Abstract
In patients with asthma, elevations of IgE correlate both with allergic inflammation of the airways and with bronchial hyperreactivity (BHR). Several investigations, using mouse models of this disease, have indicated a central role for IgE in the pathogenesis of the eosinophilic inflammation as well as in the obstructive airway physiology of BHR. Some diagnostic studies and therapeutic strategies for asthma are based on the putative role of IgE in asthma pathogenesis. Here, we use mice with a null mutation of the Cɛ locus to show that bronchial inflammation and BHR in response to allergen inhalation both can occur in the absence of IgE. We demonstrate that the eosinophilic bronchial inflammation elicited in an established mouse model of hypersensitivity to Aspergillus fumigatus (Af) is accompanied by the asthmatic physiology of BHR. Wild-type and IgE-deficient mice were sensitized intranasally with Af extract. Both groups of animals developed bronchoalveolar lavage eosinophilia and pulmonary parenchymal eosinophilia. This was accompanied by increased serum levels of total and Af-specific IgE in the wild-type animals only. This Af-sensitization protocol resulted in significant BHR in both wild-type mice and IgE-deficient mice. Interestingly, unsensitized IgE-deficient mice had increased bronchial responsiveness compared with unsensitized wild-type controls. We conclude that BHR and airways inflammation can be fully expressed via IgE-independent mechanisms. These may involve the activation of mast cells by factors other than IgE as well as a mucosal lymphocyte-mediated immune response to allergen.
Asthma is characterized by an increased tendency of bronchial smooth muscle to contract in response to irritant stimuli (bronchial hyperreactivity, BHR) (1). The airway mucosa of asthmatics who manifest BHR contains an eosinophilic and lymphocytic inflammatory infiltrate (2). The presence of eosinophils has been reported even for patients with so-called “intrinsic” asthma for whom allergic triggers of asthma flares have not been identified (3).
BHR and airway obstruction in asthmatics are associated with elevations of total and antigen-specific IgE (4, 5). Genetic analyses of families have shown that BHR and IgE levels are linked (6). Thus, in clinical practice, allergen-specific IgE, demonstrated by skin testing or in vitro assays, is generally thought to play a role in the induction of patients’ allergic airway symptoms and is used as a guide for environmental modification and immunotherapy. The concept that allergen-specific IgE initiates allergic airway symptoms has driven the development of therapeutics such as blockers of the interaction of IgE with its high-affinity receptor, FcɛRI (7, 8).
IgE might function in a number of distinct facets of the pathogenesis of airways allergy. It is well established that IgE can initiate immediate hypersensitivity reactions by triggering mast cell degranulation via FcɛRI (9). In the airways, mast cell-derived mediators released after allergen challenge lead to immediate bronchial smooth muscle constriction, bronchial edema, and mucous hypersecretion (10, 11). IgE-triggered immediate reactions often are followed by a “late-phase” response that occurs after 4–8 h. It has been postulated that chronic airway symptoms result from persistent late-phase inflammatory responses in situations of perennial allergen exposure (12).
IgE-induced activation of mast cells leads to the synthesis of cytokines (13, 14). By promoting the development of mast cells, eosinophils, and Th2 cells as well as inducing adhesion molecule expression and isotype switching to IgE, these mast cell-derived cytokines may serve to amplify local allergic inflammation. IgE also may modulate the cellular immune response to allergen by facilitating antigen uptake, processing, and presentation by B cells via CD23, thereby amplifying and regulating the immune response to allergens (15, 16).
A number of investigations based on murine models have pointed toward a central role for IgE in asthma pathogenesis. Nebulized ovalbumin (OVA) has been used to elicit specific IgE responses and bronchial hypersensitivity in mice (17). The passive transfer of IgE+ B cells has been shown to confer OVA-inducible BHR in this model (18). Mice bred for high levels of IgE production have been reported to manifest both greater BHR and more eosinophil infiltration in the respiratory epithelium after OVA sensitization and challenge than control animals (19). The inhibition of IgE with anti-IgE antibodies has been shown to lead to an attenuation of both eosinophilic airway inflammation and BHR in mice (20).
While substantial evidence exists that IgE plays an important, and sometimes necessary, role in the development of BHR and airway pathology, it is likely that a number of parallel mechanisms participate in asthma pathogenesis. Several studies have delineated roles for lymphocytes. The respiratory mucosa of asthmatic patients contains activated allergen-specific T cells (21, 22). A mouse model of BHR induced by repeated applications of picryl chloride has the features of a delayed type hypersensitivity reaction and is T cell dependent (23).
