Introduction
Positron emission tomography (PET) combined with tracer-kinetic modelling 1–5 affords the assessment of regional myocardial blood flow (MBF) in ml/g/min and, thus, extending the scope of conventional scintigraphic imaging in the non-invasive identification and characterization of coronary circulatory (dys)function in early subclinical stages of coronary artery disease (CAD).2, 6 Experimental and clinical investigations indicate the importance of the functional integrity of the coronary circulatory function to mediate antiatherosclerotic and antithrombotic effects.7, 8 Vice versa, an impairment of coronary circulatory function reflects a proatherosclerotic state with substantial diagnostic and prognostic implications.9–14 The non-invasive identification and characterization of coronary circulatory (dys)function by means of PET offers important insight in the complex nature of mechanism underlying functional alterations of the coronary circulation and, thus, may contribute to unravel the pathophysiology of the early development of the coronary atherosclerotic process. For example, by evaluating MBF responses to sympathetic stimulation with cold pressor testing (CPT) and/or hyperaemic MBF increases during pharmacologic vasodilation, and their responses to pharmaceutical intervention,15, 16 or their relation to markers of systemic inflammation, insulin resistance or adipocytokines,17–21 PET measurements of MBF may contribute to better understanding of the development of early stages of the CAD process. Conceptually, the results of such investigations with PET quantification of MBF could also provide substantial information to develop specific medical therapy strategies to prevent the initiation and/or development of CAD in coronary risk patients.
Invasive assessment of coronary vasomotor function
The investigation of coronary vasomotor (circulatory) function is usually confined to patients with chest pain syndroms undergoing coronary angiography. The determination of alterations in epicardial diameter in response to intracoronary infusion of acetylcholine, bradykinin, or substance P is obtained by computer-based measurements of the coronary diameter (QCA; quantitative coronary angiography).22,23 Endothelium-dependent vasodilators such as acetylcholine specifically stimulate the receptor-mediated release of endothelium-derived nitric oxide (NO) that induces the epicardial artery to dilate through relaxation of the vascular smooth muscle cells. An epicardial vasodilation in response to specific acetylcholine stimulation, therefore, defines normal endothelial function. In the presence of a dysfunctional endothelium of the epicardial artery, however, the concurrent smooth muscle cell constrictor effects of acetylcholine, overcome the endothelium- mediated vasodilation 23 and a lack of a vasodilation or, more commonly, a paradoxical vasoconstriction ensues.23, 24 Another attractive and more physiologic approach to assess epicardial endothelial function is to determine a flow-mediated and, thus, also endothelium-related epicardial vasodilation by QCA during hyperaemic flow increases owing to pharmacologic vasodilation with adenosine or papaverine.23,25,26 While a flow-mediated epicardial vasodilation defines normal endothelial function, an impairment or absence of the flow-mediated epicardial vasodilation is indicative for a dysfunctional vascular endothelium. In addition, substances such as nitroglycerin or sodium nitroprusside supply NO directly to the vascular smooth muscle cell layer that induces an epicardial vasodilation, independent of the functional state of the vascular endothelium.
The assessment of coronary blood flow to evaluate the responses of the coronary microcirculatory system requires an intracoronary Doppler catheter and the placement of a flow wire.23 Notably, endothelium-dependent vasodilators like acetylcholine also increase coronary blood flow, mirroring their concomitant vasodilator effects on the coronary arteriolar resistance vessels. Increases in coronary flows to acetylcholine stimulation as measured with the Doppler flow wire, therefore, reflect normal endothelium-dependent coronary arteriolar vasomotor function, while an impairment or absence of coronary flow is appreciated as endothelial dysfunction of the arteriolar vessels. Conversely, substances like papaverine or adenosine exert a vascular smooth muscle cell relaxing effect at the site of the coronary resistance (arteriolar) vessels and, consequently, induce predominantly endothelium-independent coronary flow increases.2,22, 23
PET measurements of MBF
Approaches that apply PET to determine regional MBF in ml/g/min entail intravenous administration of positron-emitting tracers of MBF such as 13N-ammonia, 15O-water, and also more recently with 82 rubidium and imaging of the radiotracer’s transit time through the central circulatory system and its extraction and retention in the myocardium.1,3–5, 27–29 The final static 15-min frame of the serially acquired image data sets is reoriented into short and long axis myocardial slices and, as in our approach, assembled into polar maps.18, 19 Regions of interest (ROI) are assigned automatically by the analysis software program to the territories of the three major coronary arteries and a 25 mm2 ROI is assigned to the left ventricular (LV) blood pool. The ROIs are then copied to all serially acquired image data sets after they have been re-sliced and assembled into polar maps using the same reorientation parameters, as applied to the final 15-min static image. Following, the arterial tracer input function and the tracer uptake into the myocardium are determined.1, 3 The resulting time activity curves from these ROIs are then fitted with tracer compartment models, which describe the exchange of radiotracer between tissue compartments and the volume of tracer distribution in each compartment, and regional MBFs are calculated in ml/g/min.1–4,6,30 Tracer kinetic models correct for the physical decay of the radioisotope, spillover of activity between the left-ventricular blood pool and the myocardium31 and for partial volume effect, assuming a uniform myocardial wall thickness of 1 cm.32 13N-ammonia and 15O-water have been widely validated against independent microsphere blood flow measurements in animals and yield reproducible values of MBF.1, 4 Similarly measurements of MBF with 13N-ammonia and 15O-water in humans yield comparable estimates of MBF.27,33 More recently, 82 rubidium is emerging as a promising and reliable flow tracer 30 but still needs to be more validated. The short physical half-time of these positron-emitting tracers (physical half-live of 13N-ammonia 9.8 minutes and of 15O-water 2.4 minutes, while only 78 seconds for 82 rubidium) allow serial evaluations of MBF in one study session.
Determinants of MBF and flow reserve
The concept of the coronary flow reserve that describes the vasodilatory capacity of the coronary circulation to increase coronary flows to 3–5 times from baseline in response to increased metabolic demand was first described by Coffman and Gregg.34 Subsequently, Mosher et al.35 added the concept of the “coronary autoregulation” that provided a more physiologic framework for the coronary flow reserve. There are a numbers of determinants of resting MBF, while the oxygen demand, as a function of heart rate, blood pressures, myocardial contractility, and ventricular preload, is the prevalent factor in the modulation of resting MBF. When the metabolic myocardial oxygen demand is constant, MBF within the range of its autoregulation is widely independent of the coronary perfusion pressure. It follows then, that within this range of coronary autoregulation, so called “plateau”, coronary flow changes little despite alterations in perfusion pressures.36 Conversely, maximal or submaximal vasodilation of the coronary arteriolar resistance vessels during pharmacologic vasodilation, the coronary flow does not underlie any more the autoregulation and changes linearly with the intracoronary perfusion pressure. In this concept, the ratio of hyperaemic to resting coronary flow defines the coronary flow reserve.
As regards the coronary flow reserve, it is worthy to note that it does not necessarily reflect the true vasodilator capacity of the coronary circulation. Coronary flow reserve can decline owing to an increase in resting flow, a decrease in maximum hyperaemic flow, or both. Factors that increase myocardial oxygen demand such as arterial hypertension, increased myocardial contractility, increased left-ventricular wall stress, and tachycardia, induce an increase in resting flow. On the other side, maximum hyperaemic coronary flow may decline in the presence of a focal flow-limiting epicardial lesion, in the presence of microvascular disease in patients with hypertension or diabetes, or as a consequence of increases in extravascular resistive forces paralleled by increases in left-ventricular pressures in patients with congestive heart failure or hypertension. Despite these limitations of the concept of the coronary flow reserve, it is a useful index to determine the functional significance or downstream effects of focal epicardial artery lesions, the functional improvement after coronary revascularization, and for the evaluation of coronary circulatory function in individuals with subclinical or clinically manifest CAD.2, 37, 38
Resting MBF
PET measured MBFs at rest in healthy volunteers has been reported to range between 0.4 to 1.2 ml/g/min.36, 39–43 The variability in these individual resting MBFs were most likely related to differences in left-ventricular myocardial workload at the time of assessment, while methodological factors may have contributed to this.44 There is a close linear correlation between resting MBF and the rate-pressure product (defined as product of systolic blood pressure and heart rate) as a index cardiac work and, thus, also metabolic oxygen demand.36, 40, 45 The continuous age-related increase in resting MBF has also been attributed to increases in cardiac work owing to alterations predominantly in systolic blood pressure.36 More controversial are the MBF values at rest between men and women. Some investigations observed higher values of resting MBF in women than in men,40, 43, 46 that was suggested to be most likely related to sex-related differences in the lipid profile, while others did not find such a difference.36
PET in the non-invasive assessment of coronary circulatory dysfunction
Approaches to assess coronary circulatory function implicate measurements of MBF with PET at rest and its responses to physiologically- or pharmacologically-induced coronary flow increases, including bicycle exercise, dobutamine stress, sympathetic stimulation with cold pressor testing (CPT), vascular smooth muscle cell relaxation at the site of the coronary arteriolar vessels,2, 5, 29, 47–50 or heterogenous responses of MBF in more apical and more basal regions of the left ventricle as a possible non-inavsive index of epicardial vasomotion.51, 52
Sympathetic stimulation with cold pressor test
Non-invasive measurements of MBF with PET at rest and its response to sympathetic stimulation by cold pressor testing (CPT) have been appreciated to entail specific information on endothelium-related coronary vasomotor function.15, 49, 53–56 Immersion of the left hand into ice water causes a sympathetically-mediated increase in heart rate and blood pressures and, thus, an increase in myocardial workload. This increase in myocardial oxygen demand, as reflected by the elevation of the myocardial workload, is associated with a vasodilation of the coronary arteriolar resistance vessels through the release of presumably adenosine as a metabolic vasodilator.57 As a decrease in coronary vascular resistance ensues, an increase in coronary inflow is observed, which in turn leads to a flow-mediated and, thus, endothelium-dependent dilation of the upstream coronary vessel segment. Consequently, an increase in cardiac work is normally paralleled by commensurate flow-mediated coronary vasodilation (Fig. 1) and an increase in MBF as measured with PET.15,17,43,49 In the presence of a functional abnormality of endothelium-related coronary vasomotion, however, the coronary flow increases during sympathetic stimulation do not mediate a flow-related vasodilation. As consequence, the vasoconstrictor effects of the sympathetically-mediated stimulation of the vascular smooth muscle cells prevail and are not overcome by a normal flow-related coronary vasodilation (Fig. 2).6,23,49 The MBFs during CPT are then attenuated, absent, or even paradoxical, which denotes an impairment of endothelium-dependent coronary vasomotor function.7, 15, 17, 49, 56, 58
Fig. 1.

