Abstract
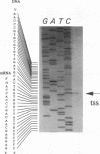
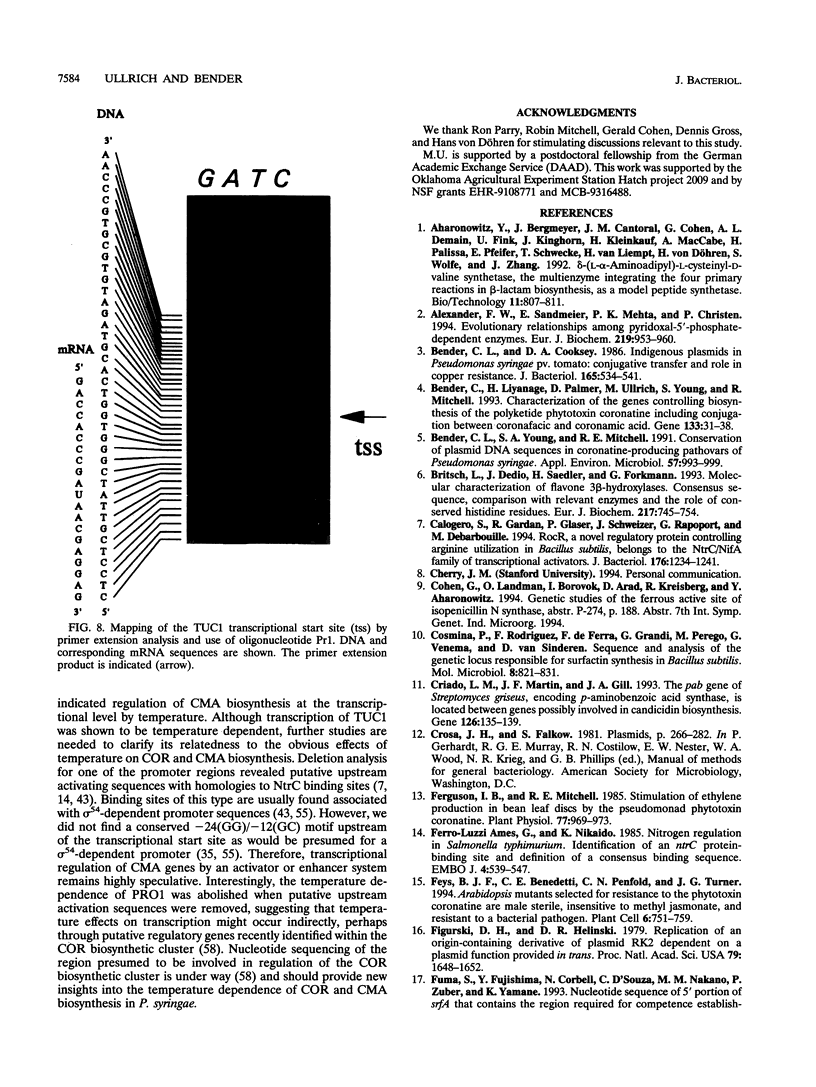
Coronamic acid (CMA), an ethylcyclopropyl amino acid derived from isoleucine, functions as an intermediate in the biosynthesis of coronatine, a chlorosis-inducing phytotoxin produced by Pseudomonas syringae pv. glycinea PG4180. The DNA required for CMA biosynthesis (6.9 kb) was sequenced, revealing three distinct open reading frames (ORFs) which share a common orientation for transcription. The deduced amino acid sequence of a 2.7-kb ORF designated cmaA contained six core sequences and two conserved motifs which are present in a variety of amino acid-activating enzymes, including nonribosomal peptide synthetases. Furthermore, CmaA contained a spatial arrangement of histidine, aspartate, and arginine residues which are conserved in the ferrous active site of some nonheme iron(II) enzymes which catalyze oxidative cyclizations. The deduced amino acid sequence of a 1.2-kb ORF designated cmaT was related to thioesterases of both procaryotic and eucaryotic origins. These data suggest that CMA assembly is similar to the thiotemplate mechanism of nonribosomal peptide synthesis. No significant similarities between a 0.9-kb ORF designated cmaU and other database entries were found. The start sites of two transcripts required for CMA biosynthesis were identified in the present study. pRG960sd, a vector containing a promoterless glucuronidase gene, was used to localize and study the promoter regions upstream of the two transcripts. Data obtained in the present study indicate that CMA biosynthesis is regulated at the transcriptional level by temperature.
