Abstract
The exquisite sensitivity of the cochlea, which mediates the transduction of sound waves into nerve impulses, depends on the endocochlear potential and requires a highly specialized environment that enables and sustains sensory function. Disturbance of cochlear homeostasis is the cause of many forms of hearing loss including the most frequently occurring syndromic and non-syndromic forms of hereditary hearing loss, Pendred syndrome and Cx26-related deafness. The occurrence of these and other monogenetic disorders illustrates that cochlear fluid homeostasis and the generation of the endocochlear potential are poorly secured by functional redundancy. This review summarizes the most prominent aspects of cochlear fluid homeostasis. It covers cochlear fluid composition, the generation of the endocochlear potential, K+ secretion and cycling and its regulation, the role of gap junctions, mechanisms of acid–base homeostasis, and Ca2+ transport.
Introduction
Of all the organs in the body, the cochlea secretes the most unusual extracellular fluid, endolymph, and generates the largest transepithelial voltage, the endocochlear potential – all to enable and sustain the most impressive mechanoreceptor, the hair cell. This review is focused on the first two fascinating superlatives, cochlear fluid homeostasis and the generation of the endocochlear potential. Other recent reviews have focused on hereditary hearing losses (Petit et al. 2001), cochlear fluid volume regulation (Salt, 2001), age-related hearing loss (Gates & Mills, 2005), ototoxicity (Rybak & Whitworth, 2005) and noise trauma (Henderson et al. 2006).
Inner ear fluids
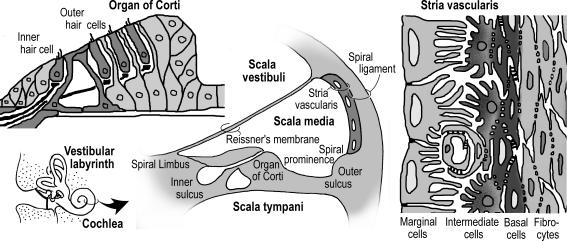
Three extracellular fluids have been identified in the cochlea: endolymph, perilymph and intrastrial fluid (Table 1; Wangemann & Schacht, 1996). Endolymph fills scala media of the cochlea (Fig. 1). Intrastrial fluid fills the small extracellular spaces within stria vascularis. Perilymph fills scala vestibuli and scala tympani and is continuous with all extracellular spaces of the cochlea except those that are filled with blood, endolymph or intrastrial fluid.
Table 1.
Composition of cochlear fluids
| Component | Unit | Endolymph scala media | Intrastrial fluid | Perilymph scala vestibuli | Perilymph scala tympani | Plasma |
|---|---|---|---|---|---|---|
| Na+ | (mm) | 1.3 | 85 | 141 | 148 | 145 |
| K+ | (mm) | 157 | 2 | 6.0 | 4.2 | 5.0 |
| Ca2+ | (mm) | 0.023 | 0.8 | 0.6 | 1.3 | 2.6 |
| Cl− | (mm) | 132 | 55 | 121 | 119 | 106 |
| HCO3− | (mm) | 31 | n.a. | 18 | 21 | 18 |
| Glucose | (mm) | 0.6 | n.a. | 3.8 | 3.6 | 8.3 |
| pH | (pH units) | 7.4 | n.a. | 7.3 | 7.3 | 7.3 |
| Protein | (mg dl−1) | 38 | n.a. | 242 | 178 | 4238 |
For references, see Wangemann & Schacht (1996)
Figure 1. Schematic diagrams illustrating the anatomy of the cochlea (centre), organ of Corti (left) and stria vascularis (right).
Endolymph is enclosed by a heterogeneous epithelium that consists in the cochlea of about 12 different epithelial cell types that include the sensory hair cells. Endolymph is an unusual extracellular fluid in that its composition is reminiscent of an intracellular fluid. K+ provides the major charge carrier for sensory transduction and is an ideal charge carrier, since it is by far the most abundant ion in the cytosol. Relative changes in the cytosolic ion composition due to transduction currents are smaller for K+ than they would be for Na+, and both K+ influx into and efflux from the sensory cells can occur passively down the electrochemical gradient, which limits the metabolic requirements. The major driving force for sensory transduction is generated by stria vascularis, a densely vascularized epithelium in the lateral wall of the cochlea. The metabolic demand for the generation of this driving force requires a dense capillary network for the delivery of O2 and glucose and the removal of CO2. Spatial separation between stria vascularis (as the source of the transduction current) and organ of Corti (housing the sensory transducer) has the advantage that the highly sensitive mechanosensor is somewhat isolated from low frequency vibrations associated with flowing blood in capillaries.
Endolymph is not only an unusual extracellular fluid for its high K+ and low Na+ concentration but also for its low Ca2+ concentration, high HCO3− concentration and low protein content. The significance of the high HCO3− concentration may be related to the need for pH buffering. The low Ca2+ concentration is critical for sensory transduction in the cochlea. Ca2+ enters the hair bundle together with K+ and is necessary for the generation of the mechano-electrical transduction current as well as for adaptation of the transduction mechanism (Ricci & Fettiplace, 1998; Holt & Corey, 2000).
The endocochlear potential
The main driving force for sensory transduction is provided by the endocochlear potential, which is generated by stria vascularis (von Békésy, 1950; Davis, 1953; Wangemann, 2002). Stria vascularis is functionally a two layered epithelium as shown in Fig. 2, with cells forming both barriers by tight junctions (Jahnke, 1975). Tight junctions among basal cells define an inner membrane facing the intrastrial space and an outer membrane facing spiral ligament. The inner membrane is connected to strial intermediate cells via gap junctions comprised of Gjb2 (Cx26) and Gjb6 (Cx30), which ensure that intermediate cells are electrically a part of the basal cell barrier (Kikuchi et al. 1995; Lautermann et al. 1998; Xia et al. 1999, 2001). The outer membrane of basal cells is connected by gap junctions to fibrocytes of the spiral ligament. This complicated architecture is a prerequisite for the generation of the endocochlear potential.
Figure 2. Schematic diagrams illustrating K+ secretion and the generation of the endocochlear potential in the cochlea.
Left, cochlea cross-section. Right, stria vascularis. Strial marginal cells and basal cells of stria vascularis are each connected among themselves by tight junctions to form two barriers. The barrier between endolymph and intrastrial fluid consists of strial marginal cells and the barrier between intrastrial fluid and the extracellular fluid spaces in spiral ligament consists of basal cells. The endocochlear potential is generated across the basal cell barrier. Intermediate cells are connected by gap junctions to the inner membrane of basal cells. The outer membrane of basal cells is connected via gap junctions to fibrocytes of spiral ligament.
The endocochlear potential is essentially a K+ equilibrium potential that is generated by the K+ channel Kcnj10 (Kir 4.1) located in the intermediate cells of stria vascularis in conjunction with the very low K+ concentration of intrastrial fluid and a normally high K+ concentration in the cytosol of intermediate cells (Takeuchi et al. 2000; Marcus et al. 2002). Consequently, the endocochlear potential can be measured across the basal cell barrier (Salt et al. 1987). Strial marginal cells contribute to the generation of the endocochlear potential in that they keep the K+ concentration in the intrastrial fluid spaces extremely low (Marcus et al. 1985; Wangemann et al. 1995; Takeuchi et al. 2000). Similarly, spiral ligament fibrocytes, which are endowed with K+ uptake mechanisms, assist the generation of the endocochlear potential by maintaining the high cytosolic K+ concentration in the intermediate cells (Schulte & Adams, 1989; Mizuta et al. 1997; Crouch et al. 1997).
