The observation that infection can precipitate an autoimmune disease dates back more than a century. The first human autoimmune disease described, paroxysmal cold hemoglobulinuria, was thought of as a late consequence of syphilis, and rheumatic fever is still associated with preceding streptococcal infection. In modern times, these associations have been attributed to molecular mimicry. In its simplest form, the concept of molecular mimicry states that antigenic determinants of infectious microorganisms resemble structures in the tissues of the host but differ enough to be recognized as foreign by the host’s immune system. It is now clear that mimicry on the molecular level is a common phenomenon; that is, many sequential and structural determinants of infectious agents simulate epitopes of host tissues (1). But, as Mackay and I remarked recently, “There are, as yet, no firm instances of molecular mimicry by microorganisms serving as initiating agents of human autoimmune disease…” (2).
Molecular mimicry in Chagas’ disease
There is probably no better candidate for investigating mimicry than the cardiomyopathy of chronic Chagas’ disease. It afflicts about 30% of the 20 million individuals infected with the protozoan Trypanosoma cruzi in the Americas. The presence of a cardiac inflammatory infiltrate in apparent absence of parasites suggests that the trypanosome initiates an autoimmune response. Indeed, a number of cross-reactive human antigens have been implicated by their reaction with sera of Chagas patients. They include, for example, a 23 kDa ribosomal protein (3), a functional epitope on the β1 adrenergic receptor (4), a 48 kDa protein found in neuronal axons (5), and a heptapeptide of cardiac myosin heavy chain (6). In this issue of the JCI, Gironès and colleagues have identified another cross-reactive antigen (Cha), a novel peptide from human cells (7). This peptide, which reacts with the sera of patients with chronic Chagas’ disease and of mice infected with T. cruzi, was found in abundance in human and mouse hearts. Cross-reaction between the mammalian and trypanosomal peptides was documented for both T and B cells. The finding that this peptide bears both B- and T-cell epitopes makes it a leading candidate for the induction of the cardiomegaly of Chagas’ disease through molecular mimicry, since it would facilitate T/B-cell cooperation (8).
This work leaves critical questions unanswered. Some patients with Chagas’ disease develop megacolon and megaesophagus due to destruction of parasympathetic ganglia, but Cha is not found in nervous tissue. No functional changes were associated with Cha-specific antibodies. On the other hand, mice immunized with a 13 amino acid ribosomal peptide of T. cruzi produced antibodies that caused functional changes in the heart without evidence of mononuclear infiltration (9). Finally, living trypanosomes induce Chagas-like lesions in the hearts of mice, but recombinant Cha does not. Thus, the question of whether molecular mimicry using a single, defined antigen of the parasite in the absence of infection actually mirrors clinical autoimmune disease remains unaddressed.
Molecular mimicry abounds
Recent insights into T-cell recognition have greatly broadened the original concept of molecular mimicry. A number of studies have shown that there is a fair measure of flexibility in the amino acid sequence acceptable for both MHC class II binding and for recognition by the T-cell receptor (10, 11). Clearly, microbial peptides with relatively limited sequence homology to myelin basic protein (MBP) can activate autoreactive T cells. Using an extensive combinatorial peptide library, Hemmer et al. (12) described differing recognition profiles of individual autoreactive T-cell clones from patients with multiple sclerosis. Li et al. (13) showed that, because of topological differences in their peptide finding sites, different MHC class II molecules can create different alignments of the same bound MBP peptide, thereby creating distinct T-cell epitopes from the same peptide. Thus, T-cell recognition is even more degenerate than previously anticipated, and primary amino acid sequence similarities provide little clue to a molecular mimic.
Further expansion of the autoimmune response probably occurs due to epitope spreading. Vanderlugt and colleagues (14) developed a murine model of relapsing encephalomyelitis, using an immunodominant epitope of the myelin antigen, proteolipid protein (PLP). In the course of the disease, T-cell clones recognizing epitopes of MBP are detected that are not present on PLP, suggesting that the release of endogenous antigen during the initial response can stimulate self-reactive T cells and play a critical role in disease progression. These data further suggest that an unrelated infection could mobilize a self-peptide and potentiate but not initiate an attack of autoimmune disease in a subclinically primed individual (15). All of these findings demonstrate why it may be difficult to identify a single organism as the etiological agent of an autoimmune disease.