Full text
PDF











Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aharonowitz Y., Bergmeyer J., Cantoral J. M., Cohen G., Demain A. L., Fink U., Kinghorn J., Kleinkauf H., MacCabe A., Palissa H. Delta-(L-alpha-aminoadipyl)-L-cysteinyl-D-valine synthetase, the multienzyme integrating the four primary reactions in beta-lactam biosynthesis, as a model peptide synthetase. Biotechnology (N Y) 1993 Jul;11(7):807–810. doi: 10.1038/nbt0793-807. [DOI] [PubMed] [Google Scholar]
- Alexander F. W., Sandmeier E., Mehta P. K., Christen P. Evolutionary relationships among pyridoxal-5'-phosphate-dependent enzymes. Regio-specific alpha, beta and gamma families. Eur J Biochem. 1994 Feb 1;219(3):953–960. doi: 10.1111/j.1432-1033.1994.tb18577.x. [DOI] [PubMed] [Google Scholar]
- Bender C. L., Cooksey D. A. Indigenous plasmids in Pseudomonas syringae pv. tomato: conjugative transfer and role in copper resistance. J Bacteriol. 1986 Feb;165(2):534–541. doi: 10.1128/jb.165.2.534-541.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bender C. L., Liyanage H., Palmer D., Ullrich M., Young S., Mitchell R. Characterization of the genes controlling the biosynthesis of the polyketide phytotoxin coronatine including conjugation between coronafacic and coronamic acid. Gene. 1993 Oct 29;133(1):31–38. doi: 10.1016/0378-1119(93)90221-n. [DOI] [PubMed] [Google Scholar]
- Bender C. L., Young S. A., Mitchell R. E. Conservation of Plasmid DNA Sequences in Coronatine-Producing Pathovars of Pseudomonas syringae. Appl Environ Microbiol. 1991 Apr;57(4):993–999. doi: 10.1128/aem.57.4.993-999.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Britsch L., Dedio J., Saedler H., Forkmann G. Molecular characterization of flavanone 3 beta-hydroxylases. Consensus sequence, comparison with related enzymes and the role of conserved histidine residues. Eur J Biochem. 1993 Oct 15;217(2):745–754. doi: 10.1111/j.1432-1033.1993.tb18301.x. [DOI] [PubMed] [Google Scholar]
- Calogero S., Gardan R., Glaser P., Schweizer J., Rapoport G., Debarbouille M. RocR, a novel regulatory protein controlling arginine utilization in Bacillus subtilis, belongs to the NtrC/NifA family of transcriptional activators. J Bacteriol. 1994 Mar;176(5):1234–1241. doi: 10.1128/jb.176.5.1234-1241.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Criado L. M., Martín J. F., Gil J. A. The pab gene of Streptomyces griseus, encoding p-aminobenzoic acid synthase, is located between genes possibly involved in candicidin biosynthesis. Gene. 1993 Apr 15;126(1):135–139. doi: 10.1016/0378-1119(93)90602-y. [DOI] [PubMed] [Google Scholar]
- Ferguson I. B., Mitchell R. E. Stimulation of ethylene production in bean leaf discs by the pseudomonad phytotoxin coronatine. Plant Physiol. 1985 Apr;77(4):969–973. doi: 10.1104/pp.77.4.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferro-Luzzi Ames G., Nikaido K. Nitrogen regulation in Salmonella typhimurium. Identification of an ntrC protein-binding site and definition of a consensus binding sequence. EMBO J. 1985 Feb;4(2):539–547. doi: 10.1002/j.1460-2075.1985.tb03662.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feys BJF., Benedetti C. E., Penfold C. N., Turner J. G. Arabidopsis Mutants Selected for Resistance to the Phytotoxin Coronatine Are Male Sterile, Insensitive to Methyl Jasmonate, and Resistant to a Bacterial Pathogen. Plant Cell. 1994 May;6(5):751–759. doi: 10.1105/tpc.6.5.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figurski D. H., Helinski D. R. Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1648–1652. doi: 10.1073/pnas.76.4.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuma S., Fujishima Y., Corbell N., D'Souza C., Nakano M. M., Zuber P., Yamane K. Nucleotide sequence of 5' portion of srfA that contains the region required for competence establishment in Bacillus subtilus. Nucleic Acids Res. 1993 Jan 11;21(1):93–97. doi: 10.1093/nar/21.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gocht M., Marahiel M. A. Analysis of core sequences in the D-Phe activating domain of the multifunctional peptide synthetase TycA by site-directed mutagenesis. J Bacteriol. 1994 May;176(9):2654–2662. doi: 10.1128/jb.176.9.2654-2662.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grundy F. J., Waters D. A., Takova T. Y., Henkin T. M. Identification of genes involved in utilization of acetate and acetoin in Bacillus subtilis. Mol Microbiol. 1993 Oct;10(2):259–271. doi: 10.1111/j.1365-2958.1993.tb01952.x. [DOI] [PubMed] [Google Scholar]
- Haydock S. F., Dowson J. A., Dhillon N., Roberts G. A., Cortes J., Leadlay P. F. Cloning and sequence analysis of genes involved in erythromycin biosynthesis in Saccharopolyspora erythraea: sequence similarities between EryG and a family of S-adenosylmethionine-dependent methyltransferases. Mol Gen Genet. 1991 Nov;230(1-2):120–128. doi: 10.1007/BF00290659. [DOI] [PubMed] [Google Scholar]
- Heaton M. P., Neuhaus F. C. Biosynthesis of D-alanyl-lipoteichoic acid: cloning, nucleotide sequence, and expression of the Lactobacillus casei gene for the D-alanine-activating enzyme. J Bacteriol. 1992 Jul;174(14):4707–4717. doi: 10.1128/jb.174.14.4707-4717.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KING E. O., WARD M. K., RANEY D. E. Two simple media for the demonstration of pyocyanin and fluorescin. J Lab Clin Med. 1954 Aug;44(2):301–307. [PubMed] [Google Scholar]
- Kenyon J. S., Turner J. G. The Stimulation of Ethylene Synthesis in Nicotiana tabacum Leaves by the Phytotoxin Coronatine. Plant Physiol. 1992 Sep;100(1):219–224. doi: 10.1104/pp.100.1.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krätzschmar J., Krause M., Marahiel M. A. Gramicidin S biosynthesis operon containing the structural genes grsA and grsB has an open reading frame encoding a protein homologous to fatty acid thioesterases. J Bacteriol. 1989 Oct;171(10):5422–5429. doi: 10.1128/jb.171.10.5422-5429.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- MacCabe A. P., van Liempt H., Palissa H., Unkles S. E., Riach M. B., Pfeifer E., von Döhren H., Kinghorn J. R. Delta-(L-alpha-aminoadipyl)-L-cysteinyl-D-valine synthetase from Aspergillus nidulans. Molecular characterization of the acvA gene encoding the first enzyme of the penicillin biosynthetic pathway. J Biol Chem. 1991 Jul 5;266(19):12646–12654. [PubMed] [Google Scholar]
- Mamer O. A., Reimer M. L. On the mechanisms of the formation of L-alloisoleucine and the 2-hydroxy-3-methylvaleric acid stereoisomers from L-isoleucine in maple syrup urine disease patients and in normal humans. J Biol Chem. 1992 Nov 5;267(31):22141–22147. [PubMed] [Google Scholar]
- Marahiel M. A. Multidomain enzymes involved in peptide synthesis. FEBS Lett. 1992 Jul 27;307(1):40–43. doi: 10.1016/0014-5793(92)80898-q. [DOI] [PubMed] [Google Scholar]
- Matsuda J., Okabe S., Hashimoto T., Yamada Y. Molecular cloning of hyoscyamine 6 beta-hydroxylase, a 2-oxoglutarate-dependent dioxygenase, from cultured roots of Hyoscyamus niger. J Biol Chem. 1991 May 25;266(15):9460–9464. [PubMed] [Google Scholar]