K+ cycling
K+ is the major charge carrier for sensory transduction and for the generation of the endocochlear potential (Konishi et al. 1978; Johnstone et al. 1989; Zidanic & Brownell, 1990). K+ in endolymph is driven by the endocochlear potential into the sensory hair cells via the apical transduction channel and out into perilymph via basolateral K+ channels including Kcnq4 (mediates the current IK,n), Kcnn2 (ISK2) and Kcnma1 (IK,f) (Kros, 1996). From perilymph, K+ is taken up by fibrocytes of the spiral ligament from where it diffuses into strial intermediate cells. K+ is released from the intermediate cells into the intrastrial space via the KCNJ10 K+ channel that generates the endocochlear potential (Marcus et al. 2002). Other K+ channels may contribute to the delivery of K+ into the intrastrial space (Takeuchi & Irimajiri, 1996; Nie et al. 2005). From the intrastrial space, K+ is taken up by strial marginal cells and secreted into endolymph (Wangemann et al. 1995). Uptake occurs via the Na+–2Cl−–K+ cotransporter Slc12a2 and the Na+,K+-ATPase Atp1a1/Atp1b2 and secretion across the apical membrane into endolymph occurs via the K+ channel Kcnq1/Kcne1 (mediates IsK) (Shen et al. 1997; Sunose et al. 1997). Cl− is recycled through the basolateral membrane via Cl− channels Clcnka (Clck-1), Clcnkb (Clck-2) and Bsnd (barttin) (Ando & Takeuchi, 2000; Sage & Marcus, 2001). Recycling of Cl− appears to be essential for K+ secretion and the generation of the endocochlear potential. Mutations of Bsnd or coincidence of mutations of Clcnka and Clcnkb cause Bartter syndrome type 4, which is characterized by renal salt wasting and deafness (Estevez et al. 2001; Birkenhager et al. 2001; Schlingmann et al. 2004).
Most segments of K+ cycling are well established and generally accepted including the pathway through stria vascularis and through hair cells. Two concepts, however, are presented for the pathway between perilymph and fibrocytes of the spiral ligament. Supported by flux and current measurements is the concept that K+ enters perilymph and flows through perilymph toward spiral ligament (Zidanic & Brownell, 1990; Salt & Ohyama, 1993). An alternative concept envisions that K+ released from the hair cells is taken up by supporting cells in the organ of Corti and funnelled via a gap junction network toward the spiral ligament and stria vascularis as well as toward the spiral limbus toward yet-undefined sites of K+ secretion (Spicer & Schulte, 1996). Cellular uptake of K+ has been envisioned to occur via KCl transporters in supporting cells of the organ of Corti. The transport direction of KCl transporters, however, is generally outwardly directed given the usual stoichiometry and ionic gradients, which precludes their involvement in an uptake of K+. In addition, the gap junctions Gjb2 (Cx26) or Gjb6 (Cx30) have been shown not to be essential for the generation of a normal endolymphatic K+ concentration and a normal cochlear endolymph volume, which argues against a necessary role of the funnel mechanism (Cohen-Salmon et al. 2002; Teubner et al. 2003).
Reabsorptive pathways contributing to K+ cycling are not limited to the pathway through the hair cells. Part of the current that is generated by stria vascularis is carried through the outer sulcus and through Reissner's membrane (Zidanic & Brownell, 1990; Salt & Ohyama, 1993). The outer sulcus has been shown to mediate Na+ and K+ absorption (Marcus & Chiba, 1999) and Reissner's membrane has been shown to be involved in Na+ absorption from endolymph (Lee & Marcus, 2003).
The essential elements of K+ secretion
Several of the transporters involved in K+ secretion and the generation of the endocochlear potential are essential in that their absence leads to a cessation of K+ secretion, a collapse of the endolymphatic compartment, and failure to generate the endocochlear potential.
The K+ channel Kcnq1/Kcne1 is essential for K+ secretion across the apical membrane of strial marginal cells and their equivalent, vestibular dark cells, in the vestibular labyrinth (Marcus & Shen, 1994; Wangemann et al. 1995). K+ secretion fails in mice that lack functional Kcnq1 or Kcne1 and endolymphatic spaces appear collapsed (Vetter et al. 1996; Lee et al. 2000; Letts et al. 2000; Casimiro et al. 2001). Severe mutations of Kcnq1 or Kcne1 cause Jervell and Lange-Nielsen syndrome, which is a rare autosomal recessive disease characterized by profound sensory-neural deafness and prolonged QT-intervals of the cardiac action potentials (Schulze-Bahr et al. 1997; Wang et al. 2002). The endolymphatic space of patients suffering from Jervell and Lange-Nielsen syndrome appears collapsed as observed in mouse models (Friedmann et al. 1966). Jervell and Lange-Nielsen syndrome precipitates arrhythmias, especially during exercise or emotional stress, that can lead to syncope and even sudden death in otherwise healthy individuals. Less severe mutations of Kcnq1 and/or Kcne1 lead to Romano-Ward syndrome, which is a more frequently observed autosomal dominant disease consisting of long-QT syndrome without deafness.
The Na+–2Cl−–K+ cotransporter Slc12a2 is expressed in strial marginal cells as well as in fibrocytes of the spiral ligament and in vestibular dark cells (Marcus et al. 1987; Wangemann & Marcus, 1990; Mizuta et al. 1997; Crouch et al. 1997). Slc12a2 is essential for the uptake of K+ from cochlear and vestibular perilymph as well as for uptake of K+ from the intrastrial space, which is essential for the generation of the endocochlear potential. Inhibition of Slc12a2 with loop-diuretic abolishes the endocochlear potential (Kusakari et al. 1978). K+ secretion fails in mice that lack Slc12a2, and endolymphatic spaces appear collapsed (Dixon et al. 1999; Delpire et al. 1999; Flagella et al. 1999).
The Na+,K+-ATPase subunits Atp1a1 and Atp1b1 or Atp1b2 are expressed in the basolateral membrane of vestibular dark cells and strial marginal cells as well as in fibrocytes of the spiral ligament (Schulte & Adams, 1989; McGuirt & Schulte, 1994). The Na+,K+-ATPase takes up K+ with high affinity and drives further uptake of K+ via the Na+–2Cl−–K+ cotransporter (Marcus & Marcus, 1987; Wangemann et al. 1995).
The K+ channel Kcnj10 is expressed in intermediate cells of stria vascularis (Ando & Takeuchi, 1999). The onset of expression during postnatal development parallels the generation of the endocochlear potential (Hibino et al. 2004). Kcnj10 was pharmacologically identified as the channel that generates the endocochlear potential (Marcus et al. 1985; Takeuchi & Ando, 1998b) and furthermore mice that lack Kcnj10 also lack the endocochlear potential (Marcus et al. 2002). Kcnj10 is not only essential for the generation of the endocochlear potential but provides a major, although not an essential, pathway for K+ cycling. The endolymphatic volume and endolymphatic K+ concentration in mice lacking Kcnj10 were found to be partly reduced, whereas the vestibular endolymphatic volume and K+ concentration, which do not depend on Kcnj10, were normal (Marcus et al. 2002).
Regulation of K+ cycling
Secretory and reabsorptive mechanisms across the heterogeneous epithelia enclosing scala media need to be well balanced to maintain a constant fluid volume. Failure to maintain this balance will result in an enlargement of the endolymphatic compartment as seen in Menière's disease and Pendred syndrome or result in a collapse of scala media as seen in Jervell and Lange-Nielsen syndrome (Yamakawa, 1938; Friedmann et al. 1966; Johnsen et al. 1986). Whether the volume of scala media is detected in the cochlea and where this sensor is located is still unclear. Secretory and reabsorptive mechanisms also need to be well tuned to maintain the intrastrial space. Failure to balance release of K+ by intermediate cells and transepithelial K+ secretion by strial marginal cells can lead to an accumulation of K+ in the intrastrial fluid space and thereby abolish the endocochlear potential. K+ secretion by strial marginal cells and vestibular dark cells is exquisitely sensitive, responding to 1 mm changes in the K+ concentration of the extracellular fluid bathing the basolateral membrane (Wangemann et al. 1996). Thus, the K+ secretory cells appear to be the sensors as well as effectors in the control of the K+ concentration at their basolateral side.
K+ cycling is modified by acoustic stimulation. Acoustic overstimulation causes a temporary threshold shift, reduces the endocochlear potential and the endolymphatic K+ concentration and alters the K+ efflux pathway from endolymph to perilymph (Salt & Konishi, 1979; Thorne et al. 2004). Sound stimulation releases ATP into endolymph and perilymph, which initiates protective mechanisms (Munoz et al. 2001). ATP released into endolymph stimulates P2X receptors in outer hair cells, Reissner's membrane and outer sulcus cells and activates reabsorptive pathways for K+ that provide a shunt to reduce the current density through the sensory hair cells (King et al. 1998; Lee et al. 2001; Zhao, 2005). In addition, ATP released into perilymph activates P2X receptors in the basolateral membrane of outer hair cells (Housley et al. 1999; Zhao et al. 2005). P2X receptors reduce outer hair cell motility, which dampens the cochlear amplifier and thereby limits the mechanical stimulation of the sensory cells. Further, ATP activates P2Y4 receptors in the apical membrane of strial marginal cells, which reduces the rate of K+ secretion and can be expected to reduce the endocochlear potential (Marcus et al. 2005).