(A) Normal coronary angiogram of the left coronary tree in the RAO view of a healthy individual without coronary risk factors. (B) Corresponding angiogram during sympathetic stimulation with CPT. (C and D) Quantitative angiographic assessment of the proximal-mid LAD segment at rest (mean diameter: 2.0mm) (C) and during CPT (mean diameter:2.5mm) (D). (With kind permission from reference 49)
Fig. 2.
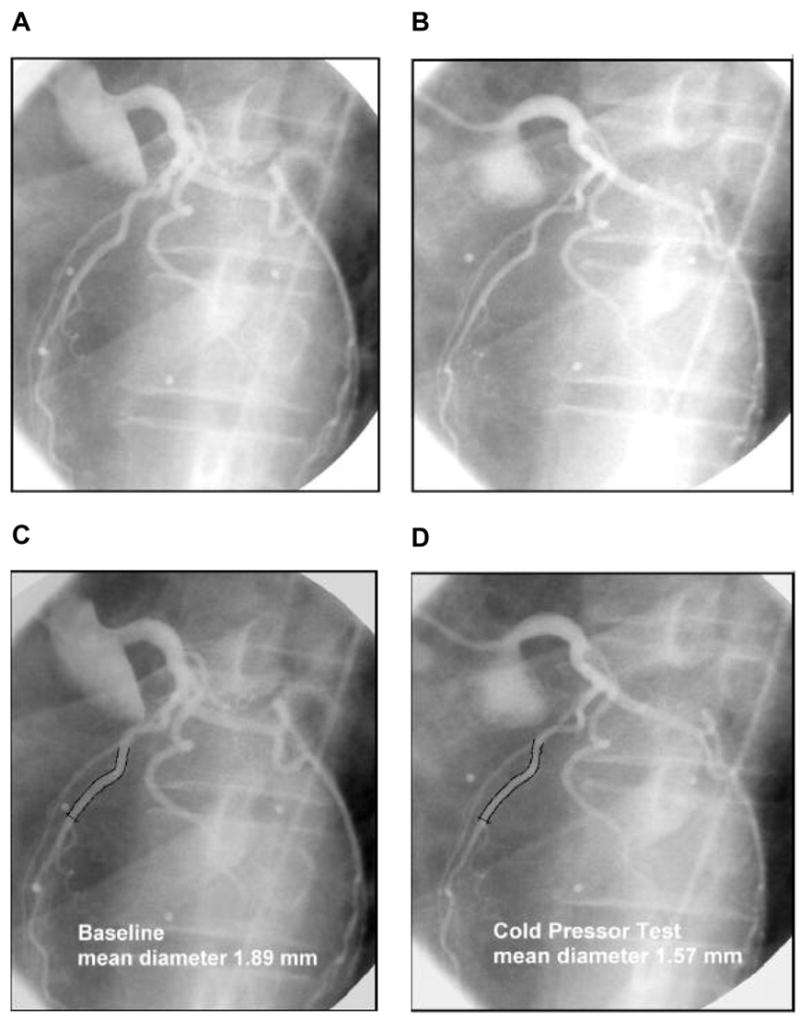
(A) Normal coronary angiogram of the left coronary artery tree in the LAO view in a chronic smkokers at rest. (B) Corresponding coronary angiogram during CPT. (C and D) Quantitative angiographic assessment of the proximal-mid LAD segment at rest (mean diameter: 1.89mm) (C) and during CPT (mean diameter:1.57mm) (D). (With kind permission from reference 49)
Several studies assessing coronary vasomotor function support the validity and value of non-invasively PET measured changes in MBF during CPT from rest as index of endothelium-dependent coronary vasomotion. For example, coronary flow increases during CPT, as assessed invasively with Doppler wire during coronary angiography, have been shown to closely correlate with the flow response to acetylcholine stimulation (Fig. 3).23, 53 Coronary flows during sympathetic stimulation with CPT, therefore, may indeed probe endothelium-related vasomotor function and, thereby, the functional integrity of the vascular wall.23,49 Notably, a similar association was also observed for between the epicardial vasomotor response to CPT and acetylcholine-stimulation, suggesting the epicardial vasomotor response to CPT to be intimately related to the the integrity of endothelial function.23,58 Further CPT-induced changes in epicardial luminal diameter, as determined by quantitative coronary angiography (QCA), and PET-measured responses of MBF to CPT closely correlated.15, 17, 49 More direct evidence for the involvement of the endothelium in the CPT-mediated MBF responses has been provided by Campisi et al.56 In chronic smokers presenting an impairment of MBF responses to CPT, intravenous infusion of L-arginine as a substrate of nitric oxide synthase (NOS) restored the MBF increase to CPT, most likely due to increases in the bioavailability of endothelium-derived nitric oxide (NO).
Fig. 3.

Association between the coronary flow response to CPT and actylcholine stimulation during coronary angiography in 12 normal control patients and in 19 patients with diffuse CAD. The CPT-induced changes in coronary vascular resistance inversely and significantly correlateds with the extent of endothelial dysfunction of the coronary arteriolar vessels as determined with acetylcholine stimulation. (With kind permission from reference 53)
There are also investigations with PET determining MBF and at the same time regional adrenergic neuronal function, using 11-labeled hydroxyepinephrine. These comparative investigations by PET could demonstrate the involvement of adrenergic neurons in the modulation of MBF responses to CPT. The uptake of 11-labeled hydroxyepinephrine in cardiac adrenergic nerve terminals is for example diminished in patients with cardiac allografts with a neuronal state of denervation or in diabetic patients with neuropathy and, thus, neuronal dysfunction.59–63 In these populations studied, reductions in myocardial 11-labeled hydroxyepinephrine retention were paralleled by a reduced MBF response to CPT. In particular, in diabetic patients MBF responses to sympathetic activation with CPT were somehow still preserved in myocardial regions with normal 11-labeled hydroxyepinephrine.62 These findings suggest indeed that both sympathetic activation and and flow-mediated vasomotor function modulate the MBF increases during stimulation with CPT.
Hyperaemic MBFs during pharmacologic vasodilation
Another interesting and more clinically used approach, is the assessment of coronary circulatory function by pharmacologically-induced hyperaemic MBF increases.2,5,44,64,65 Vascular smooth muscle-relaxing substances such as dipyridamole, adenosine, or more recently also adenosine triphosphate (ATP) or adenosine receptor agonists66 decrease the resistance to flow at the site of the coronary arteriolar resistance vessels and, thereby, induce a maximum or sub-maximum hyperemic flow increase. Since these substances induces hyperaemic flow increases through vascular smooth muscle cell relaxation, the resulting hyperaemic coronary flow increase is deemed as a measure of a predominantly endothelium-independent flow response. Inhibition of the endothelial nitric-oxide-synthase (eNOS) by intravenous infusion of L-NG-monomethyl arginine (L-NMMA) significantly reduces adenosine-induced MBF increases by 20–25% as measured with PET (Fig. 4).67, 68 Thus, shear sensitive components of the coronary endothelium contribute in part through flow-mediated coronary vasodilation to the overall hyperaemic MBF increases during pharmacologic vasodilation,23, 67, 68 which may also be seen as total integrated coronary circulatory function.
Fig. 4.

Hyperaemic MBF increase to adenosine stimulation and its alteration to the intravenous infusion of the nitric oxide (NO) synthase inhibitor NG-nitro-L-arginine methyl ester (4mg L-NAME /body weight i.v.). In the presence of L-NAME the hyperaemic MBF response was attenuated by 21% that is likely to reflect the impairment of the flow-mediated and, thus, endothelium-derived and NO-mediated vasodilation by L-NAME. (Adapted from reference 67)
Heterogeneity in longitudinal, base-to-apex MBF
A heterogeneity in longitudinal myocardial perfusion or MBFs has been described first during hyperaemic coronary flow increases during pharmacologic vasodilation in patients with diffuse CAD69 and, subsequently also with quantitative MBF measurements in individuals with coronary risk factors.51, 52 Fluid dynamic consequences of CAD related vessel stiffness and/or functional abnormalities of the epicardial conduit vessels may account for the longitudinal heterogeneity in myocardial perfusion. Based on the Hagen-Poiseuille equation,70–72 intracoronary resistance relates to the velocity of the blood flow and inversely to the fourth power of the vessel diameter. Normal functioning of the coronary vascular endothelium guarantees that increases in flow-velocity during exercise or pharmacologic vasodilation are associated with a flow- mediated dilation of the coronary circulation that balances the velocity-induced increase in coronary resistance, so that the resistance is kept low.73, 74 An abnormal structural and/or functional coronary vascular state during the early development of CAD, however, is commonly associated with an impairment of flow-mediated epicardial vasodilation. Under such condition, the resistance to higher coronary flows increases leads to a progressive proximal-to distal decline in intracoronary pressure along the epicardial artery,70 that is assumed as cause for a gradual, base-to-apex relative decline or heterogeneity in myocardial perfusion or MBF.51,52,69,70 The assessment of a heterogeneity in longitudinal myocardial perfusion or quantitatively in MBFs by means of PET, therefore, could be a promising non-invasive index of early structural and/or functional alterations of the CAD process predominantly at the site of the epicardial artery.
Reproducibility of MBFs during vasomotor stress
As PET-measured MBF responses to cold exposure and/or to pharmacologic vasodilation are increasingly applied to determine the effects of life-style modifications and/ or therapeutic interventions on coronary circulatory function,15,16, 20, 48,75–78 there is a need to establish the reproducibility of these MBF measurements. Such an analysis is necessary to assess the true measurement error of PET-measured MBFs in repeat assessments and/or to determine the sample size of future study populations needed for serial clinical PET studies. Previous studies have demonstrated that hyperaemic MBF increases during pharmacologic vasodilation can be measured reproducibly with 13N-ammonia or 15O-labeled water and PET.64,79 As recent investigations demonstrate, there is also a reliable one day reproducibility for CPT-related MBF when determined with 15O-labeled water.80 More recently and in a more extended investigation,81 the hemodynamic and MBF responses to CPT, as measured with 13N-ammonia and PET, were demonstrated to be highly reproducible in short- as well as in long-term (one day and 2–3 weeks protocol). The range of measurement errors, as denoted by the SEE for the endothelium-related ?MBF from rest to CPT, was 0.09 ml/g/min for short-term and 0.17 ml/g/min for long-term repeat measurements (Table 1). It follows then, that alterations in ?MBF in serial pharmaceutical studies that are above this range of SEE are likely to be indeed related to beneficial effects of pharmaceutical interventions on coronary endothelial function.81 The data of the latter study provided also useful information as regards the sample size needed to sufficiently power a study for serial MBF studies with CPT. According to this, a sample size of about 22 individuals with coronary risk factors is desirable for long-term-interventional pharmaceutical trial, while a sample size of 14 would be sufficient for a 1-day protocol.