The adjuvant effect
The experiments described above remind us that the generation of an immune response depends upon two types of signals: antigen-specific recognition through the T-cell and B-cell receptors and a number of non–antigen-specific, nonclonal signals. An infectious agent may provide the specific signal by molecular mimicry or by release of endogenous antigen. The inflammatory process itself enhances the nonclonal costimulatory signals necessary to mount an immune response. Lack of appropriate costimulatory signals may account for the difference between a pathogenic and a nonpathogenic autoimmune response following infection. Our own studies with Coxsackie B3–induced (CB3-induced) myocarditis exemplify such a situation. In genetically susceptible A/J mice, infection with CB3 induces autoimmune myocarditis (16). Injection of killed virus does not, suggesting that inflammation elicited by live virus provides costimulatory signals that influence subsequent immune responses. Evidence expanding this view came from experiments using less susceptible B10.A mice. If cotreated with bacterial LPS, as well as virus or cardiac myosin, the animals develop typical myocarditis. LPS acts through toll-like receptor 4 (TLR-4) and potently activates the innate immune response; particularly, it upregulates production of inflammatory cytokines such as IL-1 and TNF-α. Administration of either of these two cytokines converts a less susceptible to a more susceptible mouse. Furthermore, the production of autoimmune myocarditis in the susceptible mouse strain could be delayed or abrogated by administration of inhibitors of either of these two cytokines.
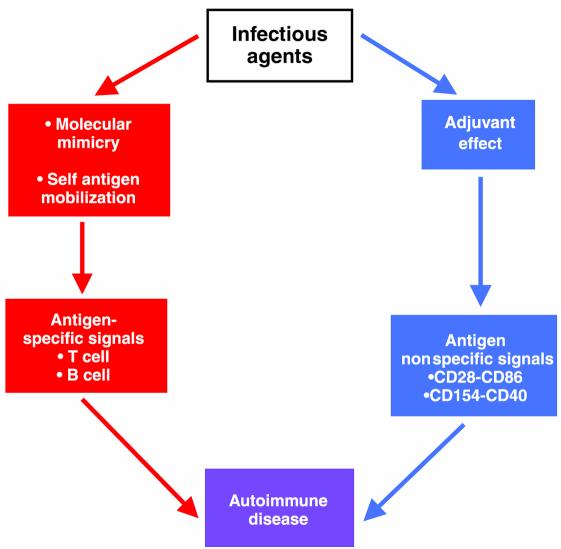
These results point to the importance of the inflammatory response itself in the generation of an autoimmune disease. They emphasize that an infectious microorganism may play two roles in the induction of disease (Figure 1). The first is to provide the requisite antigenic signal. This may come through molecular mimicry or through the release of excessive amounts of self-antigen from tissue cells during the infectious process. The second role of the infectious agent is to provide the adjuvant milieu in the form of upregulation of costimulatory molecules and other products of inflammation. The activation of antigen-presenting cells during microbial infection upregulates costimulatory molecules and secretion of inflammatory cytokines (17), thereby reducing the threshold needed for activation of T cells by the antigenic signal. This effect, which may promote protective immunity and thus benefit the host, may also prove be detrimental when it increases susceptibility to damaging autoimmune responses.
Figure 1.
An infectious agent contributes to the induction of autoimmunity in two ways. First, it provides a cognate, antigen-specific signal by molecular mimicry or by mobilizing endogenous antigen (red arrows). In parallel, it causes inflammation, generating antigen nonspecific signals (blue arrows) that potentiate immune responses through what is known as an adjuvant effect.
