Abstract
After single-lung transplantation (SLT) for emphysema, a hyperinflated (native) lung operates in parallel with a normal (transplanted) lung. The interpulmonary distribution of the changes in pleural pressure (ΔPpl) during breathing, however, is unknown. To approach the problem, two endotracheal tubes were inserted in the right and left main stem bronchi of anaesthetized dogs, one lung was passively inflated, and the values of inspiratory ΔPpl over the two lungs were assessed by measuring the changes in airway opening pressure (ΔPao) in the two tubes during occluded breaths. With single-lung inflation, ΔPao decreased in both lungs, but the decrease in the inflated lung was invariably larger than in the non-inflated lung; when transrespiratory pressure in the inflated lung was set at 30 cmH2O, ΔPao in this lung was 27.7 ± 2.0% of the value of functional residual capacity (FRC), whereas ΔPao in the non-inflated lung was 74.4 ± 4.5% (P < 0.001). This difference was abolished after the ventral mediastinal pleura was severed. The ribs in both hemithoraces were displaced cranially with inflation, such that the displacement in the contralateral hemithorax was 75% of that in the ipsilateral hemithorax, and parasternal intercostal activity remained unchanged. These observations suggest that in patients with SLT for emphysema (1) the inspiratory ΔPpl over the transplanted lung are greater than those over the native lung and (2) this difference results primarily from the greater pressure-generating ability of the inspiratory muscles, in particular the diaphragm, on the transplanted side.
Single-lung transplantation (SLT) is now a well-established treatment option for patients with advanced emphysema (Glanville & Estenne, 2003; Trulock et al. 2004); however, the functional implications of having in the pleural cavity a native (emphysematous) lung and a transplanted (normal) lung (i.e. two lungs with different sizes and different elastic properties) are not fully understood. The interpulmonary distribution of the change in pleural pressure (ΔPpl) during inspiration in particular is unknown. However, the compliance of the native lung is 2- to 3-fold greater than that of the transplanted lung. Therefore, if such patients had an equal ΔPpl over the two lungs as healthy individuals do, the native lung would expand about 2- to 3-fold more than the transplanted lung. Computed tomography studies of the volumes of the individual lungs in 10 patients with SLT for emphysema have shown, however, that the inspiratory capacity of the native lung is only 50% greater than that of the transplanted lung (Estenne et al. 1999). Studies of regional ventilation by radioisotope techniques have also shown that expansion of the native lung during resting breathing is considerably smaller than that of the transplanted lung (Gibbons et al. 1991; Chacon et al. 1998). Therefore the hypothesis must be that the mean ΔPpl over the native lung is smaller than that over the transplanted lung.
The present studies were undertaken to test this hypothesis. Single-lung inflation was thus performed in anaesthetized dogs to simulate unilateral emphysema, and the ΔPpl over the inflated and non-inflated lungs was assessed by measuring the changes in airway opening pressure (ΔPao) in the two lungs during occluded breaths. The results indicate that despite the equal activation of the inspiratory muscles on the two sides of the chest, ΔPpl over the inflated lung is substantially smaller.
Methods
The studies were carried out on 12 adult cross-breed dogs (12–31 kg) anaesthetized with pentobarbital sodium (initial dose, 30 mg kg−1i.v.), as approved by the Animal Ethics and Welfare Committee of the Brussels School of Medicine. The animals were placed in the supine position and intubated with a cuffed endotracheal tube, and a venous cannula was inserted in the forelimb to give maintenance doses of anaesthetic. The rib cage and intercostal muscles were then exposed on both sides of the chest from the first to the tenth rib by reflection of the skin and the superficial muscle layers, and the vagi were isolated through a midline incision of the neck, infiltrated with 2% lignocaine (lidocaine) and sectioned. A tracheostomy was also performed, and two endotracheal tubes (no. 5–7) were inserted in the right and left main stem bronchi. The position of the tubes was adjusted under endoscopic guidance to ensure patency of all lobar bronchi. The two tubes were then tethered to the tracheal rings above and below the site of tracheostomy to avoid any inadvertent displacement later.
Measurements
ΔPao in each lung was measured with a differential pressure transducer (Validyne Corp., Northridge, CA, USA) connected to a side port of the endobronchial tube, and the electromyograms of the parasternal and external intercostal muscles in both the right and the left hemithorax were recorded with pairs of silver hook electrodes spaced 3–4 mm apart. Each electrode pair was placed in parallel fibres and inserted in the muscle area receiving the greatest neural inspiratory drive. Implantation of the parasternal intercostal electrodes therefore was made in the third interspace in the muscle bundles situated near the sternum (De Troyer & Legrand, 1995; Legrand et al. 1996, 2001), and the external intercostal electrodes were implanted in the dorsal portion of the second interspace, immediately ventral to the rib angle (Kirkwood et al. 1982; Greer & Martin, 1990; Legrand & De Troyer, 1999). The four electromyographic (EMG) signals were processed with amplifiers (model 830/1, CWE, Ardmore, PA, USA), band-pass filtered below 100 and above 2000 Hz, and rectified before their passage through leaky integrators with a time constant of 0.2 s.