Physical and emotional stresses via the stress hormones noradrenaline and adrenaline enable a number of organ-specific responses that support a ‘fight or flight reaction’ mediated by β-adrenergic receptors. For example, β1-adrenergic receptors increase the rate and force of the heart beat and β2-adrenergic receptors open and moisturize airways and eyes. It is thus not surprising that hearing is prepared for a successful ‘fight or flight reaction’. β1-Adrenergic receptors have been found in the organ of Corti, in the outer sulcus and in stria vascularis of the cochlea (Fauser et al. 2004). Evidence is emerging that supports the concept that noradrenaline accelerates K+ cycling in the cochlea and the vestibular labyrinth. Noradrenaline stimulates the rate of K+ secretion in strial marginal cells (Sunose et al. 1997; Wangemann et al. 2000). Further, outer hair cells appear to contain β1-adrenergic receptors and cAMP has been shown to stimulate the basolateral channels including the K+ channel Kcnq4 (Jagger & Ashmore, 1999). Functional β2-adrenergic receptors have also been identified in the spiral ligament (Schimanski et al. 2001). It is conceivable, but has not yet been demonstrated, that β2-adrenergic receptors stimulate the uptake of K+ via the Na+,K+-ATPase and the Na+–2Cl−–K+ cotransporter Slc12a2 in spiral ligament fibrocytes.
Gap junctions
Two gap junction systems are generally distinguished in the cochlea (Kikuchi et al. 2000). One system includes the supporting cells of the organ of Corti and the inner and outer sulcus epithelial cells. The other system includes strial intermediate and basal cells as well as fibrocytes of the spiral ligament. Although most cells in the cochlea are connected by gap junctions, there are notable exceptions including inner and outer hair cells and strial marginal cells (Takeuchi & Ando, 1998a; Kikuchi et al. 2000).
Gap junctions, especially Gjb2 (Cx26) and Gjb6 (Cx30), which connect intermediate and basal cells of stria vascularis, are critically important for the generation of the endocochlear potential and therefore essential for hearing. Mutations of GJB2 are the most prevalent hereditary cause of childhood deafness (Zelante et al. 1997; Denoyelle et al. 1998). Mice that lack functional expression of Gjb6 lack the endocochlear potential and are profoundly deaf. The endolymphatic K+ concentration at the time of the onset of hearing is normal in these mice, illustrating that Gjb6 is not essential for K+ cycling (Teubner et al. 2003). Mutations of GJB6 are another prevalent cause of childhood deafness (Del Castillo et al. 2003).
Although gap junctions may not be essential for K+ cycling, they appear to be essential for sharing metabolites among cells (Matsunami et al. 2006). Gap junctions formed from Gjb2, which harbours mutations that cause non-syndromic deafness, maintained ionic coupling but failed to exchange biochemicals (Zhang et al. 2005). Metabolic coupling may be important in the lateral wall as well as in the organ of Corti in that the connected cells may provide a buffer system for HCO3−, free radicals, Ca2+ and metabolites (Sun et al. 2005; Zhao, 2005). Buffering of glutamate released from inner hair cells may depend on coupling between supporting cells. Glutamine synthase, a key enzyme in the detoxification of glutamate, is mainly expressed in the neighbours but not in the cells that express the glutamate uptake transporter Slc1a3 (Glast) (Eybalin et al. 1996). Consequently, Slc1a3-expressing cells are the first to undergo apoptosis in mice that lack Cx26 in this region of the cochlea (Cohen-Salmon et al. 2002).
In addition to Gjb2 and Gjb6, Gja1 (Cx43), Gja7 (Cx45) and Gje1 (Cx29) have been found in the cochlea (Ahmad et al. 2003). Gja1 and Gja7 are expressed in various cells of the developing mouse cochlea and mutations of GJA1 are associated with non-syndromic deafness of unclear aetiology (Liu et al. 2001; Cohen-Salmon et al. 2004). In adult mice, however, Gja1 expression is limited to the spiral ligament, capillaries of stria vascularis and bone of the otic capsule and Gja7 expression is limited to capillary endothelial cells (Cohen-Salmon et al. 2004). Gje1 is expressed in myelinating Schwann cells of the spiral ganglion. Mice that lack Gje1 (Cx29) have prolonged latencies in their auditory brain stem responses and acquire hearing with a delay (Tang et al. 2006). Whether mutations of GJA7 or GJE1 are responsible for human deafness is currently unclear.
Cochlear acid–base transport
The main pH buffer in endolymph appears to be HCO3−/CO2. Proteins, due to their low concentration, play a minor role (Table 1). The significance of pH homeostasis in cochlear fluids is linked to the general pH sensitivity of ion channels, transporters and metabolic enzymes. Acute acidification of cochlear fluids by experimental manoeuvres have been shown to reduce the endocochlear potential (Sterkers et al. 1984; Ikeda et al. 1987b). Acidification of cochlear fluids enhances free radical stress and hearing loss, whereas alkalinization has a protective effect (Tanaka et al. 2004). Consistently, metabolic acidosis and impairment of cochlea pH regulation are associated with hearing loss.
The endolymphatic pH appears to depend on HCO3− and H+ secretion and on carbonic anhydrase activity, although little is known about endolymphatic pH and HCO3− homeostasis (Sterkers et al. 1984; Ikeda et al. 1987b). The largest source of metabolically generated CO2 in the cochlea are marginal cells of stria vascularis (Marcus et al. 1978). Metabolically derived CO2 may be captured and converted to HCO3− by carbonic anhydrases expressed in high density in intermediate and basal cells of stria vascularis, in fibrocytes of the spiral ligament and spiral limbus and in red blood cells (Lim et al. 1983; Spicer & Schulte, 1991; Okamura et al. 1996). Fibrocytes of the spiral ligament may take up HCO3− via the Na+–HCO3− cotransporter Slc4a7 and share HCO3− via gap junctions (Bok et al. 2003).
It is conceivable that some fraction of HCO3− is cycled through endolymph. Support for the concept of HCO3− cycling comes from the observation that acoustic stimulation, which increases stria metabolism and CO2 production, causes an alkalization of endolymph, which would be consistent with secretion of HCO3− into endolymph (Ikeda et al. 1988). Several epithelial cells lining cochlear endolymph express HCO3− and H+ transporters and are thereby likely participants in the control of the endolymph pH and HCO3− concentration. Epithelial cells that express H+-ATPase or the Cl−–HCO3− exchanger Slc26a4 in their apical membrane include interdental cells of the spiral limbus, strial marginal cells, spiral prominence and outer sulcus epithelial cells and spindle cells of stria vascularis (Stankovic et al. 1997; Wangemann et al. 2004). In addition, interdental and outer sulcus epithelial cells express the Cl−–HCO3− exchanger Slc4a2 (Ae2) in their basolateral membrane (Stankovic et al. 1997).
Cochlear pH regulation appears to be important for the prevention of hearing loss. In particular, long-term effects of cochlear acidosis may limit energy metabolism, promote the generation of free radicals and activate the innate immune system. Mutations of Slc26a4 cause Pendred syndrome, which is the most common syndromic form of hereditary deafness associated with an enlarged vestibular aqueduct and euthyroid goitre (Everett et al. 1997; Reardon et al. 2000). Deafness is sometimes late in onset and provoked by stresses such as light head injury or infection (Luxon et al. 2003). Mice lacking Slc26a4 suffer from hearing loss and provide an excellent model of Pendred syndrome (Everett et al. 2001; Wangemann et al. 2004).
Mutations of subunits of H+-ATPase, including ATP6V1B1 or ATPV0A4, cause renal tubular acidosis with progressive sensorineural hearing loss (Karet et al. 1999; Stover et al. 2002). Atp6v1b1 is expressed in a limited number of tissues including the acid secreting intercalated cells of the renal distal tubule and interdental cells of the cochlea. Mutations of ATP6V1B1 are generally associated with an early onset of sensorineural hearing loss whereas mutations of ATPV0A4, which is a more commonly expressed subunit of H+-ATPases, is generally associated with a later onset in hearing loss. The aetiology of the hearing loss is unclear. Interestingly, mice lacking functional expression of Atp6v1b1 differ significantly from the human phenotype. Humans bearing mutations of ATP6V1B1 generate an abnormally alkaline urine, develop a metabolic acidosis, lose Ca2+ from bone, and suffer from growth retardation, rickets, calciuria, renal stone formation and hearing loss. In contrast, mice lacking functional expression of Atp6v1b1 did not develop metabolic acidosis although they generated a significantly more alkaline urine. In addition, mice do not suffer from calciuria, grow normally and have normal hearing (Dou et al. 2003; Finberg et al. 2005). It is conceivable that these differences are due to compensatory pH regulatory mechanisms present in the murine cochlea. Alternatively, differences may indicate that hearing loss in humans is due to alteration in cochlear Ca2+ homeostasis rather than pH regulation.