In the present study, we have used completely IgE-deficient mice to define any requisite role of IgE in the generation of BHR and eosinophilic airways inflammation after allergen exposure. These mice, which have a null mutation of the Cɛ exons encoding the IgE heavy chain, have been maintained inbred in the genetic background of the 129 strain and are identical to control 129 mice at all other loci (24). They have been shown to have no genomic sequences encoding ɛ heavy chain constant region domains and their B cells are incapable of producing either ɛ mRNA or IgE protein. As a result, neither tissue mast cells nor splenic B cells from these mice have detectable adsorbed IgE, and the animals show no response to injected anti-IgE antibodies (ref. 24 and H.C.O., unpublished data). The mice were studied using a previously described model of hypersensitivity to Aspergillus fumigatus (Af) (25, 26). Unlike OVA, which has been employed in many of the reported mouse models of asthma, Af is a naturally occurring airway allergen. Af sensitization via mucosal exposure is efficient and does not require the potentially immunomodulatory intraperitoneal or subcutaneous priming (with adjuvant) procedures used in the OVA models to evoke airways inflammation and BHR.
After intranasal exposure to extracts of Af, both wild-type and IgE-deficient mice developed heavy eosinophilic loads in bronchoalveolar lavage (BAL) fluid and in the lung parenchyma. BHR also was induced in both wild-type and IgE-deficient mice after Af treatment. Interestingly, we also noted that the airways of unsensitized IgE-deficient animals exhibited enhanced responsiveness compared with unsensitized wild-type mice.
MATERIALS AND METHODS
Reagents and Mice.
A mixture of culture filtrate and mycelial extracts of Af was the kind gift of Bayer Pharmaceuticals and was diluted to 2 mg/ml. IgE-deficient mice were derived as previously described (24). They have been maintained inbred in a 129/SVEV background.
Sensitization of Mice.
Mice were anesthetized by methoxyfluorane inhalation and 50 μl of Aspergillus antigen or 50 μl of saline was applied to the left nare. Mice were immunized three times a week for 3 weeks and were studied 12 h after the last dose.
Pulmonary Function Measurements.
Pulmonary conductance (GL) and pulmonary dynamic compliance (Cdyn) changes in response to methacholine, administered via a jugular venous catheter to anesthetized and ventilated mice in a whole-body plethysmograph, were measured as previously described (27). The output signals from 8–10 consecutive breaths were analyzed using a computerized cross-correlation method (28). The GL and Cdyn peak responses to each dose were expressed as a percentage of the baseline values before that dose.
Collection of Specimens.
For BAL, PBS, 0.8 ml containing 10% fetal calf serum and 1 mM EDTA, was infused into the trachea and then retrieved using a syringe. BAL leukocytes were counted using a hemocytometer. The leukocyte differential was determined from Diff-Quik (Baxter)-stained cytocentrifuged BAL fluid, and the absolute eosinophil count derived as the product of the leukocyte count and the eosinophil fraction.
Histologic Analysis.
Lungs were fixed in 10% formalin under mild vacuum. Five-micrometers hematoxylin- and eosin-stained sections were prepared. In addition, Luna stain was used to confirm the presence of eosinophils in the inflammatory infiltrate. Sections were incubated for 5 min in 0.9 vol of Weigert’s iron hematoxylin (0.005% acid hematoxylin and 0.6% ferric chloride in 2% HCl) with 0.1 vol of 1% Biebrich scarlet and 0.1% acid fuchsin in 1% acetic acid. After differentiation in 1% acid alcohol, slides were washed in water. Final color development was done in 0.5% lithium carbonate. These stains render eosinophils bright red on a blue background.
A grading scheme was developed to characterize the intensity of the inflammatory infiltrate in normal saline (NS)- and Af-treated animals. Lungs that showed no focal inflammation or peribronchial or perivascular inflammatory infiltrates were scored as grade 0. Those that showed one or two centrally located microscopic foci of inflammatory infiltrate were graded as 1. In grade 2 lungs, a dense inflammatory infiltrate was seen in a perivascular and peribronchial distribution originating in the center of the lung and extending along the vessels and bronchi into the middle third of the lung parenchyma. In grade 3 lesions, the perivascular and peribronchial infiltrates extended to the periphery of the lung approaching the visceral pleura. Grading was performed (by M.V.R.) on unidentified slides.
Quantification of Serum IgE Levels.
Serum IgE levels were determined by ELISA as described in the PharMingen protocol, using clone R35-188 antibodies for coating and clone R35-72 antibodies for detection.
Af-specific IgE was measured using heterologous passive cutaneous anaphylaxis (PCA). Serial dilutions of sera were injected intradermally into the shaved back of a Sprague–Dawley rat (Taconic). Twenty-four hours later, 2 mg of Af in 1 ml of 1 × PBS containing 1% Evans blue dye was injected into the tail vein and the skin was examined at 30 min for extravasation of the dye into the dermis. The PCA titer was determined as the reciprocal of the highest dilution giving a positive reaction.
RESULTS
Serum IgE Levels.
