Abstract
Background/aims
Quantitative data regarding the impact of neovascular age‐related macular degeneration (NV‐AMD) on individuals and society is a prerequisite for rational decision‐making processes when evaluating alternative treatments for the disease.
Methods
75 bilateral NV‐AMD (patients) and 91 elderly non‐AMD (controls) subjects forming the UK cohort of an international cross‐sectional, observational study were independently analysed. Subjects completed a telephone survey including the National Eye Institute Visual Function Questionnaire (NEI‐VFQ‐25), the EuroQol (EQ‐5D), the Hospital Anxiety and Depression Scale (HADS), history of falls and health resource utilisation.
Results
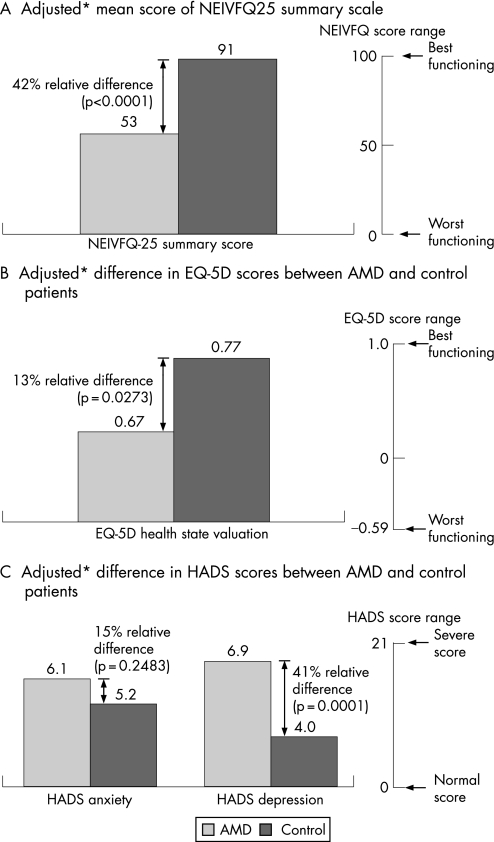
Patients with NV‐AMD reported substantially worse vision‐related functioning and overall well‐being, including higher depression scores, than controls after adjusting for age, gender and co‐morbidities (adjusted mean scores: NEI‐VFQ‐25 overall 52.7 vs 90.7, p<0.0001; EQ‐5D 0.67 vs 0.77, p = 0.0273; HADS depression 6.8 vs 4.0, p = 0.0026). Significantly more patients reported a need for assistance with daily activities compared with controls (25.3% vs 6.6%, p = 0.003). Total annual healthcare utilisation costs were more than sevenfold higher for patients with AMD compared with controls (£3,823.89 vs £517.05, respectively; p<0.0001)
Conclusions
Patients with NV‐AMD show a significant decline in quality of life and increased need for daily living assistance compared to a control population without AMD. With the availability of effective new therapies there is a need for improved early access to treatment.
Age‐related macular degeneration (AMD) is the leading cause of blindness in people over 65 years of age.1 Around 1.7% of individuals aged over 50 years of age have end stage (blinding) AMD, with the incidence rising substantially with age.2 Neovascular (“wet”) AMD (NV‐AMD) represents around 15% of cases of AMD,3 and arises due to abnormal angiogenesis beneath the retina (choroidal neovascularisation), and within the retina (retinal angiomatous proliferation).4 NV‐AMD is more sight‐threatening than the geographic (“dry”) type, accounting for 90% of AMD‐related vision loss in older people. The majority of patients with NV‐AMD progress to blindness in the affected eye within 2 years of diagnosis and there is a 43% probability of progression to NV‐AMD in the fellow eye within 5 years.5 The annual incidence of NV‐AMD is conservatively estimated at 130 per typical health authority of 500 000 patients (0.26 per 1000 patients).6 In the UK in 2005, the annual incidence of NV‐AMD was calculated to be around 24 000, with a prevalence of 243 000; this is predicted to rise to over 300 000 by 2025.7
The effect of AMD on quality of life (QOL) has been markedly underestimated.8 The loss of visual acuity (VA) associated with NV‐AMD has been shown to have a marked effect on QOL, ranging from a 17% decrement in the QOL of the average patient with mild AMD, to 60% in patients with very severe AMD, equivalent to that encountered with end‐stage prostate cancer or a catastrophic stroke.4 Additionally, patients with AMD‐related visual impairment experience problems with common activities of daily living (ADL),9,10 and in their ability to care for self and dependants.11
Detailed documentation of the many consequences of NV‐AMD and their impact on individuals and society is essential if rational policy decisions regarding these patients are to be made. Although previous studies have evaluated the effect of AMD on QOL to a certain extent, there has been no attempt to quantify the burden of NV‐AMD in its totality. The International AMD Burden of Illness Study aimed to document the humanistic and economic impacts of NV‐AMD through a simultaneous assessment of patients and a similar group of subjects not affected by the disease in five countries.12 This article reports the humanistic burden of NV‐AMD and related resource utilisation in patients and controls from the UK cohort included in the international study.