Protocol
The animals were allowed to recover for 30 min after instrumentation, after which Pao in the right and left lungs and intercostal EMG activity were recorded. The animals were spontaneously breathing throughout. Every 10–15 breaths, however, a syringe was connected either to the right or to the left endobronchial tube and the volume of the corresponding lung was increased above FRC, at which time the two endobronchial tubes were occluded. The inflation, which involved the right lung in seven animals and the left lung in five animals, was always performed during the expiratory pause and the occlusion was maintained for a single inspiratory effort. Also, at least 15–20 occluded breaths over a wide range of lung volumes, taken in random order, were recorded in each animal; four to six occluded breaths with both lungs at FRC were also obtained. Thus, we could assess the effects of progressive single-lung inflation on inspiratory muscle function without any significant change in chemical respiratory drive.
The measurements indicated that the ΔPao in the inflated lung was different from the ΔPao in the non-inflated (control) lung (see Results). After the initial procedure was completed, two additional protocols were followed.
First, although the ΔPpl was not measured in our animals, such differences in ΔPao strongly suggested that the mediastinum sustained significant transmural pressure. In three animals therefore the sternum was opened by a midline incision, and the ventral mediastinal pleura connecting the dorsal aspect of the sternum to the pericardium and separating the left from the right pleural cavity was excised. The animals were ventilated mechanically (Harvard pump, Chicago, IL, USA) throughout the procedure. After the sternum was closely sutured and any residual pneumothorax was evacuated, the animals were returned to spontaneous breathing and the procedure of single-lung inflation was repeated.
Second, in six animals, we evaluated the role of the rib cage in causing the observed interpulmonary differences in ΔPao. In each animal, a hook was thus screwed into the fourth, fifth or sixth rib in the right midaxillary line and connected, through a long inextensible thread, to a linear displacement transducer (Schaevitz Engineering, Pennsauken, NJ, USA) to measure the craniocaudal (axial) rib displacement, as previously described (De Troyer, 1992; De Troyer & Leduc, 2004). After calibration of the transducer, the animal was made apnoeic by mechanical hyperventilation, and the right lung was inflated twice by 100-ml increments to establish the passive relationship between rib motion and transrespiratory pressure. The left lung was subsequently inflated in a similar manner, after which the two lungs were inflated simultaneously.
The animals appeared to remain at a satisfactory depth of anaesthesia throughout. They did not react to painful stimuli and made no spontaneous movements other than respiratory movements both during surgery and during the measurements. Also, they had no pupillary light reflex and no corneal reflex, thus indicating a deep level of anaesthesia. Rectal temperature was kept constant between 36 and 38°C with infrared lamps. At the end of the experiment, the animals were given an overdose (30–40 mg kg−1i.v.) of anaesthetic.
Data analysis
Phasic inspiratory electrical activity in the parasternal and external intercostal muscles during each occluded breath in each animal was first quantified by measuring the peak height of the integrated EMG signals in arbitrary units. To allow comparison between the muscles in the inflated and non-inflated hemithoraces in the different animals, EMG activity in each muscle was subsequently expressed as a percentage of the activity measured during occlusion at FRC (control). Also, the inspiratory ΔPao in the inflated and non-inflated lung were measured relative to the onset of the parasternal inspiratory burst. Consequently, the ΔPao values that were considered in the calculations resulted exclusively from the contraction of inspiratory muscles and were not corrupted by the relaxation of the expiratory muscles at the end of expiration (De Troyer & Ninane, 1986). The ΔPao values in both lungs and the inspiratory EMG activities measured during the occluded breaths at the different lung volumes were then plotted against the value of Pao in the inflated lung before inspiration (i.e. the precontractile transrespiratory pressure). The relationships were fitted by quadratic regression equations, and pressures and EMG activities at fixed transrespiratory pressures at 5.0 cmH2O increments were determined from these equations by interpolation. The axial motion of the ribs during passive inflation of the ipsilateral lung, of the contralateral lung and of the two lungs simultaneously was analysed similarly.
Data were averaged over the animal group, and they are presented as means ± s.e.m. Statistical assessments of the effects of lung volume on the ΔPao values in the inflated and non-inflated lung, on the inspiratory EMG activity in the inflated and non-inflated hemithorax, and on rib displacement during passive inflation were made by analysis of variance (ANOVA) with repeated measures. Multiple comparison testing of the mean values was performed, when appropriate, using Student–Newman–Keul's tests. Statistical comparisons between the inflated and non-inflated lung (hemithorax) were made similarly. The criterion for statistical significance was taken as P < 0.05.
Results
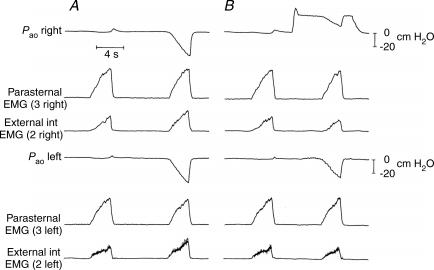
The records of Pao in the right and left lungs, and parasternal and external intercostal EMG activity obtained in a representative animal during an occluded breath with both lungs at FRC and during an occluded breath after inflation of the right lung are shown in Fig. 1. During the occluded breath at FRC (Fig. 1A), ΔPao in both lungs was −35.0 cmH2O; in agreement with previous studies (Sant'Ambrogio & Widdicombe, 1965; Shannon & Zechman, 1972; De Troyer, 1991, 1992), the inspiratory EMG activity recorded from the parasternal intercostals during this breath was unchanged relative to that recorded during the preceding unoccluded breath, whereas the activity recorded from the external intercostals was increased. In contrast, when the volume of the right lung before occlusion was increased to a transrespiratory pressure of 24.0 cmH2O (Fig. 1B), ΔPao in this lung was reduced to −13.0 cmH2O, and ΔPao in the left lung was reduced to −27.0 cmH2O. Also, parasternal intercostal EMG activity during this breath was similar to that recorded during the occluded breath at FRC, but external intercostal activity in both the left and the right hemithorax was reduced.