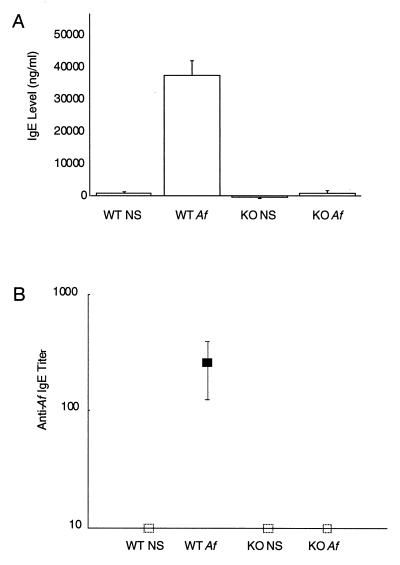
We used a mouse model of hypersensitivity to the naturally occurring airborne allergen Af to study the role of IgE in the induction of BHR and eosinophilic inflammation of the airway mucosa. Repeated intranasal administration of a protein extract of Af in BALB/c mice previously has been shown to evoke significant elevations of IgE (25, 26). To establish that Af can induce elevated IgE in the 129/sv strain background and to confirm the null phenotype of the IgE-deficient animals, we subjected wild-type and IgE-deficient mice to 3 weeks of repeated intranasal sensitization (nine doses) with Af or NS. As expected, intranasal Af sensitization induced substantially elevated IgE levels in wild-type Af-treated mice (Fig. 1A). IgE was undetectable in sera from both NS- and Af-treated IgE-deficient mice.
Figure 1.
(A) Serum IgE levels in IgE-deficient (KO) and wild-type (WT) mice after treatment with NS or Af extract. Bars represent mean values for five mice within each treatment group ± SEM. (B) Heterologous PCA determination of anti-Af IgE. Titers represent the reciprocal of the greatest serum dilution giving a positive cutaneous reaction. Mean titers ± SEM are presented for the groups described in A. Open squares represent data groups in which no sera conferred a detectable reaction (titer <10).
The presence of Af-specific IgE in sera of NS- and Af-treated mice was assayed using PCA in rat skin. This sensitive heterologous PCA assay is specific for murine IgE and gives no reaction for IgG (ref. 29 and H.O., unpublished data). Intranasal Af extract administration to wild-type mice resulted in a mean PCA titer of 140 (Fig. 1B). No Af-specific IgE was detectable in the sera from NS-treated wild-type mice or in any of the IgE-deficient mice (titers all <10).
BAL Leukocytosis and Eosinophilia.
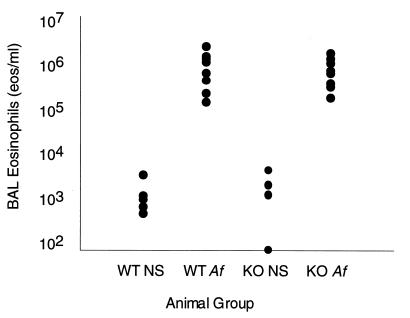
To establish whether IgE is critical to the recruitment of eosinophils to the bronchi in Af-sensitized mice, we measured BAL eosinophilia 12 h after the last allergen dose. BAL eosinophil counts were elevated by almost three orders of magnitude in Af-treated wild-type animals (mean 9.42 × 105 eosinophils per ml) compared with NS-treated controls (mean 1.02 × 103 eosinophils per ml) (Fig. 2). The intensity of BAL eosinophilia was equivalent in the Af-treated IgE-deficient mice (mean 7.69 × 105 eosinophils per ml). Leukocyte differential of the BAL specimens was also similar in animals from both groups; in particular, monocytes were the primary cell type in NS-treated animals, and eosinophils were the predominant cell type (>80%) in Af-treated mice (data not shown).
Figure 2.
Eosinophil counts (eosinophils per ml) in BAL fluid from IgE-deficient (KO) and wild-type (WT) mice treated with NS or with Af extract. Each group includes 8–10 mice, and the data are combined from two independently performed experiments. (P < 0.01 for NS- vs. Af-treated groups for both WT and KO mice)
Lung Histopathology.
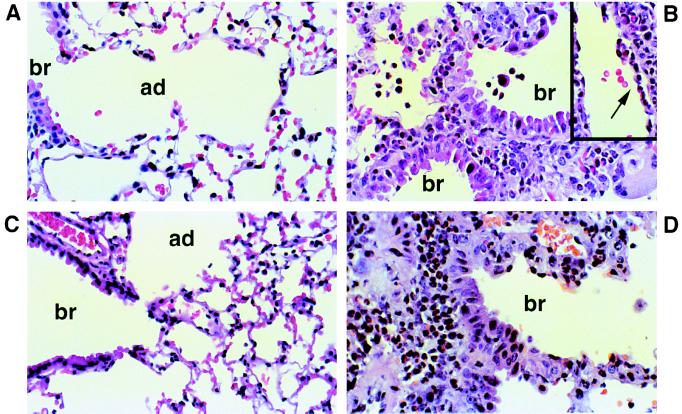
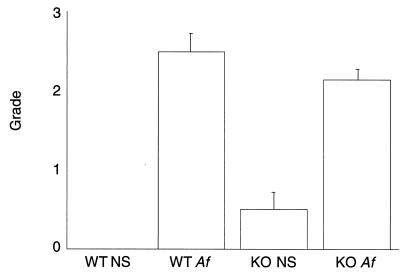
The extent and anatomy of the allergic airways inflammation induced by intranasal Af exposure was further studied by histologic analysis. The lungs of wild-type mice treated with NS appeared normal (Fig. 3A) and were uniformly scored as grade 0 (Fig. 4). In contrast, wild-type mice treated with Af showed dense inflammatory infiltrates (Fig. 3B). These infiltrates were predominantly present as peribronchial and perivascular cuffs that extended from the center of the lung to almost reach the visceral pleura in three cases (grade 3). The remaining three animals showed grade 2 perivascular and peribronchial infiltrates (mean grade 2.5; Fig. 4).