Cochlear Ca2+ transport
The endolymphatic Ca2+ concentration is critical for normal auditory function. Microphonic potentials and transduction currents generated by the sensory cells are blocked by elevated endolymphatic Ca2+ concentrations and suppressed by reduced Ca2+ concentrations (Tanaka et al. 1980; Ohmori, 1985). Reduced endolymphatic Ca2+ concentrations are also suspected to be the cause for hearing loss associated with vitamin D deficiency and hypoparathyroidism, two conditions that are associated with low plasma Ca2+ concentrations (Brookes, 1983; Ikeda et al. 1987a). Consistent with those observations, the endolymphatic Ca2+ concentration is pathologically low in deaf-waddler mice and pathologically high in a surgically derived model of Menière's disease (Ninoyu & Meyer zum Gottesberge, 1986; Wood et al. 2004).
Ca2+ absorption may occur through paracellular and transcellular pathways and may at least in part be driven by the endocochlear potential (Ikeda et al. 1987c). Transcellular pathways may include uptake of Ca2+ from endolymph via Ca2+ permeable TRP channels (Ecac) and export into perilymph via Ca2+-ATPases and Na+–Ca2+ exchangers, as described in the vestibular labyrinth (Yamauchi et al. 2005).
Ca2+ secretion into endolymph may occur via Ca2+-ATPases rather than Na+–Ca2+ exchangers (Ikeda & Morizono, 1988). In particular, the Pmca2 Ca2+-ATPase Atp2b2 appears to be involved in Ca2+ secretion based on the finding that deaf-waddler mice bear a loss-of-function mutation in Atp2b2 (Street et al. 1998; Wood et al. 2004). Based on the limited expression of Atp2b2 in the cochlea, it is conceivable that Reissner's membrane secretes Ca2+ into endolymph. Support for this concept comes from the finding that the Ca2+ concentration in perilymph of scala vestibuli is lower than in scala tympani (Table 1). In addition, Atp2b2 is expressed in the hair bundles of the sensory cells. The role of this Ca2+-ATPase may be restricted to the removal of Ca2+ from the bundle (Apicella et al. 1997).
Conclusion
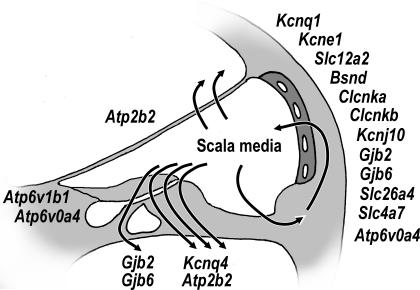
Cellular homeostasis, homeostasis of cochlear fluids and the generation of the endocochlear potential are critically important for enabling and sustaining cochlear function. The importance of homeostasis is underscored by the fact that mutations of genes such as GJB2, GJB6, SLC26A4, BSND, KCNQ1, KCNE1 and KCNQ4 lead to deafness in humans and that spontaneous mutations of Kcne1 and Atp2b2, targeted mutations of Gjb2, and null mutations of Slc26a4, Slc12a2, Slc4a7, Gjb6, Kcnq1, Kcne1 and Kcnj10 lead to deafness in mouse models (Fig. 3).
Figure 3. Schematic diagram illustrating the approximate location of expression of genes known or suspected to be associated with deafness.
Data were collated from knowledge obtained in mice and humans.
Acknowledgments
The author thanks Daniel C. Marcus for critically reading the manuscript. Support by NIH-R01-DC01098 is gratefully acknowledged.
References
- Ahmad S, Chen S, Sun J, Lin X. Connexins 26 and 30 are co-assembled to form gap junctions in the cochlea of mice. Biochem Biophys Res Commun. 2003;307:362–368. doi: 10.1016/s0006-291x(03)01166-5. [DOI] [PubMed] [Google Scholar]
- Ando M, Takeuchi S. Immunological identification of an inward rectifier K+ channel (Kir4.1) in the intermediate cell (melanocyte) of the cochlear stria vascularis of gerbils and rats. Cell Tissue Res. 1999;298:179–183. doi: 10.1007/s004419900066. [DOI] [PubMed] [Google Scholar]
- Ando M, Takeuchi S. mRNA encoding ‘ClC-K1, a kidney Cl− channel’ is expressed in marginal cells of the stria vascularis of rat cochlea: its possible contribution to Cl− currents. Neurosci Lett. 2000;284:171–174. doi: 10.1016/s0304-3940(00)01021-1. [DOI] [PubMed] [Google Scholar]
- Apicella S, Chen S, Bing R, Penniston JT, Llinas R, Hillman DE. Plasmalemmal ATPase calcium pump localizes to inner and outer hair bundles. Neuroscience. 1997;79:1145–1151. doi: 10.1016/s0306-4522(97)00035-3. [DOI] [PubMed] [Google Scholar]
- Birkenhager R, Otto E, Schurmann MJ, Vollmer M, Ruf EM, Maier-Lutz I, Beekmann F, Fekete A, Omran H, Feldmann D, Milford DV, Jeck N, Konrad M, Landau D, Knoers NV, Antignac C, Sudbrak R, Kispert A, Hildebrandt F. Mutation of BSND causes Bartter syndrome with sensorineural deafness and kidney failure. Nat Genet. 2001;29:310–314. doi: 10.1038/ng752. [DOI] [PubMed] [Google Scholar]
- Bok D, Galbraith G, Lopez I, Woodruff M, Nusinowitz S, BeltrandelRio H, Huang W, Zhao S, Geske R, Montgomery C, Van Slightenhorst I, Friddle C, Platt K, Sparks MJ, Pushkin A, Abuladze N, Ishiyama A, Dukkipati R, Liu W, Kurtz I. Blindness and auditory impairment caused by loss of the sodium bicarbonate cotransporter NBC3. Nat Genet. 2003;34:313–319. doi: 10.1038/ng1176. [DOI] [PubMed] [Google Scholar]
- Brookes GB. Vitamin D deficiency – a new cause of cochlear deafness. J Laryngol Otol. 1983;97:405–420. doi: 10.1017/s0022215100094330. [DOI] [PubMed] [Google Scholar]
- Casimiro MC, Knollmann BC, Ebert SN, Vary JC, Jr, Greene AE, Franz MR, Grinberg A, Huang SP, Pfeifer K. Targeted disruption of the Kcnq1 gene produces a mouse model of Jervell and Lange–Nielsen Syndrome. Proc Natl Acad Sci U S A. 2001;98:2526–2531. doi: 10.1073/pnas.041398998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen-Salmon M, Maxeiner S, Kruger O, Theis M, Willecke K, Petit C. Expression of the connexin43- and connexin45-encoding genes in the developing and mature mouse inner ear. Cell Tissue Res. 2004;316:15–22. doi: 10.1007/s00441-004-0861-2. [DOI] [PubMed] [Google Scholar]
- Cohen-Salmon M, Ott T, Michel V, Hardelin JP, Perfettini I, Eybalin M, Wu T, Marcus DC, Wangemann P, Willecke K, Petit C. Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death. Curr Biol. 2002;12:1106–1111. doi: 10.1016/s0960-9822(02)00904-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crouch JJ, Sakaguchi N, Lytle C, Schulte BA. Immunohistochemical localization of the Na-K-Cl co-transporter (NKCC1) in the gerbil inner ear. J Histochem Cytochem. 1997;45:773–778. doi: 10.1177/002215549704500601. [DOI] [PubMed] [Google Scholar]
- Davis H. Energy into nerve impulses: the inner ear. Adv Sci. 1953;9:420–425. [Google Scholar]
- Del Castillo I, Moreno-Pelayo MA, Del Castillo FJ, Brownstein Z, Marlin S, Adina Q, Cockburn DJ, Pandya A, Siemering KR, Chamberlin GP, Ballana E, Wuyts W, Maciel-Guerra AT, Alvarez A, Villamar M, Shohat M, Abeliovich D, Dahl HH, Estivill X, Gasparini P, Hutchin T, Nance WE, Sartorato EL, Smith RJ, Van Camp G, Avraham KB, Petit C, Moreno F. Prevalence and evolutionary origins of the del (GJB6-D13S1830) mutation in the DFNB1 locus in hearing-impaired subjects: a multicenter study. Am J Hum Genet. 2003;73:1452–1458. doi: 10.1086/380205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delpire E, Lu J, England R, Dull C, Thorne T. Deafness and imbalance associated with inactivation of the secretory Na-K-2Cl co-transporter. Nat Genet. 1999;22:192–195. doi: 10.1038/9713. [DOI] [PubMed] [Google Scholar]