Figure 1. Effects of single-lung inflation on the pressure changes in the individual lungs in a representative animal.
A, shows the traces of airway opening pressure (Pao) in the right and the left lung during an unimpeded breath and an occluded breath with both lungs at resting end-expiration (FRC); the traces of EMG activity in the parasternal intercostals (3rd space) and external intercostals (2nd space) in the right and left hemithoraces are also shown. B, also shows an unimpeded breath at FRC followed by an occluded breath; between the two breaths, however, the right lung was passively inflated to a transrespiratory pressure of 24 cmH2O. Note that during the occluded breath after right lung inflation, the change in Pao (ΔPao) in the right lung is much smaller than that during the occluded breath at FRC; ΔPao in the left lung is greater than that in the right lung but smaller than that at FRC. Note also the decrease in external intercostal activity on both sides of the chest during the occluded breath after inflation.
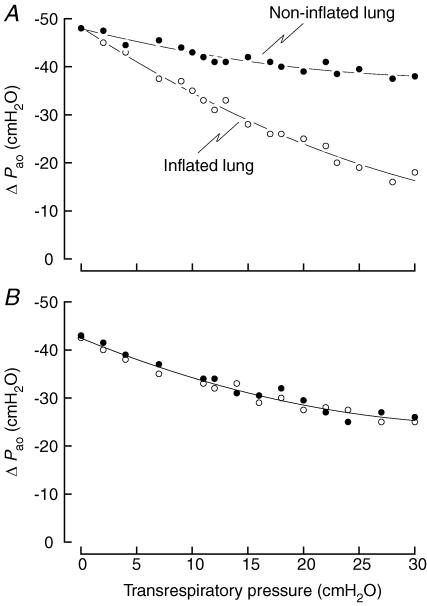
With single-lung inflation, ΔPao in the inflated lung decreased progressively in all animals, as shown in Fig. 2. Concomitantly, ΔPao in the non-inflated lung decreased in 11 of 12 animals and remained unchanged in one animal. Therefore for the group as a whole, as transrespiratory pressure in the inflated lung was increased from 0 to 30 cmH2O, ΔPao in this lung decreased from −31.6 ± 3.1 to −9.0 ± 1.1 cmH2O (P < 0.001), and ΔPao in the non-inflated lung decreased from −31.2 ± 3.1 to −22.4 ± 2.0 cmH2O (P < 0.001). Thus, at 30 cmH2O transrespiratory pressure, ΔPao in the inflated lung was 27.7 ± 2.0% of the control FRC value, whereas in the contralateral lung, ΔPao was 74.4 ± 4.5% of the control value.
Figure 2. Effect of gradual single-lung inflation on airway opening pressure in the individual lungs.
Values of ΔPao (mean ± s.e.m) in the inflated (○) and non-inflated (•) lungs obtained from 12 animals during single-lung inflation. The right lung was inflated in seven animals, and the left lung was inflated in five animals The values of transrespiratory pressure along the abscissa refer only to the inflated lung.
Single-lung inflation did not induce any consistent alteration in parasternal intercostal EMG activity in either the ipsilateral or the contralateral hemithorax. However, external intercostal activity in both hemithoraces decreased progressively, as shown in Fig. 3. As transrespiratory pressure in the inflated lung was increased to 30 cmH2O, activity in the inflated hemithorax thus decreased, on average, to 46.3 ± 5.3% of the value obtained during occlusion at FRC, and activity in the contralateral hemithorax decreased to 53.8 ± 7.4% of the FRC value (P < 0.001 for both). At no transrespiratory pressure, did the difference between the two hemithoraces reach the level of statistical significance.
Figure 3. Effect of single-lung inflation on external intercostal EMG activity in the two hemithoraces.
Values of external Intercostal EMG activity (mean ± s.e.m) in the Ipsilateral (○) and contralateral (•) hemithoraces obtained from 12 animals during single-lung inflation. These values are expressed as percentages of the activity recorded at FRC.
The role played by the decrease in external intercostal activity in causing the reduction in ΔPao during inflation was tested in four animals by sectioning bilaterally the external intercostal muscles from the costochondral junction to the spine in interspaces 1–7. The results of this procedure are shown in Fig. 4. When the right (two animals) or left (two animals) lung was inflated to 30 cmH2O with all the intercostal muscles intact (Fig. 4A), ΔPao in the inflated lung decreased to 27.6 ± 7.1% of the control FRC value, and EMG activity in the ipsilateral external intercostals decreased to 52.9 ± 13.3% of control. ΔPao in the non-inflated lung simultaneously decreased to 64.1 ± 6.9% of control, and activity in the contralateral external intercostals decreased to 62.9 ± 12.4%. Similar decreases in ΔPao, however, occurred after section of the muscles, such that ΔPao in the inflated and non-inflated lungs at 30 cmH2O was 29.3 ± 3.9% and 65.0 ± 5.3% of the FRC value, respectively (Fig. 4B).
