Abstract
What is already known about this subject
Carnitine is essential for fatty acid metabolism, and is obtained from the diet and endogenous synthesis.
Patients with renal failure who are on chronic haemodialysis may require carnitine supplementation.
It is known that carnitine depletion in these patients may take years to develop, and that measurements of plasma carnitine may not be predictive of loss of carnitine from muscle, the main pool of carnitine in the body.
What this study adds
This paper adds a quantitative understanding of the time-course of the relationship between haemodialysis and the plasma concentration of L-carnitine, with particular reference to how the carnitine pools in the body re-equilibrate after a dialysis session.
It also provides insight into the time-course of the depletion of L-carnitine in these patients.
Aims
Patients requiring chronic haemodialysis may develop a secondary carnitine deficiency through dialytic loss of L-carnitine. A previous report has described the plasma concentrations of L-carnitine in 12 such patients under baseline conditions and after L-carnitine administration (20 mg kg−1). A three-compartment pharmacokinetic model was developed to describe these data to make inferences about carnitine supplementation in these patients.
Methods
L-carnitine removal was mediated solely by intermittent haemodialysis, which was incorporated into the model as an experimentally derived dialysis clearance value that was linked to an on-off pulse function. Data were described by a model with a central compartment linked to ‘fast’- and ‘slow’-equilibrating peripheral compartments.
Results
The model adequately described the changing plasma concentrations of endogenous L-carnitine in individual haemodialysis patients. Based on pooled data (mean ± SD; n = 12), the volume of the central compartment was 10.09 ± 0.72 l and the transfer rate constants into and out of the slowly equilibrating pool were 0.100 ± 0.018 h−1 and 0.00014 ± 0.00016 h−1, respectively. The turnover time of L-carnitine in the slow pool (which was assumed to represent muscle) was approximately 300 days. The model was in general agreement with separate data on the measured loss of carnitine from muscle in dialysis patients.
Conclusions
Haemodialysis causes rapid reductions in plasma L-carnitine concentrations with each dialysis session. Plasma concentrations are restored between sessions by redistribution from peripheral compartments. However, during chronic haemodialysis, the ongoing dialytic loss of L-carnitine may lead to a slow depletion of the compound, contributing to a possible secondary deficiency.
Keywords: haemodialysis, L-carnitine, modelling, muscle, pharmacokinetics
Introduction
Carnitine (levocarnitine [(R)-3-carboxy-2-hydroxy-N,N,N-trimethyl-1-propanaminium, inner salt]) is an endogenous compound that facilitates the transfer of long-chain fatty acids into the matrix of mitochondria, thereby delivering these substrates for β-oxidation [1]. In humans, L-carnitine is derived from dietary components and synthesized from the essential amino acids lysine and methionine in the liver and kidneys [2]. The body's L-carnitine pool (which includes L-carnitine and its short-, medium- and long-chain esters) resides primarily within skeletal and myocardial muscle, with the remainder being present in the liver, kidneys and other tissues [3]. Plasma contains only a small fraction of the total body pool of L-carnitine [4].
The healthy human kidney acts to conserve the body's L-carnitine stores via extensive (>95%) carrier-mediated tubular reabsorption [4]. However, L-carnitine is efficiently removed by haemodialysis and patients with end-stage renal disease (ESRD) undergoing chronic haemodialysis have low plasma concentrations of L-carnitine [5]. Low concentrations of L-carnitine in skeletal muscles have also been found in these patients [6, 7]. A recent study evaluated the pharmacokinetics of endogenous and exogenous L-carnitine in patients with ESRD undergoing chronic haemodialysis [8]. The patients were studied under baseline conditions, after single and multiple (9 weeks) intravenous (i.v.) doses of L-carnitine and during a 6-week wash-out period. The dose used was 20 mg kg−1 at the end of each dialysis session. In the USA, the prescribing information for the use of L-carnitine in dialysis recommends a starting dose of 10–20 mg kg−1 dry body weight; the dose may be adjusted based on trough (predialysis) L-carnitine concentration to approximately 5 mg kg−1 after about 1 month of therapy. The study showed that the removal of L-carnitine by haemodialysis was highly efficient – about 74% of L-carnitine was cleared from blood during a single passage through the dialyser. The half-life of the compound in plasma during haemodialysis (about 2 h) was significantly less than the average duration of the dialysis procedure (typically 3–4 h), leading to a 60–80% reduction in plasma L-carnitine concentrations during a single haemodialysis session. Under baseline conditions, plasma L-carnitine concentrations slowly returned to the predialysis level during the interdialysis interval, which was typically 44 or 68 h [8].
The aim of the present study was to develop a mathematical model to describe the pharmacokinetics of L-carnitine in ESRD patients undergoing chronic haemodialysis before and after a supplementary i.v. dose of exogenous carnitine. A number of compartmental pharmacokinetic models of L-carnitine have been reported [9–13]. However, they are not directly applicable to dialysis patients because they do not consider endogenous L-carnitine formation. Furthermore, they do not account for the reduced clearance of the compound in patients with ESRD and the intermittent nature of the efficient removal of the compound from the body during haemodialysis.
Methods
Pharmacokinetic data
The study that generated the datasets used in the current modelling has been described in detail elsewhere [8]. The study was approved by the Human Research Ethics Committees of the University of South Australia and the Royal Adelaide Hospital. The subjects (six male, six female) had been on maintenance haemodialysis for a minimum of 6 months and underwent three haemodialysis treatments per week throughout the study (Table 1). Patients had not received L-carnitine supplementation prior to entering the study. The study included a baseline observation period, during which blood samples were collected after a single i.v. dose of sterile normal saline. This was followed by a single dose component in which a sterile solution for injection of L-carnitine (Carnitor® 1 g in 5 ml; Sigma-tau Pharmaceuticals, Inc., Gaithersburg, MD, USA) in a volume of 0.1 ml kg−1 (corresponding to a dose of 20 mg kg−1), was administered intravenously via the patient's venous return line after it had been disconnected from the dialyser. Plasma samples were analysed for L-carnitine using a validated high-performance liquid chromatography method [8]. The limit of quantification was 0.5 µmol l−1, the accuracy ranged from −8% to 11% and the reproducibility was within 9%. The esterification of L-carnitine to acylcarnitines, of which acetylcarnitine is the most abundant, was not considered as a separate compartment, since the process is reversible and considered in equilibrium with the total body carnitine homeostasis.