Figure 3.
Lung histology from wild-type (WT) and IgE-deficient mice (KO) after treatment with NS or Af. (×600.) (A) WT mouse treated with NS. (B) WT mouse treated with intranasal Af extract shows a peribronchiolar inflammatory infiltrate consisting predominantly of eosinophils, admixed with lymphocytes. Involvement of the bronchiolar epithelium with associated epithelial damage is present. The airspaces contain numerous eosinophils and histiocytes. Vessels are surrounded by a cuff of inflammatory cells and contain marginating eosinophils with migration into the vessel wall (arrow in Inset). (C) KO mouse treated with NS. (D) KO mouse treated with intranasal Af shows essentially the same findings as those seen in WT mice. Br, bronchiole; ad, alveolar duct. These panels depict fields representative of the histology obtained from six or seven mice per group.
Figure 4.
Intensity of pulmonary infiltrate in wild-type (WT) and IgE-deficient (KO) mice after treatment with NS or Af extract. The grading scheme is described in detail in the text. Each group contained six or seven animals. The average grade of the four groups of mice ± SEM is presented.
The inflammatory infiltrate consisted predominantly of eosinophils with a significant number of admixed lymphocytes and occasional histiocyte-like cells. The infiltrate expanded the interstitium and was associated with edema. At the level of the terminal bronchioles, numerous eosinophilic cells were seen in the submucosal layers with occasional involvement of the columnar epithelium (Fig. 3B) associated with epithelial damage. In many vessels eosinophils were seen marginating and migrating through the vessel walls (Fig. 3B Inset). Eosinophils and histiocytes were also present in bronchiolar and alveolar airspaces. The eosinophilic nature of the infiltrate was confirmed by Luna stain (data not shown).
IgE-deficient mice treated with NS showed a grade 0 score for three animals and a grade 1 score for the remaining three animals (mean grade 0.5; Fig. 4). In grade 1 animals, only one or two centrally located small foci of inflammatory cells were seen. No perivascular or peribronchial cuffs were noted. None of the animals in this group showed significant inflammatory infiltrates at the boundary between the terminal bronchiole and alveolar ducts (Fig. 3C). The histologic findings were essentially the same as those seen in wild-type mice treated with NS.
The IgE-deficient mice treated with Af showed features quite similar to those seen in identically treated wild-type animals (Fig. 3D). The distribution of the infiltrate was scored as grade 3 in one specimen; the remaining six were grade 2 (mean 2.1; Fig. 4).
Pulmonary Function Studies.
To determine whether the Af sensitization protocol used in this model induces BHR and to assess the role of IgE in this phenomenon, we determined GL and Cdyn following graded i.v. methacholine administration in sedated, ventilated mice placed in a full-body plethysmograph.
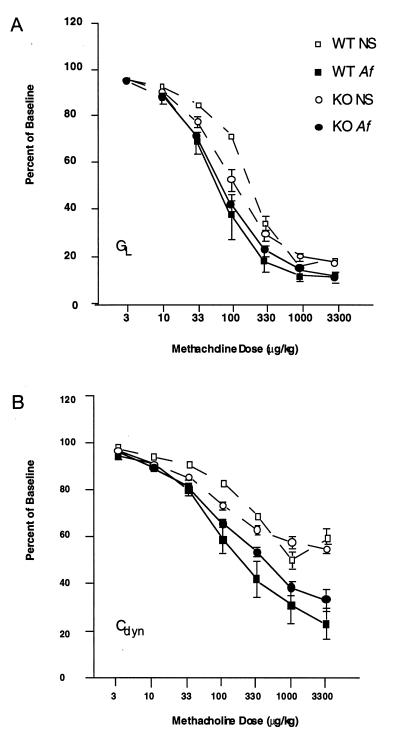
Both wild-type and IgE-deficient Af-treated mice had diminutions in airway conductance and dynamic compliance at lower doses of cholinergic agonist than did NS-treated controls (Fig. 5). In addition, they exhibited greater maximal airway constriction in comparison with NS controls. This is manifested by a leftward and downward shift of the methacholine dose–response curves (Fig. 5). The data obtained from these experiments were subjected to two-way ANOVA with the treatment group and logarithm of methacholine dose as independent variables and the maximally decreased GL or Cdyn values expressed as percentages of preinfusion values. The shift in airways responsiveness observed after Af treatment, as measured by either GL or Cdyn, was highly significant for both wild-type and IgE-deficient mice (P < 0.001 in each case by ANOVA; Fig. 5).
Figure 5.