- Denoyelle F, Lina-Granade G, Plauchu H, Bruzzone R, Chaib H, Levi-Acobas F, Weil D, Petit C. Connexin 26 gene linked to a dominant deafness. Nature. 1998;393:319–320. doi: 10.1038/30639. [DOI] [PubMed] [Google Scholar]
- Dixon MJ, Gazzard J, Chaudhry SS, Sampson N, Schulte BA, Steel KP. Mutation of the Na-K-Cl co-transporter gene Slc12a2 results in deafness in mice. Hum Mol Genet. 1999;8:1579–1584. doi: 10.1093/hmg/8.8.1579. [DOI] [PubMed] [Google Scholar]
- Dou H, Finberg K, Cardell EL, Lifton R, Choo D. Mice lacking the B1 subunit of H+-ATPase have normal hearing. Hear Res. 2003;180:76–84. doi: 10.1016/s0378-5955(03)00108-4. [DOI] [PubMed] [Google Scholar]
- Estevez R, Boettger T, Stein V, Birkenhager R, Otto E, Hildebrandt F, Jentsch TJ. Barttin is a Cl− channel β-subunit crucial for renal Cl− reabsorption and inner ear K+ secretion. Nature. 2001;414:558–561. doi: 10.1038/35107099. [DOI] [PubMed] [Google Scholar]
- Everett LA, Belyantseva IA, Noben-Trauth K, Cantos R, Chen A, Thakkar SI, Hoogstraten-Miller SL, Kachar B, Wu DK, Green ED. Targeted disruption of mouse Pds provides insight about the inner-ear defects encountered in Pendred syndrome. Hum Mol Genet. 2001;10:153–161. doi: 10.1093/hmg/10.2.153. [DOI] [PubMed] [Google Scholar]
- Everett LA, Glaser B, Beck JC, Idol JR, Buchs A, Heyman M, Adawi F, Hazani E, Nassir E, Baxevanis AD, Sheffield VC, Green ED. Pendred syndrome is caused by mutations in a putative sulphate transporter gene (PDS) Nat Genet. 1997;17:411–422. doi: 10.1038/ng1297-411. [DOI] [PubMed] [Google Scholar]
- Eybalin M, Norenberg MD, Renard N. Glutamine synthetase and glutamate metabolism in the guinea pig cochlea. Hear Res. 1996;101:93–101. doi: 10.1016/s0378-5955(96)00136-0. [DOI] [PubMed] [Google Scholar]
- Fauser C, Schimanski S, Wangemann P. Localization of β1-adrenergic receptors in the cochlea and the vestibular labyrinth. J Membr Biol. 2004;201:25–32. doi: 10.1007/s00232-004-0703-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finberg KE, Wagner CA, Bailey MA, Paunescu TG, Breton S, Brown D, Giebisch G, Geibel JP, Lifton RP. The B1-subunit of the H+ ATPase is required for maximal urinary acidification. Proc Natl Acad Sci U S A. 2005;102:13616–13621. doi: 10.1073/pnas.0506769102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flagella M, Clarke LL, Miller ML, Erway LC, Giannella RA, Andringa A, Gawenis LR, Kramer J, Duffy JJ, Doetschman T, Lorenz JN, Yamoah EN, Cardell EL, Shull GE. Mice lacking the basolateral Na-K-2Cl cotransporter have impaired epithelial chloride secretion and are profoundly deaf. J Biol Chem. 1999;274:26946–26955. doi: 10.1074/jbc.274.38.26946. [DOI] [PubMed] [Google Scholar]
- Friedmann I, Fraser GR, Froggatt P. Pathology of the ear in the cardioauditory syndrome of Jervell and Lange-Nielsen (recessive deafness with electrocardiographic abnormalities) J Laryngol Otol. 1966;80:451–470. doi: 10.1017/s002221510006552x. [DOI] [PubMed] [Google Scholar]
- Gates GA, Mills JH. Presbycusis. Lancet. 2005;366:1111–1120. doi: 10.1016/S0140-6736(05)67423-5. [DOI] [PubMed] [Google Scholar]
- Henderson D, Bielefeld EC, Harris KC, Hu BH. The role of oxidative stress in noise-induced hearing loss. Ear Hear. 2006;27:1–19. doi: 10.1097/01.aud.0000191942.36672.f3. [DOI] [PubMed] [Google Scholar]
- Hibino H, Higashi-Shingai K, Fujita A, Iwai K, Ishii M, Kurachi Y. Expression of an inwardly rectifying K+ channel, Kir5.1, in specific types of fibrocytes in the cochlear lateral wall suggests its functional importance in the establishment of endocochlear potential. Eur J Neurosci. 2004;19:76–84. doi: 10.1111/j.1460-9568.2004.03092.x. [DOI] [PubMed] [Google Scholar]
- Holt JR, Corey DP. Two mechanisms for transducer adaptation in vertebrate hair cells. Proc Natl Acad Sci U S A. 2000;97:11730–11735. doi: 10.1073/pnas.97.22.11730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Housley GD, Kanjhan R, Raybould NP, Greenwood D, Salih SG, Jarlebark L, Burton LD, Setz VC, Cannell MB, Soeller C, Christie DL, Usami S, Matsubara A, Yoshie H, Ryan AF, Thorne PR. Expression of the P2X2 receptor subunit of the ATP-gated ion channel in the cochlea: implications for sound transduction and auditory neurotransmission. J Neurosci. 1999;19:8377–8388. doi: 10.1523/JNEUROSCI.19-19-08377.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeda K, Kobayashi T, Kusakari J, Takasaka T, Yumita S, Furukawa Y. Sensorineural hearing loss associated with hypoparathyroidism. Laryngoscope. 1987a;97:1075–1079. doi: 10.1288/00005537-198709000-00014. [DOI] [PubMed] [Google Scholar]
- Ikeda K, Kusakari J, Takasaka T. Ionic changes in cochlear endolymph of the guinea pig induced by acoustic injury. Hear Res. 1988;32:103–110. doi: 10.1016/0378-5955(88)90081-0. [DOI] [PubMed] [Google Scholar]
- Ikeda K, Kusakari J, Takasaka T, Saito Y. Early effects of acetazolamide on anionic activities of the guinea pig endolymph: evidence for active function of carbonic anhydrase in the cochlea. Hear Res. 1987b;31:211–216. doi: 10.1016/0378-5955(87)90189-4. [DOI] [PubMed] [Google Scholar]
- Ikeda K, Kusakari J, Takasaka T, Saito Y. The Ca2+ activity of cochlear endolymph of the guinea pig and the effect of inhibitors. Hear Res. 1987c;26:117–125. doi: 10.1016/0378-5955(87)90040-2. [DOI] [PubMed] [Google Scholar]
- Ikeda K, Morizono T. Calcium transport mechanism in the endolymph of the chinchilla. Hear Res. 1988;34:307–311. doi: 10.1016/0378-5955(88)90010-x. [DOI] [PubMed] [Google Scholar]
- Jagger DJ, Ashmore JF. The fast activating potassium current, IK,f, in guinea-pig inner hair cells is regulated by protein kinase A. Neurosci Lett. 1999;263:145–148. doi: 10.1016/s0304-3940(99)00128-7. [DOI] [PubMed] [Google Scholar]
- Jahnke K. The fine structure of freeze-fractured intercellular junctions in the guinea pig inner ear. Acta Otolaryngol (Stockh) 1975;336(Suppl.):1–40. [PubMed] [Google Scholar]
- Johnsen T, Jorgensen MB, Johnsen S. Mondini cochlea in Pendred's syndrome. A histological study. Acta Otolaryngol. 1986;102:239–247. doi: 10.3109/00016488609108673. [DOI] [PubMed] [Google Scholar]
- Johnstone BM, Patuzzi R, Syka J, Sykova E. Stimulus-related potassium changes in the organ of Corti of guinea-pig. J Physiol. 1989;408:77–92. doi: 10.1113/jphysiol.1989.sp017448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karet FE, Finberg KE, Nelson RD, Nayir A, Mocan H, Sanjad SA, Rodriguez-Soriano J, Santos F, Cremers CW, Di Pietro A, Hoffbrand BI, Winiarski J, Bakkaloglu A, Ozen S, Dusunsel R, Goodyer P, Hulton SA, Wu DK, Skvorak AB, Morton CC, Cunningham MJ, Jha V, Lifton RP. Mutations in the gene encoding B1 subunit of H+-ATPase cause renal tubular acidosis with sensorineural deafness. Nat Genet. 1999;21:84–90. doi: 10.1038/5022. [DOI] [PubMed] [Google Scholar]