Figure 4. Effects of external intercostal muscle section on airway opening pressure during single-lung inflation.
Values of ΔPao (mean ± s.e.m) in the inflated (○) and non-inflated (•) lungs obtained from four animals during gradual single-lung inflation before (A) and after (B) section of the external intercostal muscles in interspaces 1–7. Note that the interpulmonary difference in ΔPao was unchanged after section of the muscles.
On the other hand, severing the ventral mediastinal pleura had a dramatic effect on ΔPao during single-lung inflation, as shown by the results obtained in a representative animal in Fig. 5. Specifically, after the ventral mediastinal pleura was excised, the volume dependence of ΔPao in the non-inflated lung was identical to that in the inflated lung, including when transrespiratory pressure in this lung was set at 30 cmH2O.
Figure 5. Effect of mediastinal excision on airway opening pressure during single-lung inflation.
The data shown are the ΔPao values recorded in the left (○) and right (•) lungs during inflation of the left lung in a representative animal before (A) and after (B) excision of the ventral mediastinal pleura. Note that the difference in ΔPao between the two lungs is eliminated after excision.
Figure 6 compares the axial displacement of the ribs during passive inflation of the contralateral lung with that during passive inflation of the ipsilateral lung. Although the ribs moved cranially with inflation of either lung, their displacement during contralateral lung inflation was consistently smaller than that during ipsilateral lung inflation. Thus, with right lung inflation from 0 to 30 cmH2O transrespiratory pressure, the cranial rib displacement was 8.4 ± 0.9 mm, whereas with left lung inflation, it was only 6.4 ± 0.5 mm (P < 0.001). Rib displacement during bilateral lung inflation, however, was substantially larger (P < 0.001) and averaged 13.4 ± 1.3 mm at 30 cmH2O transrespiratory pressure.
Figure 6. Effect of single-lung inflation on rib cage expansion.
Values of cranial rib displacement (mean ± s.e.m) obtained from six animals during passive Inflation of the ipsilateral (○) and contralateral lung (•). The cranial rib displacement observed during bilateral lung inflation is shown for comparison ( ).
).
Discussion
Studies of the mechanics of the diaphragm in animals (Marshall, 1962; Kim et al. 1976; Minh et al. 1976; De Troyer et al. 2005) and in humans (Danon et al. 1979; Smith & Bellemare, 1987) have clearly established that the fall in Ppl obtained in response to a given stimulation of the phrenic nerves decreases rapidly as lung volume is passively increased above FRC and muscle length is decreased. Furthermore, increasing lung volume displaces the ribs cranially and makes them orientated more transversally relative to the sagittal midplane. As a result, the ability of the parasternal and external intercostals to produce a fall in Ppl is also reduced (De Troyer & Leduc, 2004; Leduc & De Troyer, 2006). It would be expected therefore that the pressure-generating ability of the inspiratory muscles in the ipsilateral hemithorax would be decreased with single-lung inflation. In addition, single-lung inflation causes, through the rise in Ppl over the ipsilateral lung, a shift of the mediastinum towards the opposite side as well as a caudal displacement of the ipsilateral hemidiaphragm leading to a rise in abdominal pressure. Therefore, pleural pressure over the contralateral, non-inflated lung should also increase, and indeed, both indirect and direct Ppl measurements in dogs by Hubmayr & Margulies (1992) have shown that during passive single-lung inflation, the pressure rise over the contralateral lung was, on average, 70% of the pressure rise over the inflated lung. Consequently, it would also be expected that single-lung inflation would induce expansion of the contralateral hemithorax and cause a reduction in the pressure-generating ability of the inspiratory muscles in this hemithorax.
In agreement with this prediction, single-lung inflation in our animals did adversely affect the ΔPao in both lungs (Figs 1 and 2). Of more importance, it affected the inflated and non-inflated lungs very differently. Thus, when one lung was inflated to a transrespiratory pressure of 30 cmH2O, ΔPao in this lung was only −9.0 cmH2O or 28% of the control FRC value, whereas ΔPao in the contralateral lung was −22.4 cmH2O or 73% of the control value. Because these ΔPao values were obtained with no airflow, they also represent the mean ΔPpl over the two lungs. The conclusion must be drawn therefore that with single-lung inflation in the dog, the inspiratory ΔPpl over the inflated lung is smaller than that over the contralateral lung.
The ventral mediastinal pleura in the dog consists of a thin transparent sheet of connective tissue (Evans & Christensen, 1979). Therefore, the finding that the inspiratory Ppl swings at 30 cmH2O transrespiratory pressure were −9.0 cmH2O over the inflated lung and −22.4 cmH2O over the contralateral lung may seem surprising, yet this difference is most probably an underestimate of the real pressure difference across the mediastinum. Thus, although pleural pressures in the right and left pleural spaces were not measured in this study, it is reasonable to assume that with a transrespiratory pressure of 30.0 cmH2O at end-expiration, the mean Ppl over the inflated lung was approximately +10.0 cmH2O. Consequently, at the peak of the occluded breath, Ppl over this lung was (+10–9) or +1.0 cmH2O. On the other hand, the observation of Hubmayr & Margulies (1992) would indicate that mean end-expiratory Ppl over the contralateral lung was only +7.0 cmH2O. Also, whereas these investigators occluded the airway of this lung before inflation, in the current study the airway was occluded after inflation was completed. In our animals at end-expiration, the rise in Ppl over the contralateral lung was probably smaller, for example +4.0 cmH2O, so at the peak of the occluded breath, Ppl over this lung was ∼ (+ 4–22) or −18.0 cmH2O. In other words, the pressure difference across the mediastinum was nearly 20.0 cmH2O, and when the ventral mediastinal pleura was severed, such that the two lungs were in a single pleural cavity, such a difference was no longer observed (Fig. 5). This is unequivocal evidence that the mediastinum in the dog, although delicate, is able to sustain significant pressure differences, and the question then arises as to how the inspiratory muscles can generate a substantially greater ΔPpl over the non-inflated than over the inflated lung.