- Mehta P. K., Christen P. Homology of pyridoxal-5'-phosphate-dependent aminotransferases with the cobC (cobalamin synthesis), nifS (nitrogen fixation), pabC (p-aminobenzoate synthesis) and malY (abolishing endogenous induction of the maltose system) gene products. Eur J Biochem. 1993 Jan 15;211(1-2):373–376. doi: 10.1111/j.1432-1033.1993.tb19907.x. [DOI] [PubMed] [Google Scholar]
- Merrick M. J. In a class of its own--the RNA polymerase sigma factor sigma 54 (sigma N). Mol Microbiol. 1993 Dec;10(5):903–909. doi: 10.1111/j.1365-2958.1993.tb00961.x. [DOI] [PubMed] [Google Scholar]
- Myllylä R., Günzler V., Kivirikko K. I., Kaska D. D. Modification of vertebrate and algal prolyl 4-hydroxylases and vertebrate lysyl hydroxylase by diethyl pyrocarbonate. Evidence for histidine residues in the catalytic site of 2-oxoglutarate-coupled dioxygenases. Biochem J. 1992 Sep 15;286(Pt 3):923–927. doi: 10.1042/bj2860923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer D. A., Bender C. L. Effects of Environmental and Nutritional Factors on Production of the Polyketide Phytotoxin Coronatine by Pseudomonas syringae pv. Glycinea. Appl Environ Microbiol. 1993 May;59(5):1619–1626. doi: 10.1128/aem.59.5.1619-1626.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patriarca E. J., Chiurazzi M., Manco G., Riccio A., Lamberti A., De Paolis A., Rossi M., Defez R., Iaccarino M. Activation of the Rhizobium leguminosarum glnII gene by NtrC is dependent on upstream DNA sequences. Mol Gen Genet. 1992 Sep;234(3):337–345. doi: 10.1007/BF00538692. [DOI] [PubMed] [Google Scholar]
- Poulose A. J., Rogers L., Cheesbrough T. M., Kolattukudy P. E. Cloning and sequencing of the cDNA for S-acyl fatty acid synthase thioesterase from the uropygial gland of mallard duck. J Biol Chem. 1985 Dec 15;260(29):15953–15958. [PubMed] [Google Scholar]
- Raibaud A., Zalacain M., Holt T. G., Tizard R., Thompson C. J. Nucleotide sequence analysis reveals linked N-acetyl hydrolase, thioesterase, transport, and regulatory genes encoded by the bialaphos biosynthetic gene cluster of Streptomyces hygroscopicus. J Bacteriol. 1991 Jul;173(14):4454–4463. doi: 10.1128/jb.173.14.4454-4463.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reznikoff W. S. The Tn5 transposon. Annu Rev Microbiol. 1993;47:945–963. doi: 10.1146/annurev.mi.47.100193.004501. [DOI] [PubMed] [Google Scholar]
- Rich J. J., Willis D. K. A single oligonucleotide can be used to rapidly isolate DNA sequences flanking a transposon Tn5 insertion by the polymerase chain reaction. Nucleic Acids Res. 1990 Nov 25;18(22):6673–6676. doi: 10.1093/nar/18.22.6673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rusnak F., Sakaitani M., Drueckhammer D., Reichert J., Walsh C. T. Biosynthesis of the Escherichia coli siderophore enterobactin: sequence of the entF gene, expression and purification of EntF, and analysis of covalent phosphopantetheine. Biochemistry. 1991 Mar 19;30(11):2916–2927. doi: 10.1021/bi00225a027. [DOI] [PubMed] [Google Scholar]
- Salmeron J. M., Staskawicz B. J. Molecular characterization and hrp dependence of the avirulence gene avrPto from Pseudomonas syringae pv. tomato [corrected]. Mol Gen Genet. 1993 May;239(1-2):6–16. doi: 10.1007/BF00281595. [DOI] [PubMed] [Google Scholar]