GL (A) and Cdyn (B)in IgE-deficient (KO) and wild-type (WT) mice after treatment with NS or Af extract. Treatment groups are WT NS (□), WTAf (▪), KO NS (○), and KO Af (•). After the establishment of baseline values, GL and Cdyn were determined after graded doses of i.v. methacholine. Individual points on each curve indicate the mean for each treatment group ± SEM (n = 4 for WT groups and 8–9 for KO groups). A two-way ANOVA was performed for the data used to derive these curves (treatment group and methacholine dose as independent variables and percent of baseline as dependent variable). Both GL and Cdyn responses to methacholine were significantly greater (P < 0.001) in Af-treated mice compared with NS-treated mice of the same (WT or KO) genotype. Both GL and Cdyn responses to methacholine were also significantly greater (P < 0.025) in NS-treated KO mice compared with NS-treated WT mice.
To further quantitate this difference in the bronchial responses of NS-treated mice, the mean methacholine dose required to cause a 50% decrease in GL (ED50 GL) or a 30% decrease in Cdyn (ED30 Cdyn) was calculated (Table 1). In each case, comparison of these estimates of airway responsiveness also revealed significantly greater responsiveness in Af-sensitized IgE-deficient as well as wild-type mice compared with the nonsensitized control mice.
Table 1.
GL and Cdyn responses to i.v. methacholine
| Group | ED50GL,* μg/kg | 95% C.I.,† μg/kg | P‡ | ED30Cdyn,* μg/kg | 95% C.I.,† μg/kg | P‡ |
|---|---|---|---|---|---|---|
| WT NS | 200 | 167.5–237.7 | 291.7 | 269.8–315.5 | ||
| WT Af | 69.2 | 38.3–124.7 | 0.014 | 62.7 | 33.9–117.5 | 0.029§ |
| KO NS | 120.2 | 86.7–165.2 | 177.8 | 123.6–255.9 | ||
| KO Af | 77.6 | 61.5–95.7 | 0.046 | 72.4 | 55.7–94.2 | 0.001 |
| WT NS | 200 | 167.5–237.7 | 291.7 | 269.8–315.5 | ||
| KO NS | 120.2 | 86.7–165.2 | 0.071 | 177.8 | 123.6–255.9 | 0.199§ |
ED50 GL and ED30 Cdyn represent the geometric mean methacholine dose required to cause a 50% decrease in GL or a 30% decrease in Cdyn. These values were obtained by graphical interpolation of dose–response curves obtained on individual mice.
The 95% confidence intervals for the mean ED50 GL and ED30 Cdyn values. These were calculated from the logarithmic ED50 GL or ED30 Cdyn values ± 2 times the geometric SEM.
Significance of differences in ED50 GL and ED30 Cdyn values between mouse groups indicated. These values represent results of the Student t test except where indicated by
, when data failed normality tests and the nonparametric Mann–Whitney U test was applied.
Surprisingly, the NS-treated IgE-deficient mice had more bronchial reactivity than NS-treated wild-type mice. This was a statistically significant shift (P = 0.022 for GL and P = 0.012 for Cdyn by ANOVA) and occurred reproducibly and in the absence of significant differences in BAL eosinophilia or lung histopathology. The ED50 GL was 200 μg/kg for wild-type NS-treated mice and only 120.2 μg/kg for NS-treated IgE-deficient animals. A similar decrement was observed for ED30 Cdyn; 291 μg/kg for wild-type mice and 177.8 μg/kg for IgE-deficient animals. These data demonstrate significant BHR in unsensitized IgE-deficient mice when compared with wild-type mice in the same genetic background.
DISCUSSION
Airway inflammation and BHR are central features of asthma and have been induced in a number of animal models. We have studied mice with a targeted deletion of the IgE heavy chain locus and wild-type mice in an identical genetic background to compare asthmatic pathology and pathophysiology in the absence and presence of IgE. As expected, Af treatment induced elevations in total and antigen-specific IgE only in wild-type mice. Despite the absence of elevated IgE, however, IgE-deficient mice developed intense eosinophilic airways exudates, of the same magnitude as wild-type animals as measured in BAL. The pulmonary histology of the IgE-deficient Af-treated mice was also indistinguishable from that of wild-type Af-sensitized animals. We conclude that IgE is not required for the generation of marked allergic airways inflammation in response to inhaled allergen in this mouse model.
Furthermore, the induction of BHR associated with this inflammation occurred even in the absence of IgE. Both mutant and wild-type mice displayed similar GL methacholine responses after Af treatment (Fig. 5A). The apparent relative shift in methacholine responsiveness in Af-treated vs. NS-treated animals was a bit less dramatic for the IgE-deficient mice compared with wild-type animals, but this is accounted for by the increased bronchial reactivity of NS-treated mutant mice. We would interpret these data to indicate that IgE is not required to induce the expression of Af-induced BHR. The Cdyn methacholine responses for Af-treated wild-type mice were somewhat more intense than those of IgE-deficient animals (Fig. 5B). This finding suggests that although IgE is not required for allergen-induced BHR, the physiologic response may be slightly increased in the presence of IgE antibodies.