- Kikuchi T, Kimura RS, Paul DL, Adams JC. Gap junctions in the rat cochlea: immunohistochemical and ultrastructural analysis. Anat Embryol (Berl) 1995;191:101–118. doi: 10.1007/BF00186783. [DOI] [PubMed] [Google Scholar]
- Kikuchi T, Kimura RS, Paul DL, Takasaka T, Adams JC. Gap junction systems in the mammalian cochlea. Brain Res Brain Res Rev. 2000;32:163–166. doi: 10.1016/s0165-0173(99)00076-4. [DOI] [PubMed] [Google Scholar]
- King M, Housley GD, Raybould NP, Greenwood D, Salih SG. Expression of ATP-gated ion channels by Reissner's membrane epithelial cells. Neuroreport. 1998;9:2467–2474. doi: 10.1097/00001756-199808030-00008. [DOI] [PubMed] [Google Scholar]
- Konishi T, Hamrick PE, Walsh PJ. Ion transport in guinea pig cochlea. I. Potassium and sodium transport. Acta Otolaryngol (Stockh) 1978;86:22–34. doi: 10.3109/00016487809124717. [DOI] [PubMed] [Google Scholar]
- Kros CJ. Physiology of mammalian hair cells. In: Dallos P, Popper AN, Fay R, editors. Springer Handbook of Auditory Research: the Cochlea. New York: Springer; 1996. pp. 319–385. [Google Scholar]
- Kusakari J, Kambayashi J, Ise I, Kawamoto K. Reduction of the endocochlear potential by the new ‘loop’ diuretic, bumetanide. Acta Otolaryngol (Stockh) 1978;86:336–341. doi: 10.3109/00016487809107512. [DOI] [PubMed] [Google Scholar]
- Lautermann J, ten Cate WJ, Altenhoff P, Grummer R, Traub O, Frank H, Jahnke K, Winterhager E. Expression of the gap-junction connexins 26 and 30 in the rat cochlea. Cell Tissue Res. 1998;294:415–420. doi: 10.1007/s004410051192. [DOI] [PubMed] [Google Scholar]
- Lee JH, Chiba T, Marcus DC. P2X2 receptor mediates stimulation of parasensory cation absorption by cochlear outer sulcus cells and vestibular transitional cells. J Neurosci. 2001;21:9168–9174. doi: 10.1523/JNEUROSCI.21-23-09168.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JH, Marcus DC. Endolymphatic sodium homeostasis by Reissner's membrane. Neuroscience. 2003;119:3–8. doi: 10.1016/s0306-4522(03)00104-0. [DOI] [PubMed] [Google Scholar]
- Lee MP, Ravenel JD, Hu RJ, Lustig LR, Tomaselli G, Berger RD, Brandenburg SA, Litzi TJ, Bunton TE, Limb C, Francis H, Gorelikow M, Gu H, Washington K, Argani P, Goldenring JR, Coffey RJ, Feinberg AP. Targeted disruption of the Kvlqt1 gene causes deafness and gastric hyperplasia in mice. J Clin Invest. 2000;106:1447–1455. doi: 10.1172/JCI10897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letts VA, Valenzuela A, Dunbar C, Zheng QY, Johnson KR, Frankel WN. A new spontaneous mouse mutation in the Kcne1 gene. Mamm Genome. 2000;11:831–835. doi: 10.1007/s003350010178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim DJ, Karabinas C, Trune DR. Histochemical localization of carbonic anhydrase in the inner ear. Am J Otolaryngol. 1983;4:33–42. doi: 10.1016/s0196-0709(83)80005-2. [DOI] [PubMed] [Google Scholar]
- Liu XZ, Xia XJ, Adams J, Chen ZY, Welch KO, Tekin M, Ouyang XM, Kristiansen A, Pandya A, Balkany T, Arnos KS, Nance WE. Mutations in GJA1 (connexin 43) are associated with non-syndromic autosomal recessive deafness. Hum Mol Genet. 2001;10:2945–2951. doi: 10.1093/hmg/10.25.2945. [DOI] [PubMed] [Google Scholar]
- Luxon LM, Cohen M, Coffey RA, Phelps PD, Britton KE, Jan H, Trembath RC, Reardon W. Neuro-otological findings in Pendred syndrome. Int J Audiol. 2003;42:82–88. doi: 10.3109/14992020309078339. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Chiba T. K+ and Na+ absorption by outer sulcus epithelial cells. Hear Res. 1999;134:48–56. doi: 10.1016/s0378-5955(99)00074-x. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Liu J, Lee JH, Scherer EQ, Scofield MA, Wangemann P. Apical membrane P2Y4 purinergic receptor controls K+ secretion by strial marginal cell epithelium. Cell Commun Signal. 2005;3:1–8. doi: 10.1186/1478-811X-3-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcus NY, Marcus DC. Potassium secretion by nonsensory region of gerbil utricle in vitro. Am J Physiol. 1987;253:F613–F621. doi: 10.1152/ajprenal.1987.253.4.F613. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Marcus NY, Greger R. Sidedness of action of loop diuretics and ouabain on nonsensory cells of utricle: a micro-Ussing chamber for inner ear tissues. Hear Res. 1987;30:55–64. doi: 10.1016/0378-5955(87)90183-3. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Rokugo M, Thalmann R. Effects of barium and ion substitutions in artificial blood on endocochlear potential. Hear Res. 1985;17:79–86. doi: 10.1016/0378-5955(85)90133-9. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Shen Z. Slowly activating, voltage-dependent K+ conductance is apical pathway for K+ secretion in vestibular dark cells. Am J Physiol. 1994;267:C857–C864. doi: 10.1152/ajpcell.1994.267.3.C857. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Thalmann R, Marcus NY. Respiratory rate and ATP content of stria vascularis of guinea pig in vitro. Laryngoscope. 1978;88:1825–1835. doi: 10.1288/00005537-197811000-00011. [DOI] [PubMed] [Google Scholar]
- Marcus DC, Wu T, Wangemann P, Kofuji P. KCNJ10 (Kir4.1) potassium channel knockout abolishes endocochlear potential. Am J Physiol Cell Physiol. 2002;282:C403–C407. doi: 10.1152/ajpcell.00312.2001. [DOI] [PubMed] [Google Scholar]
- Matsunami T, Suzuki T, Hisa Y, Takata K, Takamatsu T, Oyamada M. Gap junctions mediate glucose transport between GLUT1-positive and -negative cells in the spiral limbus of the rat cochlea. Cell Commun Adhes. 2006;13:93–102. doi: 10.1080/15419060600631805. [DOI] [PubMed] [Google Scholar]
- McGuirt JP, Schulte BA. Distribution of immunoreactive α and β subunit isoforms of Na,K-ATPase in the gerbil inner ear. J Histochem Cytochem. 1994;42:843–853. doi: 10.1177/42.7.8014467. [DOI] [PubMed] [Google Scholar]
- Mizuta K, Adachi M, Iwasa KH. Ultrastructural localization of the Na-K-Cl-cotransporter in the lateral wall of the rabbit cochlear duct. Hear Res. 1997;106:154–162. doi: 10.1016/s0378-5955(97)00010-5. [DOI] [PubMed] [Google Scholar]
- Munoz DJ, Kendrick IS, Rassam M, Thorne PR. Vesicular storage of adenosine triphosphate in the guinea-pig cochlear lateral wall and concentrations of ATP in the endolymph during sound exposure and hypoxia. Acta Otolaryngol. 2001;121:10–15. doi: 10.1080/000164801300006209. [DOI] [PubMed] [Google Scholar]