Methods
This was a cross‐sectional, observational study of patients with bilateral subfoveal NV‐AMD recruited from five retina specialist offices/clinics, and non‐AMD controls recruited from five general practitioner offices/clinics in the UK. Bilateral subfoveal NV‐AMD was required as QOL and functional status impairment have been shown to be correlated with VA in the better‐seeing eye.13,14,15 Controls had best‐corrected Snellen VA of ⩾20/40 in the better eye and were free from serious ocular pathologies. All subjects were ⩾50 years of age. Exclusion criteria included participation in investigational drug studies within 30 days prior to the survey.
UK ethics committee approval was obtained and no medical interventions or invasive procedures were required by the study protocol.
Potential subjects were identified during routine office visits. Written informed consent was obtained and subjects undertook a standardised 60‐minute telephone survey administered by trained interviewers. A case report form was completed by review of the subject's medical record. Recorded data (within the previous 24 months) included basic demographics, VA, previous diagnostic tests, treatments for NV‐AMD, prescribed equipment for eyesight and vision rehabilitation, concomitant illnesses, and other ophthalmologic diagnoses/co‐morbidities.
Study survey
The survey included three validated QOL questionnaires and 12 study‐specific questions. The National Eye Institute Visual Function Questionnaire (NEI‐VFQ‐25), a 25‐item vision‐specific QOL assessment relevant to multiple eye conditions, reflects 11 vision‐related constructs plus a single general health rating question.16 Scores on the 12 resulting subscales range from 0 to 100, where 100 represents optimal QOL; the summary score is the average of the 11 vision‐related subscale scores. The EuroQol (EQ‐5D) assesses general health‐related QOL across five dimensions: mobility, self‐care, performance of usual activities, pain or discomfort, and anxiety or depression.17 Health state valuation scores, calculated using relative weights (scoring coefficients) for each item, range from –0.594 to 1.00, with a higher score indicating better QOL. The Hospital Anxiety and Depression Scale (HADS) is designed to detect the presence and severity of anxiety and depression mood symptoms.18 The HADS includes 14 items, seven assessing anxiety and seven assessing depression; subscales scores range from 0 to 21, with higher scores representing more symptoms and poorer emotional well‐being. Study‐specific questions were developed to assess the occurrence of accidents/falls and related injuries, and health resource utilisation (HRU).
Endpoints and statistical analysis
The primary endpoint was the impact of NV‐AMD on NEI‐VFQ‐25 summary scores in patients and compared with controls. Other endpoints included QOL burden using the EQ‐5D and HADS, and differences in HRU. The effect of VA level on QOL was also investigated.
Summary statistics were calculated including means and standard deviations (SD) for continuous variables and frequency distributions for categorical variables. VA was analysed as a continuous variable in decimal units. Mean (95% confidence interval [CI]) QOL scores (NEI‐VFQ‐25, EQ‐5D, HADS) and continuous HRU variables were compared between patients and controls using analysis of covariance (ANCOVA) models with outcomes modelled as a function of the study group controlling for age, gender, race and co‐morbidities. Mean QOL scores across the range of VA in the better‐seeing eye (normal: better than 20/40; mild: 20/40 to better than 20/80; moderate: 20/80 to better than 20/200; severe: 20/200 to better than 20/400; and near blindness: 20/400 or worse) were compared using ANCOVA models adjusting for covariates.