References
- 1.Oldstone MBA. Molecular mimicry and immune-mediated diseases. FASEB J. 1998;12:1255–1265. doi: 10.1096/fasebj.12.13.1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rose NR, Mackay IR. Molecular mimicry: a critical look at exemplary instances in human diseases. Cell Mol Life Sci. 2000;57:542–551. doi: 10.1007/PL00000716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bonfa E, Viana VST, Barreto ACP, Yoshinari NH, Cossermelli W. Autoantibodies in Chagas’ disease. J Immunol. 1993;150:3917–3923. [PubMed] [Google Scholar]
- 4.Ferrari I, et al. Molecular mimicry between the immunodominant ribosomal protein P0 of Trypanosoma cruzi and a functional epitope on the human β1-adrenergic receptor. J Exp Med. 1995;182:59–65. doi: 10.1084/jem.182.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Van Voorhis WC, Schlekewy L, Trong HL. Molecular mimicry by Trypanosoma cruzi: the F1-160 epitope that mimics mammalian nerve can be mapped to a 12-amino acid peptide. Proc Natl Acad Sci USA. 1991;88:5993–5997. doi: 10.1073/pnas.88.14.5993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kalil J, Cunha-Neto E. Autoimmunity in Chagas’ disease cardiomyopathy: fulfilling the criteria at last? Parasitol Today. 1996;12:396–399. doi: 10.1016/0169-4758(96)10058-2. [DOI] [PubMed] [Google Scholar]
- 7.Gironès N, et al. Dominant T- and B-cell epitopes in an autoantigen linked to Chagas’ disease. J Clin Invest. 2001;107:985–993. doi: 10.1172/JCI10734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liang B, Mamula MJ. Molecular mimicry and the role of B lymphocytes in the processing of autoantigens. Cell Mol Life Sci. 2000;57:561–568. doi: 10.1007/PL00000718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Motrán CC, Fretes RE, Cerbán FM, Rivarola HW, Vottero de Cima E. Immunization with the C-terminal region of Trypanosoma cruzi ribosomal P1 and P2 proteins induces long-term duration cross-reactive antibodies with heart functional and structural alterations in young and aged mice. Clin Immunol. 2000;97:89–94. doi: 10.1006/clim.2000.4919. [DOI] [PubMed] [Google Scholar]
- 10.Hausmann S, Martin M, Gauthier L, Wucherpfennig KW. Structural features of autoreactive TCR that determine the degree of degeneracy in peptide recognition. J Immunol. 1999;162:338–344. [PubMed] [Google Scholar]
- 11.Barnaba V, Sinigaglia F. Molecular mimicry and T cell-mediated autoimmune disease. J Exp Med. 1999;185:1529–1531. doi: 10.1084/jem.185.9.1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hemmer B, et al. Identification of high potency microbial and self ligands for a human autoreactive class II-restricted T cell clone. J Exp Med. 1997;185:1651–1659. doi: 10.1084/jem.185.9.1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li Y, Li H, Martin R, Mariuzza RA. Structural basis for the binding of an immunodominant peptide from myelin basic protein in different registers by two HLA-DR2 proteins. J Mol Biol. 2000;304:177–188. doi: 10.1006/jmbi.2000.4198. [DOI] [PubMed] [Google Scholar]
- 14.Vanderlugt CL, et al. Pathologic role and temporal appearance of newly emerging autoepitopes in relapsing experimental autoimmune encephalomyelitis. J Immunol. 2000;164:670–678. doi: 10.4049/jimmunol.164.2.670. [DOI] [PubMed] [Google Scholar]
- 15.Rao VP, Kajon AE, Spindler KR, Carayonniotis G. Involvement of epitope mimicry in potentiation but not initiation of autoimmune disease. J Immunol. 1999;162:5888–5893. [PubMed] [Google Scholar]
- 16.Lane JR, Neumann DA, Lafond-Walker A, Herskowitz A, Rose NR. Role of IL-1 and tumor necrosis factor in coxsackie virus-induced autoimmune myocarditis. J Immunol. 1993;151:1682–1690. [PubMed] [Google Scholar]
- 17.Manickasingham SP, Anderton SM, Burkhart C, Wraith DC. Qualitative and quantitative effects of CD28/B7-mediated costimulation on naive T cells in vitro. J Immunol. 1998;161:3827–3835. [PubMed] [Google Scholar]