- Nie L, Gratton MA, Mu KJ, Dinglasan JN, Feng W, Yamoah EN. Expression and functional phenotype of mouse ERG K+ channels in the inner ear: potential role in K+ regulation in the inner ear. J Neurosci. 2005;25:8671–8679. doi: 10.1523/JNEUROSCI.1422-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ninoyu O, Meyer zum Gottesberge AM. Changes in Ca++ activity and DC potential in experimentally induced endolymphatic hydrops. Arch Otorhinolaryngol. 1986;243:106–107. doi: 10.1007/BF00453759. [DOI] [PubMed] [Google Scholar]
- Ohmori H. Mechano-electrical transduction currents in isolated vestibular hair cells of the chick. J Physiol. 1985;359:189–217. doi: 10.1113/jphysiol.1985.sp015581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamura HO, Sugai N, Suzuki K, Ohtani I. Enzyme-histochemical localization of carbonic anhydrase in the inner ear of the guinea pig and several improvements of the technique. Histochem Cell Biol. 1996;106:425–430. doi: 10.1007/BF02473302. [DOI] [PubMed] [Google Scholar]
- Petit C, Levilliers J, Hardelin JP. Molecular genetics of hearing loss. Annu Rev Genet. 2001;35:589–645. doi: 10.1146/annurev.genet.35.102401.091224. [DOI] [PubMed] [Google Scholar]
- Reardon W, OMahoney CF, Trembath R, Jan H, Phelps PD. Enlarged vestibular aqueduct: a radiological marker of pendred syndrome, and mutation of the PDS gene. Q J Med. 2000;93:99–104. doi: 10.1093/qjmed/93.2.99. [DOI] [PubMed] [Google Scholar]
- Ricci AJ, Fettiplace R. Calcium permeation of the turtle hair cell mechanotransducer channel and its relation to the composition of endolymph. J Physiol. 1998;506:159–173. doi: 10.1111/j.1469-7793.1998.159bx.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rybak LP, Whitworth CA. Ototoxicity: therapeutic opportunities. Drug Discov Today. 2005;10:1313–1321. doi: 10.1016/S1359-6446(05)03552-X. [DOI] [PubMed] [Google Scholar]
- Sage CL, Marcus DC. Immunolocalization of ClC-K chloride channel in strial marginal cells and vestibular dark cells. Hear Res. 2001;160:1–9. doi: 10.1016/s0378-5955(01)00308-2. [DOI] [PubMed] [Google Scholar]
- Salt AN. Regulation of endolymphatic fluid volume. Ann N Y Acad Sci. 2001;942:306–312. doi: 10.1111/j.1749-6632.2001.tb03755.x. [DOI] [PubMed] [Google Scholar]
- Salt AN, Konishi T. Effects of noise on cochlear potentials and endolymph potassium concentration recorded with potassium-selective electrodes. Hear Res. 1979;1:343–363. doi: 10.1016/0378-5955(79)90005-4. [DOI] [PubMed] [Google Scholar]
- Salt AN, Melichar I, Thalmann R. Mechanisms of endocochlear potential generation by stria vascularis. Laryngoscope. 1987;97:984–991. [PubMed] [Google Scholar]
- Salt AN, Ohyama K. Accumulation of potassium in scala vestibuli perilymph of the mammalian cochlea. Ann Otol Rhinol Laryngol. 1993;102:64–70. doi: 10.1177/000348949310200112. [DOI] [PubMed] [Google Scholar]
- Schimanski S, Scofield MA, Wangemann P. Functional β2-adrenergic receptors are present in non-strial tissues of the lateral wall in the gerbil cochlea. Audiol Neurootol. 2001;6:124–136. doi: 10.1159/000046819. [DOI] [PubMed] [Google Scholar]
- Schlingmann KP, Konrad M, Jeck N, Waldegger P, Reinalter SC, Holder M, Seyberth HW, Waldegger S. Salt wasting and deafness resulting from mutations in two chloride channels. N Engl J Med. 2004;350:1314–1319. doi: 10.1056/NEJMoa032843. [DOI] [PubMed] [Google Scholar]
- Schulte BA, Adams JC. Distribution of immunoreactive Na+,K+-ATPase in gerbil cochlea. J Histochem Cytochem. 1989;37:127–134. doi: 10.1177/37.2.2536055. [DOI] [PubMed] [Google Scholar]
- Schulze-Bahr E, Wang Q, Wedekind H, Haverkamp W, Chen Q, Sun Y, Rubie C, Hordt M, Towbin JA, Borggrefe M, Assmann G, Qu X, Somberg JC, Breithardt G, Oberti C, Funke H. KCNE1 mutations cause jervell and Lange–Nielsen syndrome. Nat Genet. 1997;17:267–268. doi: 10.1038/ng1197-267. [DOI] [PubMed] [Google Scholar]
- Shen Z, Marcus DC, Sunose H, Chiba T, Wangemann P. IsK channel in strial marginal cell: Voltage-dependence, ion selectivity, inhibition by 293B and sensitivity to clofilium. Audit Neurosci. 1997;3:215–230. [PMC free article] [PubMed] [Google Scholar]
- Spicer SS, Schulte BA. Differentiation of inner ear fibrocytes according to their ion transport related activity. Hear Res. 1991;56:53–64. doi: 10.1016/0378-5955(91)90153-z. [DOI] [PubMed] [Google Scholar]
- Spicer SS, Schulte BA. The fine structure of spiral ligament cells relates to ion return to the stria and varies with place-frequency. Hear Res. 1996;100:80–100. doi: 10.1016/0378-5955(96)00106-2. [DOI] [PubMed] [Google Scholar]
- Stankovic KM, Brown D, Alper SL, Adams JC. Localization of pH regulating proteins H+ATPase and Cl−/HCO3− exchanger in the guinea pig inner ear. Hear Res. 1997;114:21–34. doi: 10.1016/s0378-5955(97)00072-5. [DOI] [PubMed] [Google Scholar]
- Sterkers O, Saumon G, Tran Ba Huy P, Ferrary E, Amiel C. Electrochemical heterogeneity of the cochlear endolymph: effect of acetazolamide. Am J Physiol. 1984;246:F47–F53. doi: 10.1152/ajprenal.1984.246.1.F47. [DOI] [PubMed] [Google Scholar]
- Stover EH, Borthwick KJ, Bavalia C, Eady N, Fritz DM, Rungroj N, Giersch AB, Morton CC, Axon PR, Akil I, Al Sabban EA, Baguley DM, Bianca S, Bakkaloglu A, Bircan Z, Chauveau D, Clermont MJ, Guala A, Hulton SA, Kroes H, Li VG, Mir S, Mocan H, Nayir A, Ozen S, Rodriguez SJ, Sanjad SA, Tasic V, Taylor CM, Topaloglu R, Smith AN, Karet FE. Novel ATP6V1B1 and ATP6V0A4 mutations in autosomal recessive distal renal tubular acidosis with new evidence for hearing loss. J Med Genet. 2002;39:796–803. doi: 10.1136/jmg.39.11.796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Street VA, McKee-Johnson JW, Fonseca RC, Tempel BL, Noben-Trauth K. Mutations in a plasma membrane Ca2+-ATPase gene cause deafness in deafwaddler mice. Nat Genet. 1998;19:390–394. doi: 10.1038/1284. [DOI] [PubMed] [Google Scholar]
- Sun J, Ahmad S, Chen S, Tang W, Zhang Y, Chen P, Lin X. Cochlear gap junctions coassembled from Cx26 and 30 show faster intercellular Ca2+ signaling than homomeric counterparts. Am J Physiol Cell Physiol. 2005;288:C613–C623. doi: 10.1152/ajpcell.00341.2004. [DOI] [PubMed] [Google Scholar]
- Sunose H, Liu J, Marcus DC. cAMP increases K+ secretion via activation of apical IsK/KvLQT1 channels in strial marginal cells. Hear Res. 1997;114:107–116. doi: 10.1016/s0378-5955(97)00152-4. [DOI] [PubMed] [Google Scholar]
- Takeuchi S, Ando M. Dye-coupling of melanocytes with endothelial cells and pericytes in the cochlea of gerbils. Cell Tissue Res. 1998a;293:271–275. doi: 10.1007/s004410051118. [DOI] [PubMed] [Google Scholar]
- Takeuchi S, Ando M. Inwardly rectifying K+ currents in intermediate cells in the cochlea of gerbils: a possible contribution to the endocochlear potential. Neurosci Lett. 1998b;247:175–178. doi: 10.1016/s0304-3940(98)00318-8. [DOI] [PubMed] [Google Scholar]
