Abstract
Decreased placental oxygenation and increased oxidative stress are implicated in the development of preeclampsia. Oxidative stress arises from imbalance between pro-versus anti-oxidants and can lead to biological oxidation and apoptosis. Because pregnant women living at high altitude (3100 m, HA) have lowered arterial PO2 and an increased incidence of preeclampsia, we hypothesized that HA placentas would have decreased anti-oxidant enzyme activity, increased oxidative stress (lipid peroxidation, protein oxidation and nitration) and greater trophoblast apoptosis than low-altitude (LA) placentas. We measured enzymatic activities, lipid and protein oxidation and co-factor concentrations by spectrophotometric techniques and ELISA in 12 LA and 18 HA placentas. Immunohistochemistry (IHC) was used to evaluate nitrated proteins and specific markers of apoptosis (activated caspase 3 and M30). Superoxide dismutase activity was marginally lower (p=0.05), while glutathione peroxidase activity (p<0.05), thioredoxin concentrations (p<.005) and thioredoxin reductase activity p< 0.01 were all reduced in HA placentas. Decreased anti-oxidant activity was not associated with increased oxidative stress: lipid peroxide content and protein carbonyl formation were lower at HA (p<.01). We found greater nitrotyrosine residues in the syncytiotrophoblast at 3100 m (p<.05), but apoptosis did not differ between altitudes. Our data suggest that hypoxia does not increase placental oxidative stress in vivo. Nitrative stress may be a consequence of hypoxia but does not appear to contribute to increased apoptosis. Lowered placental concentrations of anti-oxidants may contribute to the susceptibility of women living at HA to the development of preeclampsia, but are unlikely to be etiological.
Introduction
Preeclampsia is a leading cause of maternal and fetal mortality, pre-term birth and neonatal intensive care unit admissions. Epidemiological, clinical and molecular studies support that placental hypoxia is involved in preeclampsia. The incidence of the syndrome is doubled at high altitude (>2700 m) [1-3]. Human and experimental animal studies at >3000 m support that the blood entering the intervillous space has a PO2 approximately 20% lower than at sea level [4]. Uterine blood flow and oxygen delivery are reduced by ∼33%, even in normal human pregnancy at high altitude [5]. Finally, molecular studies show that global patterns of gene expression are similar in high altitude versus preeclamptic placentas and in placental explants subjected to 3% oxygen in organ culture [6]. The high altitude placenta is thus a valuable model to distinguish hypoxia-related biochemical pathways uniquely associated with preeclampsia versus benign or adaptive responses to hypoxia.
Oxidative stress, an imbalance between the cellular generation of reactive oxygen species (ROS) and the capacity of anti-oxidants to prevent oxidative damage, may play a pivotal role in preeclampsia (reviewed in [7]). Oxidative stress can result in lipid peroxidation (which compromises mitochrondrial ATP production and can stimulate pro-apoptotic events), protein carbonylation or nitration/nitrosylation (which alters protein conformation and function), and accumulation of DNA damage. ROS are primarily a byproduct of aerobic respiration, and are involved in normal extracellular and intracellular signaling processes [8]. Excess ROS can be produced by enzymatic processes involving oxidases, while excessive mitochondrial ROS production can cause damage which escalates ROS production by the mitochondrial electron transport chain, leading to further dysfunction. ROS can also increase where there are decreased concentrations of non-protein ROS-scavenging antioxidants (Vitamins A, C, E, and metabolic co-factors such as glutathione and thioredoxin) or decreased production of antioxidant enzymes Antioxidant enzymes defend against free radical damage; their regulation depends mainly on the oxidant status of the cell [9]. Antioxidant enzymes are reduced in preeclamptic placentas [10] and oxidative stress is increased in placentas of mothers with preeclampsia [10, 11].