Table 1.
Measured and calculated parameter values for each patient
| Patient | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | |
| Duration of haemodialysis (h)* | 3.5 | 3.5 | 3 | 4 | 4 | 3, 3.5, 4 | 4.5 | 3.5 | 4.5 | 4 | 4 | 4 |
| CLdial (l h−1) (baseline conditions) | 13.5 | 14.1 | 14.3 | 12.7 | 14.7 | 13.7 | 13.8 | 14.1 | 12.6 | 11.5 | 12.1 | 14.2 |
| CLdial (l h−1) (first dose of L-carnitine) | 12.2 | 12.3 | 17.4 | 12.2 | 16.0 | 13.5 | 12.3 | 13.0 | 13.5 | 12.2 | 12.6 | 13.2 |
| Dose of L-carnitine (mmol) at 20 mgkg−1 | 5.83 | 8.69 | 5.09 | 9.68 | 9.43 | 8.19 | 8.07 | 7.82 | 7.32 | 6.95 | 6.21 | 7.07 |
| Average output rate (umol h−1)† | 6.54 | 7.27 | 4.00 | 9.96 | 11.52 | 9.79 | 10.49 | 4.70 | 11.18 | 7.43 | 5.87 | 9.58 |
| Baseline average plasma concen-tration of L-carnitine (umol.l−1) | 16.56 | 14.46 | 13.75 | 22.77 | 23.64 | 27.54 | 12.10 | 14.45 | 27.24 | 25.10 | 15.45 | 20.50 |
| Initial amount of L-carnitine in compartment 2 (umol) | 422 | 409 | 324 | 575 | 510 | 621 | 392 | 423 | 761 | 678 | 360 | 516 |
| Initial amount of L-carnitine in compartment 3 (mmol) | 41.0 | 39.8 | 31.5 | 55.9 | 49.6 | 60.3 | 38.1 | 41.1 | 74.0 | 66.0 | 35.0 | 50.1 |
In all patients, there were three dialysis sessions per week.
This was assumed to be equal to the total input rate from all sources at steady state.
General modelling methods
Models were constructed as differential equations using the Scientist for Windows software package (Version 2.01; Micromath Scientific Software, Salt Lake City, UT, USA). The models were compiled as a series of differential equations and were solved using the Episode Stiff integration routine. For the least squares curve-fitting, the best fit was determined as that with the highest Model Selection Criteria (MSC) and without redundant parameters. The MSC is essentially an inverse Akaike Information Criterion scaled to compensate for datasets of different magnitudes and is unique to the Scientist Package. The MSC adjusts for the number of parameters in the model using Equation 1,
 |
(1) |
where Yobs is the observed value, Ycal is the calculated value, wi is a weighting term, p is the number of parameters and n is the number of data points. The higher the MSC, the better the fit. Generally, a MSC >5 indicates an extremely good fit, while a MSC < 1 indicates a poor fit. All data were given y−1 weighting for curve-fitting.
Initial model development was conducted using mean plasma concentration vs. time data for ‘exogenous’ L-carnitine after single dose administration. This dataset was obtained by subtracting the mean plasma concentration of L-carnitine under baseline conditions from that after the i.v. dose of L-carnitine at the corresponding sampling time post dialysis. This dataset was useful for initial model development, since it did not require estimates of initial carnitine quantities in the various compartments (as exogenous levels could be assumed to be zero) and endogenous production could be ignored. The dataset did not include the plasma concentrations for the haemodialysis period, during which there was a dramatic fall in plasma L-carnitine concentrations. This initial development step identified a simple three-compartment mamillary model as being the most appropriate for describing the disposition of intravenously dosed ‘exogenous’ L-carnitine.
Intermittent clearance
A unique feature of L-carnitine disposition in the ESRD patients is that elimination of the compound from the body occurs almost exclusively during the haemodialysis period. This required the development of a method for incorporating intermittent clearance into the pharmacokinetic model. This was achieved by linking clearance to a pulse function for which the start, stop and height of the pulse (clearance) could be specified. The dialysis clearance value (CLDIAL) that was used in the model was calculated for each patient from the rate of removal of L-carnitine via the dialysate divided by the concentration of L-carnitine in plasma entering the dialyser, as described previously [8]. The dialysis clearance values for individual patients, and time schedules, are reported in Table 1.
Dosing and endogenous production
Exogenous L-carnitine administration was incorporated into the model as bolus input into the central compartment. Unlike conventional drugs, L-carnitine is an endogenous compound that normally enters the body via endogenous synthesis and dietary intake. For the pharmacokinetic model, the overall rate of L-carnitine input into the body (biosynthesis plus absorption from the diet) was incorporated as a constant zero order rate of input into the central compartment. To estimate this input rate for individual subjects, it was assumed that under baseline conditions (where no supplementary L-carnitine was being administered) the rate of loss of L-carnitine from the body by haemodialysis was equal to the average rate of endogenous L-carnitine input. This assumption is based on the fact that only patients who had been on haemodialysis for a least 6 months were enrolled in the study. The average length of time that the patients had been receiving dialysis was 3 years and 11 months (± 58 months); therefore, the patients were likely to be in a state of pseudo-equilibrium with respect to endogenous L-carnitine mass balance. Under these circumstances, there will be fluctuations in the carnitine pools of the body with each dialysis session, but there will be no net change in the average amount in each pool. Thus, under these steady-state conditions the rate of input of L-carnitine into the body is equal to the rate of loss from the body. Further, since there appears to be no significant contribution of metabolism or nonrenal excretion to the clearance of the L-carnitine, loss via dialysate is likely to represent the major route of L-carnitine elimination in these patients. Therefore, for each patient, the measured amount of total L-carnitine recovered in outflow dialysate under baseline conditions [8] was multiplied by three (dialysis sessions per week) to calculate the total weekly loss, and this value was used to estimate the average hourly endogenous input rate of L-carnitine. Values for individual subjects ranged from 4.00 to 11.52 µmol h−1 and are reported in Table 1.