- Schlumbohm W., Stein T., Ullrich C., Vater J., Krause M., Marahiel M. A., Kruft V., Wittmann-Liebold B. An active serine is involved in covalent substrate amino acid binding at each reaction center of gramicidin S synthetase. J Biol Chem. 1991 Dec 5;266(34):23135–23141. [PubMed] [Google Scholar]
- Tai M. H., Chirala S. S., Wakil S. J. Roles of Ser101, Asp236, and His237 in catalysis of thioesterase II and of the C-terminal region of the enzyme in its interaction with fatty acid synthase. Proc Natl Acad Sci U S A. 1993 Mar 1;90(5):1852–1856. doi: 10.1073/pnas.90.5.1852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thöny B., Hennecke H. The -24/-12 promoter comes of age. FEMS Microbiol Rev. 1989 Dec;5(4):341–357. doi: 10.1016/0168-6445(89)90028-4. [DOI] [PubMed] [Google Scholar]
- Tokita K., Hori K., Kurotsu T., Kanda M., Saito Y. Effect of single base substitutions at glycine-870 codon of gramicidin S synthetase 2 gene on proline activation. J Biochem. 1993 Oct;114(4):522–527. doi: 10.1093/oxfordjournals.jbchem.a124210. [DOI] [PubMed] [Google Scholar]
- Turgay K., Krause M., Marahiel M. A. Four homologous domains in the primary structure of GrsB are related to domains in a superfamily of adenylate-forming enzymes. Mol Microbiol. 1992 Feb;6(4):529–546. doi: 10.1111/j.1365-2958.1992.tb01498.x. [DOI] [PubMed] [Google Scholar]
- Ullrich M., Guenzi A. C., Mitchell R. E., Bender C. L. Cloning and expression of genes required for coronamic Acid (2-ethyl-1-aminocyclopropane 1-carboxylic Acid), an intermediate in the biosynthesis of the phytotoxin coronatine. Appl Environ Microbiol. 1994 Aug;60(8):2890–2897. doi: 10.1128/aem.60.8.2890-2897.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Eede G., Deblaere R., Goethals K., Van Montagu M., Holsters M. Broad host range and promoter selection vectors for bacteria that interact with plants. Mol Plant Microbe Interact. 1992 May-Jun;5(3):228–234. doi: 10.1094/mpmi-5-228. [DOI] [PubMed] [Google Scholar]
- Weckermann R., Fürbass R., Marahiel M. A. Complete nucleotide sequence of the tycA gene coding the tyrocidine synthetase 1 from Bacillus brevis. Nucleic Acids Res. 1988 Dec 23;16(24):11841–11841. doi: 10.1093/nar/16.24.11841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiler E. W., Kutchan T. M., Gorba T., Brodschelm W., Niesel U., Bublitz F. The Pseudomonas phytotoxin coronatine mimics octadecanoid signalling molecules of higher plants. FEBS Lett. 1994 May 23;345(1):9–13. doi: 10.1016/0014-5793(94)00411-0. [DOI] [PubMed] [Google Scholar]
- Witkowski A., Naggert J., Witkowska H. E., Randhawa Z. I., Smith S. Utilization of an active serine 101----cysteine mutant to demonstrate the proximity of the catalytic serine 101 and histidine 237 residues in thioesterase II. J Biol Chem. 1992 Sep 15;267(26):18488–18492. [PubMed] [Google Scholar]
- Xiao Y., Lu Y., Heu S., Hutcheson S. W. Organization and environmental regulation of the Pseudomonas syringae pv. syringae 61 hrp cluster. J Bacteriol. 1992 Mar;174(6):1734–1741. doi: 10.1128/jb.174.6.1734-1741.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young S. A., Park S. K., Rodgers C., Mitchell R. E., Bender C. L. Physical and functional characterization of the gene cluster encoding the polyketide phytotoxin coronatine in Pseudomonas syringae pv. glycinea. J Bacteriol. 1992 Mar;174(6):1837–1843. doi: 10.1128/jb.174.6.1837-1843.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Döhren H. The organization of multifunctional peptide and depsipeptide synthetases. Biochem Soc Trans. 1993 Feb;21(1):214–217. doi: 10.1042/bst0210214. [DOI] [PubMed] [Google Scholar]