Acute (hours to days), chronic (weeks) and lifelong high-altitude hypoxic exposure increases circulating markers of oxidative stress in humans [12, 13]. The elevated incidence of preeclampsia at high altitude, and the broad similarity in gene expression between preeclamptic and high-altitude placentas [6] led us to the hypothesis that high-altitude placentas would have altered pro- versus anti-oxidant defenses, resulting in increased oxidative damage. In this comparison of high versus low altitude placentas, we focused first on the levels of key endogenous anti-oxidants including superoxide dismutase (total SOD activity), glutathione peroxidase, thioredoxin and thioredoxin reductase. The accumulation of lipid peroxides and protein carbonyls was measured as an indicator of oxidative damage. We also examined the degree of protein nitration, a by-product of nitric oxide (NO) and superoxide interaction leading to conversion to peroxynitrite, as increased nitration is commonly noted in preeclampsia [14]. As oxidative stress is often considered as the event precipitating the increased placental apoptosis observed in preeclampsia [15] we further tested whether differences in oxidative stress would translate into increased apoptosis.
Methods
Subjects
Participants were pregnant women residing at 1600 m (n=12, 625 mmHg barometric pressure) and 3100 m (n=18, 530 mmHg barometric pressure) Participants resided at their respective altitudes of residence for a minimum of three months prior to conception, throughout pregnancy and delivery. All 1600 m subjects were born and raised at 1600 m (n=3) or lower. One high-altitude mother was native to 3100 m, 4 were native to towns ≥2400 m and the remainder were born and raised at low altitude. All gave informed consent to procedures approved by the ethics committees of the participating institutions. We included only women who were primiparous, healthy, and without conditions predisposing to preeclampsia such as renal disease, diabetes or obesity. Women were excluded if they had an abnormal oral glucose tolerance test or developed other complications of pregnancy.
Sample collection and preparation
Placentas were collected immediately following delivery and washed in cold PBS until the majority of blood was cleared. Fresh tissue was sampled from multiple sites within the villous core and flash frozen in liquid nitrogen. Randomly chosen blocks of full depth tissue were collected for paraffin embedding. Placental extracts were prepared and protein measured as previously described [10, 16].
Assays
Total superoxide dismutase (SOD) activity was evaluated via a spectrophotometric assay based upon the inhibition of pyrogallol oxidation; one unit of SOD activity is the amount that causes 50% inhibition pyrogallol oxidation. SOD activity was measured every 5 min over 1 hr at 405 nm, as previously detailed [10, 16], and expressed as units per milligram of protein (U/mg). The inter- and intra-assay variation were 1.40% and 0.35%, respectively.
Glutathione peroxidase activity was determined spectrophotometrically by coupling the oxidation of glutathione and NADPH using glutathione reductase, as we have previously reported [10]. Glutathione peroxidase activity was standardized against protein concentrations and expressed as moles/min/mg protein. The inter – and intra-assay variation were 0.43% and 1.25%, respectively.
Thioredoxin concentrations were determined using a previously described double-antibody capture-linked immunoabsorbent assay (ELISA) [10, 16] and expressed as ng/mg of extracted protein. The inter- and intra-assay variation were 1.14% and 2.96% respectively.
The activity of thioredoxin reductase was measured using the thioredoxin reductase and NADPH dependent reduction of insulin with and without thioredoxin [10, 16]. The inter- and intra-assay variation were 3.66% and 2.36% respectively.
The degree of lipid peroxidation in placental extracts was determined using a Lipid Peroxidation Assay Kit (Calbiochem). The assay evaluates lipid peroxidation levels through the reaction of malondialdehyde (MDA) and 4-hydroxy-2(E)-nonenal (4-HNE) with the chromogenic reagent 1-methyl-2-phenylindole at 45°C, which results in the production of a stable chromophore with maximal absorbance at 586 nm. A standard curve was created using 4-HNE, samples were measured in duplicate and lipid peroxide concentrations are expressed as μM MDA + 4-HNE/mg protein. Inter- and intra-assay variation were 3.71% and 3.8% respectively.
Placental extracts were analyzed for protein carbonyl concentrations by ELISA. This assay measures protein carbonyls in biological samples after reaction with 2,4-dinitrophenyl hydrazine (DNPH) [10, 16]. Each sample was analyzed in triplicate and samples were quantified by comparison with oxidized BSA standards. Inter- and intra-assay variation were 2.6% and 6.7% respectively.