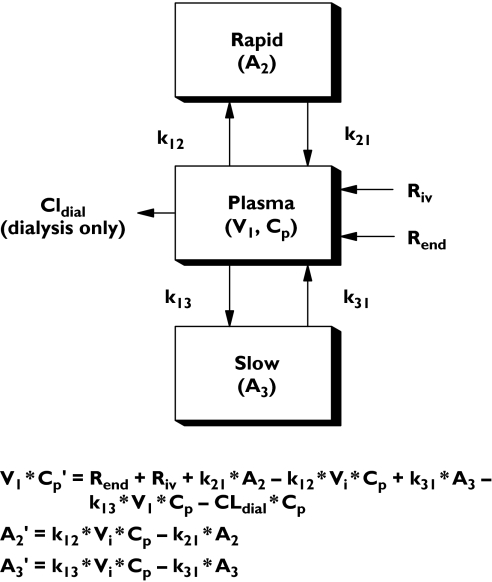
Model equations and initial values
The final pharmacokinetic model and the critical differential equations are given in Figure 1. An important requirement for integrating differential equations is that the initial values of all variables (e.g. concentrations and/or amounts of drug in each compartment) must be specified. While it is safe to assume these values are zero when a drug is administered for the first time, this is not the case for endogenous compounds such as L-carnitine. Although the concentration of L-carnitine in plasma (it was assumed that this represented the concentration in the central compartment) was measured for each patient, the initial values for the content of L-carnitine in the two peripheral compartments needed to be estimated from the known distribution pattern of endogenous L-carnitine in the human body.
Figure 1.
The pharmacokinetic model used to describe L-carnitine in end-stage renal disease patients undergoing intermittent haemodialysis. V1 is the volume of the central compartment with concentration Cp. A2 and A3 are the amounts in the peripheral compartments, as governed by the rate constants k12, k21, and k13 and k31, respectively. CLDIAL is the clearance that occurred intermittently (only during the dialysis periods). Riv is the intermittent rate of administration of exogenous carnitine corresponding to intravenous doses; Rend is the constant rate of endogenous carnitine input from dietary and metabolic sources. The rapidly and slowly equilibrating peripheral compartments were nominally attributed to liver and muscle, respectively. Cp′, A2′ and A3′ are notation for derivatives with respect to time
The distribution of L-carnitine within the body of healthy individuals has been reported by Brass [14]. While the normal extracellular fluid concentration of total L-carnitine was reported to be 40 µmol l−1, the normal amounts in rapidly equilibrating (e.g. liver) and slowly equilibrating (e.g. muscle) peripheral pools were 1.3 and 126.4 mmol, respectively. Assuming that the haemodialysis patients were at, or near, steady state with respect to L-carnitine disposition prior to starting our study, and assuming first-order exchange kinetics between plasma and muscle, it follows that the tissue pools of L-carnitine would have been depleted to the same extent as the corresponding concentrations in the central compartment. The initial L-carnitine content of the rapidly and slowly equilibrating peripheral compartments were therefore calculated using the following relationships:
 |
(2) |
 |
(3) |
where Cav,baseline is the average concentration of L-carnitine in plasma (compartment 1) in the haemodialysis patients (µmol l−1). This average plasma concentration was calculated by trapezoidal integration of the measured concentrations over the interdialysis period (from the end of the mid-week dialysis treatment until the start of the next, third dialysis session of the week), divided by the duration of the interval (typically 43–44 h). The values for individual patients ranged from 12.1 to 27.5 µmol l−1 (Table 1), which are substantially lower that the plasma concentrations that are considered to be representative of healthy humans, about 40 µmol l−1[11]. The derived initial values for the amount of L-carnitine in compartments 2 and 3 are given for each patient in Table 1.
Modelling baseline and single-dose datasets
The three-compartment model with endogenous production and intermittent clearance (Figure 1) was fitted simultaneously to the baseline and single-dose dataset for each patient and for pooled (average) data. Fixed parameters for each patient were the dialysis clearance value, calculated as described above and in Evans et al.[8], the start and stop times of each haemodialysis session (three sessions per week) and the rate of endogenous production. Initial values were the estimated amounts of L-carnitine within the two peripheral compartments (Table 1) and the measured initial plasma concentration of L-carnitine at t = 0. Fitted parameters (Table 2) were the volume of the central compartment (V1) and the rate constants (h−1) for the transfer of L-carnitine into and out of the rapidly equilibrating (k12 and k21, respectively) and slowly equilibrating (k13 and k31, respectively) peripheral compartments. The model parameters were also expressed as derived values that provide a better insight into the kinetic processes governing L-carnitine disposition. The apparent distribution volumes of the two peripheral compartments were calculated using Equations 4 and 5:
| (4) |
| (5) |
High apparent distribution volumes are associated with large quantities of L-carnitine stored in the compartment at steady state. The turnover time (T) of L-carnitine within each compartment was calculated as the reciprocal of the efflux rate constant, as follows:
| (6) |
| (7) |
The turnover time represents the mean time that a molecule spends within the compartment after entering it. A high turnover time indicates that the L-carnitine in the compartment is only slowly exchanged with carnitine in the central compartment. Equilibration is approximately 66% complete after one turnover time and 95% complete after three turnover times.