IgE-independent eosinophilic inflammation and BHR could arise by alternative pathways of immediate hypersensitivity as well as by delayed type hypersensitivity mechanisms. It is well known that immediate hypersensitivity reactions can be elicited in mice by IgE-independent pathways. Cultured mast cells can be triggered to degranulate by a number of non-IgE factors, including IgG, complement components C3a and C5a, and “histamine-releasing factors” (30, 31). Both wild-type and IgE-deficient mice produce Af-specific IgG after intranasal sensitization (data not shown). In the past it had been shown that the cutaneous hypersensitivity could be transferred from donor mouse to recipient by IgG (32, 33). Allergen-specific mast cell degranulation in vivo as well as full-blown active systemic anaphylaxis can be induced in the absence of IgE (24). We have been unable to detect histamine in the BAL of Af-treated wild-type or IgE-deficient mice (data not shown). The sensitivity of the radioimmunoassay may be inadequate for this greatly diluted fluid, so we do not believe that a role for mast cells is excluded.
In addition to the sequelae of IgE-independent mast cell activation, airways inflammation and BHR in IgE-deficient mice likely arise by pathways that do not involve mast cells at all. Both eosinophilic airways inflammation and allergen-induced BHR have been observed in mast cell-deficient W/Wv mice (20, 34). A number of human and murine studies now document a prominent role for a T cell response to allergen in the induction of airway mucosal inflammation and BHR. Antigen-specific T cells are clearly expanded in the airways of asthmatic humans and in mice subjected to inhaled allergen exposure (21, 22, 35). BHR and inflammation can be transferred from immune to naive animals by T cells alone (23, 36).
We recognize that our observations are not necessarily applicable to human asthma. Human eosinophils, unlike their murine counterparts, express the high-affinity IgE receptor, FcɛRI, so that IgE might directly influence their function in allergic inflammation (37). Human mast cells have not been shown to be as sensitive to activation via FcγR as rodent cells.
Coyle and colleagues recently have suggested that IgE plays a significant role in the recruitment of eosinophils to the lungs of mice immunized and challenged with allergen (20). Treatment with an anti-IgE antibody resulted in a significant attenuation of eosinophil recruitment. These findings appear to contradict our results. This may be attributable to the in vivo effects of the anti-IgE reagent used as well as to differences in the sensitization and challenge regimens or to strain differences affecting responses to allergic stimuli. Although, the “nonanaphylactogenic” anti-IgE used by Coyle for this work cannot interact with FcɛRI-bound IgE, and therefore cannot activate mast cells, its ability to recognize IgE bound to B cells via the low-affinity IgE receptor, CD23, has not been examined. B cells that are IgE+ by virtue of CD23-bound “cytophilic” IgE are quite prevalent and could be functionally affected by crosslinking of their CD23 in a way that influences an ongoing allergic response. Furthermore, injected anti-IgE may give rise to circulating IgE immune complexes whose impact on immune responses is unknown. Thus, the injection of an anti-IgE reagent might give rise to consequences that are not directly related to the diminution of serum IgE. This study employed a priming step using alum as adjuvant. Perhaps the eosinophilic airways inflammation induced after systemic immunization with a Th2-promoting adjuvant is more dependent on IgE than that evoked after mucosal exposure alone.
Interestingly, the group of Gelfand has described the induction of eosinophilic airways inflammation and BHR by mucosal immunization alone using nebulized OVA in BALB/c mice (17). They have shown that BHR, measured by an ex vivo assay, and eosinophilic inflammation after OVA challenge can be passively transferred to naive recipients by IgE or IgE+ B cells from animals sensitized to OVA only by inhalation (18). IgE-independent mechanisms involving the generation of allergen-specific CD8+ cells also may participate in pathogenesis in this model (38).
One unexpected finding in our study was that IgE-deficient animals had a higher level of baseline bronchial reactivity (measured in NS-treated controls) than did the wild-type NS-treated animals. A similar observation previously has been made for interleukin (IL)-4-deficient mice that also have an inability to produce IgE (39). We did not find evidence of increased baseline eosinophilic inflammation in the IgE-deficient mice. Their BAL eosinophil counts were not significantly different from those of wild-type mice. On histologic examination, some of the animals in this group had scattered microscopic foci of inflammation resulting in scores of grade 1 for three of six animals, but the distinction between grades 0 and 1 is so slight that we hesitate to conclude that this represents a significant difference. It is conceivable that the absence of IgE in the IgE and IL-4 “knockouts” removes a negative-feedback mechanism by which factor(s) that enhance bronchial reactivity are regulated. Alternatively, IgE itself might modulate the reactivity of airways in response to nonspecific stimuli.
Our findings, together with those of other investigators, are consistent with the existence of parallel pathways of eosinophil recruitment to the allergic airway and induction of BHR in asthma. IgE may be required for pathogenesis in some circumstances while functioning as an accessory or regulatory factor in others. The factors that govern the relative contribution of IgE to bronchial eosinophilia and BHR remain to be elucidated, but it is likely that they will include the physical nature of the immunizing allergen, its route of administration, and the influences of a variety of independent background genes (40). We expect that additional studies with well characterized mouse mutants in distinct strain backgrounds, adopting diverse immunization protocols, will further define the determinants of airway allergy.