Immunohistochemistry
IHC was performed on a subset of 12 placentas from each altitude. Samples representing 3 different locations/placenta were randomly chosen for IHC analyses. We examined activated caspase-3 (rabbit monoclonal, Cell Signaling, Danvers MA), M30, the cleaved epitope of cytokeratin 18 that results from caspase-3 induced apoptosis (mouse monoclonal, M30 CytoDEATH, Roche, NJ), and nitrotyrosine (clone 1A6 mouse monoclonal, Upstate Biochemical, Charlottesville VA). Endogenous peroxidase was blocked (3% H2O2 in methanol) and antigen retrieval was performed by incubating slides in DAKO Target Retrieval Solution for 30 minutes at 95°C (DAKO, Carpenteria, CA). All primary and secondary antibody incubations were conducted at 37°C in a humidified chamber (activated caspase 3, 1:200; M30, 1:50 for 2.5 hours; nitrotyrosine 1:100 for 2 hours). All slides were incubated with specific biotinylated secondary antibodies for 1 hour (Sigma, St. Louis MO), followed by detection using the Vectastain ABC Kit for 45 min. Color was developed for 10 minutes with NovaRed substrate solution (Vector Laboratories, Burlingame, CA), and counterstaining and mounting was according to standard protocols.
For an additional positive control we treated cultured BeWo choriocarcinoma cells (seeded in 2-well Lab-Tek slides at 105 cells per well) with 2.5 uM camptothecin (Sigma, St Louis, MO) to induce apoptosis. Cells were fixed on the slide with cold ethanol/acetone mixture (1:1) and IHC performed as described above.
Image analysis
IHC data were analyzed using a digital analysis program (Image Pro, Media Cybernetics Silver Springs, MD) which permitted quantification of the percentage of cells positively stained for activated caspase 3 and M30. Positive controls (tonsil for Caspase-3, colon carcinoma for M30 and kidney and ischemic brain for nitrotyrosine) and negative controls (omission of primary antibody) were run for each batch of slides. The number of fields to be examined per slide was set at 12 after preliminary analyses showed that variability in the readings stabilized at 12 fields, and did not differ from observations of 16, 24, or 32 fields.
Statistical Analysis
Results are shown as mean ± SD. Maternal and infant characteristics and placental measures of oxidative stress were compared between low and high altitude using Student's t test. Correlation analysis was performed by linear or non-linear regression. The caspase 3 and M30 IHC results were analyzed using the Mann-Whitney U test. Evaluation of nitrotyrosine staining was based on a qualitative scale of −, +, ++, +++ representing no, weak, moderate or strong staining. Conversion of the qualitative data to ordinal data (0-3) was used for the nitrotyrosine IHC analyses in order to permit statistical analysis using the Mann-Whitney U [17]. Consistent with the requirements of the Mann-Whitney U, the variances were similar between the low versus high altitude samples. These data are reported as medians and inter-quartile ranges. Data are reported as significant where p < 0.05.
Results
Subject characteristics
Participating women were similar in demographic and obstetric characteristics at low versus high altitude (Table 1). Birth weight was lower at high altitude, despite similarity in placental weight, birth length and gestational age.
Table 1.
Maternal and Infant Characteristics (means ± SD)
| 1600 m n=12 | 3100 m n=18 | |
|---|---|---|
| Age (years) | 29 ± 3 | 28 ± 7 |
| Height (cm) | 169 ± 5 | 166 ± 7 |
| Pre-pregnant weight (kg) | 62 ± 6 | 63 ± 10 |
| BMI (kg/m2) | 22 ± 2 | 23 ± 3 |
| Weight gain with pregnancy (kg) | 14 ± 5 | 15 ± 5 |
| Birth weight (grams) | 3365±382 | 3132±335* |
| Gestational Age (weeks) | 39.4 ± 1.7 | 39.7 ± 1.2 |
| Baby Length (cm) | 50.0 ± 3.0 | 49.6 ± 3.2 |
| Placental weight (grams) | 551 ±166 | 625 ± 83 |
| Ratio M/F | 9M/3F | 12M/6F |
p < 0.05
Endogenous anti-oxidants
The activities of anti-oxidant enzymes in placental tissues from low vs. high altitude are illustrated in Figure 1. Comparison of endogenous SOD between altitudes revealed marginally greater SOD activity at low altitude (2.0 ± 0.7 U/mg protein) than at high altitude (1.4 ± 0.6 U/mg protein, p = 0.05, Figure 1A). Glutathione peroxidase (Figure 1B) activity was greater at low altitude (16.4 ± 3.5) than at high altitude (11.9 ± 4.1 mmoles/min/mg protein, p < 0.05). The placental concentration of thioredoxin was greater at low altitude (122.1 ± 34.6) than at high altitude (80.4 ± 24.4 ng/mg protein, p < 0.005, Figure 1C). Consistent with this, thioredoxin reductase activity was twice as high in low (16.5 ± 7.8) vs. high altitude placentas (8.9 ± 5.4 U/mg protein, p< 0.01, Figure 1D). However, the decreased anti-oxidant activity at high altitude did not appear to result in oxidative stress, as the placental content of lipid peroxides (μM MDA+4HNE/mg protein) was 2-fold less in the high altitude (7.6 ± 4.2) than the low altitude placentas (15.7 ± 9.8, p < 0.01, Figure 2A). We also observed significantly less protein carbonyl formation in placentas from high altitude (80.5 ± 24.4 U/mg, p < 0.01, Figure 2B) versus low altitude (122.1 ± 34.6 U/mg, p < 0.01, Figure 2B) In summary, anti-oxidant substrates and activity were decreased at high altitude, but this did not translate into increased oxidative stress.