Unit costs for healthcare utilisation were based on the Personal Social Services Research Unit19 with rates inflated for 2005 and the British National Formulary.20
Statistical significance was evaluated at the 0.05 level, with no adjustments for multiple comparisons. All analyses were performed using PC‐SAS version 9.1 (SAS Institute, Cary, North Carolina, USA).
Results
Demographic and clinical characteristics
Data were collected between April and October 2005 for 75 NV‐AMD patients and 91 controls. Subject demographic and clinical characteristics are summarised in table 1. Significant differences in age (p<0.0001) and race (p = 0.0087) existed between the two groups. The presence of co‐morbid disease was lower in the patient than in the control group (65.3% vs 84.6%, respectively; p = 0.0038). A higher percentage of NV‐AMD patients had vision‐related co‐morbidities (p = 0.0029), while a higher percentage of controls had other co‐morbidities including anxiety disorder (p = 0.0085), headache (p = 0.0393) and asthma (p = 0.021).
Table 1 Demographic and clinical characteristics of study sample.
| Characteristics | NV‐AMD (n = 75) | Control (n = 91) |
|---|---|---|
| Age, years | ||
| Mean (SD) | 79.6 (6.43) | 65.3 (8.53) |
| Median | 80 | 65 |
| Range | 60–92 | 50–86 |
| Gender, n (%) | ||
| Male | 28 (37.3) | 46 (50.5) |
| Female | 47 (62.7) | 45 (49.5) |
| Race, n (%) | ||
| Caucasian | 75 (100) | 76 (83.5) |
| Black | 0 (0) | 1 (1.1) |
| Asian | 0 (0) | 4 (4.4) |
| Other | 0 (0) | 7 (7.7) |
| Missing | 0 (0) | 3 (3.3) |
| Living situation in the past 12 months, n (%) | ||
| Living with spouse or family member | 46 (61.3) | 71 (78.0) |
| Living alone | 26 (34.7) | 19 (20.9) |
| Living in assisted living facility or nursing home | 3 (4.0) | 1 (1.1) |
| Current best‐corrected visual acuity in better‐seeing eye | ||
| Mean (SD) | 0.26 (0.19) | 0.91 (0.24) |
| Median | 0.17 | 1.0 |
| Range | 0.04–0.67 | 0.50–1.5 |
| Co‐morbid diseases, n (%)*,† | ||
| Cataract | 14 (18.7) | 6 (6.6) |
| Glaucoma | 3 (4.0) | 0 (0.0) |
| Headache | 0 (0.0) | 5 (5.5) |
| Anxiety disorder | 0 (0.0) | 8 (8.8) |
| Asthma | 1 (1.3) | 9 (9.9) |
| Chronic neck/back pain | 3 (4.0) | 12 (13.2) |
NV‐AMD = neovascular age‐related macular degeneration; SD = standard deviation.
*Categories are not mutually exclusive. †Not all co‐morbid diseases assessed are listed. Differences between study groups are statistically significant (p<0.05) for all listed co‐morbidities. Other co‐morbid diseases assessed included depression, diabetes, chronic obstructive pulmonary disease, arthritis, heart disease, stroke and sleep disturbance.
QOL differences between study groups
NV‐AMD patients had significantly reduced QOL as measured by all validated questionnaires (fig 1). Patients had significantly poorer NEI‐VFQ‐25 scores in all subscales except for General Health and Ocular Pain compared with controls (table 2).
Figure 1 Adjusted mean differences in QOL scores by NV‐AMD and control subjects. *Adjusted for age, race, vision‐related co‐morbidities and other co‐morbidities.