Table 2.
Fitted and derived parameters for the three-compartment model that was used to describe the pharmacokinetics of L-carnitine in end-stage renal disease patients undergoing haemodialysis
| Patient | MSC | V1 (l) | k12 (h−1) | k21 (h−1) | k13 (h−1) | k31 (h−1) | V2app (l) | V3app (l) | T2 (h) | T3 (h) | Ac,base (µmol) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.97 | 11.84 | 0.127 | 0.118 | 0.082 | 0.000056 | 12.74 | 17426 | 8.47 | 17949 | 196.1 |
| 2 | 1.76 | 11.29 | 0.166 | 0.07 | 0.056 | 0.000091 | 26.77 | 6940 | 14.29 | 10977 | 163.3 |
| 3 | 2.5 | 7.04 | 0.328 | 0.237 | 0.179 | 0.000205 | 9.74 | 6139 | 4.22 | 4871 | 96.8 |
| 4 | 3.13 | 11.8 | 0.083 | 0.196 | 0.138 | 0.000438 | 5.00 | 3718 | 5.10 | 2283 | 268.7 |
| 5 | 2.15 | 12.92 | 0.191 | 0.091 | 0.126 | 0.000393 | 27.12 | 4147 | 10.99 | 2547 | 305.4 |
| 6 | 2.22 | 11.88 | 0.142 | 0.105 | 0.084 | 0.000078 | 16.07 | 12741 | 9.52 | 12768 | 327.2 |
| 7 | 4.46 | 5.52 | 4.422 | 2.763 | 0.388 | 0.001685 | 8.83 | 1271 | 0.36 | 594 | 66.8 |
| 8 | 2.13 | 9.81 | 0.126 | 0.104 | 0.098 | 0.000083 | 11.89 | 11649 | 9.62 | 12117 | 141.8 |
| 9 | 2.33 | 7.93 | 0.249 | 0.059 | 0.039 | 0.000076 | 33.47 | 4073 | 16.95 | 13170 | 216.0 |
| 10 | 3.11 | 8.19 | 0.271 | 0.138 | 0.092 | 0.092243 | 16.08 | 8 | 7.25 | 11 | 205.6 |
| 11 | 1.52 | 7.66 | 0.178 | 0.058 | 0.061 | 0.000008 | 23.51 | 55261 | 17.24 | 118266 | 118.3 |
| 12 | 1.64 | 11.88 | 0.116 | 0.065 | 0.094 | 0.000128 | 21.20 | 8710 | 15.38 | 7800 | 243.5 |
| Median | 2.19 | 10.55 | 0.170 | 0.100 | 0.090 | 0.000110 | 16.07 | 6539 | 9.57 | 9388 | 200.8 |
| Pooled data* | 2.26 | 10.09 | 0.162 | 0.106 | 0.100 | 0.00014 | 15.42 | 7207 | 9.43 | 7143 | 214.7 |
| Pooled SD | 0.72 | 0.07 | 0.07 | 0.018 | 0.00016 | 12.22 | 5834 | 6.23 | 1286 | 15.3 |
The symbols used for the fitted and derived parameters are described in the text.
Values obtained by fitting model to pooled plasma concentration vs. time data.
The amount of L-carnitine in the central pool, under baseline conditions (Ac,base), was estimated from the estimated volume of the central compartment and the measured average concentration of L-carnitine in plasma, according to Equation 8:
| (8) |
Comparison with L-carnitine muscle concentrations in patients
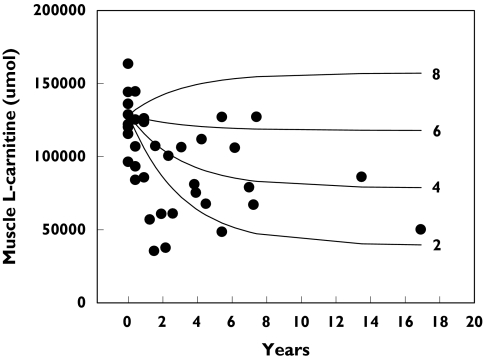
The model was used to simulate the time course of the muscle content of L-carnitine in a typical patient with a normal carnitine pool [40 µmol l−1 in plasma, 900 µmol in compartment 2, 128 000 µmol in compartment 3 (muscle)], who had commenced regular dialysis after a sudden loss of renal function. An average continuous clearance of 0.367 µmol h−1 was used, which was shown to approximate to three dialysis sessions per week as per the patients in Table 1. The endogenous production of carnitine was set at 2, 4, 6 and 8 µmol h−1. The simulations were compared with published data on the muscle concentration of L-carnitine in patients who had been on dialysis between 0 and 17 years (28 measurements in 36 patients) [15]. The muscle concentration was converted to muscle content assuming a body mass of 70 kg, which consisted of 43% muscle [14].
Results
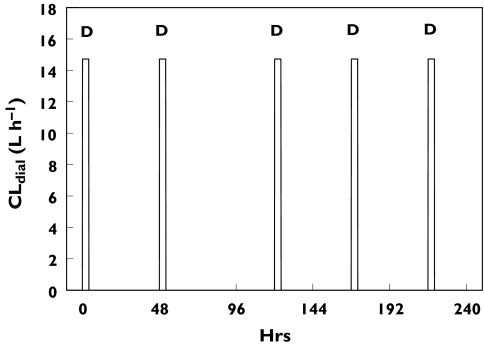
A critical feature of the pharmacokinetic model was that of intermittent clearance, which was incorporated as a series of pulse functions that could be programmed for each individual patient depending on their dialysis schedule and the calculated dialysis clearance. An example of the time course of clearance for one particular patient is shown in Figure 2. By incorporating this pulsed clearance, it was possible to accommodate the dramatic change in the pharmacokinetics of L-carnitine that occurs when haemodialysis is commenced and stopped.