Figure 1.
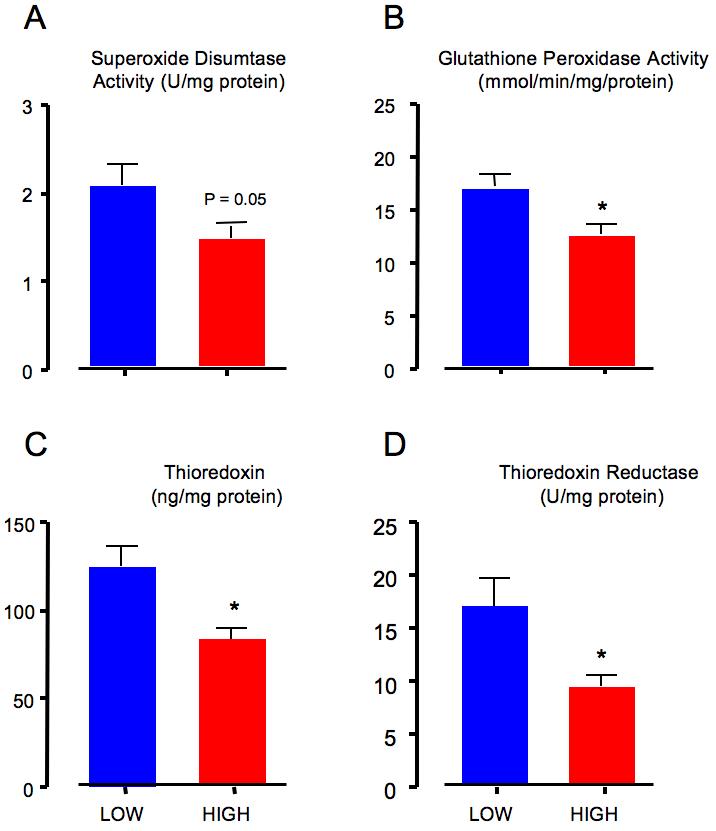
A: Superoxide dismutase activity was marginally (p=0.05) greater in the low altitude (blue bar, range 1.19-2.99) than high altitude placentas (red bar, range 0.56 – 0.91).
B: Glutathione peroxidase activity was greater (p = 0.01) in the low altitude (blue bar, range 11.63-21.89) than high altitude placentas (red bar, range 5.32 – 18.79).
C. Thioredoxin content was greater (p = 0.002) in low altitude (blue bar, range 75.58 – 176.8) than high altitude placentas (red bar, range = 41.20-122.0).
D. Thioredoxin reductase activity was also greater (p = 0.009) in low altitude (blue bar, range 6.22 – 27.01) than high altitude placentas (red bar, range 1.69 – 19.04).
Figure 2.