Table 2 Differences in NEI‐VFQ‐25 subscales.
| NEI VFQ‐25 subscales | AMD (n = 75) | Control (n = 91) | p |
|---|---|---|---|
| Adjusted mean score | Adjusted mean score | ||
| General Health | 45.61 | 53.85 | 0.1622 |
| General Vision | 39.35 | 77.57 | <0.0001 |
| Ocular Pain | 85.33 | 89.38 | 0.1901 |
| Near Activities | 33.24 | 87.85 | <0.0001 |
| Distance Activities | 46.62 | 91.36 | <0.0001 |
| Vision‐specific: | |||
| Social function | 60.47 | 95.38 | <0.0001 |
| Mental health | 43.18 | 92.16 | <0.0001 |
| Role difficulties | 47.47 | 92.61 | <0.0001 |
| Dependency | 49.39 | 94.88 | <0.0001 |
| Driving | 12.59 | 76.61 | <0.0001 |
| Colour Vision | 81.98 | 100.14 | <0.0001 |
| Peripheral Vision | 68.00 | 92.45 | <0.0001 |
| Composite Score | 52.67 | 90.67 | <0.0001 |
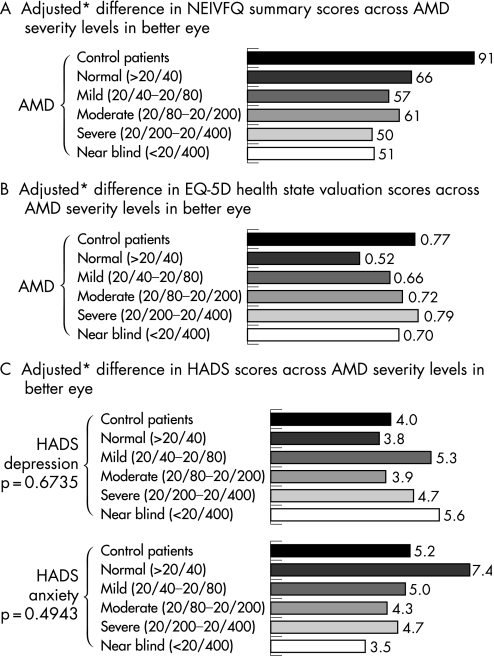
Impact of reduced VA on QOL measures
There was no significant association between decreasing vision‐specific NEI‐VFQ‐25 summary scores and the severity of vision loss (fig 2A). There was no apparent relationship between severity of vision loss and EQ‐5D health state valuation scores or HADS depression or anxiety subscale scores (fig 2B,C).
Figure 2 Adjusted mean differences in QOL scores across AMD severity levels in better eye. *Adjusted for age, race, vision‐related co‐morbidities and other co‐morbidities.
AMD‐associated co‐morbidities
Among subjects who fell (13% for both groups), the mean number of falls was significantly higher in the AMD group than in control patients (2.20 vs 1.17, respectively; p = 0.0442). The number of falls was highest in those patients with severe AMD compared with moderate or mild disease (25% vs 16.7% and 8.3%, respectively, of reported falls in the AMD group).
Health resource utilisation
Compared with controls, AMD patients reported significantly more visits to ophthalmologists (38.7% vs 15.4%; p = 0.0023), and optometrists (62.7% vs 40.7%; p = 0.0147). Almost three‐quarters of patients (72%) were referred for low vision rehabilitation, 44% attended rehabilitation, 46.7% were prescribed vision‐enhancing equipment, and 21.3% received social benefits and transportation subsidies. Table 3 summarises treatments received by AMD patients in the past 24 months.
Table 3 Treatments received by patients in the previous 24 months.
| NV‐AMD treatment | Past 12 months | Past 13–24 months | ||
|---|---|---|---|---|
| Better eye | Worse eye | Better eye | Worse eye | |
| Verteporfin, n (%) | 15 (20.0) | 4 (5.3) | 7 (9.3) | 6 (8.0) |
| Mean (SD) number of treatments* | 1.1 (0.3) | 2.0 (0.7) | 1.2 (0.4) | 1.5 (1.2) |
| Simultaneous verteporfin treatment in both eyes, n (%) | 2 (2.7) | 1 (1.3) | ||
| Mean (SD) number of treatments* | 1.0 (0.0) | 1.0 (n/a) | ||
| IVT corticosteroids | 4 (5.3) | 1 (1.3) | 1 (1.3) | 0 (0.0) |
| Mean (SD) number of treatments* | 1.3 (0.5) | 1.0 (n/a) | 1.0 (n/a) | n/a |
| PDT with IVT corticosteroids | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
IVT, intravitreal; NV‐AMD = neovascular age‐related macular degeneration; PDT, photodynamic therapy; SD = standard deviation.
