized transduction with the bacteriophages Mx4 or Mx8 as indicated (13). DK8601 (aglB1 ΔpilA∷Tcr; Mx4(DK10407) × DK1217→Tcr), DK8602 (aglB1Δtgl∷Tcr; Mx8(DK10405) × DK1217→Tcr), DK8603 (ΔmglBA ΔpilA∷Tcr; Mx4(DK10407) × DK6204→Tcr), DK8604 (ΔmglBA Δtgl∷Tcr; Mx8(DK10405) × DK6204→Tcr), DK8605 (aglB1 pilAp-gfp(Kmr; kanamycin resistance); Mx4(DK10546) × DK1217→Kmr), DK8606 (aglB1 ΔpilA∷Tcr pilAp-gfp(Kmr); Mx4(DK10407) × DK8605→Tcr), DK8607 (aglB1 Δtgl pilAp-gfp(Kmr); Mx4(DK10546) × DK8602→Kmr), DK8608 (Δtgl∷Tcr frzD∷Tn5Ω224 (Kmr) (18); Mx4(DZF3460) × DK10405→Kmr), DK8609 (ΔpilAr frzD∷Tn5Ω224 (Kmr); Mx4(DZF3460) × DK10410→Kmr). The in vitro construction of the pilAp-gfp(Kmr) transcriptional fusion was constructed by E. Licking (Stanford University) and will be described elsewhere. Bacteria were grown in casitone-Tris (CTT) medium (13).
Stimulation Assay for Pili.
To enumerate pili, cells were treated as previously described (19). Briefly, at various times cells were harvested and pili were sheared off cells by vortexing. Cells were separated from pili in suspension by centrifugation. The pili were precipitated by the addition of MgCl2, then resuspended in sample buffer, boiled, and fractionated by SDS/PAGE. To detect PilA protein, the gels were blotted and the blots probed with a 1:2,000 dilution of anti-PilA serum (20).
Microscopy.
A Nikon Eclipse E800 microscope equipped with an array of objectives, including a Plan Fluor 10× (numerical aperture 0.30) and 20× (numerical aperture 0.50) was used for phase contrast. For fluorescence microscopy a Chroma FITC-HYQ filter set was added to visualize green fluorescent protein-labeled cells. Color images were captured with an Optronics Engineering DEI-750T cooled three-chip colored charge-coupled device video camera and recorded on a Panasonic TQ-3031F optical disc recorder. To prepare figures, images were transferred from the optical disc recorder to a Power Macintosh where they were converted to black and white images by using Abode Photoshop 3.0, and then printed on a Fujix Pictrography 3000 printer.
RESULTS
Tgl Stimulation Requires a Solid Surface.
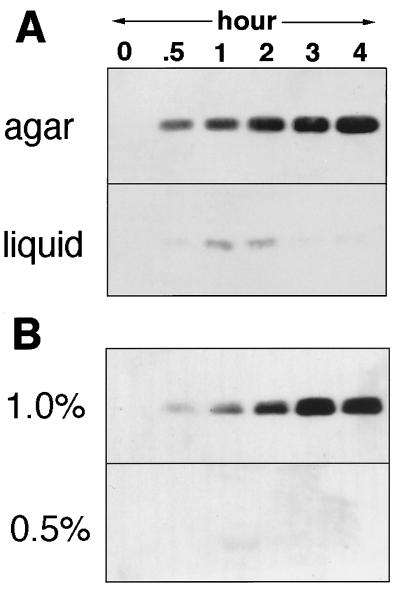
Formerly stimulation had to be assayed by swarming (1, 6), but recently a faster and more direct assay that detects Tgl stimulation by means of pilus production has been described (19). Using this assay, stimulation in liquid cultures was compared with stimulation on a solid surface of 1.0% agar. The donor and recipient genotypes were chosen so that pili arise only from the recipient cells, which carry a deletion mutation in the tgl gene (Δtgl). The donor cells in the mixture carry a ΔpilA mutation, which prevents synthesis of the pilin monomer. As shown in Fig. 1A, Tgl stimulation is readily detected in Western blots when recipient (Δtgl pilA+) cells are mixed with donor (tgl+ ΔpilA) cells on agar. If the same cell mixture was placed in a liquid culture and the culture was rotated at 300 rpm on an eccentric shaker to prevent the cells from settling, no stimulation was observed (the minor spots at 1 and 2 hr were variable and represent experimental background). Prolonging the incubation to 24 hr in liquid failed to increase the stimulation signal above that evident at 4 hr.