Table 1.
MBF and hemodynamics at measurements 1 (M 1) and 2 (M 2) on the same day and measurement 3 (M 3) after 2 weeks for all study participants (n=20)
| M l | M 2 | M 3 | Mean difference
|
||
|---|---|---|---|---|---|
| M 1-2 | M 1–3 | ||||
| MBF in ml/g/min | |||||
| At rest | 0.67±0.19 | 0.66±0.15 | 0.63±0.18 | 0.09±0.10 | 0.10±0.10 |
| During CPT | 0.88±0.21 | 0.85±0.20 | 0.82±0.21 | 0.11±0.09 | 0.14±0.10 |
| Δ Change to CPT | 0.21±0.17 | 0.19±0.16 | 0.19±0.14 | 0.08±0.05 | 0.19±0.10 |
| Hemodynamics at rest | |||||
| Heart rate (beat/min) | 61±7 | 62±9 | 61±9 | 2.5±2.2 | 7.1±5.0 |
| SBP(mmHg) | 116±12 | l20±15 | 115±13 | 5.5±7.6 | 6.6±5.6 |
| DBP(mmHg) | 71±7 | 73±6 | 69±8 | 3.0±2.6 | 5.5±5.3 |
| RPP(mmHg/min) | 7,113 ±1,161 | 7,349±1,157 | 6,936±986 | 430±445 | 789±691 |
| Hemodynamics during CPT | |||||
| Heart rate(beat/min) | 68±8 | 67±7 | 67±9 | 3.5±2.4 | 5.8±5.1 |
| SBP (mmHg) | 148±22 | 152±22 | 149±23 | 5.8±10 | 8.3±11 |
| DBP (mmHg) | 86±11 | 87±12 | 85±13 | 2.7±2.1 | 8.5±6.9 |
| RPP (mmHg/min) | 9,982±1,798 | 10,160±1,456 | 9,935±1,259 | 724±543 | 1,470±1,011 |
| Δ RPP (mmHg/min) | 2,869±1,666 | 2,811±1,300 | 2,999±1,740 | 762±5l7 | 1,046±857 |
SBP systolic blood pressure, DBP diastolic blood pressure, RPP rate-pressure product
P≤0.05 for difference by paired t-test
(With kind permission from reference 81)
In interpreting the data of reproducibility studies, Bland and Altman82 proposed a repeatability coefficient (RPC) that is widely used to demonstrate the agreement between repeat measurements. The RPC can be used for a direct comparison of the precision of PET-determined MBFs between different studies (Table 2). As regards the RPC for the endothelium- related ? MBF to CPT, it was observed to be 0.18 ml/g/min for the short-term and 0.27 ml/g/min for the long-term reproducibility measurements with 13N-ammonia and PET.81 Both values were found to be lower than for the short-term RPC of CPT-related MBFs as measured with 15O-labeled water in a 1-day study protocol80 and also for hyperaemic MBF increases reported in previous studies,47,64,83 which have been reported between 0.49 and 1.34 ml/g/min (Table 2).
Table 2.
MBF repeatability coefficient in different studies
| Schindler et al. (present study) | Siegrest et al.[26] | Kaufmann et al.[25] | Wyss et al.[39] | Jagathesan et al. [40] | |||
|---|---|---|---|---|---|---|---|
| Radiotraser | 13N-ammonia | 15O-water | 15O-water | 15O-water | 15O-water | ||
| Period | ST(1 day) | LT(2 wk) | ST(1 day) | ST(1 day) | ST(1 day) | LT(2 wk) | |
| MBF at baseline | 0.26 | 0.26 | n.a. | 0.17 | 0.26 | 0.30a | 0.26b |
| MBF during CPT | 0.28 | 0.31 | 0.41 | n.a. | n.a. | n.a. | n.a. |
| Δ MBF during CPT | 0.18 | 0.27 | n.a. | n.a. | n.a. | n.a. | n.a. |
| MBF during adenosine | n.a. | n.a. | n.a. | 0.94 | 1.34 | n.a. | n.a. |
| MBF during bicycle exercise | n.a. | n.a. | n.a. | n.a. | 0.82 | n.a. | n.a. |
| MBF during dobutamine | n.a. | n.a. | n.a. | n.a. | n.a. | 0.49 | 0.58 |
ST short term, LT long term, n.a. not assessed
Ischemic myocardium
Remote myocardium
(With kind permission from reference 81)
Coronary circulatory function and cardiovascular events
The causes of coronary circulatory dysfunction in patients with coronary risk factors are certainly multifactorial, while an increased vascular production of reactive oxygen species (ROS) derived from superoxide-producing endothelial enzymes, such as NAD(P)H oxidase, xanthine oxidase, or uncoupled NO synthase, leading to a reduction in the bioavailability of endothelium-derived NO is thought to be a common final pathway underlying abnormal coronary circulatory function.84 An impairment of endothelium-dependent coronary circulatory function is commonly associated with other active processes such as inflammation, proliferation or apoptosis, and the expression of vascular cellular adhesion molecules (ICAM). This so called “endothelial activation” may reflect the initial injury of the vascular wall that may initiate and contribute to the development and progression of the atherosclerotic process. Such “endothelial activation” plays an important role in the pathogenesis of acute coronary syndromes, which is characterized by coronary plaque vulnerability, paradoxical vasoconstriction paralleled by endothelial dysfunction which is likely to contribute to plaque rupture,85,86 and increased thrombogenicity due to a loss of a potent antithrombotic endothelial surface.87 An impairment of coronary circulatory function, therefore, is likely to reflect in part the vulnerability of a plaque to rupture, which may explain the independent predictive value of coronary circulatory dysfunction for future cardiovascular events.9–11,88 It may be concluded, that functional alterations of coronary circulatory function appear to reflect on-going processes that modify the functional status of the arterial wall and may provide a rationale for the independent predictive value of functional abnormalities in coronary circulatory function for future cardiovascular events (Fig. 5).10 Such functional alterations of the coronary circulation appear to be superior to the assessment of CAD-related structural alterations of the epicardial wall in the prediction of the future cardiovascular clinical outcome.89–93
Fig. 5.
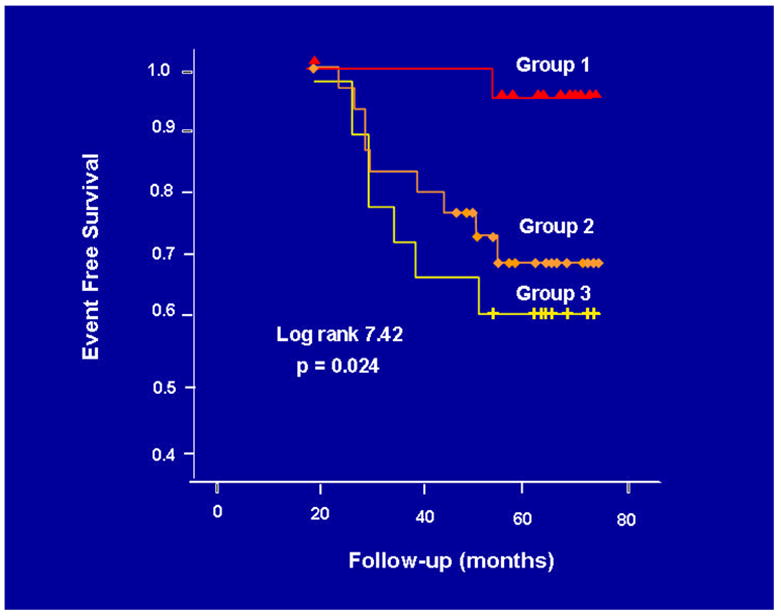
Prognostic value of PET-measured, endothelium-related MBF responses to sympathetic stimulation with CPT. The Kaplan-Meier analysis demonstrates an association between the incidence of cardiovascular events and the degree of the dimished MBF response to CPT (group 1: ?MBF ≥40%; group 2: ?MBF<40% and; group 3: ?MBF≤0%). (With kind permission from reference 9)
PET-determined coronary circulatory function and vascular redox state
Measurements of MBF at rest and during various forms of vasomotor stress have contributed new “in vivo” mechanistic insight in the mechanisms affecting coronary circulatory function in individuals with coronary risk factors. For example, Kaufmann et al.77 could demonstrate that acute intravenous antioxidant intervention with vitamin C, in order to reduce the oxidative stress burden in smokers, significantly increased hyperaemic MBF increases to pharmacologic vasodilation. The latter findings strongly suggest reactive oxygen species (ROS) as prevalent cause for the impairment of the total coronary vasodilator function in chronic smokers. In individuals with familial hypercholesterolemia or with secondary hypercholesterolemia hyperaemic MBFs during pharmacologic vasodilation are commonly reduced when compared to age-matched controls.94–96 Notably, the hyperaemic flow increases and MFR were inversely correlated with the severity of abnormal plasma lipid levels. Despite the well known detrimental effects of total cholesterol plasma levels on the coronary circulation, it appears that not the only total cholesterol but also LDL-cholesterol is a major determinant of a reduced coronary vasodilatory capacity as determined with PET.94 Conversely, restoring tetrahydrobiopterin (BH4) deficiency in hypercholesterolemic individuals normalized adenosine-stimulated hyperaemic flow responses.76 Such finding points out that BH4 deficiency may also contribute to coronary circulatory dysfunction, most likely through an uncoupling of endothelial NOS97 with a further increases in ROS in hypercholesterolemia.
Of note, previous investigations with PET measurements of MBF at rest and the response to CPT 15 demonstrated heterogenous responses in endothelium-related MBFs during CPT to short- and long-term antioxidant intervention with vitamin C in patients with different coronary risk factors such as smoking, arterial hypertension and hypercholesterolemia (Fig. 6). Such “in vivo” findings by PET imaging 15 may contrast experimental investigations that proposed increases in ROS as main common pathway underlying endothelial dysfunction 84, 98 and, thus, may suggest rather complex mechanism which may account for abnormalities in coronary circulatory function.23 As regards chronic smokers, short- and long-term antioxidant vitamin C challenges improved abnormalities in MBF responses to CPT, while no such beneficial effect was observed in individuals with hypercholesterolemia (Fig. 6). This may suggest that abnormalities in endothelium-related coronary vasomotion in smokers is predominantly mediated by the release of ROS, while other mechanism appear to prevail in the presence of hypercholesterolemia. For example, selective targeting of G-protein-dependent signal transduction by oxidized LDL results in a diminished receptor-mediated stimulation of endothelial NO production.99 Interestingly, in hypertensive patients short-term vitamin C challenges did not lead to an improvement in of impaired MBF responses to CPT. In contrast, after long-term application of vitamin C over 3 months a normalized endothelium-related MBF response to CPT was observed, which was also sustained after a 2 years follow-up. The delayed onset of the beneficial effect of vitamin C challenges on endothelium-related MBF responses to cold exposure remain uncertain but may be related to an improvement of the endothelial redox equilibrium resulting in an increase expression of endothelial nitric-oxide synthase (eNOS), or prevention of eNOS uncoupling through enhanced bioavailability of tetrahydrobiopterin (BH4).97, 100, 101
Fig. 6.