- Takeuchi S, Ando M, Kakigi A. Mechanism generating endocochlear potential: role played by intermediate cells in stria vascularis. Biophys J. 2000;79:2572–2582. doi: 10.1016/S0006-3495(00)76497-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeuchi S, Irimajiri A. Maxi-K+ channel in plasma membrane of basal cells dissociated from the stria vascularis of gerbils. Hear Res. 1996;95:18–25. doi: 10.1016/0378-5955(96)00016-0. [DOI] [PubMed] [Google Scholar]
- Tanaka Y, Asanuma A, Yanagisawa K. Potentials of outer hair cells and their membrane properties in cationic environments. Hear Res. 1980;2:431–438. doi: 10.1016/0378-5955(80)90079-9. [DOI] [PubMed] [Google Scholar]
- Tanaka F, Whitworth CA, Rybak LP. Round window pH manipulation alters the ototoxicity of systemic cisplatin. Hear Res. 2004;187:44–50. doi: 10.1016/s0378-5955(03)00330-7. [DOI] [PubMed] [Google Scholar]
- Tang W, Zhang Y, Chang Q, Ahmad S, Dahlke I, Yi H, Chen P, Paul DL, Lin X. Connexin29 is highly expressed in cochlear Schwann cells, and it is required for the normal development and function of the auditory nerve of mice. J Neurosci. 2006;26:1991–1999. doi: 10.1523/JNEUROSCI.5055-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teubner B, Michel V, Pesch J, Lautermann J, Cohen-Salmon M, Sohl G, Jahnke K, Winterhager E, Herberhold C, Hardelin JP, Petit C, Willecke K. Connexin30 (Gjb6)-deficiency causes severe hearing impairment and lack of endocochlear potential. Hum Mol Genet. 2003;12:13–21. doi: 10.1093/hmg/ddg001. [DOI] [PubMed] [Google Scholar]
- Thorne PR, Munoz DJ, Housley GD. Purinergic modulation of cochlear partition resistance and its effect on the endocochlear potential in the guinea pig. J Assoc Res Otolaryngol. 2004;5:58–65. doi: 10.1007/s10162-003-4003-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vetter DE, Mann JR, Wangemann P, Liu Z, McLaughlin KJ, Lesage F, Marcus DC, Lazdunski M, Heinemann SF, Barhanin J. Inner ear defects induced by null mutation of isk gene. Neuron. 1996;17:1251–1264. doi: 10.1016/s0896-6273(00)80255-x. [DOI] [PubMed] [Google Scholar]
- von Békésy G. DC potentials and energy balance of the cochlear partition. J Acoust Soc Am. 1950;22:576–582. [Google Scholar]
- Wang Z, Li H, Moss AJ, Robinson J, Zareba W, Knilans T, Bowles NE, Towbin JA. Compound heterozygous mutations in KvLQT1 cause Jervell and Lange–Nielsen syndrome. Mol Genet Metab. 2002;75:308–316. doi: 10.1016/S1096-7192(02)00007-0. [DOI] [PubMed] [Google Scholar]
- Wangemann P. K+ cycling and the endocochlear potential. Hear Res. 2002;165:1–9. doi: 10.1016/s0378-5955(02)00279-4. [DOI] [PubMed] [Google Scholar]
- Wangemann P, Itza EM, Albrecht B, Wu T, Jabba SV, Maganti RJ, Lee JH, Everett LA, Wall SM, Royaux IE, Green ED, Marcus DC. Loss of KCNJ10 protein expression abolishes endocochlear potential and causes deafness in Pendred syndrome mouse model. BMC Medicine. 2004;2:30. doi: 10.1186/1741-7015-2-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wangemann P, Liu J, Marcus DC. Ion transport mechanisms responsible for K+ secretion and the transepithelial voltage across marginal cells of stria vascularis in vitro. Hear Res. 1995;84:19–29. doi: 10.1016/0378-5955(95)00009-s. [DOI] [PubMed] [Google Scholar]
- Wangemann P, Liu J, Shimozono M, Schimanski S, Scofield MA. K+ secretion in strial marginal cells is stimulated via β1-adrenergic receptors but not via β2-adrenergic or vasopressin receptors. J Membr Biol. 2000;175:191–202. doi: 10.1007/s00232001067. [DOI] [PubMed] [Google Scholar]
- Wangemann P, Marcus DC. K+-induced swelling of vestibular dark cells is dependent on Na+ and Cl− and inhibited by piretanide. Pflugers Arch. 1990;416:262–269. doi: 10.1007/BF00392062. [DOI] [PubMed] [Google Scholar]
- Wangemann P, Schacht J. Homeostasic mechanisms in the cochlea. In: Dallos P, Popper AN, Fay R, editors. Springer Handbook of Auditory Research: The Cochlea. New York: Springer; 1996. pp. 130–185. [Google Scholar]
- Wangemann P, Shen Z, Liu J. K+-induced stimulation of K+ secretion involves activation of the IsK channel in vestibular dark cells. Hear Res. 1996;100:201–210. doi: 10.1016/0378-5955(96)00127-x. [DOI] [PubMed] [Google Scholar]
- Wood JD, Muchinsky SJ, Filoteo AG, Penniston JT, Tempel BL. Low endolymph calcium concentrations in deafwaddler2J mice suggest that PMCA2 contributes to endolymph calcium maintenance. J Assoc Res Otolaryngol. 2004;5:99–110. doi: 10.1007/s10162-003-4022-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia A, Katori Y, Oshima T, Watanabe K, Kikuchi T, Ikeda K. Expression of connexin 30 in the developing mouse cochlea. Brain Res. 2001;898:364–367. doi: 10.1016/s0006-8993(01)02216-8. [DOI] [PubMed] [Google Scholar]
- Xia A, Kikuchi T, Hozawa K, Katori Y, Takasaka T. Expression of connexin 26 and Na,K-ATPase in the developing mouse cochlear lateral wall: functional implications. Brain Res. 1999;846:106–111. doi: 10.1016/s0006-8993(99)01996-4. [DOI] [PubMed] [Google Scholar]
- Yamakawa K. Über pathologische Veränderungen bei einem Menière-Kranken. J Otolaryngol Soc Jpn. 1938;44:181–182. [Google Scholar]
- Yamauchi D, Raveendran NN, Pondugula SR, Kampalli SB, Sanneman JD, Harbidge DG, Marcus DC. Vitamin D upregulates expression of ECaC1 mRNA in semicircular canal. Biochem Biophys Res Commun. 2005;331:1353–1357. doi: 10.1016/j.bbrc.2005.04.053. [DOI] [PubMed] [Google Scholar]
- Zelante L, Gasparini P, Estivill X, Melchionda S, D'Agruma L, Govea N, Mila M, Monica MD, Lutfi J, Shohat M, Mansfield E, Delgrosso K, Rappaport E, Surrey S, Fortina P. Connexin26 mutations associated with the most common form of non-syndromic neurosensory autosomal recessive deafness (DFNB1) in Mediterraneans. Hum Mol Genet. 1997;6:1605–1609. doi: 10.1093/hmg/6.9.1605. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Tang W, Ahmad S, Sipp JA, Chen P, Lin X. Gap junction-mediated intercellular biochemical coupling in cochlear supporting cells is required for normal cochlear functions. Proc Natl Acad Sci U S A. 2005;102:15201–15206. doi: 10.1073/pnas.0501859102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao HB. Connexin26 is responsible for anionic molecule permeability in the cochlea for intercellular signalling and metabolic communications. Eur J Neurosci. 2005;21:1859–1868. doi: 10.1111/j.1460-9568.2005.04031.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao HB, Yu N, Fleming CR. Gap junctional hemichannel-mediated ATP release and hearing controls in the inner ear. Proc Natl Acad Sci U S A. 2005;102:18724–18729. doi: 10.1073/pnas.0506481102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zidanic M, Brownell WE. Fine structure of the intracochlear potential field. I. The silent current. Biophys J. 1990;57:1253–1268. doi: 10.1016/S0006-3495(90)82644-8. [DOI] [PMC free article] [PubMed] [Google Scholar]