Figure 1.
Tgl stimulation of pilus production. Immunoblots of pili sheared from cells and separated by SDS/PAGE, then probed with anti-PilA serum. DK10410 (A+ tgl+ ΔpilA) donor cells were mixed with DK10405 (A+ Δtgl pilA+) recipient cells at a ratio of 1:2, respectively, and incubated for the time indicated. (A) Cells were incubated on either 1/2 CTT agar (1.0%) plates or in 1/2 CTT liquid medium (13) at a calculated cell density of 100 Klett units. (B) Cells were incubated on 1/2 CTT plates with either 1.0 or 0.5% agar at a calculated cell density of 500 Klett units.
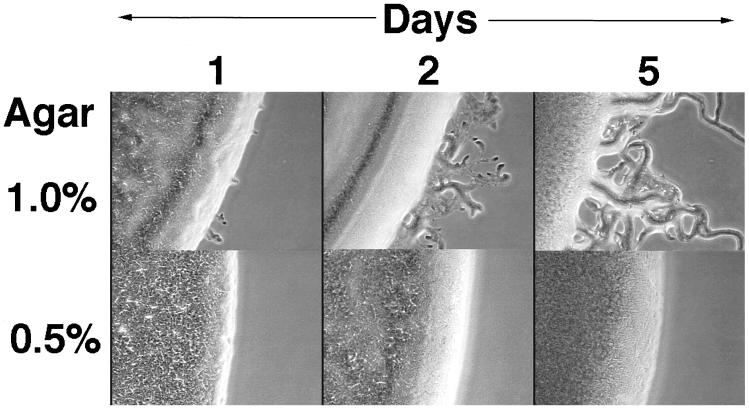
One difference between liquid and a solid surface is in the way cells can contact each other: on a solid surface the elongated cells tend to align and thus make more contacts, which also can persist longer, whereas in agitated liquid culture, the cells are unable to align and cell contacts though frequent are fleeting. Conditions that are intermediate between a liquid and a firm surface were examined. No stimulation of pilus production was found in 0.5% (semisolid) agar, in sharp contrast to the rapid and substantial stimulation seen on 1.0% agar (Fig. 1B). This result was particularly intriguing, given that 0.5% agar enhances S-motile swarming by 3- to 5-fold above that on 1.0% agar (3, 21). To pursue this unexpected distinction between S-motility itself and the stimulation of S-motility, we attempted to detect the stimulation of S-motility by swarm expansion on 0.5% agar. To eliminate background from A-motile swarming, the donor and recipient cells carried the same (nonstimulatable) mutation (aglB1) in the A-motility system. In addition, the donor strain contained a ΔpilA mutation, rendering both donor and recipient strains A−S− and thus nonmotile. In such mixtures only the pilA+ recipient strain would have the potential to be stimulated. Consistent with the results obtained with the pilus assembly assay (Fig. 1B), no stimulation of S-motile swarming was observed on 0.5% agar, even after 5 days (Fig. 2). Swarming assays such as this are known from previous experience to be highly sensitive (11, 13). However, stimulation of S-motility is clearly evident when the same cell mixture was incubated on 1.0% agar (Fig. 2). Again the firmness of the agar surface is seen to have a dramatic effect on stimulation. Noting that equal volumes of the same cell suspension were applied to the 0.5 and 1.0% agar, it is unlikely that the absence of stimulation in liquid (Fig. 1) or on soft agar is caused by a difference in the concentration of cells.