*Among subjects who received the treatment.
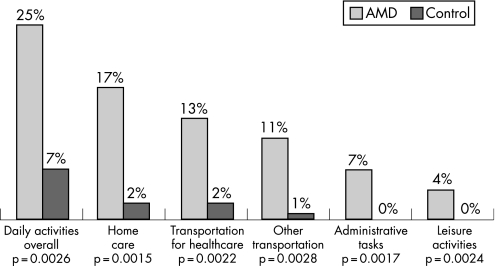
A significantly higher percentage of patients required assistance across a range of ADL compared with controls; 3.8 times more patients received assistance with ADL in general (fig 3). More patients with severe VA (35.0%) or near‐blindness (66.7%) required ADL‐related assistance compared with patients with mild (16.7%) or moderate VA (11.1%).
Figure 3 Patients receiving assistance for daily activities in AMD and control subjects.
Annual cost of NV‐AMD patient
Total annual costs (UK£) of subjects with NV‐AMD were more than seven times higher than those of controls (table 4).
Table 4 Mean health resource utilisation cost between study groups.
| Health utilisation cost | Costs per year (UK£) | ||
|---|---|---|---|
| AMD (n = 75) | Control (n = 91) | p | |
| Direct vision‐related medical cost* | 1553.14 | 44.76 | <0.0001 |
| Direct non‐vision‐related medical cost† | 431.01 | 412.72 | 0.9213 |
| Direct non‐medical related cost‡ | 1839.74 | 59.57 | 0.0012 |
| Total cost per patient | 3823.89 | 517.05 | <0.0001 |
*Direct vision‐related medical costs includes retina specialist evaluation, diagnostic test, and previous treatment of AMD, visual rehabilitation, eye‐sight equipment, eye doctor visits, eye glasses or contact lenses. †Direct non‐vision‐related medical costs represent a summary of fall‐related injuries, other accident‐related injuries, depression/anxiety treatment, and non‐vision related medical treatment. ‡Direct non‐medical related costs include living situation costs (ie, government‐sponsored assisted living facilities or nursing homes), assistance for received daily activities, and social benefit received.
Direct vision‐related costs
The greatest contributor to this outcome was diagnostic testing (table 5), of which fluorescein angiography represented the highest annual costs (£463.33). Only 3% of total annual costs were attributable to previous AMD therapy and these were largely due to single‐eye treatment with verteporfin (£83.33); intravitreal corticosteroids contributed £21.74 per year.
Table 5 Health resource utilisation cost for UK AMD patients.
| Health utilisation cost | Costs per year (UK£) (n = 75) (% total costs) |
|---|---|
| Direct vision‐related medical cost | 1553.14 (40.6) |
| Total visual rehabilitation‐related costs | 30.72 (0.8) |
| Total eye sight equipment‐related costs | 270.69 (7.1) |
| Total costs for eye doctor visits | 100.81 (2.6) |
| Total eye glasses or contact lenses‐related costs | 13.60 (0.4) |
| Total retina specialist evaluation costs | 300.00 (7.8) |
| Total diagnostic test‐related costs | 725.00 (19.0) |
| Total previous treatment of AMD‐related costs | 112.31 (2.9) |
| Direct non‐vision‐related medical cost | 431.01 (11.3) |
| Total fall‐related costs | 25.27 (0.7) |
| Total other accident‐related costs | 3.19 (0.1) |
| Total non‐vision‐related medical treatment‐related costs | 351.43 (9.2) |
| Total depression/anxiety treatment‐related costs | 51.12 (1.3) |
| Direct non‐medical‐related cost | 1839.74 (48.1) |
| Total living situation‐related costs | 536.88 (14.0) |
| Total costs of assistance for daily activities received | 1102.85 (28.8) |
| Total amount of social benefits received for visual disability | 200.01 (5.2) |
| Total cost per patient | 3823.89 (100.0) |
Direct non‐vision‐related medical costs
Mean direct non‐vision‐related medical cost constituted only 11% of total costs in the UK (table 4). The main cost driver for this category of cost was non‐vision‐related medical treatment, which was primarily due to hospital admission costs (£298.67).