Figure 2.
An example of the output of the algorithm used for intermittent clearance. In this instance, the algorithm described the haemodialysis for patient 5, who received five periods of dialysis (CLDIAL, 14.7 l h−1 for 4 h) during the modelled period. Each period is marked with the symbol ‘D’
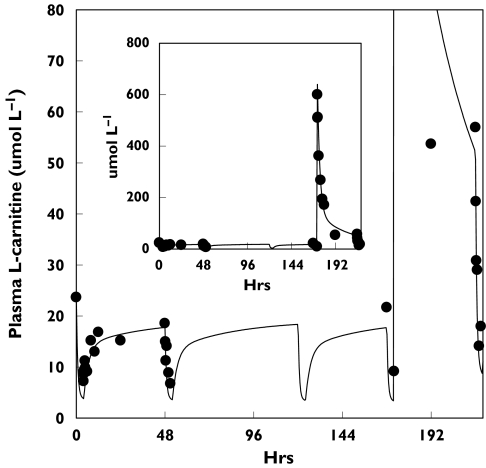
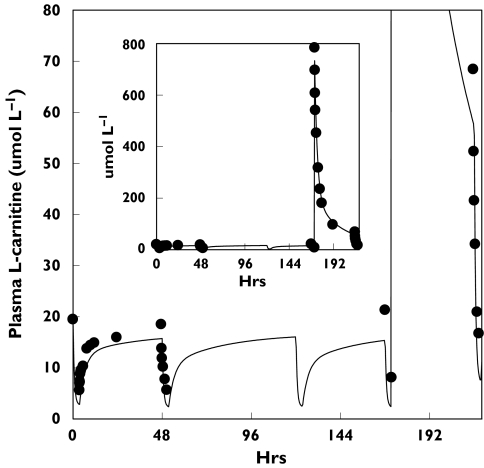
The pharmacokinetic model was capable of describing plasma L-carnitine in each patient during the baseline period and after the first dose of L-carnitine. In all cases, convergence was achieved within a reasonable time and the MSC values ranged between 1.52 and 4.46, indicating a satisfactory to good overall fit. Using values for the initial amounts in the peripheral compartments that differed widely from those calculated using Equations 2 and 3 resulted in a poorer fit to the data, providing some support for the underlying assumptions of these equations. The line of best fit for a representative patient is presented in Figure 3, whereas the fit for the pooled data is given in Figure 4.
Figure 3.
Plasma concentration vs. time profiles for L-carnitine in a representative end-stage renal disease patient (patient 5) under baseline conditions and after a single intravenous dose of L-carnitine (20 mg kg−1). The symbols represent measured concentrations, and the continuous line represents the concentrations predicted by the fitted model
Figure 4.
Plasma concentration vs. time profiles for L-carnitine in 12 end-stage renal disease patients under baseline conditions and after a single intravenous dose of L-carnitine (20 mg kg−1). The symbols represent pooled concentrations from the 12 patients, and the continuous line represents the concentrations predicted by the fitted model
The fitted and derived pharmacokinetic parameters for each patient are presented in Table 2 together with the parameters obtained from fitting the pooled data. There was considerable interpatient variability, in particular, values obtained for patient 10, which were much lower than those obtained in all other patients. However, the median parameter values for all patients were similar in magnitude to those obtained by fitting the model to the pooled data (Table 2).
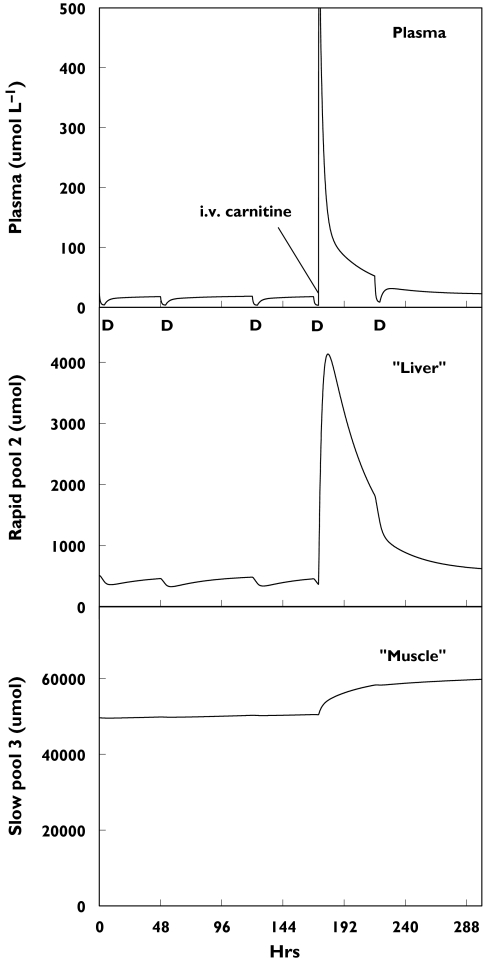
The pharmacokinetic model, using parameters from the analysis of pooled data, was used to simulate the content of the two peripheral compartments during routine dialysis and after a single dose of L-carnitine (Figure 5). This figure shows that during a single haemodialysis session under baseline conditions, the dramatic fall in plasma L-carnitine concentrations (approximately 80%) coincides with a fall of about 30% in the amount of L-carnitine in the rapidly equilibrating compartment. Over the same time, the amount of L-carnitine in the slow-equilibrating pool changes negligibly. After the end of dialysis, the concentrations in plasma and the rapidly exchanging compartment increase, but again, negligible change is observed in the slowly equilibrating pool. The situation is reversed after i.v. administration of L-carnitine. There is the expected large increase in plasma concentrations, followed by a multiexponential decline. The additional L-carnitine is initially transferred into the rapidly equilibrating pool, and thereafter transfers into the slow-equilibrating pool, which continues to increase for some time after the carnitine administration.