Figure 2.
Tgl stimulation of S-motility on 1.0 and 0.5% agar. Nonmotile DK8601 (aglB1 tgl+ ΔpilA) donor cells were mixed with nonmotile DK8602 (aglB1 Δtgl pilA+) recipient cells at a ratio of 1:1 at a cell density of 500 Klett units. Ten microliters of this cell mixture was placed on 1/2 CTT plates with either 1.0 or 0.5% agar. The peninsula-like flares of cells arise from the spot edge as a result of S-motility.
Motility Enhances Stimulation.
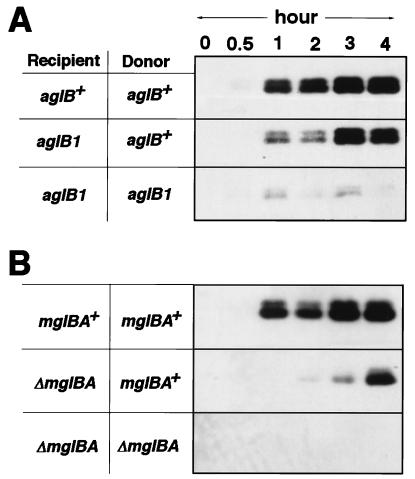
The above observations suggested that A-motility might enhance Tgl stimulation because stimulation can be obtained when cells are on a firm agar surface, which favors A-motility, but not on soft agar, which decreases A-motility. To investigate the possibility that motility can play a role in stimulation, mutant donor and recipient strains that lacked A-motility (aglB1) were compared with strains that retained A-motility (aglB+). Fig. 3A shows that when both donor (tgl+ ΔpilA aglB1) and recipient (Δtgl pilA+ aglB1) cells carry the aglB1 mutation and hence lack A-motility as well as S-motility, stimulation of pilus production is very poor compared with aglB+ donor and recipient cells, which retain A-motility (Fig. 3A). When one of the strains was A+ and the other (the recipient) was A−, stimulation was observed, although the time required for pilus formation was longer than when both donor and recipient cells retained A-motility. When, in a converse experiment, the donor was A− and the recipient A+ there was also stimulation of pilus assembly (data not shown). The absence of A-motility in both donor and recipient allows only weak stimulation (Fig. 3A), which is evident only after a 1- to 2-day delay (Fig. 2).
Figure 3.
Motility enhances Tgl stimulation of pilus production. Pili assays were described in the legend to Fig. 1 on 1/2 CTT (1.0%) agar plates. (A) Effect of A-motility on Tgl stimulation. Isogenic donor (DK10407 or DK8601) and recipient (DK10405 or DK8602) strains were either A+ (aglB+) or A− (aglB1) as indicated. (B) Effect of loss of mgl on stimulation. Isogenic donor (DK10407 or DK8603) and recipient (DK10405 or DK8604) strains were either A+ (mglBA+) or A− (ΔmglBA) as indicated.
To eliminate motility in ways other than use of aglB1, other mutations were used. Null mutations in mglA block both A- and S-motility (16). Although mgl mutant cells have been observed to oscillate back and forth there is no net translocation; the oscillations are at high frequency and have small amplitude. As a consequence mgl mutants form colonies with sharp edges (16). Although ΔmglBA mutants lack S-motility they do have some pili, though fewer than mgl+ cells (unpublished results). As described in Fig. 3B, when Δmgl donor (tgl+ ΔpilA ΔmglBA) and recipient (Δtgl pilA+ ΔmglBA) cells were mixed, stimulation of pilus formation was very poor over the 4-hr time course of the experiment. However, when an A+ donor (tgl+ ΔpilA mglBA+) was mixed with a Δmgl recipient (Δtgl pilA+ ΔmglBA) there was stimulation, but only after a delay of several hours (Fig. 3B).