Effects of short-term and long-term antioxidant intervention on endothelium-related MBF responses to CPT. The graphic demonstrates contrasting MBF responses to challenges in vitamin C in hypercholesterolemic patients, smokers and hypertensive patients. (With kind permission from reference 15)
Insulin-resistance affecting the coronary circulation
In recent years, obesity, which frequently associated with the insulin-resistance syndrome and systemic microinflammation, has been recognized as an increasing risk factor of cardiovascular morbidity and mortality.102 The exact mechanisms by which obesity initiates and accelerates coronary vascular disease remain to be investigated. Studying coronary circulatory function with PET demonstrated that insulin resistant patients without traditional coronary risk factors may present abnormalities in coronary circulatory function.20 In these individuals with insulin resistance, the endothelium-related MBF response to CPT was diminished, while hyperaemic MBF during dipyridamole stimulation was still preserved. Consequently, the functional abnormality in this group of insulin-resistant individuals was confined to the coronary vascular endothelium. Notably, while initial stages of the vascular injury may only involve the endothelium,17,56,75,103 more advanced stages of coronary risk factors states such as increases in oxidative stress burden, may also lead to an impairment in smooth muscle cell vasodilator function.97 For example, functional abnormality of the coronary circulation in individuals with increasing body weight progresses from an impairment in endothelium-dependent MBF response to CPT in overweight to an impairment of the predominantly endothelium-independent hyperaemic MBFs during dipyridamole stimulation in obesity (Fig. 7A and 7B).18 Similar observations were reported by Prior et al.,19 as they observed a progressive worsening of functional abnormalities of endothelium-dependent vasomotion to manifest with increasing severity of insulin-resistance and carbohydrate intolerance, while attenuated endothelium-independent hyperaemic flow responses to pharmacologic vasodilation paralleled the more clinically evident metabolic abnormalities in diabetes. In support of the latter findings, patients with type 1 and type 2 diabetes mellitus revealed a comparable marked impairment of hyperaemic MBFs 60,104–108 that was related to euglycemic control and that also correlated inversely with plasma glucose concentrations averaged over several months.
Fig. 7.


(A) Myocardial blood flow (MBF) at rest, during cold pressor testing (CPT), and during pharmacologic vasodilation with dipyridamole for the three study groups. The dipyridamole-stimulated MBF was lower in overweight than in controls, but not significantly. In obesity the hyperaemic MBFs during dipyridamole stimulation were lowest. (B) Change of endothelium-related MBF during CPT (?MBF) for the three study groups. As can be appreciated, there is a progressive decrease of the endothelium-related MBF response to CPT from control, to overweight and obesity. (With kind permission from reference 18).
In a more complex setting, the assessment of coronary circulatory function in overweight individuals with PET could demonstrate that increases in body weight, paralleled by an increase in plasma markers of the insulin-resistance syndrome and chronic inflammation, was independently associated with abnormal coronary circulatory function.18 These findings provided first evidence that that obesity is an alternate mediator of coronary vascular disease rather than epiphenomenon related to other traditional coronary risk factors.18,109 Of interest, the role of adipocytokines such as leptin. adiponectin or grhelin affecting the coronary circulation in humans is controversial and remains to be investigated.110,111 For example, in “in vitro” studies, leptin released from the adipose tissue, may stimulate prothatherosclerotic effects, such as increases in ROS in cultured human endothelial cells, acceleration of vascular cell calcification, smooth muscle cell proliferation, and migration.112 On the other hand, leptin may induce both endothelium-dependent and -independent vasodilation, 113–115 indicating NO-mediated antiatherosclerotic effects.116 In view of these contrasting observations, PET-imaging in obese individuals demonstrated a significant and positive association between leptin plasma levels and endothelium-related MBFs to CPT (r=0.37, p<0.036). Increases in leptin plasma levels, therefore, were associated with relatively higher endothelium-related MBF increases to CPT. This positive association might be suggestive of a beneficial effect of leptin and/or leptin-related but still undetermined factors on the coronary endothelium to counteract the adverse effects of increases in body weight on coronary vasomotor function.18 As the recent investigations demonstrate,18 analysing PET-measurements of MBF and plasma markers of various coronary risk factors may be a unique means to tease out the adverse or beneficial effects of various factors on coronary circulatory function in complex “in vivo” conditions. While experimental investigations are most important to investigate a direct cause-effect relation,84, 98, 117 “in vivo” imaging with PET may contribute to denote important associations between coronary risk factors and coronary vascular state.
Monitoring Responses to Therapeutic Intervention
Based on the central role of coronary circulatory dysfunction for the development and progression of atherosclerosis, improvement of abnormal endothelial-dependent coronary vasomotion by a variety of interventions, such as angiotensin-converting-enzyme-inhibitors, 118 beta-hydroxymethylglutaryl-coenzyme A reductase inhibitors,78,119 hormone replacement therapy in postmenopausal women,75 euglycemic controls in diabetes and physical exercise,21, 120 has become a primary therapeutic goal in the prevention of the atherosclerotic process. Notably, Fichtlscherer et al.121 reported that a normalization of endothelial function of the forearm circulation in patients having suffered an acute coronary syndrome was paralleled by an event-free survival, but not in those who did not show a restoration of forearm vasomotor function to standard therapy. Similar findings were reported by Modena et al.122 in hypertensive postmenopausal women. An improvement in brachial artery flow-mediated and, thus, endothelium-dependent vasodilation after institution of medical therapy to control arterial hypertension, resulted in an improved clinical outcome as compared to those postmenopausal women who did not respond. Although low in numbers, these preliminary results support the evolving concept that improvement in vasomotor function in the peripheral and coronary circulation could indeed mediate an improvement in clinical outcome. If this holds true in future clinical studies, then the assessment of coronary circulatory function by PET imaging could be a promising and unique tool to successfully guide preventive medical therapy in the development and/or progression of the CAD process.
In this regard, long-term estrogen replacement in postmenopausal women without traditional coronary risk factors may improve endothelium-related MBF responses to CPT, while no such effect was observed for the short-term administration,75,123,124 or in postmenopausal women with traditional coronary risk factors.75 Also, beta-hydroxymethylglutaryl-coenzyme A reductase inhibitors (simvastatin) beneficially altered the lipid profile in hypercholesterolemic patients, that was accompanied by a significant increase in dipyridamole-stimulated mean hyperaemic MBF from 1.82±0.36 ml/g/min at baseline to 2.38±0.58ml/g/min at follow-up as measured with 13N-ammonia and PET.119 Such an improvement in coronary circulatory function in hypercholesterolemic patients after lipid-lowering therapy with beta-hydroxymethylglutaryl-coenzyme A reductase inhibitors was confirmed by other investigations.125–127 Interestingly, studying coronary circulatory function with PET demonstrated that insulin resistant patients without traditional coronary risk factors may present abnormalities in coronary vasomotor function.20 More excitingly, insulin-sensitizing thiazolidinedione therapy normalized abnormal MBF responses to CPT in 25 individuals with insulin resistance. More recently, the effects of glucose lowering therapy on coronary circulatory function in type 2 diabetic patients were studied as well.21 Three months of glucose-lowering treatment with glyburide and metformin significantly improved the coronary endothelium-mediated vasomotor function (Fig. 8A). Of note, the decrease in plasma glucose levels significantly correlated with the improvement in endothelium-related MBF responses to CPT and, thus, an improvement in coronary vasomotor (dys)function (r=0.67, p<0.01) (Fig. 8B). This association suggests a direct adverse effect of elevated plasma glucose, apart from the adverse effects of the insulin resistance syndrome, on diabetes-related coronary vascular disease in a preclinical state of CAD.
Fig. 8.


(A) Effects of glucose lowering-therapy with glyburide and metformin on endothelium-related ?MBF to CPT in type 2 diabetes mellitus patients. In type 2 diabetic patients with euglycemic control after 3 months of glucose lowering treatment with glucose plasma levels ≤126mg/dl (group of responders) the endothelium-mediated MBF response to CPT significantly increased comparable to controls, while in patients with glucose plasma levels >126mg/dl (group of non- responders) virtually no change in ?MBF to CPT was observed. (B) Association of the endothelium-related ?MBF to CPT and the change in fasting plasma glucose concentration as defined as difference in ?MBF and ?Glucose decrease between 3 months follow-up and baseline measurements. (With kind permission from reference 21)
Conclusions and comment
Combining cardiac PET perfusion imaging with tracer kinetic models affords the non-invasive assessment of regional MBF in ml/g/min that offers important “in vivo” insight in the complex nature of mechanism underlying functional alterations of the coronary circulation. Thus, cardiac PET imaging may contribute to unravel the pathophysiology of the early development of the atherosclerotic process. Such “in vivo” imaging with PET may denote important associations between coronary risk factors and coronary circulatory function that may complement or even contrast experimental studies that investigate direct cause-effect relationships. Further, by assessing MBFs at rest and during vasomotor stress, the functional consequences of structural and/or functional alterations in the coronary circulation may be identified before hemodynamically significant obstructive CAD may manifest. The identifications of such early structural and/or functional abnormalities of the coronary circulation appears to carry important diagnostic and prognostic information. Whether an improvement or even restoration of abnormal functional alterations of coronary circulatory function due to preventive medical intervention also results in an improved clinical outcome in these patients warrants further investigations.
Acknowledgments
Research Grants: This work was supported in part by NIH grant HL 33177 from the National Heart, Lung and Blood Institute, Bethesda, Maryland, USA.