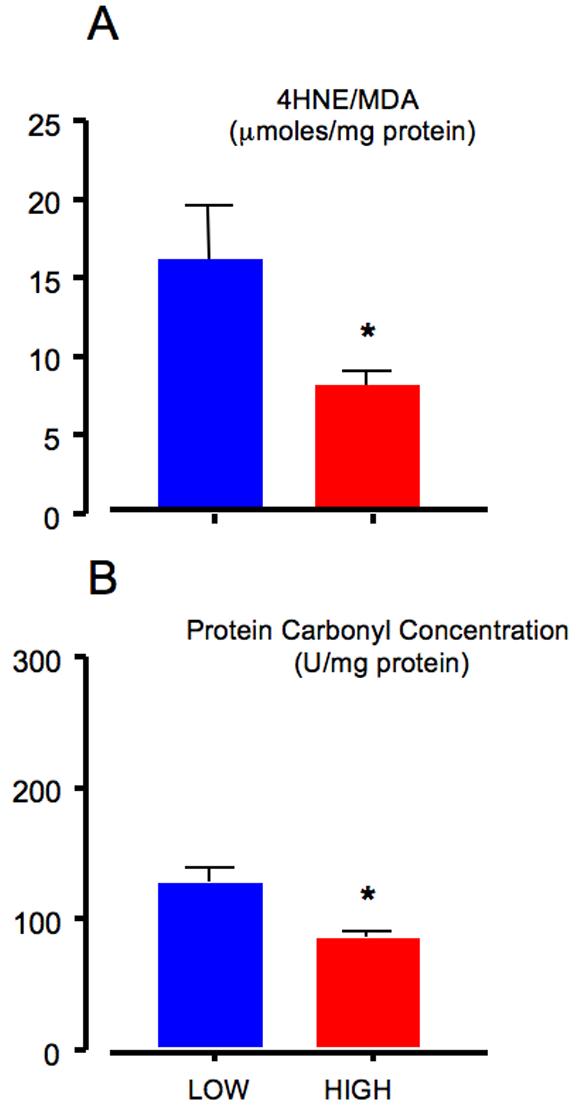
A: Oxidative stress, as indicated by lipid peroxidation, was greater (p = 0.007) in low altitude placentas (blue bar, range 4.89 – 29.81) when compared with high altitude placentas (red bar, range 3.42 – 21.22).
B: Oxidative stress, as reflected in protein carbonylation, was greater (p=0.002) in low altitude (blue bar, range 75.6 – 176.8) than high altitude placentas (red bar, range 41.2-122.0).
Nitrotyrosine residues
IHC for nitrotyrosine revealed no difference between low and high altitude in the qualitative staining for nitrated proteins in endothelium or villous stroma. However staining was greater in the syncytiotrophoblast and extravillous trophoblast at high altitude (p < 0.05, Table 2, Figure 3). Thus, despite less oxidative stress as measured by lipid peroxidation and protein carbonylation in high altitude placentas, there was evidence for increased nitrative stress in the trophoblast.
Table 2.
IHC analysis of nitrated proteins (medians and inter-quartile range)
| 1600 m n=12 | 3100 m n=12 | |
|---|---|---|
| Syncytiotrophoblast | 1.0 (1.5) | 2.5 (1.0)* |
| Intermediate trophoblast | 2.0 (1.0) | 3 (1.0)* |
| Stroma | 1.0 (0.5) | 2.0 (2.0) |
| Endothelium | 1.0 (1.0) | 1.5 (1.0) |
p < 0.05, Mann-Whiteny U
Figure 3.