Another type of motility mutant, frzD, was examined. frzD alleles are dominant mutations in the frzCD gene (18), which are expected to produce constitutive activation of the frz two-component signal transduction pathway. At the molecular level these mutations in the carboxyl end of frzCD are analogous to the signaling locked mutations in the methyl-accepting chemotaxis proteins of Escherichia coli (22). frzD mutants, like mgl mutants, have a hyper-reversal phenotype that limits their movements to a small fraction of a cell’s length before reversing; as a consequence they are unable to swarm (23). As with the aglB1 and ΔmglBA mutations, we found that when the donor (DK8609) and recipient (DK8608) cells carried a frzD (Tn5Ω224) mutation there was very poor stimulation of pilus production (data not shown). The results with aglB1, ΔmglAB, and frzD strains can be summarized by saying that as the amplitude of cell displacement is decreased, there is a corresponding decrease in efficiency of stimulation.
Cells Become Aligned as a Consequence of Translocating Movements.
The experiments above show that the ability of donor, recipient, or both to move on a surface greatly enhances Tgl stimulation. High-frequency oscillation without net cell translocation is insufficient. This effect is not limited to one gene nor specific set of motility genes, because three fundamentally different types of motility mutations had a similar effect on stimulation. Moreover, when A-motile strains (A+S−) are mixed in liquid or placed on soft agar where they translocate poorly, they also do not stimulate. For technical reasons, having to do with a necessity for both donor and recipient to be S− and pili− for stimulation to be detected by pilus assembly or S-motile swarming, the effect of S-motility on stimulation has not been investigated. The soft agar experiments show that a complete A-motility system is not sufficient for stimulation; significant displacement of cells evidently is needed to enhance stimulation.
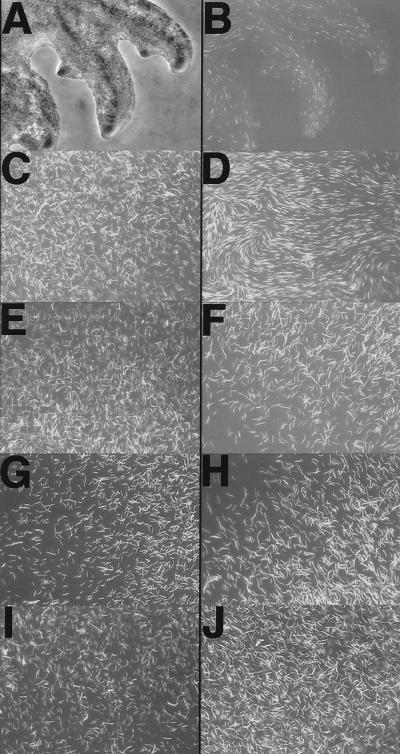
It might be expected that movements of densely packed elongated cells would tend to align. To monitor the extent of alignment within a dense population of moving cells, a small fraction of fluorescent cells were added to a relatively dense population of nonfluorescent cells. Cells were tagged with a transcription fusion to the green fluorescent protein. Cell orientations were monitored by fluorescence microscopy. Fig. 4A is a phase contrast image of a cluster of S-motility flares at the edge of an A−S+ swarm, and Fig. 4B is the fluorescence image of the same cluster. Fig. 4B reveals the nature and extent of alignment of the cells within these representative S-motility flares. Cells are highly aligned with each other and with the axes not only of the primary flare but also its secondary branches. This high degree of cell alignment is characteristic of S-motility.
Figure 4.