Footnotes
Conflict of interest disclosure: The authors do not have any conflict of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Kuhle WG, Porenta G, Huang SC, Buxton D, Gambhir SS, Hansen H, et al. Quantification of regional myocardial blood flow using 13N-ammonia and reoriented dynamic positron emission tomographic imaging. Circulation. 1992;86(3):1004–1017. doi: 10.1161/01.cir.86.3.1004. [DOI] [PubMed] [Google Scholar]
- 2.Kaufmann PA, Camici PG. Myocardial blood flow measurement by PET: technical aspects and clinical applications. J Nucl Med. 2005;46(1):75–88. [PubMed] [Google Scholar]
- 3.Schelbert HR, Phelps ME, Huang SC, MacDonald NS, Hansen H, Selin C, et al. N-13 ammonia as an indicator of myocardial blood flow. Circulation. 1981;63(6):1259–1272. doi: 10.1161/01.cir.63.6.1259. [DOI] [PubMed] [Google Scholar]
- 4.Bergmann SR, Fox KA, Rand AL, McElvany KD, Welch MJ, Markham J, et al. Quantification of regional myocardial blood flow in vivo with H215O. Circulation. 1984;70(4):724–733. doi: 10.1161/01.cir.70.4.724. [DOI] [PubMed] [Google Scholar]
- 5.Camici PG, Crea F. Coronary microvascular dysfunction. N Engl J Med. 2007;356(8):830–840. doi: 10.1056/NEJMra061889. [DOI] [PubMed] [Google Scholar]
- 6.Schindler TH, Schelbert HR. Measurements of myocardial blood flow and monitoring therapy. In: Zaret BL, Beller GA, editors. Nuclear Cardiology - State of The Art and Future Directions. 3. Mosby; Philadelphia: 2005. pp. 399–412. [Google Scholar]
- 7.Ganz P, Vita JA. Testing endothelial vasomotor function: nitric oxide, a multipotent molecule. Circulation. 2003;108(17):2049–2053. doi: 10.1161/01.CIR.0000089507.19675.F9. [DOI] [PubMed] [Google Scholar]
- 8.Widlansky ME, Gokce N, Keaney JF, Jr, Vita JA. The clinical implications of endothelial dysfunction. J Am Coll Cardiol. 2003;42(7):1149–1160. doi: 10.1016/s0735-1097(03)00994-x. [DOI] [PubMed] [Google Scholar]
- 9.Schindler TH, Nitzsche EU, Schelbert HR, Olschewski M, Sayre J, Mix M, et al. Positron emission tomography-measured abnormal responses of myocardial blood flow to sympathetic stimulation are associated with the risk of developing cardiovascular events. J Am Coll Cardiol. 2005;45(9):1505–1512. doi: 10.1016/j.jacc.2005.01.040. [DOI] [PubMed] [Google Scholar]
- 10.Lerman A, Zeiher AM. Endothelial function: cardiac events. Circulation. 2005;111(3):363–368. doi: 10.1161/01.CIR.0000153339.27064.14. [DOI] [PubMed] [Google Scholar]
- 11.Schindler TH, Hornig B, Buser PT, Olschewski M, Magosaki N, Pfisterer M, et al. Prognostic value of abnormal vasoreactivity of epicardial coronary arteries to sympathetic stimulation in patients with normal coronary angiograms. Arterioscler Thromb Vasc Bio. 2003;23(3):495–501. doi: 10.1161/01.ATV.0000057571.03012.F4. [DOI] [PubMed] [Google Scholar]
- 12.Schachinger V, Britten MB, Zeiher AM. Prognostic impact of coronary vasodilator dysfunction on adverse long-term outcome of coronary heart disease. Circulation. 2000;101(16):1899–1906. doi: 10.1161/01.cir.101.16.1899. [DOI] [PubMed] [Google Scholar]
- 13.Britten MB, Zeiher AM, Schachinger V. Microvascular dysfunction in angiographically normal or mildly diseased coronary arteries predicts adverse cardiovascular long-term outcome. Coron Art Dis. 2004;15(5):259–264. doi: 10.1097/01.mca.0000134590.99841.81. [DOI] [PubMed] [Google Scholar]
- 14.Cecchi F, Olivotto I, Gistri R, Lorenzoni R, Chiriatti G, Camici PG. Coronary microvascular dysfunction and prognosis in hypertrophic cardiomyopathy. N Engl J Med. 2003;349(11):1027–1035. doi: 10.1056/NEJMoa025050. [DOI] [PubMed] [Google Scholar]
- 15.Schindler TH, Nitzsche EU, Munzel T, Olschewski M, Brink I, Jeserich M, et al. Coronary vasoregulation in patients with various risk factors in response to cold pressor testing: contrasting myocardial blood flow responses to short- and long-term vitamin C administration. J Am Coll Cardiol. 2003;42(5):814–822. doi: 10.1016/s0735-1097(03)00851-9. [DOI] [PubMed] [Google Scholar]
- 16.Hattori N, Schnell O, Bengel FM, Rihl J, Nekolla SG, Drzezga AE, et al. Deferoxamine improves coronary vascular responses to sympathetic stimulation in patients with type 1 diabetes mellitus. Eur J Nucl Med Mol Imaging. 2002;29(7):891–898. doi: 10.1007/s00259-002-0799-0. [DOI] [PubMed] [Google Scholar]
- 17.Schindler TH, Nitzsche EU, Olschewski M, Magosaki N, Mix M, Prior JO, et al. Chronic inflammation and impaired coronary vasoreactivity in patients with coronary risk factors. Circulation. 2004;110(9):1069–1075. doi: 10.1161/01.CIR.0000140264.56496.76. [DOI] [PubMed] [Google Scholar]
- 18.Schindler TH, Cardenas J, Prior JO, Facta AD, Kreissl MC, Zhang XL, et al. Relationship between increasing body weight, insulin resistance, inflammation, adipocytokine leptin, and coronary circulatory function. J Am Coll Cardiol. 2006;47(6):1188–1195. doi: 10.1016/j.jacc.2005.10.062. [DOI] [PubMed] [Google Scholar]
- 19.Prior JO, Quinones MJ, Hernandez-Pampaloni M, Facta AD, Schindler TH, Sayre JW, et al. Coronary circulatory dysfunction in insulin resistance, impaired glucose tolerance, and type 2 diabetes mellitus. Circulation. 2005;111(18):2291–2298. doi: 10.1161/01.CIR.0000164232.62768.51. [DOI] [PubMed] [Google Scholar]
- 20.Quinones MJ, Hernandez-Pampaloni M, Schelbert H, Bulnes-Enriquez I, Jimenez X, Hernandez G, et al. Coronary vasomotor abnormalities in insulin-resistant individuals. Ann Intern Med. 2004;140(9):700–708. doi: 10.7326/0003-4819-140-9-200405040-00009. [DOI] [PubMed] [Google Scholar]
- 21.Schindler TH, Facta AD, Prior JO, Cadenas J, Hsueh WA, Quinones MJ, et al. Improvement in coronary vascular dysfunction produced with euglycaemic control in patients with type 2 diabetes. Heart. 2007;93(3):345–349. doi: 10.1136/hrt.2006.094128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cohn JN, Quyyumi AA, Hollenberg NK, Jamerson KA. Surrogate markers for cardiovascular disease: functional markers. Circulation. 2004;109(25 Suppl 1):IV31–46. doi: 10.1161/01.CIR.0000133442.99186.39. [DOI] [PubMed] [Google Scholar]
- 23.Drexler H. Endothelial dysfunction: clinical implications. Prog Cardiovasc Dis. 1997;39(4):287–324. doi: 10.1016/s0033-0620(97)80030-8. [DOI] [PubMed] [Google Scholar]
- 24.Ludmer PL, Selwyn AP, Shook TL, Wayne RR, Mudge GH, Alexander RW, et al. Paradoxical vasoconstriction induced by acetylcholine in atherosclerotic coronary arteries. N Engl J Med. 1986;315(17):1046–1051. doi: 10.1056/NEJM198610233151702. [DOI] [PubMed] [Google Scholar]
- 25.Zeiher AM, Drexler H, Wollschlager H, Just H. Modulation of coronary vasomotor tone in humans. Progressive endothelial dysfunction with different early stages of coronary atherosclerosis. Circulation. 1991;83(2):391–401. doi: 10.1161/01.cir.83.2.391. [DOI] [PubMed] [Google Scholar]
- 26.Rossen JD, Quillen JE, Lopez AG, Stenberg RG, Talman CL, Winniford MD. Comparison of coronary vasodilation with intravenous dipyridamole and adenosine. J Am Coll Cardiol. 1991;18(2):485–491. doi: 10.1016/0735-1097(91)90604-8. [DOI] [PubMed] [Google Scholar]
- 27.Nitzsche EU, Choi Y, Czernin J, Hoh CK, Huang SC, Schelbert HR. Noninvasive quantification of myocardial blood flow in humans. A direct comparison of the [13N]ammonia and the [15O]water techniques. Circulation. 1996;93(11):2000–2006. doi: 10.1161/01.cir.93.11.2000. [DOI] [PubMed] [Google Scholar]
- 28.Mullani NA, Goldstein RA, Gould KL, Marani SK, Fisher DJ, O'Brien HA, et al. Myocardial perfusion with rubidium-82. I. Measurement of extraction fraction and flow with external detectors. J Nucl Med. 1983;24(10):898–906. [PubMed] [Google Scholar]
- 29.Di Carli MF, Hachamovitch R. New technology for noninvasive evaluation of coronary artery disease. Circulation. 2007;115(11):1464–1480. doi: 10.1161/CIRCULATIONAHA.106.629808. [DOI] [PubMed] [Google Scholar]
- 30.Sampson UK, Dorbala S, Limaye A, Kwong R, Di Carli MF. Diagnostic accuracy of rubidium-82 myocardial perfusion imaging with hybrid positron emission tomography/computed tomography in the detection of coronary artery disease. J Am Coll Cardiol. 2007;49(10):1052–1058. doi: 10.1016/j.jacc.2006.12.015. [DOI] [PubMed] [Google Scholar]
- 31.Weinberg IN, Huang SC, Hoffman EJ, Araujo L, Nienaber C, Grover-McKay M, Dahlbom M, Schelbert H. Validation of PET-acquired input functions for cardiac studies. J Nucl Med. 1988;29(2):241–247. [PubMed] [Google Scholar]
- 32.Gambhir SS, Schwaiger M, Huang SC, Krivokapich J, Schelbert HR, Nienaber CA, et al. Simple noninvasive quantification method for measuring myocardial glucose utilization in humans employing positron emission tomography and fluorine-18 deoxyglucose. J Nucl Med. 1989;30(3):359–366. [PubMed] [Google Scholar]