Acknowledgments
We thank Dr. Raif Geha for his critical reading of the manuscript. P.D.M. is supported by National Institutes of Health Grant T32HD-07321. T.R.M. is supported by National Institutes of Health Grant HL36110. H.C.O. is the recipient of National Institutes of Health Grant KO8 AI01253 and the American Academy of Allergy, Asthma and Immunology Education and Research Trust Award.
Footnotes
Abbreviations: BHR, bronchial hyperreactivity; Af, Aspergillus fumigatus; NS, normal saline; BAL, bronchoalveolar lavage; PCA, passive cutaneous anaphylaxis; GL, pulmonary conductance; Cdyn, pulmonary dynamic compliance; OVA, ovalbumin.
References
- 1.McFadden E R, Gilbert I A. N Engl J Med. 1992;327:1928–1937. doi: 10.1056/NEJM199212313272708. [DOI] [PubMed] [Google Scholar]
- 2.Hogg J C. J Allergy Clin Immunol. 1993;92:1–4. doi: 10.1016/0091-6749(93)90029-f. [DOI] [PubMed] [Google Scholar]
- 3.Bentley A M, Menz G, Storz C, Robinson D S, Bradley B, Jeffery P K, Durham S R, Kay A B. Am Rev Respir Dis. 1992;146:500–506. doi: 10.1164/ajrccm/146.2.500. [DOI] [PubMed] [Google Scholar]
- 4.Burrows B, Martinez F D, Halonen M, Barbee R A, Cline M G. N Engl J Med. 1989;320:271–277. doi: 10.1056/NEJM198902023200502. [DOI] [PubMed] [Google Scholar]
- 5.Sears M R, Burrows B, Flannery E M, Herbison G P, Hewitt C J, Holdaway M D. N Engl J Med. 1991;325:1067–1071. doi: 10.1056/NEJM199110103251504. [DOI] [PubMed] [Google Scholar]
- 6.Postma D S, Bleecker E R, Amelung P J, Holroyd K J, Xu J, Panhuysen C I, Meyers D A, Levitt R C. N Engl J Med. 1995;333:894–900. doi: 10.1056/NEJM199510053331402. [DOI] [PubMed] [Google Scholar]
- 7.Saban R, Haak F M, Zine M, Ridgway J, Gorman C, Presta L G, Bjorling D, Saban M, Jardieu P. J Allergy Clin Immunol. 1994;94:836–843. doi: 10.1016/0091-6749(94)90151-1. [DOI] [PubMed] [Google Scholar]
- 8.Shields R L, Whether W R, Zioncheck K, O’Connell L, Fendly B, Presta L G, Thomas D, Saban R, Jardieu P. Int Arch Allergy Immunol. 1995;107:308–312. doi: 10.1159/000237010. [DOI] [PubMed] [Google Scholar]
- 9.Galli S J. N Engl J Med. 1993;328:257–265. doi: 10.1056/NEJM199301283280408. [DOI] [PubMed] [Google Scholar]
- 10.Kaliner M. J Allergy Clin Immunol. 1989;83:510–520. doi: 10.1016/0091-6749(89)90031-6. [DOI] [PubMed] [Google Scholar]
- 11.Norel X, Haye L I, Labat C, Benveniste J, Brink C. Int Arch Allergy Appl Immunol. 1991;96:368–375. doi: 10.1159/000235524. [DOI] [PubMed] [Google Scholar]
- 12.Cartier A, Thomson N C, Frith P A, Roberts R, Hargreave F E. J Allergy Clin Immunol. 1982;70:170–177. doi: 10.1016/0091-6749(82)90038-0. [DOI] [PubMed] [Google Scholar]
- 13.Plaut M, Pierce J H, Watson C J, Hanley-Hyde J, Nordan R P, Paul W E. Nature (London) 1989;339:64–67. doi: 10.1038/339064a0. [DOI] [PubMed] [Google Scholar]
- 14.Burd P R, Rogers H W, Gordon J R, Martin C A, Jayaraman S, Wilson S D, Dvorak A M, Galli S J, Dorf M E. J Exp Med. 1989;170:245–257. doi: 10.1084/jem.170.1.245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sutton B J, Gould H J. Nature (London) 1993;366:421–428. doi: 10.1038/366421a0. [DOI] [PubMed] [Google Scholar]
- 16.Kehry M R, Yamashita L C. Proc Natl Acad Sci USA. 1989;86:7556–7560. doi: 10.1073/pnas.86.19.7556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Renz H, Smith H R, Henson J E, Ray B S, Irvin C G, Gelfand E W. J Allergy Clin Immunol. 1992;89:1127–1138. doi: 10.1016/0091-6749(92)90296-e. [DOI] [PubMed] [Google Scholar]