Motility and cell orientation visualized by fluorescence microscopy of green fluorescent protein (GFP)-labeled cells. GFP+ cells were mixed 1 to 50 with nonfluorescent cells and placed on 1/2 CTT plates (1 ml in 50 × 9-mm Petri dish) and incubated at 33°C. (A) Phase contrast image of S-motility flares (DK1217 and DK8605). (B) Fluorescent image of A. (C) Fluorescent image of A-motile (DK10410) cells mixed with nonmotile nonstimulatable GFP+ (DK8606) cells immediately after the cell suspension fluid had dried on the 1.0% agar. (D) Same as C after 4 hr. (E) Same as C, except DK8601 was mixed with nonmotile DK8606 (aglB1 ΔpilA). (F) Same as E after 4 hr. (G) Same as C, except DK8601 was mixed with nonmotile DK8603 (ΔmglBA ΔpilA). (H) Same as G after 4 hr. (I) Same as C, except cell suspension was placed on 0.5% agar. (J) Same as I after 4 hr.
Cell alignment is also characteristic of A-motile cells (below). To monitor the extent to which A-motility can align cells, a gfp-tagged strain that is nonswarming (DK8606; aglB1 ΔpilA pilAp-gfp) was mixed with an untagged but A-motile strain (DK10410; A+ ΔpilA). To provide resolution of individual (fluorescent) cells within the relatively dense populations, gfp cells were diluted 1 to 50 with nonfluorescent DK10410. Fig. 4C shows that immediately after a cell suspension had been deposited on agar and the fluid of the droplet had soaked into the gel, cells were in helter-skelter orientations. However, 4 hr later (Fig. 4D) the A-motile but untagged cells had brought the nonswarming DK8606 cells into parallel arrangements, and by implication were themselves aligned in the same way. Because of their high density, it is not possible to visualize individual cells in phase contrast. However, when the same fluorescent indicator strain, DK8606, was mixed with the A−S− strain DK8601, which is aglB1 ΔpilA, or with DK8603 cells (ΔmglBA ΔpilA) the tagged cells remained disoriented after 4 hr, like their 0-hr samples. The aligning effect of motility can be seen by comparing Fig. 4 F and H with D. A similar effect of A-motility on cell alignment was found when a tgl mutant strain (DK8607; aglB1 Δtgl pilAp-gfp) was mixed with DK10410 or DK8601 (not shown). Clearly, movement of A-motile cells can align nonmotile cells.
Stimulation did not occur on soft agar (Fig. 1), and soft agar diminishes the displacement of A-motile cells. The ability of A-motile cells to align and orient themselves and admixed nonmotile cells on 0.5% (soft) agar was assessed. Comparison of I and J in Fig. 4 shows the ability of the same mixture of A-motile and nonmotile cells used in C and D to align on 0.5% agar. After 4 hr there was much less change in relative cell alignment of the gfp indicator cells on 0.5% agar (J), than on 1.0% agar (D). Recalling that the indicator cells represent only 2% of the total cells in these microscopic fields, there are no significant gaps between cells. The correlation between cell alignment and Tgl stimulation implies that alignment plays a critical role.
DISCUSSION
M. xanthus cells are about 10 times longer than they are wide, and they are functionally asymmetric. Their gliding movements are constrained to the direction of their longitudinal axis, although they move as frequently in one direction as the other. Their pili are located at the pole(s), and in 95% of cells at one pole (6, 9). During fruiting body development, cells can detect whether they are inside a fruiting body, where they proceed to differentiate into spores, or are outside a fruiting body, where they remain peripheral rods (24). During development, C signaling and rippling require cell alignment (15, 25). In this study, we have shown a strong correlation between cell orientation and the type of cell–cell interaction necessary for Tgl stimulation—a transient cell–cell transfer of phenotype. Conditions that promote cell alignment are shown to greatly enhance Tgl stimulation, whereas conditions that lead to less extensive alignment show feeble, slow, or no stimulation. These experiments also reveal a very high degree of alignment in the flares of S-motile swarming.