- 33.Bol A, Melin JA, Vanoverschelde JL, Baudhuin T, Vogelaers D, De Pauw M, et al. Direct comparison of [13N]ammonia and [15O]water estimates of perfusion with quantification of regional myocardial blood flow by microspheres. Circulation. 1993;87(2):512–525. doi: 10.1161/01.cir.87.2.512. [DOI] [PubMed] [Google Scholar]
- 34.Coffman JD, Gregg DE. Reactive hyperemia characteristics of the myocardium. Am J Physiol. 1960;199:1143–1149. doi: 10.1152/ajplegacy.1960.199.6.1143. [DOI] [PubMed] [Google Scholar]
- 35.Mosher P, Ross J, Jr, McFate PA, Shaw RF. Control of Coronary Blood Flow by an Autoregulatory Mechanism. Circ Res. 1964;14:250–259. doi: 10.1161/01.res.14.3.250. [DOI] [PubMed] [Google Scholar]
- 36.Czernin J, Muller P, Chan S, Brunken RC, Porenta G, Krivokapich J, et al. Influence of age and hemodynamics on myocardial blood flow and flow reserve. Circulation. 1993;88(1):62–69. doi: 10.1161/01.cir.88.1.62. [DOI] [PubMed] [Google Scholar]
- 37.Morita K, Tsukamoto T, Naya M, Noriyasu K, Inubushi M, Shiga T, et al. Smoking cessation normalizes coronary endothelial vasomotor response assessed with 15O-water and PET in healthy young smokers. J Nucl Med. 2006;47(12):1914–1920. [PubMed] [Google Scholar]
- 38.Tsukamoto T, Morita K, Naya M, Katoh C, Inubushi M, Kuge Y, et al. Myocardial flow reserve is influenced by both coronary artery stenosis severity and coronary risk factors in patients with suspected coronary artery disease. Eur J Nucl Med Mol Imaging. 2006;33(10):1150–1156. doi: 10.1007/s00259-006-0082-x. [DOI] [PubMed] [Google Scholar]
- 39.Bergmann SR, Herrero P, Markham J, Weinheimer CJ, Walsh MN. Noninvasive quantitation of myocardial blood flow in human subjects with oxygen-15-labeled water and positron emission tomography. J Am Coll Cardiol. 1989;14(3):639–652. doi: 10.1016/0735-1097(89)90105-8. [DOI] [PubMed] [Google Scholar]
- 40.Chareonthaitawee P, Kaufmann PA, Rimoldi O, Camici PG. Heterogeneity of resting and hyperemic myocardial blood flow in healthy humans. Cardiovasc Res. 2001;50(1):151–161. doi: 10.1016/s0008-6363(01)00202-4. [DOI] [PubMed] [Google Scholar]
- 41.Tamaki N, Yonekura Y, Senda M, Kureshi SA, Saji H, Kodama S, et al. Myocardial positron computed tomography with 13N-ammonia at rest and during exercise. Eur J Nucl Med. 1985;11(6–7):246–251. doi: 10.1007/BF00279078. [DOI] [PubMed] [Google Scholar]
- 42.Senneff MJ, Geltman EM, Bergmann SR. Noninvasive delineation of the effects of moderate aging on myocardial perfusion. J Nucl Med. 1991;32(11):2037–2042. [PubMed] [Google Scholar]
- 43.Prior JO, Schindler TH, Facta AD, Hernandez-Pampaloni M, Campisi R, Dahlbom M, et al. Determinants of myocardial blood flow response to cold pressor testing and pharmacologic vasodilation in healthy humans. Eur J Nucl Med Mol Imaging. 2007;34(1):20–27. doi: 10.1007/s00259-006-0193-4. [DOI] [PubMed] [Google Scholar]
- 44.Sawada S, Muzik O, Beanlands RS, Wolfe E, Hutchins GD, Schwaiger M. Interobserver and interstudy variability of myocardial blood flow and flow-reserve measurements with nitrogen 13 ammonia-labeled positron emission tomography. J Nucl Cardiol. 1995;2(5):413–422. doi: 10.1016/s1071-3581(05)80029-7. [DOI] [PubMed] [Google Scholar]
- 45.Krivokapich J, Smith GT, Huang SC, Hoffman EJ, Ratib O, Phelps ME, et al. 13N ammonia myocardial imaging at rest and with exercise in normal volunteers. Quantification of absolute myocardial perfusion with dynamic positron emission tomography. Circulation. 1989;80(5):1328–1337. doi: 10.1161/01.cir.80.5.1328. [DOI] [PubMed] [Google Scholar]
- 46.Duvernoy CS, Meyer C, Seifert-Klauss V, Dayanikli F, Matsunari I, Rattenhuber J, et al. Gender differences in myocardial blood flow dynamics: lipid profile and hemodynamic effects. J Am Coll Cardiol. 1999;33(2):463–470. doi: 10.1016/s0735-1097(98)00575-0. [DOI] [PubMed] [Google Scholar]
- 47.Wyss CA, Koepfli P, Mikolajczyk K, Burger C, von Schulthess GK, Kaufmann PA. Bicycle exercise stress in PET for assessment of coronary flow reserve: repeatability and comparison with adenosine stress. J Nucl Med. 2003;44(2):146–154. [PubMed] [Google Scholar]
- 48.Namdar M, Koepfli P, Grathwohl R, Siegrist PT, Klainguti M, Schepis T, et al. Caffeine decreases exercise-induced myocardial flow reserve. J Am Coll Cardiol. 2006;47(2):405–410. doi: 10.1016/j.jacc.2005.08.064. [DOI] [PubMed] [Google Scholar]
- 49.Schindler TH, Nitzsche EU, Olschewski M, Brink I, Mix M, Prior J, et al. PET-Measured Responses of MBF to Cold Pressor Testing Correlate with Indices of Coronary Vasomotion on Quantitative Coronary Angiography. J Nucl Med. 2004;45(3):419–428. doi: 10.1097/00006231-200404000-00092. [DOI] [PubMed] [Google Scholar]
- 50.Jagathesan R, Barnes E, Rosen SD, Foale RA, Camici PG. Comparison of myocardial blood flow and coronary flow reserve during dobutamine and adenosine stress: Implications for pharmacologic stress testing in coronary artery disease. J Nucl Cardiol. 2006;13(3):324–332. doi: 10.1016/j.nuclcard.2006.03.017. [DOI] [PubMed] [Google Scholar]
- 51.Hernandez-Pampaloni M, Keng FY, Kudo T, Sayre JS, Schelbert HR. Abnormal longitudinal, base-to-apex myocardial perfusion gradient by quantitative blood flow measurements in patients with coronary risk factors. Circulation. 2001;104(5):527–532. doi: 10.1161/hc3001.093503. [DOI] [PubMed] [Google Scholar]
- 52.Schindler TH, Facta AD, Prior JO, Campisi R, Inubushi M, Kreissl MC, et al. PET-measured heterogeneity in longitudinal myocardial blood flow in response to sympathetic and pharmacologic stress as a non-invasive probe of epicardial vasomotor dysfunction. Eur J Nucl Med Mol Imaging. 2006;33(10):1140–1149. doi: 10.1007/s00259-006-0069-7. [DOI] [PubMed] [Google Scholar]
- 53.Zeiher AM, Drexler H, Wollschlager H, Just H. Endothelial dysfunction of the coronary microvasculature is associated with coronary blood flow regulation in patients with early atherosclerosis. Circulation. 1991;84(5):1984–1992. doi: 10.1161/01.cir.84.5.1984. [DOI] [PubMed] [Google Scholar]
- 54.Nabel EG, Ganz P, Gordon JB, Alexander RW, Selwyn AP. Dilation of normal and constriction of atherosclerotic coronary arteries caused by the cold pressor test. Circulation. 1988;77(1):43–52. doi: 10.1161/01.cir.77.1.43. [DOI] [PubMed] [Google Scholar]
- 55.Zeiher AM, Drexler H, Saurbier B, Just H. Endothelium-mediated coronary blood flow modulation in humans. Effects of age, atherosclerosis, hypercholesterolemia, and hypertension. J Clin Invest. 1993;92(2):652–662. doi: 10.1172/JCI116634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Campisi R, Czernin J, Schoder H, Sayre JW, Schelbert HR. L-Arginine normalizes coronary vasomotion in long-term smokers. Circulation. 1999;99(4):491–497. doi: 10.1161/01.cir.99.4.491. [DOI] [PubMed] [Google Scholar]
- 57.Sato A, Terata K, Miura H, Toyama K, Loberiza FR, Jr, Hatoum OA, et al. Mechanism of vasodilation to adenosine in coronary arterioles from patients with heart disease. Am J Physiol. 2005;288(4):H1633–1640. doi: 10.1152/ajpheart.00575.2004. [DOI] [PubMed] [Google Scholar]
- 58.Zeiher AM, Drexler H, Wollschlaeger H, Saurbier B, Just H. Coronary vasomotion in response to sympathetic stimulation in humans: importance of the functional integrity of the endothelium. J Am Coll Cardiol. 1989;14(5):1181–1190. doi: 10.1016/0735-1097(89)90414-2. [DOI] [PubMed] [Google Scholar]
- 59.Schwaiger M, Kalff V, Rosenspire K, Haka MS, Molina E, Hutchins GD, et al. Noninvasive evaluation of sympathetic nervous system in human heart by positron emission tomography. Circulation. 1990;82(2):457–464. doi: 10.1161/01.cir.82.2.457. [DOI] [PubMed] [Google Scholar]
- 60.Di Carli MF, Bianco-Batlles D, Landa ME, Kazmers A, Groehn H, Muzik O, et al. Effects of autonomic neuropathy on coronary blood flow in patients with diabetes mellitus. Circulation. 1999;100(8):813–819. doi: 10.1161/01.cir.100.8.813. [DOI] [PubMed] [Google Scholar]
- 61.Di Carli MF, Tobes MC, Mangner T, Levine AB, Muzik O, Chakroborty P, et al. Effects of cardiac sympathetic innervation on coronary blood flow. N Engl J Med. 1997;336(17):1208–1215. doi: 10.1056/NEJM199704243361703. [DOI] [PubMed] [Google Scholar]
- 62.Stevens MJ, Raffel DM, Allman KC, Dayanikli F, Ficaro E, Sandford T, et al. Cardiac sympathetic dysinnervation in diabetes: implications for enhanced cardiovascular risk. Circulation. 1998;98(10):961–968. doi: 10.1161/01.cir.98.10.961. [DOI] [PubMed] [Google Scholar]