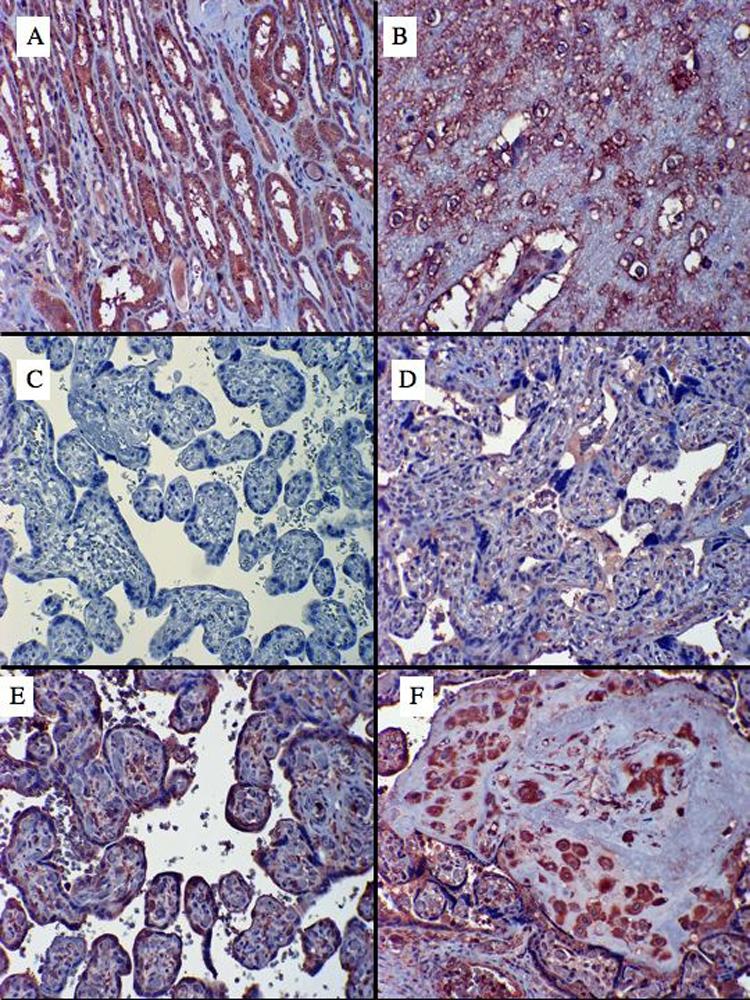
A: Kidney was used as positive control for nitrotyrosine staining (positive staining is a dark reddish brown). Note the intense staining in the renal tubules.
B: Ischemic brain was used as a second positive control.
C. Placenta with the primary antibody for nitrotyrosine omitted.
D. A low altitude placenta showing staining of serum within blood vessels and the intervillous space, some stromal staining, but a relative absence of syncytiotrophoblast staining.
E. A high altitude placenta showing intense syncytiotrophoblast staining.
F. A high altitude placenta showing strong staining in the intermediate (extravillous) trophoblast.
Markers of apoptosis
We next examined whether differences in oxidative stress might have translated into differences in placental tissue apoptosis. In the control tissues, activated caspase-3 was detected in 0.06±0.08% of tonsil cells (Figure 4A) while the negative control (Figure 4B) showed no staining. In the camptothecin-treated BeWo cells, 0.13 ± 0.12% of cells were positive (data not shown). Levels in placenta were more than an order of magnitude lower; at low altitude 0.005 ± 0.004% of cells (Figure 4C) were positive, while at high altitude 0.006 ± 0.003% of cells were positively stained (Figure 4D, p=NS).
Figure 4.
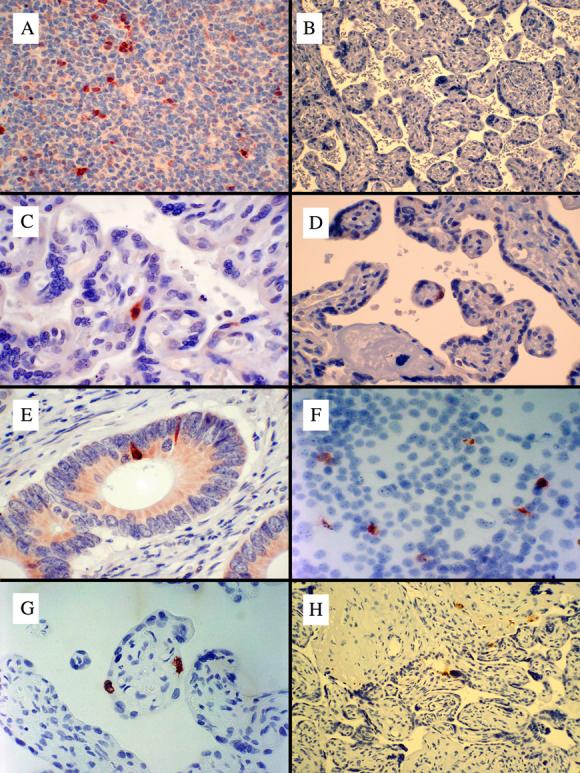
A: Tonsil was used as a positive control tissue for immunohistochemical detection of Activtaed Caspase-3. Positive staining is indicated by the reddish brown color in the cytoplasm of positively stained cells.
B: Placenta with primary antibody for caspase 3 omitted. Placenta with primary antibody omitted for M30 had the same appearance (data not shown).
C: A low altitude placenta showing a rare positively stained cell in the syncytiotrophoblast.