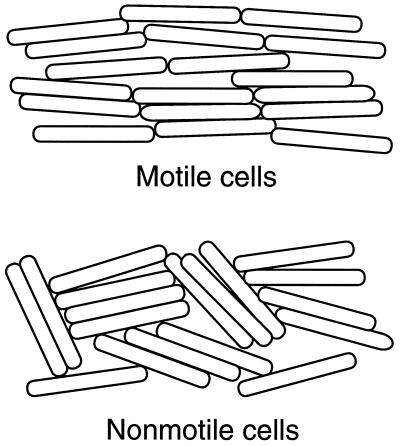
In this paper motility is shown to greatly enhance Tgl stimulation of pilus assembly. Motility in either or both the donor and recipient cells can provide the enhancement; however, when both donor and recipient are motile, the enhancement is strongest. Because three functionally different classes of motility mutants had similar effects on stimulation, an effect of a specific gene or class of genes appears unlikely. Add the older observation that donor cells need to be alive for stimulation to occur (1); dead cells have never been observed to move. Because stimulation is not evident in agitated liquid cultures, or on soft agar, we propose a model that is illustrated in Fig. 5. Here, motility provides a force that aligns cells. The gliding movements of long rod-shaped cells always in the direction of their long axis may work in the way that combing and brushing aligns hair. Myxobacterial cells are cohesive and tend to stay in side-by-side contact with each other even at moderate cell densities. As suggested in this drawing, movement-generated cell alignments probably increase the number and the lifetime of contacts between cell poles, without appreciable changes in the number or lifetimes of side-by-side contacts or end-to-side contacts. End-to-end contacts are likely to play a particularly important role in stimulation. Side-to-side cell contacts appear to be insufficient for stimulation, because such are very common in randomly oriented cells, implied by Fig. 4. End-to-end interactions are rare when cells are randomly oriented, but become much more frequent when cells are aligned, as indicated in Fig. 5, where the cell density is the same in the motile and nonmotile populations. Type IV pili are polarly localized. Because Tgl, a minor cell protein (12), is required for pilus biogenesis (6, 19) it is plausible that the Tgl protein and/or its activity is localized to the cell poles where it can facilitate pilus assembly. If Tgl activity is polarly localized, end-to-end interactions between donor and recipient cells might well be required for Tgl stimulation.
Figure 5.
Model for the role of motility in cell alignment and Tgl stimulation. The number of cells per unit area are intended to be the same in the upper and lower parts of the diagram.
One might be tempted to think that Tgl functions as an extracellular signal for pilus biogenesis and S-motility. However, tgl+ cells assemble pili even when grown in dilute liquid cultures, which renders a cell-to-cell signaling role unattractive. The fact that tgl mutants express normal levels of PilA protein rules out Tgl as a signal for pilin expression (19). Instead, the Tgl protein may function as an integral part of pilus biogenesis. During stimulation a small amount of Tgl protein might be transferred from donor to recipient cells where it would participate in biogenesis. Alternatively, Tgl might remain on donor cells where it could act as a chaperone for pilus assembly in the recipient cells. A chaperone is known to be essential for the assembly of P pili (26). On that view Tgl protein would not be a component of the assembled pilus and thus would not have to be transferred between cells.
Acknowledgments
This work was supported by a grant from the National Science Foundation (MCB 9423182) (to D.K.). D.W. was a recipient of an American Cancer Society postdoctoral fellowship (PF-4138).