Figure 5.
Simulated amounts of L-carnitine in the three compartments of the pharmacokinetic model in patient 5 undergoing intermittent haemodialysis three times per week
The simulation of the normal loss of carnitine from muscle with the onset of dialysis is shown in Figure 6. It is clear that, for the overall clearance chosen, the development of depleted muscle stores of carnitine was dependent on the rate of endogenous production (via diet and synthesis). When loss did occur, the time course of the loss was in general agreement with the observed muscle content data, suggesting that the rate constants of the model, particularly for the muscle compartment, were reasonable estimates.
Figure 6.
The predicted and observed muscle contents of L-carntine when an individual with normal carnitine pools is placed on dialysis. The symbols show the muscle carnitine contents estimated from measures of muscle carnitine concentration in dialysis patients before and during continuous dialysis for up to 17 years. The lines show the muscle content predicted using the model with initially normal carnitine pools and with parameter values and dialysis conditions set at average values and with endogenous production set at either 2, 4, 6 or 8 µmol h−1. The extent of depletion of the muscle carnitine depended on the balance between the loss of carnitine via dialysis and the rate of inputs (from diet, endogenous synthesis and supplement)
Discussion
The disposition of L-carnitine, in patients with ESRD undergoing chronic haemodialysis, was adequately described by a three-compartment model in which the substrate was cleared from the central compartment during haemodialysis. The model allowed the dialysis conditions for each individual patient to be programmed via a pulse function. It was therefore possible to incorporate the haemodialysis regimen that is tailored for each individual patient. The model also incorporated endogenous input of L-carnitine into the central compartment.
There have been a number of previous attempts to describe the pharmacokinetics of L-carnitine using compartmental models [11, 12]. The most significant of these earlier studies was that of Rebouche and Engel in 1984 [11]. These workers administered a tracer dose of methyl-3H-L-carnitine to six healthy subjects as well as one patient with primary muscle L-carnitine deficiency and another four patients with primary systemic carnitine deficiency. The use of radiolabelled L-carnitine permitted the exogenous material to be distinguished from endogenous L-carnitine. Interestingly, a three-compartment model similar to that described in the current study was suitable for describing the disappearance profile of methyl-3H-L-carnitine. The turnover time for methyl-3H-L-carnitine in the rapidly equilibrating peripheral compartment of Rebouche and Engel was 11.6 h, which compares favourably with the 9.4 h found for the pooled data in the current study. However, the turnover time values for the slow-equilibrating peripheral compartment are remarkably different: 191 h [11]vs. 7413 h (Table 2). Whether this reflects differences between healthy individuals and ESRD patients in the retention of L-carnitine in skeletal muscle is not known. It is tempting to speculate that the muscle depletion that has been reported to occur in ESRD patients receiving chronic haemodialysis may in some way lead to stronger retention of the remaining compound. However, the difference may also be due to imprecision in the estimation of the rate constants and turnover times for the slow-equilibrating compartment.
Rebouche and Engel [11] estimated the amount of L-carnitine in the central pool to range between 419 and 1327 µmol. In the current study, the values obtained under baseline conditions (product of average plasma concentration and the volume of the central compartment) were substantially lower, ranging between 67 and 305 µmol (Table 2). Indeed, the values found in the ESRD patients are closer in magnitude to the range of values (69–158 µmol) reported for four patients with primary systemic carnitine deficiency [11].
Uematsu et al.[13] used a three-compartment open model to describe the pharmacokinetics of i.v. L-carnitine. However, their model did not incorporate endogenous synthesis. Instead, the plasma concentrations were corrected for the presence of endogenous L-carnitine prior to modelling. Interestingly, the volume of the central compartment (0.108–0.198 l kg−1 at a mean weight of 68.3 kg) was comparable to that determined in the current study (Table 2). Their clearance value was about 0.1 l h−1, or 1–2 ml min−1, and about 90% of the dose was recovered in urine (0–24 h). The fact that L-carnitine was cleared from the body of the healthy subjects almost exclusively by renal excretion supports the assumption used in the current modelling, that nondialytic elimination of L-carnitine in ESRD patients was negligible.
Segre et al.[12] used a three-compartment model to describe the pharmacokinetics of i.v. L-carnitine in healthy subjects, whereas Harper et al.[10] and Glöggler et al.[9] found that a two-compartment model was sufficient. However, these latter studies measured plasma L-carnitine only for a relatively short time period (< 10 h) after dosing, meaning that slow kinetic events would be not be discernible.
A consistent finding from all attempts to model the disposition of L-carnitine is that the volume of the central compartment corresponds to the approximate volume of the extracellular fluid component of the body. Another consistent finding, which is in keeping with the known distribution of L-carnitine, is the presence of a deep compartment that equilibrates relatively slowly with L-carnitine in plasma. The fact that >98% of the total body store resides outside of plasma [14, 16] substantiates the existence of this pool. The ratios between the microrate constants for the movement of L-carnitine into and out of the peripheral compartment are 1.53 for compartment 2 and 714 for compartment 2 (pooled parameters). The fact that the efflux rate constants are lower than the corresponding influx constant is in keeping with the involvement of a carrier-mediated process for the uptake of L-carnitine into tissues such as the liver and muscle (L-carnitine is not bound to blood or tissue components).