- 63.Pop-Busui R, Kirkwood I, Schmid H, Marinescu V, Schroeder J, Larkin D, et al. Sympathetic dysfunction in type 1 diabetes: association with impaired myocardial blood flow reserve and diastolic dysfunction. J Am Coll Cardiol. 2004;44(12):2368–2374. doi: 10.1016/j.jacc.2004.09.033. [DOI] [PubMed] [Google Scholar]
- 64.Kaufmann PA, Gnecchi-Ruscone T, Yap JT, Rimoldi O, Camici PG. Assessment of the reproducibility of baseline and hyperemic myocardial blood flow measurements with 15O-labeled water and PET. J Nucl Med. 1999;40(11):1848–1856. [PubMed] [Google Scholar]
- 65.Go V, Bhatt MR, Hendel RC. The diagnostic and prognostic value of ECG-gated SPECT myocardial perfusion imaging. J Nucl Med. 2004;45(5):912–921. [PubMed] [Google Scholar]
- 66.Kubo S, Tadamura E, Toyoda H, Mamede M, Yamamuro M, Magata Y, et al. Effect of caffeine intake on myocardial hyperemic flow induced by adenosine triphosphate and dipyridamole. J Nucl Med. 2004;45(5):730–738. [PubMed] [Google Scholar]
- 67.Buus NH, Bottcher M, Hermansen F, Sander M, Nielsen TT, Mulvany MJ. Influence of nitric oxide synthase and adrenergic inhibition on adenosine-induced myocardial hyperemia. Circulation. 2001;104(19):2305–2310. doi: 10.1161/hc4401.098293. [DOI] [PubMed] [Google Scholar]
- 68.Tawakol A, Forgione MA, Stuehlinger M, Alpert NM, Cooke JP, Loscalzo J, Fischman AJ, Creager MA, Gewirtz H. Homocysteine impairs coronary microvascular dilator function in humans. J Am Coll Cardiol. 2002;40(6):1051–1058. doi: 10.1016/s0735-1097(02)02069-7. [DOI] [PubMed] [Google Scholar]
- 69.Gould KL, Nakagawa Y, Nakagawa K, Sdringola S, Hess MJ, Haynie M, et al. Frequency and clinical implications of fluid dynamically significant diffuse coronary artery disease manifest as graded, longitudinal, base-to-apex myocardial perfusion abnormalities by noninvasive positron emission tomography. Circulation. 2000;101(16):1931–1939. doi: 10.1161/01.cir.101.16.1931. [DOI] [PubMed] [Google Scholar]
- 70.De Bruyne B, Hersbach F, Pijls NH, Bartunek J, Bech JW, Heyndrickx GR, et al. Abnormal epicardial coronary resistance in patients with diffuse atherosclerosis but "Normal" coronary angiography. Circulation. 2001;104(20):2401–2406. doi: 10.1161/hc4501.099316. [DOI] [PubMed] [Google Scholar]
- 71.Lim MJ, Kern MJ. Coronary pathophysiology in the cardiac catheterization laboratory. Curr Probl Cardiol. 2006;31(8):493–550. doi: 10.1016/j.cpcardiol.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 72.Kern MJ. Coronary physiology revisited : practical insights from the cardiac catheterization laboratory. Circulation. 2000;101(11):1344–1351. doi: 10.1161/01.cir.101.11.1344. [DOI] [PubMed] [Google Scholar]
- 73.Drexler H, Zeiher AM, Wollschlager H, Meinertz T, Just H, Bonzel T. Flow-dependent coronary artery dilatation in humans. Circulation. 1989;80(3):466–474. doi: 10.1161/01.cir.80.3.466. [DOI] [PubMed] [Google Scholar]
- 74.Cox DA, Vita JA, Treasure CB, Fish RD, Alexander RW, Ganz P, Selwyn AP. Atherosclerosis impairs flow-mediated dilation of coronary arteries in humans. Circulation. 1989;80(3):458–465. doi: 10.1161/01.cir.80.3.458. [DOI] [PubMed] [Google Scholar]
- 75.Campisi R, Nathan L, Pampaloni MH, Schoder H, Sayre JW, Chaudhuri G, et al. Noninvasive assessment of coronary microcirculatory function in postmenopausal women and effects of short-term and long-term estrogen administration. Circulation. 2002;105(4):425–430. doi: 10.1161/hc0402.102860. [DOI] [PubMed] [Google Scholar]
- 76.Wyss CA, Koepfli P, Namdar M, Siegrist PT, Luscher TF, Camici PG, et al. Tetrahydrobiopterin restores impaired coronary microvascular dysfunction in hypercholesterolaemia. Eur J Nucl Med Mol Imaging. 2005;32(1):84–91. doi: 10.1007/s00259-004-1621-y. [DOI] [PubMed] [Google Scholar]
- 77.Kaufmann PA, Gnecchi-Ruscone T, di Terlizzi M, Schafers KP, Luscher TF, Camici PG. Coronary heart disease in smokers: vitamin C restores coronary microcirculatory function. Circulation. 2000;102(11):1233–1238. doi: 10.1161/01.cir.102.11.1233. [DOI] [PubMed] [Google Scholar]
- 78.Wielepp P, Baller D, Gleichmann U, Pulawski E, Horstkotte D, Burchert W. Beneficial effects of atorvastatin on myocardial regions with initially low vasodilatory capacity at various stages of coronary artery disease. Eur J Nucl Med Mol Imaging. 2005;32(12):1371–1377. doi: 10.1007/s00259-005-1828-6. [DOI] [PubMed] [Google Scholar]
- 79.Nagamachi S, Czernin J, Kim AS, Sun KT, Bottcher M, Phelps ME, et al. Reproducibility of measurements of regional resting and hyperemic myocardial blood flow assessed with PET. J Nucl Med. 1996;37(10):1626–1631. [PubMed] [Google Scholar]
- 80.Siegrist PT, Gaemperli O, Koepfli P, Schepis T, Namdar M, Valenta I, et al. Repeatability of cold pressor test-induced flow increase assessed with H(2)(15)O and PET. J Nucl Med. 2006;47(9):1420–1426. [PubMed] [Google Scholar]
- 81.Schindler TH, Zhang XL, Prior JO, Cadenas J, Dahlbom M, Sayre J, et al. Assessment of intra- and interobserver reproducibility of rest and cold pressor test-stimulated myocardial blood flow with (13)N-ammonia and PET. Eur J Nucl Med Mol Imaging. 2007 Mar 3; doi: 10.1007/s00259-007-0378-5. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 82.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986;1(8476):307–310. [PubMed] [Google Scholar]
- 83.Jagathesan R, Kaufmann PA, Rosen SD, Rimoldi OE, Turkeimer F, Foale R, et al. Assessment of the long-term reproducibility of baseline and dobutamine-induced myocardial blood flow in patients with stable coronary artery disease. J Nucl Med. 2005;46(2):212–219. [PubMed] [Google Scholar]
- 84.Cai H, Harrison DG. Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res. 2000;87(10):840–844. doi: 10.1161/01.res.87.10.840. [DOI] [PubMed] [Google Scholar]
- 85.Tomai F, Crea F, Gaspardone A, Versaci F, Ghini AS, Chiariello L, et al. Unstable angina and elevated c-reactive protein levels predict enhanced vasoreactivity of the culprit lesion. Circulation. 2001;104(13):1471–1476. doi: 10.1161/hc3801.096354. [DOI] [PubMed] [Google Scholar]
- 86.Faxon DP, Fuster V, Libby P, Beckman JA, Hiatt WR, Thompson RW, et al. Atherosclerotic Vascular Disease Conference: Writing Group III: pathophysiology. Circulationn. 2004;109(21):2617–2625. doi: 10.1161/01.CIR.0000128520.37674.EF. [DOI] [PubMed] [Google Scholar]
- 87.Topol EJ, Yadav JS. Recognition of the importance of embolization in atherosclerotic vascular disease. Circulation. 2000;101(5):570–580. doi: 10.1161/01.cir.101.5.570. [DOI] [PubMed] [Google Scholar]
- 88.Bonetti PO, Lerman LO, Lerman A. Endothelial dysfunction: a marker of atherosclerotic risk. Arterioscler Thromb Vasc Biol. 2003;23(2):168–175. doi: 10.1161/01.atv.0000051384.43104.fc. [DOI] [PubMed] [Google Scholar]
- 89.Rozanski A, Gransar H, Wong ND, Shaw LJ, Miranda-Peats R, Polk D, et al. Clinical outcomes after both coronary calcium scanning and exercise myocardial perfusion scintigraphy. J Am Coll Cardiol. 2007;49(12):1352–1361. doi: 10.1016/j.jacc.2006.12.035. [DOI] [PubMed] [Google Scholar]
- 90.Nishimura RA, Lerman A, Chesebro JH, Ilstrup DM, Hodge DO, Higano ST, et al. Epicardial vasomotor responses to acetylcholine are not predicted by coronary atherosclerosis as assessed by intracoronary ultrasound. J Am Coll Cardiol. 1995;26(1):41–49. doi: 10.1016/0735-1097(95)00142-m. [DOI] [PubMed] [Google Scholar]
- 91.Schuijf JD, Wijns W, Jukema JW, Decramer I, Atsma DE, de Roos A, et al. A comparative regional analysis of coronary atherosclerosis and calcium score on multislice CT versus myocardial perfusion on SPECT. J Nucl Med. 2006;47(11):1749–1755. [PubMed] [Google Scholar]
- 92.Madjid M, Toutouzas K, Stefanadis C, Willerson JT, Casscells SW. Coronary thermography for detection of vulnerable plaques. J Nucl Cardiol. 2007;14(2):244–249. doi: 10.1016/j.nuclcard.2007.01.036. [DOI] [PubMed] [Google Scholar]
- 93.Cury RC, Nieman K, Shapiro MD, Nasir K, Cury RC, Brady TJ. Comprehensive cardiac CT study: evaluation of coronary arteries, left ventricular function, and myocardial perfusion--is it possible? J Nucl Cardiol. 2007;14(2):229–243. doi: 10.1016/j.nuclcard.2007.01.035. [DOI] [PubMed] [Google Scholar]
- 94.Kaufmann PA, Gnecchi-Ruscone T, Schafers KP, Luscher TF, Camici PG. Low density lipoprotein cholesterol and coronary microvascular dysfunction in hypercholesterolemia. J Am Coll Cardiol. 2000;36(1):103–109. doi: 10.1016/s0735-1097(00)00697-5. [DOI] [PubMed] [Google Scholar]
- 95.Yokoyama I, Ohtake T, Momomura S, Nishikawa J, Sasaki Y, Omata M. Reduced coronary flow reserve in hypercholesterolemic patients without overt coronary stenosis. Circulation. 1996;94(12):3232–3238. doi: 10.1161/01.cir.94.12.3232. [DOI] [PubMed] [Google Scholar]