Romaniuk et al. (1992) have previously shown that bilateral lung inflation in anaesthetized dogs commonly induces a decrease in the inspiratory EMG activity recorded from the rostral external intercostal muscles during occluded breaths as well as a small decrease in the inspiratory activity recorded from the parasternal intercostals. The authors also suggested that this decrease in EMG activity was the result of the decrease in muscle length and the consequent reduction in spindle afferent input. The possibility therefore existed that single-lung inflation would induce a smaller reduction in intercostal inspiratory activity in the contralateral than the ipsilateral hemithorax, leading to a greater ΔPpl over the non-inflated lung. This possibility, however, can be discarded on the basis of several observations. First, no alteration in parasternal intercostal activity in either the ipsilateral or the contralateral hemithorax was seen in our animals. Second, although most animals did show a progressive reduction in external intercostal activity, the magnitude of the reduction in the contralateral hemithorax was similar to that in the ipsilateral hemithorax (Figs 1 and 3). Third, and more important, the difference in ΔPpl between the two lungs remained unchanged after the external intercostals on both sides of the chest were severed (Fig. 4). In addition, as is the case for the parasternal intercostals (De Troyer, 1991), neural drive to the diaphragm is primarily governed by supraspinal control mechanisms, and measurements of motoneurone synchronization in cats by Vaughan & Kirkwood (1997) have shown that phrenic motoneurones and parasternal intercostal motoneurones receive common monosynaptic inputs. The lack of alteration in parasternal intercostal activity with single-lung inflation suggests therefore that diaphragmatic activity was also unaltered. Consequently, the interpulmonary differences in ΔPpl should be related to mechanical, rather than neural mechanisms.
As previously discussed, single-lung inflation produces a shift of the mediastinum towards the contralateral lung and induces a rise in Ppl over this lung, but this pressure rise is smaller than that over the inflated lung (Hubmayr & Margulies, 1992). On this basis, it could be argued that the rib elevation in the contralateral hemithorax would be smaller than that in the ipsilateral hemithorax and therefore that the pressure-generating ability of the inspiratory intercostals in the contralateral hemithorax would be better preserved. The comparison between the rib displacements observed during unilateral and bilateral lung inflation indicates, however, that this mechanism cannot be the main determinant of the interpulmonary differences in ΔPpl observed during occluded breaths. Thus, when one lung was inflated to 30 cmH2O, the cranial rib displacement in the ipsilateral hemithorax was similar to that seen during bilateral inflation to 15 cmH2O, and the rib displacement in the contralateral hemithorax corresponded to bilateral inflation to 10 cmH2O (Fig. 6). As we found recently (Leduc & De Troyer, 2006), the rib displacement associated with a bilateral lung inflation to 15 and 10 cmH2O reduces the pressure-generating ability of the canine parasternal intercostals to 43% and 54% of the value at FRC, respectively; similar figures were previously obtained during simultaneous activation of the parasternal and external intercostals (DiMarco et al. 1990). Also, in the dog, the pressure generated by these muscles at FRC is, on average, − 16 cmH2O (DiMarco et al. 1990; De Troyer, 2005), and the pressures developed by the muscles on the left and right sides of the sternum are known to be essentially additive (Cappello & De Troyer, 2000). Therefore, reasonable estimates for the pressures generated by the inspiratory intercostals in the ipsilateral and contralateral hemithorax after single-lung inflation to 30 cmH2O would be 0.43 × −16/2 cmH2O (i.e. −3.4 cmH2O) and 0.54 × −16/2 cmH2O (i.e. −4.3 cmH2O), respectively. The implication of this result is that the interpulmonary differences in ΔPpl observed in our animals are primarily determined by the action of the diaphragm.
By causing a rise in Ppl over the contralateral lung, single-lung inflation should both expand the contralateral rib cage and displace the contralateral hemidiaphragm caudally so as to shorten its muscle fibres. However, such a shortening should be opposed by the rise in abdominal pressure resulting from the caudal displacement of the ipsilateral hemidiaphragm. Moreover, the caudal displacement of the ipsilateral hemidiaphragm should also cause a shift and a tilt of the central tendon towards the inflated side and pull on the contralateral hemidiaphragm. As a result, it would be expected that single-lung inflation would produce a marked shortening of the ipsilateral hemidiaphragm but little or no change in length of the contralateral hemidiaphragm. The observation by Cassart et al. (1999) that SLT for emphysema elicits a prominent cranial displacement of the hemidiaphragm on the transplanted side is fully consistent with the idea that the ipsilateral hemidiaphragm in our animals was significantly shorter than the contralateral hemidiaphragm, and the graphical analysis displayed in Fig. 7 indicates that such a difference in length might induce a large difference in pressure-generating ability.