- 18.Lack G, Oshiba A, Bradley K L, Loader J E, Amran D, Larsen G L, Gelfand E W. Am J Respir Crit Care Med. 1995;152:1765–1773. doi: 10.1164/ajrccm.152.6.8520735. [DOI] [PubMed] [Google Scholar]
- 19.Eum S, Haile S, Lefort J, Huerre M, Vargaftig B B. Proc Natl Acad Sci USA. 1995;92:12290–12294. doi: 10.1073/pnas.92.26.12290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Coyle A J, Wagner K, Bertrand C, Tsuyuki S, Bews J, Heusser C. J Exp Med. 1996;183:1303–1310. doi: 10.1084/jem.183.4.1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Azzawi M, Bradley B, Jeffery P K, Frew A J, Wardlaw A J, Knowles G, Assoufi B, Collins J V, Durham S, Kay A B. Am Rev Respir Dis. 1990;142:1407–1413. doi: 10.1164/ajrccm/142.6_Pt_1.1407. [DOI] [PubMed] [Google Scholar]
- 22.Gerblich A A, Campbell A E, Schuyler M R. N Engl J Med. 1984;310:1349–1352. doi: 10.1056/NEJM198405243102103. [DOI] [PubMed] [Google Scholar]
- 23.Garssen, J., Nijkamp, F. P., Van Der Vliet, H. & Van Loveren, H. (1993) Chest 103, Suppl. 2, 129S–130S. [DOI] [PubMed]
- 24.Oettgen H C, Martin T R, Wynshaw-Boris A, Deng C X, Drazen J M, Leder P. Nature (London) 1994;370:367–370. doi: 10.1038/370367a0. [DOI] [PubMed] [Google Scholar]
- 25.Kurup V P, Mauze S, Choi H, Seymour B W, Coffman R L. J Immunol. 1992;148:3783–3788. [PubMed] [Google Scholar]
- 26.Kurup V P, Choi H, Murali P S, Coffman R L. J Leukocyte Biol. 1994;56:593–598. doi: 10.1002/jlb.56.5.593. [DOI] [PubMed] [Google Scholar]
- 27.Martin T R, Gerard N P, Galli S J, Drazen J M. J Appl Physiol. 1988;64:2318–2323. doi: 10.1152/jappl.1988.64.6.2318. [DOI] [PubMed] [Google Scholar]
- 28.Kroeker J P. Biol Cybern. 1977;27:221–227. doi: 10.1007/BF00344143. [DOI] [PubMed] [Google Scholar]
- 29.Ovary Z, Caiazza S S, Kojima S. Int Arch Allergy Appl Immunol. 1975;48:16–21. doi: 10.1159/000231289. [DOI] [PubMed] [Google Scholar]
- 30.Fox P C, Basciano L K, Siraganian R P. J Immunol. 1982;129:314–319. [PubMed] [Google Scholar]
- 31.Katz H R, Raizman M B, Gartner C S, Scott H C, Benson A C, Austen K F. J Immunol. 1992;148:868–871. [PubMed] [Google Scholar]
- 32.Vaz E M, Silveira I H S, Dodd V M B, Monteiro M R, Vaz N M. Int Arch Allergy. 1970;39:459–468. [PubMed] [Google Scholar]
- 33.Nussenzweig R S, Merryman C, Benacerraf B. J Exp Med. 1964;120:315–328. doi: 10.1084/jem.120.2.315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Brusselle G G, Kips J C, Tavernier J H, van der Heyden J, Cuvelier C A, Pauwels R A, Bluethmann H. Clin Exp Allergy. 1994;24:73–80. doi: 10.1111/j.1365-2222.1994.tb00920.x. [DOI] [PubMed] [Google Scholar]
- 35.Renz H, Saloga J, Bradley K L, Loader J E, Greenstein J L, Larsen G, Gelfand E W. J Immunol. 1993;151:1907–1917. [PubMed] [Google Scholar]
- 36.Garssen J, Van Loveren H, Van Der Vliet H, Bot H, Nijkamp F P. Am Rev Respir Dis. 1993;147:307–313. doi: 10.1164/ajrccm/147.2.307. [DOI] [PubMed] [Google Scholar]
- 37.Gounni A S, Lamkhioued B, Ochiai K, Tanaka Y, Delaporte E, Capron A, Kinet J P, Capron M. Nature (London) 1994;367:183–186. doi: 10.1038/367183a0. [DOI] [PubMed] [Google Scholar]
- 38.Hamelmann E, Oshiba A, Paluh J, Bradley K, Loader J, Potter T A, Larsen G L, Gelfand E W. J Exp Med. 1996;183:1719–1729. doi: 10.1084/jem.183.4.1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brusselle G, Kips J, Joos G, Bluethmann H, Pauwels R. Am J Respir Cell Mol Biol. 1995;12:254–259. doi: 10.1165/ajrcmb.12.3.7873190. [DOI] [PubMed] [Google Scholar]
- 40.Drazen J M, Arm J P, Austen K F. J Exp Med. 1996;183:1–5. doi: 10.1084/jem.183.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]