- 96.Pitkanen OP, Nuutila P, Raitakari OT, Porkka K, Iida H, Nuotio I, et al. Coronary flow reserve in young men with familial combined hyperlipidemia. Circulation. 1999;99(13):1678–1684. doi: 10.1161/01.cir.99.13.1678. [DOI] [PubMed] [Google Scholar]
- 97.Munzel T, Daiber A, Ullrich V, Mulsch A. Vascular consequences of endothelial nitric oxide synthase uncoupling for the activity and expression of the soluble guanylyl cyclase and the cGMP-dependent protein kinase. Arterioscler Thromb Vasc Biol. 2005;25(8):1551–1557. doi: 10.1161/01.ATV.0000168896.64927.bb. [DOI] [PubMed] [Google Scholar]
- 98.Munzel T, Keaney JF., Jr Are ACE inhibitors a "magic bullet" against oxidative stress? Circulation. 2001;104(13):1571–1574. doi: 10.1161/hc3801.095585. [DOI] [PubMed] [Google Scholar]
- 99.Ohgushi M, Kugiyama K, Fukunaga K, Murohara T, Sugiyama S, Miyamoto E, et al. Protein kinase C inhibitors prevent impairment of endothelium-dependent relaxation by oxidatively modified LDL. Arterioscler Thromb. 1993;13(10):1525–1532. doi: 10.1161/01.atv.13.10.1525. [DOI] [PubMed] [Google Scholar]
- 100.May JM. How does ascorbic acid prevent endothelial dysfunction? Free Radic Biol Med. 2000;28(9):1421–1429. doi: 10.1016/s0891-5849(00)00269-0. [DOI] [PubMed] [Google Scholar]
- 101.Heller R, Unbehaun A, Schellenberg B, Mayer B, Werner-Felmayer G, Werner ER. L-ascorbic acid potentiates endothelial nitric oxide synthesis via a chemical stabilization of tetrahydrobiopterin. J Biol Chem. 2001;276(1):40–47. doi: 10.1074/jbc.M004392200. [DOI] [PubMed] [Google Scholar]
- 102.Eckel RH, Daniels SR, Jacobs AK, Robertson RM. America's children: a critical time for prevention. Circulation. 2005;111(15):1866–1868. doi: 10.1161/01.CIR.0000163655.15190.FB. [DOI] [PubMed] [Google Scholar]
- 103.Campisi R, Czernin J, Schoder H, Sayre JW, Marengo FD, Phelps ME, et al. Effects of long-term smoking on myocardial blood flow, coronary vasomotion, and vasodilator capacity. Circulation. 1998;982:119–125. doi: 10.1161/01.cir.98.2.119. [DOI] [PubMed] [Google Scholar]
- 104.Yokoyama I, Momomura S, Ohtake T, Yonekura K, Nishikawa J, Sasaki Y, et al. Reduced myocardial flow reserve in non-insulin-dependent diabetes mellitus. J Am Coll Cardiol. 1997;30(6):1472–1477. doi: 10.1016/s0735-1097(97)00327-6. [DOI] [PubMed] [Google Scholar]
- 105.Pitkanen OP, Nuutila P, Raitakari OT, Ronnemaa T, Koskinen PJ, Iida H, et al. Coronary flow reserve is reduced in young men with IDDM. Diabetes. 1998;47(2):248–254. doi: 10.2337/diab.47.2.248. [DOI] [PubMed] [Google Scholar]
- 106.Di Carli MF, Janisse J, Grunberger G, Ager J. Role of chronic hyperglycemia in the pathogenesis of coronary microvascular dysfunction in diabetes. J Am Coll Cardiol. 2003;41(8):1387–1393. doi: 10.1016/s0735-1097(03)00166-9. [DOI] [PubMed] [Google Scholar]
- 107.Srinivasan M, Herrero P, McGill JB, Bennik J, Heere B, Lesniak D, et al. The effects of plasma insulin and glucose on myocardial blood flow in patients with type 1 diabetes mellitus. J Am Coll Cardiol. 2005;46(1):42–48. doi: 10.1016/j.jacc.2005.03.056. [DOI] [PubMed] [Google Scholar]
- 108.Herrero P, Peterson LR, McGill JB, Matthew S, Lesniak D, Dence C, et al. Increased myocardial fatty acid metabolism in patients with type 1 diabetes mellitus. J Am Coll Cardiol. 2006;47(3):598–604. doi: 10.1016/j.jacc.2005.09.030. [DOI] [PubMed] [Google Scholar]
- 109.Al Suwaidi J, Higano ST, Holmes DR, Jr, Lennon R, Lerman A. Obesity is independently associated with coronary endothelial dysfunction in patients with normal or mildly diseased coronary arteries. J Am Coll Cardiol. 2001;37(6):1523–1528. doi: 10.1016/s0735-1097(01)01212-8. [DOI] [PubMed] [Google Scholar]
- 110.Matsuzawa Y. Therapy Insight: adipocytokines in metabolic syndrome and related cardiovascular disease. Nat Clin Pract. 2006;3(1):35–42. doi: 10.1038/ncpcardio0380. [DOI] [PubMed] [Google Scholar]
- 111.Avogaro A, de Kreutzenberg SV. Mechanisms of endothelial dysfunction in obesity. Clin Chim Acta. 2005;360(1–2):9–26. doi: 10.1016/j.cccn.2005.04.020. [DOI] [PubMed] [Google Scholar]
- 112.Bouloumie A, Marumo T, Lafontan M, Busse R. Leptin induces oxidative stress in human endothelial cells. FASEB J. 1999;13(10):1231–1238. [PubMed] [Google Scholar]
- 113.Vecchione C, Maffei A, Colella S, Aretini A, Poulet R, Frati G, et al. Leptin effect on endothelial nitric oxide is mediated through Akt-endothelial nitric oxide synthase phosphorylation pathway. Diabetes. 2002;51(1):168–173. doi: 10.2337/diabetes.51.1.168. [DOI] [PubMed] [Google Scholar]
- 114.Lembo G, Vecchione C, Fratta L, Marino G, Trimarco V, d'Amati G, et al. Leptin induces direct vasodilation through distinct endothelial mechanisms. Diabetes. 2000;49(2):293–297. doi: 10.2337/diabetes.49.2.293. [DOI] [PubMed] [Google Scholar]
- 115.Matsuda K, Teragawa H, Fukuda Y, Nakagawa K, Higashi Y, Chayama K. Leptin causes nitric-oxide independent coronary artery vasodilation in humans. Hypertens Res. 2003;26(2):147–152. doi: 10.1291/hypres.26.147. [DOI] [PubMed] [Google Scholar]
- 116.Winters B, Mo Z, Brooks-Asplund E, Kim S, Shoukas A, Li D, et al. Reduction of obesity, as induced by leptin, reverses endothelial dysfunction in obese (Lep(ob)) mice. J Appl Physiol. 2000;89(6):2382–2390. doi: 10.1152/jappl.2000.89.6.2382. [DOI] [PubMed] [Google Scholar]
- 117.Hink U, Li H, Mollnau H, Oelze M, Matheis E, Hartmann M, et al. Mechanisms underlying endothelial dysfunction in diabetes mellitus. Circ Res. 2001;88(2):E14–22. doi: 10.1161/01.res.88.2.e14. [DOI] [PubMed] [Google Scholar]
- 118.Mancini GB, Henry GC, Macaya C, O'Neill BJ, Pucillo AL, Carere RG, et al. Angiotensin-converting enzyme inhibition with quinapril improves endothelial vasomotor dysfunction in patients with coronary artery disease. The TREND (Trial on Reversing ENdothelial Dysfunction) Study. Circulation. 1996;94(3):258–265. doi: 10.1161/01.cir.94.3.258. [DOI] [PubMed] [Google Scholar]
- 119.Baller D, Notohamiprodjo G, Gleichmann U, Holzinger J, Weise R, Lehmann J. Improvement in coronary flow reserve determined by positron emission tomography after 6 months of cholesterol-lowering therapy in patients with early stages of coronary atherosclerosis. Circulation. 1999;99(22):2871–2875. doi: 10.1161/01.cir.99.22.2871. [DOI] [PubMed] [Google Scholar]
- 120.Czernin J, Barnard RJ, Sun KT, Krivokapich J, Nitzsche E, Dorsey D, et al. Effect of short-term cardiovascular conditioning and low-fat diet on myocardial blood flow and flow reserve. Circulation. 1995;92(2):197–204. doi: 10.1161/01.cir.92.2.197. [DOI] [PubMed] [Google Scholar]
- 121.Fichtlscherer S, Breuer S, Zeiher AM. Prognostic value of systemic endothelial dysfunction in patients with acute coronary syndromes: further evidence for the existence of the "vulnerable" patient. Circulation. 2004;110(14):1926–1932. doi: 10.1161/01.CIR.0000143378.58099.8C. [DOI] [PubMed] [Google Scholar]
- 122.Modena MG, Bonetti L, Coppi F, Bursi F, Rossi R. Prognostic role of reversible endothelial dysfunction in hypertensive postmenopausal women. J Am Coll Cardiol. 2002;40(3):505–510. doi: 10.1016/s0735-1097(02)01976-9. [DOI] [PubMed] [Google Scholar]
- 123.Peterson LR, Eyster D, Davila-Roman VG, Stephens AL, Schechtman KB, Herrero P, et al. Short-term oral estrogen replacement therapy does not augment endothelium-independent myocardial perfusion in postmenopausal women. Am Heart J. 2001;142(4):641–647. doi: 10.1067/mhj.2001.118111. [DOI] [PubMed] [Google Scholar]
- 124.Duvernoy CS, Rattenhuber J, Seifert-Klauss V, Bengel F, Meyer C, Schwaiger M. Myocardial blood flow and flow reserve in response to short-term cyclical hormone replacement therapy in postmenopausal women. J Gend Specif Med. 2001;4(3):21–27. 47. [PubMed] [Google Scholar]
- 125.Guethlin M, Kasel AM, Coppenrath K, Ziegler S, Delius W, Schwaiger M. Delayed response of myocardial flow reserve to lipid-lowering therapy with fluvastatin. Circulation. 1999;99(4):475–481. doi: 10.1161/01.cir.99.4.475. [DOI] [PubMed] [Google Scholar]
- 126.Huggins GS, Pasternak RC, Alpert NM, Fischman AJ, Gewirtz H. Effects of short-term treatment of hyperlipidemia on coronary vasodilator function and myocardial perfusion in regions having substantial impairment of baseline dilator reverse. Circulation. 1998;98(13):1291–1296. doi: 10.1161/01.cir.98.13.1291. [DOI] [PubMed] [Google Scholar]
- 127.Janatuinen T, Laaksonen R, Vesalainen R, Raitakari O, Lehtimaki T, Nuutila P, et al. Effect of lipid-lowering therapy with pravastatin on myocardial blood flow in young mildly hypercholesterolemic adults. J Cardiovasc Pharmacol. 2001;38(4):561–568. doi: 10.1097/00005344-200110000-00008. [DOI] [PubMed] [Google Scholar]