Figure 7. Graphical analysis of the force developed by the diaphragm during single-lung inflation.
The thin continuous lines are the force–length relationships for the maximally active and the passive diaphragm, the thick continuous line is the force–length relationship for the submaximally active diaphragm, and the dashed lines are the load curves describing the load imposed by the lung and chest wall on the two hemidiaphragms. Force is expressed as a fraction of maximum, and muscle length is expressed as a fraction of optimal length (Lo). The force generated by each hemidiaphragm during inspiration is given by the intersection of the load curve with the submaximally active length–tension curve (•). If after single-lung inflation, the ipsilateral hemidiaphragm (inflated) before contraction were shorter than the contralateral hemidiaphragm (non-inflated), it would generate less force during inspiration.
The thin continuous lines in this figure are the active and passive length–tension curves obtained from isolated diaphragmatic bundles (Kim et al. 1976; McCully & Faulkner, 1983; Farkas & Rochester, 1988); active force in the diaphragm, as for any skeletal muscle, decreases gradually as muscle length decreases, and it approaches zero when muscle length is ∼40% of the optimal force-producing length (Lo). The animals in this study, however, were breathing spontaneously, so activation of the diaphragm was submaximal, rather than maximal. Therefore, the active length–tension curve was shifted downwards (thick continuous line), such that force during isometric contraction at Lo would be reduced, for example to 0.4 of maximal force. As we have previously pointed out, however, the length of the diaphragm and the force developed by the muscle during contraction is also determined by the load imposed on the muscle by the lung and the chest wall (De Troyer et al. 2005). In fact, the particular values of length and force that eventually occur when the diaphragm contracts are given by the intersection of the active length–tension curve and the load curve, and the dashed lines in Fig. 7 represent the two load curves corresponding to the ipsilateral and contralateral hemidiaphragms during contraction. These load curves were assumed to be parallel and were established on the basis of two elements. First, because in supine dogs the relaxed diaphragm at FRC is close to Lo (Road et al. 1986; Farkas & Rochester, 1988), the load curve for the contralateral hemidiaphragm would intersect the curve corresponding to the passive diaphragm at Lo. Second, measurements of muscle length by sonomicrometry and radiography have shown that the canine diaphragm shortens by ∼10% of its relaxation length during inspiration at FRC (Easton et al. 1989; Sprung et al. 1989). At peak inspiration therefore muscle length in the contralateral hemidiaphragm was taken as 0.9 Lo, so the force developed by this muscle would be 0.33 of maximal force. On the other hand, if single-lung inflation caused a large shortening in the ipsilateral hemidiaphragm, for example to 0.75 Lo, the force developed by the muscle at peak inspiration would be only 0.18 of maximal force. In other words, the ratio of the force generated by the ipsilateral hemidiaphragm to the force generated by the contralateral hemidiaphragm would be 0.57, and this ratio would be decreased further if the active tension for the diaphragm in situ showed a steeper dependence on muscle length at short muscle lengths, as reported by Hubmayr et al. (1990) and Boriek et al. (1997).
The model investigated in this study differs from the situation in patients with SLT for emphysema in several respects. First, the animals had two lungs with similar elastic properties, whereas in patients, the inflated (native) lung is substantially more compliant than the non-inflated (transplanted) lung. Consequently, the elastic load imposed on the hemidiaphragm on the non-inflated side should be greater than that imposed on the hemidiaphragm on the inflated side. Even though the contribution of the lung to the total load on the diaphragm cannot be defined, the load curve for this hemidiaphragm (Fig. 7) might be less steep, and this would enhance the difference between the forces developed by the two hemidiaphragms. In addition, the mediastinum in humans is stiffer than in the dog, and it becomes even stiffer after thoracic surgery (Lin et al. 2004). On this basis, one would predict that patients with SLT for emphysema also have a larger ΔPpl over the non-inflated lung than over the inflated lung. Whereas inflation in the animals was produced acutely, however, emphysema develops slowly over many years and may induce remodelling in the diaphragm. Studies in emphysematous hamsters have shown in particular that chronic inflation causes, through the chronic muscle shortening, a loss of sarcomeres in series along the diaphragmatic muscle fibres (Farkas & Roussos, 1983; Kelsen et al. 1983). Measurements of the pressure changes produced by phrenic nerve stimulation in emphysematous patients have suggested that such a length adaptation also occurs in humans (Similowski et al. 1991). It is possible therefore that in patients with SLT for emphysema, the adverse effects of muscle length and load on the force developed by the hemidiaphragm on the native side are, in part, compensated for by a smaller number of sarcomeres.
Acknowledgments
The authors are grateful to M. Cappello for his help in sectioning the ventral mediastinal pleura and to the Fonds National de la Recherche Scientifique (FNRS, Belgium) for its financial support (Grant 3.4558.06 F).