The current study has found that although the plasma concentrations of L-carnitine change substantially during a single haemodialysis session (Figures 3 and 4), there is a comparatively minor change in the predicted concentrations of the compound in the two peripheral compartments, particularly in compartment 3, which has been allocated to represent skeletal muscle (Figure 5). The relatively slow equilibration of L-carnitine between the central and the peripheral compartments means that it may not be feasible to make an assessment of the ‘carnitine status’ of a patient from plasma concentrations alone. For example, one can envisage a situation in which a patient may have high plasma L-carnitine concentrations due to the recent administration of an i.v. dose of L-carnitine, but skeletal muscle concentrations could still be much lower than normal. Conversely, a low L-carnitine concentration in plasma at the end of a haemodialysis session does not imply a deficiency within skeletal muscle. The long turnover time of L-carnitine in the slow equilibrating pools also implies that it would take many months, if not years, to deplete or replenish this peripheral compartment to a significant amount. Indeed, with an average efflux rate constant of 0.00014 h−1 for the muscle compartment, it would take about 200 days for a 50% change in muscle content to occur if the rate of input (or clearance) was altered. This was supported by the comparison of the model predictions with muscle contents in patients (Figure 6). In general, the loss of carnitine from muscle was predicted to occur over a period of years when there are slight mismatches between loss due to dialysis and gain due to endogenous production. This is in keeping with the concepts that the development of symptoms of L-carnitine deficiency in ESRD patients receiving haemodialysis may not be apparent until that patient has been treated for some time [5], and that changes in the skeletal muscle content of L-carnitine require many months of treatment with pharmaceutical formulations [14].
As is always the case when applying a model to pharmacokinetic data, it is important to evaluate the assumptions on which the model was based, and to consider the consequences if any of these assumptions are violated. In the current model, a number of fundamental assumptions were made. First, it was assumed that haemodialysis was the only method for the removal of L-carnitine from the body of patients with ESRD. This assumption is based on the premise that L-carnitine is normally removed from the body primarily by renal excretion, that there was no residual renal function in these patients [8] and the fact that the metabolic clearance of L-carnitine is negligible [4]. Although L-carnitine undergoes acylation to acetyl-L-carnitine and other short- and long-chain esters, this is a reversible metabolic process that is functionally incorporated into the model within the ‘distribution’ rate constants. The acylation of L-carnitine was therefore not modelled separately in the kinetic analysis. When the model was modified by incorporation of a first-order elimination rate constant from the central compartment (representing residual clearance), there was no overall improvement in the quality of the fit and the rate constant was poorly defined.
The calculated endogenous input rate in the 12 haemodialysis patients (overall mean of 8.2 µmol h−1) was markedly lower than the value of >20 µmol h−1 reported by Rebouche and Engel [11] in normal patients, but is compatible with their reported values in patients with systemic carnitine deficiency (3–7 µmol h−1). The lower than normal input rate is consistent with a lower dietary L-carnitine intake in dialysis patients on a low-protein diet, and possibly lower biosynthesis due to the impaired kidneys. In the model, endogenous input of L-carnitine was added to the central compartment – a trial in which the production was added to a peripheral compartment showed no improvement in fit. Attempts were made to determine the endogenous input rate of L-carnitine as a fitted parameter, but this led to imprecise estimates of input, suggesting that it could not be reliably determined by modelling of the present data.
Another fundamental assumption implicit in the final model is that the movement of L-carnitine into and out of the peripheral compartments could be described by first-order rate constants. The uptake of L-carnitine from plasma into liver and muscle has been reported to be a carrier-mediated process that allows the compound to accumulate against a significant concentration gradient. Bremer [1] reported km values for these saturable uptake processes of 500 µmol l−1 in isolated muscle and 5000 µmol l−1 in liver cells. If the concentration bathing these tissues greatly exceeds this km value, then the uptake would be expected to demonstrate nonlinear characteristics. However, more recently, lower km values have been reported in isolated muscle cell membrane vesicle preparations and other models [17]. However, even above the km values, uptake by systems with lower affinities may mean that the membrane transport of the compound is a mix of apparent zero- and first-order processes. The highest plasma concentration achieved in the present study was approximately 1200 µmol l−1, immediately after the i.v. dose of L-carnitine, which is sufficient to suggest that nonlinearity may be an issue for skeletal muscle uptake. A rate constant for uptake into a peripheral compartment ‘e.g. k13’ can be made nonlinear by replacing it with the term [Vmax/(km + Cp)]/V1, where Vmax is the maximum possible transport rate and km is the plasma concentration at which the transport rate is half maximal. Models were constructed with nonlinear uptake for both the liver and muscle compartments of the pharmacokinetic model, with various initial values of km (suggested by the literature). However, there was no improvement in model fit by assuming nonlinear kinetics and this approach was not pursued further. It is important to keep in mind that using an in vitro study of human cultured muscle, Martinuzzi et al.[18] have shown that at very high concentrations of L-carnitine, >10 mmol l−1, a significant uptake into the cell, probably via passive diffusion, was still seen. Therefore, based on these results, it could be predicted that the transport of L-carnitine into muscle in vivo is not saturated at the supraphysiological concentrations obtained following i.v. administration.
The model also assumed that the dialysis clearance value determined during one of the three weekly dialysis sessions was applicable to the other dialysis sessions of the same week. This assumption is justified, in that the dialysis schedule for all patients enrolled in this study remained constant throughout the study. Further, it had been shown previously that the dialysis clearance of L-carnitine remained unchanged over a 10-week observation period [8].
In summary, a three-compartment pharmacokinetic model was shown to be capable of describing the changing plasma concentrations of L-carnitine in patients with ESRD receiving chronic treatment with haemodialysis. The model was able to describe the pharmacokinetics of endogenous and exogenous L-carnitine administered intravenously at the end of a haemodialysis session. The model may prove to be useful for assessing the relative changes in the plasma and tissue concentrations of L-carnitine in ESRD patients who have developed a secondary carnitine deficiency caused by the imbalance that occurs between the input of L-carnitine (via endogenous synthesis and dietary intake) and its efficient removal by haemodialysis.