Direct non‐medical‐related cost
Direct non‐medical‐related costs were the main costs associated with AMD patients in the UK and were significantly higher in AMD patients than in controls (table 3). The main contributor to these costs was assistance for daily living received and, of this, home care was associated with the largest cost (£562.56, 14.7% of total).
Discussion
This study was the first to simultaneously investigate the consequences of NV‐AMD through clinical assessments and patient‐reported QOL and HRU measures. In this cohort of patients from the UK, bilateral NV‐AMD was associated with substantially worse QOL, poorer vision‐related functioning, and more symptoms of depression compared to control subjects without AMD. Patients with NV‐AMD required significantly more assistance with ADL, and utilised significantly more health resources than control subjects, with mean annual costs of more than seven times that of subjects without AMD. In an average health authority population of around 500 000, the prevalence of NV‐AMD is estimated at around 1946 patients.6 Based on the figures from this study, this represents a total societal cost for such a health authority of over £7.4 million per year, compared with only £1.0 million for equivalent patients without NV‐AMD.
The adjusted mean NEI‐VFQ‐25 scale score for patients was significantly worse than for controls and comparable to that reported in bilateral NV‐AMD patients in other studies.21,22 Patients with lower VA levels generally reported worse NEI‐VFQ‐25 scores, although differences in individual subscales did not reach statistical significance, with the exception of the ability to drive. Along with worse vision‐related functioning, patients reported significantly more symptoms of depression compared with controls. Depression scores tended to worsen with decreasing VA, in agreement with a reported association between poorer VA in the better eye and higher (worse) anxiety and depression scores in patients with NV‐AMD.15 Approximately 30% of patients with AMD suffer from depression and this is a major cause of disability irrespective of the degree of vision loss or general medical problems, even in those patients with mild depressive symptoms.23,24
No relationship was seen between EQ‐5D health state valuation scores and VA level in patients, although overall EQ‐5D was significantly worse than controls. This finding is consistent with another AMD study assessing the EQ‐5D by VA level in which no discernable trend was identified,25 suggesting that the EQ‐5D may not be a suitable measure in this population. In fact, in the elderly population, the EQ‐5D has been found to be of most use where a substantial change in health is expected,26 which may imply that the EQ‐5D is not sufficiently sensitive to measure difference across VA levels.
Patients with NV‐AMD in this study needed significantly more ADL‐related assistance than controls, including home care, transportation, administrative tasks and leisure activities. The percentage of patients receiving ADL assistance was more than three times that of controls, and the cost of ADL assistance was the greatest single contributor to the annual health resource utilisation costs for AMD patients. As would be expected, the need for ADL‐related assistance was highest in patients with the most severe levels of VA compared with patients with milder degrees of visual impairment. The number of falls in the NV‐AMD group was significantly more than in controls, although this difference did not lead to a greater incidence of fall‐related fractures in these patients. The number of falls was highest in those patients with severe VA, occurring in a quarter of patients compared with only 8% of those with mild VA. Poor VA has been shown to approximately double an individual's risk of falling, and poor vision may be responsible for 25% to 50% of all falls in the visually impaired.27,28 Taken together, the increase in need for ADL‐related assistance and increased risk of falls with decreasing VA suggest that preserving vision in NV‐AMD patients through earlier intervention may help preserve their independence and functioning.
Patients included in this study were on average almost 15 years older than controls. However, a post‐hoc analysis on age‐matched subgroups (⩾70 years, n = 30, mean age 75; ⩾75 years, n = 11, mean age 80) found no relationship between age and QOL measures.