References
- Boriek AM, Rodarte JR, Wilson TA. Kinematics and mechanics of midcostal diaphragm of dog. J Appl Physiol. 1997;83:1068–1075. doi: 10.1152/jappl.1997.83.4.1068. [DOI] [PubMed] [Google Scholar]
- Cappello M, De Troyer A. Interaction between the left and right intercostal muscles in airway pressure generation. J Appl Physiol. 2000;88:817–820. doi: 10.1152/jappl.2000.88.3.817. [DOI] [PubMed] [Google Scholar]
- Cassart M, Verbandt Y, de Francquen P, Gevenois PA, Estenne M. Diaphragm dimensions after single-lung transplantation for emphysema. Am J Respir Crit Care Med. 1999;159:1992–1997. doi: 10.1164/ajrccm.159.6.9812052. [DOI] [PubMed] [Google Scholar]
- Chacon RA, Corris PA, Dark JH, Gibson GJ. Comparison of the functional results of single lung transplantation for pulmonary fibrosis and chronic airway obstruction. Thorax. 1998;53:43–49. doi: 10.1136/thx.53.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danon J, Druz WS, Goldberg NB, Sharp JT. Function of the isolated paced diaphragm and the cervical accessory muscles in C1 quadriplegics. Am Rev Respir Dis. 1979;119:909–919. doi: 10.1164/arrd.1979.119.6.909. [DOI] [PubMed] [Google Scholar]
- De Troyer A. Differential control of the inspiratory intercostal muscles during airway occlusion in the dog. J Physiol. 1991;439:73–88. doi: 10.1113/jphysiol.1991.sp018657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Troyer A. The electro-mechanical response of canine inspiratory intercostal muscles to increased resistance: the cranial rib-cage. J Physiol. 1992;451:445–461. doi: 10.1113/jphysiol.1992.sp019172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Troyer A. Interaction between the diaphragm and intercostal muscles in lung expansion. J Appl Physiol. 2005;98:795–803. doi: 10.1152/japplphysiol.00632.2004. [DOI] [PubMed] [Google Scholar]
- De Troyer A, Cappello M, Scillia P. Effect of inflation on the interaction between the left and right hemidiaphragms. J Appl Physiol. 2005;99:1301–1307. doi: 10.1152/japplphysiol.00192.2005. [DOI] [PubMed] [Google Scholar]
- De Troyer A, Leduc D. Effect of inflation on the coupling between the ribs and the lung in dogs. J Physiol. 2004;555:481–488. doi: 10.1113/jphysiol.2003.057026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Troyer A, Legrand A. Inhomogenous activation of the parasternal intercostals during breathing. J Appl Physiol. 1995;79:55–62. doi: 10.1152/jappl.1995.79.1.55. [DOI] [PubMed] [Google Scholar]
- De Troyer A, Ninane V. Triangularis sterni: a primary muscle of breathing in the dog. J Appl Physiol. 1986;60:14–21. doi: 10.1152/jappl.1986.60.1.14. [DOI] [PubMed] [Google Scholar]
- DiMarco AF, Romaniuk J, Supinski GS. Mechanical action of the interosseous intercostal muscles as a function of lung volume. Am Rev Respir Dis. 1990;142:1041–1046. doi: 10.1164/ajrccm/142.5.1041. [DOI] [PubMed] [Google Scholar]
- Easton PA, Fitting JW, Arnoux R, Guerraty A, Grassino AE. Recovery of diaphragm function after laparotomy and chronic sonomicrometer implantation. J Appl Physiol. 1989;66:613–621. doi: 10.1152/jappl.1989.66.2.613. [DOI] [PubMed] [Google Scholar]
- Estenne M, Cassart M, Poncelet P, Gevenois PA. Volume of graft and native lung after single-lung transplantation for emphysema. Am J Respir Crit Care Med. 1999;159:641–645. doi: 10.1164/ajrccm.159.2.9802017. [DOI] [PubMed] [Google Scholar]
- Evans HE, Christensen GC. Miller's Anatomy of the Dog. 2. Philadelphia: W.B. Saunders; 1979. [Google Scholar]
- Farkas GA, Rochester DF. Functional characteristics of canine costal and crural diaphragm. J Appl Physiol. 1988;65:2253–2260. doi: 10.1152/jappl.1988.65.5.2253. [DOI] [PubMed] [Google Scholar]
- Farkas GA, Roussos C. Diaphragm in emphysematous hamsters: sarcomere adaptability. J Appl Physiol. 1983;54:1635–1640. doi: 10.1152/jappl.1983.54.6.1635. [DOI] [PubMed] [Google Scholar]
- Gibbons WJ, Levine SM, Bryan CL, Segarra J, Calhoon JH, Trinkle JK, Jenkinson SG. Cardiopulmonary exercise responses after single lung transplantation for severe obstructive lung disease. Chest. 1991;100:106–111. doi: 10.1378/chest.100.1.106. [DOI] [PubMed] [Google Scholar]
- Glanville AR, Estenne M. Indications, patient selection and timing of referral for lung transplantation. Eur Respir J. 2003;22:845–852. doi: 10.1183/09031936.03.00039003. [DOI] [PubMed] [Google Scholar]
- Greer JJ, Martin TP. Distribution of muscle fiber types and EMG activity in cat intercostal muscle. J Appl Physiol. 1990;69:1208–1211. doi: 10.1152/jappl.1990.69.4.1208. [DOI] [PubMed] [Google Scholar]
- Hubmayr RD, Margulies SS. Effects of unilateral hyperinflation on the interpulmonary distribution of pleural pressure. J Appl Physiol. 1992;73:1650–1654. doi: 10.1152/jappl.1992.73.4.1650. [DOI] [PubMed] [Google Scholar]