ABBREVIATIONS
- A-motility
adventurous motility
- S-motility
social motility
- CTT
casitone-Tris
- KmR
kanamycin resistance
- TcR
tetracycline resistance
References
- 1.Hodgkin J, Kaiser D. Mol Gen Genet. 1979;171:177–191. [Google Scholar]
- 2.Kaiser D, Crosby C. Cell Motil. 1983;3:227–245. [Google Scholar]
- 3.Shi W, Zusman D R. Proc Natl Acad Sci USA. 1993;90:3378–3382. doi: 10.1073/pnas.90.8.3378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hartzell P L, Youderian P. Arch Microbiol. 1995;164:309–323. doi: 10.1007/BF02529977. [DOI] [PubMed] [Google Scholar]
- 5.Wu S S, Wu J, Kaiser D. Mol Microbiol. 1997;23:109–121. doi: 10.1046/j.1365-2958.1997.1791550.x. [DOI] [PubMed] [Google Scholar]
- 6.Kaiser D. Proc Natl Acad Sci USA. 1979;76:5952–5956. doi: 10.1073/pnas.76.11.5952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wu S S, Kaiser D. Mol Microbiol. 1995;18:547–558. doi: 10.1111/j.1365-2958.1995.mmi_18030547.x. [DOI] [PubMed] [Google Scholar]
- 8.Wu S S. Ph.D. thesis. Stanford, CA: Stanford University; 1997. [Google Scholar]
- 9.MacRae T H, McCurdy H D. Can J Microbiol. 1976;22:1589–1593. doi: 10.1139/m76-234. [DOI] [PubMed] [Google Scholar]
- 10.Ward M J, Zusman D R. Mol Microbiol. 1997;24:885–893. doi: 10.1046/j.1365-2958.1997.4261783.x. [DOI] [PubMed] [Google Scholar]
- 11.Rodriguez-Soto J P, Kaiser D. J Bacteriol. 1997;179:4361–4371. doi: 10.1128/jb.179.13.4361-4371.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rodriguez-Soto J P, Kaiser D. J Bacteriol. 1997;179:4372–4381. doi: 10.1128/jb.179.13.4372-4381.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hodgkin J, Kaiser D. Proc Natl Acad Sci USA. 1977;74:2938–2942. doi: 10.1073/pnas.74.7.2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hodgkin J, Kaiser D. Mol Gen Genet. 1979;171:167–176. [Google Scholar]
- 15.Kim S K, Kaiser D. Science. 1990;249:926–928. doi: 10.1126/science.2118274. [DOI] [PubMed] [Google Scholar]
- 16.Hartzell P, Kaiser D. J Bacteriol. 1991;173:7625–7635. doi: 10.1128/jb.173.23.7625-7635.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wu S S, Kaiser D. J Bacteriol. 1996;178:5817–5821. doi: 10.1128/jb.178.19.5817-5821.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McBride M J, Weinberg R A, Zusman D R. Proc Natl Acad Sci USA. 1989;86:424–428. doi: 10.1073/pnas.86.2.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wall D, Wu S S, Kaiser D. J Bacteriol. 1998;180:759–761. doi: 10.1128/jb.180.3.759-761.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu S S, Kaiser D. J Bacteriol. 1997;179:7748–7758. doi: 10.1128/jb.179.24.7748-7758.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.MacNeil S D, Mouzeyan A, Hartzell P L. Mol Microbiol. 1994;14:785–795. doi: 10.1111/j.1365-2958.1994.tb01315.x. [DOI] [PubMed] [Google Scholar]
- 22.Ames P, Parkinson J S. Cell. 1988;55:817–826. doi: 10.1016/0092-8674(88)90137-7. [DOI] [PubMed] [Google Scholar]
- 23.Blackhart B D, Zusman D R. Proc Natl Acad Sci USA. 1985;82:8767–8770. doi: 10.1073/pnas.82.24.8767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.O’Connor K A, Zusman D R. J Bacteriol. 1991;173:3318–3333. doi: 10.1128/jb.173.11.3318-3333.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sager B, Kaiser D. Genes Dev. 1994;8:2793–2804. doi: 10.1101/gad.8.23.2793. [DOI] [PubMed] [Google Scholar]
- 26.Hultgren S J, Jacob-Dubuisson F, Jones C H, Brenden C I. Adv Protein Chem. 1993;44:99–123. doi: 10.1016/s0065-3233(08)60565-3. [DOI] [PubMed] [Google Scholar]