There are several limitations to this study. First, practice participation was voluntary, and therefore it is possible that sites which agreed to participate may differ from those sites which declined. As patient participation was also voluntary, the clinical/sociodemographic characteristics of NV‐AMD patients who declined to take part are not known. Finally, the study did not evaluate the significant elderly patient population in nursing homes. As a substantial percentage of such patients may have lost independence due to ocular disease,29 our findings may underestimate the full societal impact of NV‐AMD.
In conclusion, this study provides clear evidence of the negative effect of bilateral, subfoveal NV‐AMD on the emotional health and functional ability of sufferers, and the major impact of the disease on providers and society overall. Patients reported substantially worse QOL, poorer vision‐related functioning, more depressive symptoms, more falls, greater dependency on caregivers, and higher HRU compared with controls without AMD. The association between deteriorating VA and continuous decrease in QOL and increase in need for ADL‐related assistance argues for the early detection and treatment of NV‐AMD to arrest VA loss and preserve the patient's independent functioning and well‐being. This is especially important in the context of the expected major increase in the elderly population due to changing demographics. Much progress has been made in the management of AMD; effective new therapies such as pegaptanib (Macugen) and ranibizumab (Lucentis) are now available and early treatment will help to reduce the overall costs to both the individual and to society. It is, therefore, essential that awareness of the burden of NV‐AMD is raised to ensure that the significant impact of the disease is given full consideration when managing these patients.
Abbreviations
ADL - activities of daily living
AMD - age‐related macular degeneration
EQ‐5D - EuroQol questionnaire
HADS - Hospital Anxiety and Depression Scale
HRU - health resource utilisation
NEI‐VFQ‐25 - National Eye Institute Visual Function Questionnaire
NV‐AMD - neovascular age‐related macular degeneration
QOL - quality of life
VA - visual acuity
Footnotes
Funding: Support for this study was provided by Pfizer Inc., New York, USA.
Competing interests: None declared.
References
- 1.Ambati J, Ambati B K, Yoo S H.et al Age‐related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv Ophthalmol 200348257–293. [DOI] [PubMed] [Google Scholar]
- 2.Arnold J J, Sark S H. Age related macular degeneration. BMJ 2000321741–744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ferris 3rd F L, Fine S L, Hyman L. Age‐related macular degeneration and blindness due to neovascular maculopathy. Arch Ophthalmol 19841021640–1642. [DOI] [PubMed] [Google Scholar]
- 4.Brown G C, Brown M M, Sharma S.et al The burden of age‐related macular degeneration: a value‐based medicine analysis. Trans Am Ophthalmol Soc 2005103173–186. [PMC free article] [PubMed] [Google Scholar]
- 5.Age‐Related Eye Disease Study Research Group A randomized, placebo‐controlled, clinical trial of high‐dose supplementation with vitamins C and E, beta carotene, and zinc for age‐related macular degeneration and vision loss: AREDS report no. 8. Arch Opthalmol 20011191417–1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Meads C, Salas C, Roberts T.et al Clinical effectiveness and cost‐utility of photodynamic therapy for wet age‐related macular degeneration: a systemic review and economic evaluation. Health Technol Assess 20037 [DOI] [PubMed] [Google Scholar]
- 7.Mattson Jack Group Epidemiology of age‐related macular degeneration. St Louis, Missouri: Mattson Jack Group, 2003, [Data on file].
- 8.Brown M M, Brown G C, Sharma S.et al The burden of age‐related macular degeneration: a value‐based analysis. Curr Opin Ophthalmol 200617257–266. [DOI] [PubMed] [Google Scholar]
- 9.Mangione C M, Lee P P, Pitts J.et al Psychometric properties of the National Eye Institute Visual Function Questionnaire (NEI‐VFQ). NEI‐VFQ Field Test Investigators. Arch Ophthalmol 19981161496–1504. [DOI] [PubMed] [Google Scholar]
- 10.Scott I U, Smiddy W E, Schiffman J.et al Quality of life of low‐vision patients and the impact of low‐vision services. Am J Ophthalmol 199912854–56. [DOI] [PubMed] [Google Scholar]
- 11.Stevenson M R, Hart P M, Montgomery A M.et al Reduced vision in older adults with age related macular degeneration interferes with ability to care for self and impairs role as carer. Br J Ophthalmol 2004881125–1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Soubrane G, Creuss A, Lotery A.et al Burden of Illness, Visual Impairment, and Health Resource Utilization of Neovascular Age‐Related Macular Degeneration Patients: Results from a Five‐Country Cross‐Sectional Study. Arch Ophthalmol. In press [DOI] [PMC free article] [PubMed]
- 13.Brown M M, Brown G C, Sharma S J.et al Quality of life associated with visual loss: a time trade‐off utility analysis comparison with medical health status. Ophthalmology 20031101076–1081. [DOI] [PubMed] [Google Scholar]
- 14.Knur K K, Reichel M B, Gäbler P.et al Quality of life in patients with age‐related macular degeneration: a prospective study. Presented at the Annual Meeting of the Association for Research in Vision and Ophthalmology; 25–29April 2004; Fort Lauderdale, Florida, USA.