- Hubmayr RD, Sprung J, Nelson S. Determinants of transdiaphragmatic pressure in dogs. J Appl Physiol. 1990;69:2050–2056. doi: 10.1152/jappl.1990.69.6.2050. [DOI] [PubMed] [Google Scholar]
- Kelsen SG, Wolanski T, Supinski GS, Roesmann U. The effect of elastase-induced emphysema on diaphragmatic muscle structure in hamsters. Am Rev Respir Dis. 1983;127:330–334. doi: 10.1164/arrd.1983.127.3.330. [DOI] [PubMed] [Google Scholar]
- Kim MJ, Druz WS, Danon J, Machnach W, Sharp JT. Mechanics of the canine diaphragm. J Appl Physiol. 1976;41:369–382. doi: 10.1152/jappl.1976.41.3.369. [DOI] [PubMed] [Google Scholar]
- Kirkwood PA, Sears TA, Stagg D, Westgaard RH. The spatial distribution of synchronisation of intercostal motoneurones in the cat. J Physiol. 1982;327:137–155. doi: 10.1113/jphysiol.1982.sp014224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leduc D, De Troyer A. The effect of lung inflation on the inspiratory action of the canine parasternal intercostals. J Appl Physiol. 2006;100:858–863. doi: 10.1152/japplphysiol.00739.2005. [DOI] [PubMed] [Google Scholar]
- Legrand A, Brancatisano A, Decramer M, De Troyer A. Rostrocaudal gradient of electrical activation in the parasternal intercostal muscles of the dog. J Physiol. 1996;495:247–254. doi: 10.1113/jphysiol.1996.sp021589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Legrand A, De Troyer A. Spatial distribution of external and internal intercostal activity in dogs. J Physiol. 1999;518:291–300. doi: 10.1111/j.1469-7793.1999.0291r.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Legrand A, Goldman S, Damhaut P, De Troyer A. Heterogeneity of metabolic activity in the canine parasternal intercostals during breathing. J Appl Physiol. 2001;90:811–815. doi: 10.1152/jappl.2001.90.3.811. [DOI] [PubMed] [Google Scholar]
- Lin KC, Dizner-Golab A, Thurer RL, Loring SH. Mediastinal and chest wall limitations to asymmetry of lung inflation. J Appl Physiol. 2004;96:999–1004. doi: 10.1152/japplphysiol.00807.2003. [DOI] [PubMed] [Google Scholar]
- McCully KK, Faulkner JA. Length-tension relationship of mammalian diaphragm muscles. J Appl Physiol. 1983;54:1681–1686. doi: 10.1152/jappl.1983.54.6.1681. [DOI] [PubMed] [Google Scholar]
- Marshall R. Relationships between stimulus and work of breathing at different lung volumes. J Appl Physiol. 1962;17:917–921. [Google Scholar]
- Minh VD, Dolan GF, Konopka RF, Moser KM. Effect of hyperinflation on inspiratory function of the diaphragm. J Appl Physiol. 1976;40:67–73. doi: 10.1152/jappl.1976.40.1.67. [DOI] [PubMed] [Google Scholar]
- Road J, Newman S, Derenne JP, Grassino A. In vivo length-force relationship of canine diaphragm. J Appl Physiol. 1986;30:63–70. doi: 10.1152/jappl.1986.60.1.63. [DOI] [PubMed] [Google Scholar]
- Romaniuk JR, Supinski G, Di Marco AF. Relationship between parasternal and external intercostal muscle length and load compensatory responses in dogs. J Physiol. 1992;449:441–455. doi: 10.1113/jphysiol.1992.sp019095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sant'Ambrogio G, Widdicombe JG. Respiratory reflexes acting on the diaphragm and inspiratory intercostal muscles of the rabbit. J Physiol. 1965;180:766–779. doi: 10.1113/jphysiol.1965.sp007730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon R, Zechman FW. The reflex and mechanical response of the inspitatory muscles to an increased airflow resistance. Respir Physiol. 1972;16:51–69. doi: 10.1016/0034-5687(72)90088-6. [DOI] [PubMed] [Google Scholar]
- Similowski T, Yan S, Gauthier AP, Macklem PT, Bellemare F. Contractile properties of the human diaphragm during chronic hyperinflation. N Engl J Med. 1991;325:917–923. doi: 10.1056/NEJM199109263251304. [DOI] [PubMed] [Google Scholar]
- Smith J, Bellemare F. Effect of lung volume on in vivo contraction characteristics of human diaphragm. J Appl Physiol. 1987;62:1893–1900. doi: 10.1152/jappl.1987.62.5.1893. [DOI] [PubMed] [Google Scholar]
- Sprung J, Deschamps C, Hubmayr RD, Walters BJ, Rodarte JR. In vivo regional diaphragm function in dogs. J Appl Physiol. 1989;67:655–662. doi: 10.1152/jappl.1989.67.2.655. [DOI] [PubMed] [Google Scholar]
- Trulock EP, Edwards LB, Taylor DO, Boucek MM, Keck BM, Hertz MI. The registry of the International Society for Heart and Lung Transplantation: twenty-first official adult lung and heart-lung transplant report. J Heart Lung Transplant. 2004;23:804–815. doi: 10.1016/j.healun.2004.05.013. [DOI] [PubMed] [Google Scholar]
- Vaughan CW, Kirkwood PA. Evidence from motoneurone synchronisation for disynaptic pathways in the control of inspiratory motoneurones in the cat. J Physiol. 1997;503:673–691. doi: 10.1111/j.1469-7793.1997.673bg.x. [DOI] [PMC free article] [PubMed] [Google Scholar]