Competing interests
G.F. is an executive employee of Sigma-tau Pharmaceuticals Inc. Sigma-tau is the manufacturer of Carnitor® (levocarnitine) injection for the treatment of carnitine deficiency in end-stage reneal disease patients undergoing chronic haemodialysis.
Supported by Sigma-tau Pharmaceuticals Inc, Gaithersburg, MD 20877, USA.
References
- 1.Bremer J. Carnitine – metabolism and functions. Physiol Rev. 1983;63:1420–80. doi: 10.1152/physrev.1983.63.4.1420. [DOI] [PubMed] [Google Scholar]
- 2.Vaz FM, Wanders RJA. Carnitine biosynthesis in mammals. Biochem J. 2000;361:417–29. doi: 10.1042/0264-6021:3610417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rebouche CJ. Kinetics, pharmacokinetics, and regulation of L-carnitine and acetyl-L-carnitine metabolism. Ann NY Acad Sci. 2004;1033:30–41. doi: 10.1196/annals.1320.003. [DOI] [PubMed] [Google Scholar]
- 4.Evans AM, Fornasini G. Pharmacokinetics of L-carnitine. Clin Pharmacokinet. 2003;42:941–67. doi: 10.2165/00003088-200342110-00002. [DOI] [PubMed] [Google Scholar]
- 5.Ahmad S. L-carnitine in dialysis patients. Semin Dialysis. 2001;14:209–17. doi: 10.1046/j.1525-139x.2001.00055.x. [DOI] [PubMed] [Google Scholar]
- 6.Debska-Slizien A, Kawecka A, Wojnarowski K, Prajs J, Malgorzewicz S, Kunicka D, Zdrojewski Z, Lysiak-Szydlowska W, Lipinski J, Rutkowski B. Correlation between plasma carnitine, muscle carnitine and glycogen levels in maintenance hemodialysis patients. Int J Artif Organs. 2000;23:90–6. [PubMed] [Google Scholar]
- 7.Hiatt WR, Koziol BJ, Shapiro JI, Brass EP. Carnitine metabolism during exercise in patients on chronic hemodialysis. Kidney Int. 1992;41:1613–9. doi: 10.1038/ki.1992.233. [DOI] [PubMed] [Google Scholar]
- 8.Evans AM, Faull R, Fornasini G, Lemanowicz E, Longo A, Pace S, Nation RL. Pharmacokinetics of L-carnitine in patients with end-stage renal disease undergoing long term dialysis. Clin Pharm Ther. 2000;68:238–49. doi: 10.1067/mcp.2000.108850. [DOI] [PubMed] [Google Scholar]
- 9.Glöggler A, Bulla M, Fürst P. Kinetics of intravenously administered carnitine in haemodialysed children. J Pharmaceut Biomed Anal. 1990;3:411–4. doi: 10.1016/0731-7085(90)80068-z. [DOI] [PubMed] [Google Scholar]
- 10.Harper P, Elwin C-E, Cederblad G. Pharmacokinetics of bolus intravenous and oral doses of L-carnitine in healthy subjects. Eur J Clin Pharmacol. 1988;35:69–75. doi: 10.1007/BF00555510. [DOI] [PubMed] [Google Scholar]
- 11.Rebouche CJ, Engel AG. Kinetic compartmental analysis of carnitine metabolism in the human carnitine deficiency syndromes. Evidence for alterations in tissue carnitine transport. J Clin Invest. 1984;73:857–67. doi: 10.1172/JCI111281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Segre G, Bianchi E, Corsi M, D'Iddio S, Ghirardi O, Maccari F. Plasma and urine pharmacokinetics of free and of short-chain carnitine after administration of carnitine in man. Arzneimittelforschung. 1988;38:1830–4. [PubMed] [Google Scholar]
- 13.Uematsu T, Itaya T, Nishimoto M, Takiguchi Y, Mizuno A, Nakashima M, Yoshinobu K, Hasebe T. Pharmacokinetics and safety of L-carnitine infused i.v. in healthy subjects. Eur J Clin Pharmacol. 1988;34:213–6. doi: 10.1007/BF00614562. [DOI] [PubMed] [Google Scholar]
- 14.Brass EP. Pharmacokinetic considerations for the therapeutic use of carnitine in hemodialysis patients. Clin Ther. 1995;17:176–85. doi: 10.1016/0149-2918(95)80017-4. [DOI] [PubMed] [Google Scholar]
- 15.Evans AM, Faull RJ, Nation RL, Prasad S, Elias T, Reuter SE, Fornasini G. Impact of hemodialysis on endogenous plasma and muscle carnitine levels in patients with end-stage renal disease. Kidney Int. 2004;66:1527–34. doi: 10.1111/j.1523-1755.2004.00916.x. [DOI] [PubMed] [Google Scholar]
- 16.Mitchell M. Carnitine metabolism in human subjects II. Values of carnitine in biological fluids and tissues of ‘normal’ subjects. Am J Clin Nutr. 1978;31:481–91. doi: 10.1093/ajcn/31.3.481. [DOI] [PubMed] [Google Scholar]
- 17.Angelini C, Vergani L, Martinuzzi A. Clinical and biochemical aspects of carnitine deficiency and insufficiency: transport defects and inborn errors of beta-oxidation. Crit Rev Clin Lab Sci. 1992;29:217–42. doi: 10.3109/10408369209114601. [DOI] [PubMed] [Google Scholar]
- 18.Martinuzzi A, Vergani L, Rosa M, Angelini C. L-carnitine uptake in differentiating human cultured muscle. Biochimica Biophysica Acta. 1991;1095:217–22. doi: 10.1016/0167-4889(91)90102-4. [DOI] [PubMed] [Google Scholar]