- 15.Dong L M, Childs A L, Mangione C M.et al Health‐ and vision‐related quality of life among patients with choroidal neovascularization secondary to age‐related macular degeneration at enrollment in randomized trials of submacular surgery: SST report No. 4. Am J Ophthalmol 200413891–108. [DOI] [PubMed] [Google Scholar]
- 16.Mangione C M, Lee P P, Gutierrez P R.et al Development of the 25‐item National Eye Institute Visual Function Questionnaire. Arch Ophthalmol 20011191050–1058. [DOI] [PubMed] [Google Scholar]
- 17.EuroQol Group EuroQol: a new facility for the measurement of health related quality of life. Health Policy 199016199–208. [DOI] [PubMed] [Google Scholar]
- 18.Zigmond A, Snaith R P. The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand 198367361–370. [DOI] [PubMed] [Google Scholar]
- 19.Curtis L, Netten A.Unit Costs of Health and Social Services 2004. Kent: Personal Social Services Research Unit (PSSRU) University of Kent, 2004
- 20. British National Formulary 2005. http://www.bnf.org (accessed 1 July 2005)
- 21.Hawkins B S, Miskala P H, Bass E B.et al Surgical removal vs observation for subfoveal choroidal neovascularization, either associated with the ocular histoplasmosis syndrome or idiopathic: II. Quality‐of‐life findings from a randomized clinical trial: SST Group H Trial: SST Report No. 10. Arch Ophthalmol 20041221616–1628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lindblad A S, Clemons T E. Responsiveness of the National Eye Institute Visual Function Questionnaire to progression to advanced age‐related macular degeneration, vision loss, and lens opacity: AREDS Report No. 14. Arch Ophthalmol 20051231207–1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Casten R J, Rovner B W, Tasman W. Age‐related macular degeneration and depression: a review of recent research. Curr Opin Ophthalmol 200415181–183. [DOI] [PubMed] [Google Scholar]
- 24.Rovner B W, Casten R J, Hegel M T.et al Minimal depression and vision function in age‐related macular degeneration. Ophthalmology 20061131743–1747. [DOI] [PubMed] [Google Scholar]
- 25.Espallargues M, Czoski‐Murray C, Bansback N.et al The impact of age‐related macular degeneration on health status utility values. Invest Ophthalmol Vis Sci 2005464016–4023. [DOI] [PubMed] [Google Scholar]
- 26.Haywood K L, Garratt A M, Fitzpatrick R. Quality of life in older people: a structured review of generic self‐assessed health instruments. Qual Life Res 2005141651–1668. [DOI] [PubMed] [Google Scholar]
- 27.Coleman A L, Stone K, Ewing S K.et al Higher risk of multiple falls among elderly women who lose visual acuity. Ophthalmology 2004111857–862. [DOI] [PubMed] [Google Scholar]
- 28.Harwood R H. Visual problems and falls. Age Ageing 200130(Suppl)13–18. [DOI] [PubMed] [Google Scholar]
- 29.Eichenbaum J W, Burton W B, Eichenbaum G M.et al The prevalence of eye disease in nursing home and non‐nursing home geriatric populations. Arch Gerontol Geriatr 199928191–204. [DOI] [PubMed] [Google Scholar]