D: A high altitude placenta showing an equally rare positively stained cell in the syncytiotrophoblast.
E: Adenocarcinoma of the colon, used as a positive control for M30, showed rare epithelial cells with strong staining (brownish red) with faint staining in the apical cyctoplasm of other epithelial cells.
F. BeWo cells treated with camptothecin were used as an additional positive controls for M30 (note the much greater frequency of positive staining than in the normal placentas).
G. A low altitude placenta showing rare positive syncytiotrophoblast staining for M30. H. A high altitude placenta showing rare positive staining for M30 in the syncytiotrophoblast.
M30, a downstream marker of caspase-3-related apoptosis, was present in 0.3 ± 0.2% of the control tissue, colon adenocarcinoma (Figure 4E). The camptothecin-treated BeWo cells (Figure 4F) showed 6.3±2.6% positive staining for M30. There was positive staining in 0.03±0.02% of cells in low altitude placentas (Figure 4G) and 0.04±0.03% of cells in high altitude placenta (Figure 4H, p=NS). In summary, there were no differences in levels of apoptosis between the high versus low altitude placentas.
Oxidative stress, infant sex and gestational age
There was no difference in the placentas of male versus female infants in the measures reported above. There was no relationship between gestational age or placental weight and the dependent variables.
Discussion
The hypothesis that placentas subjected to high-altitude hypoxia would have increased oxidative stress was not supported. Instead endogenous placental anti-oxidant activity was lower, lipid peroxidation and protein carbonylation were reduced and no differences in apoptosis were found. Nitrotyrosine residues were increased in the high altitude placental trophoblast. Thus increased nitrative stress, but not oxidative stress, is associated with chronic hypoxia in the human placenta. Our findings are summarized in Figure 5. We conclude that mild chronic hypoxia does not increase placental lipid and protein oxidation in vivo, and that syncytiotrophoblast nitration is not necessarily associated with increased apoptosis.
Figure 5.
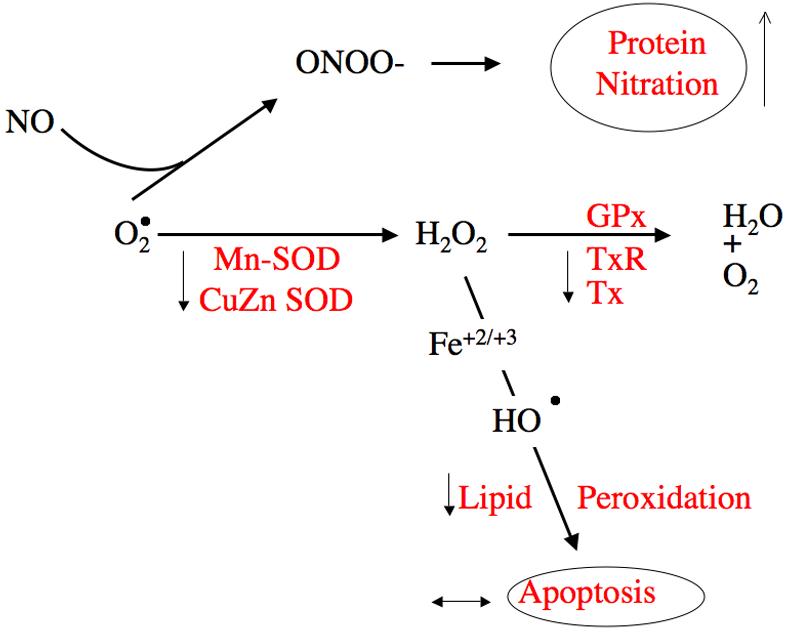
The components of oxidative stress measured in the present study are shown in red. The hypothesis predicted that high altitude placentas would show decreased endogenous anti-oxidants such as total superoxide dismutase (SOD), a reduction in glutathione peroxidase and in thioredoxin reductase, and in thioredoxin, the substrate for thioredoxin reductase, predictions confirmed by our measurements. Due to reduced anti-oxidant protection we expected increased, but instead observed decreased lipid-peroxidation and protein carbonylation and no difference in apoptosis. Despite reduced lipid peroxidation and protein carbonylation, there was increased protein nitrosylation, specifically in the syncytiotrophoblast.
The relevance of a control sample obtained at 1600 m as opposed to sea level is of concern in interpreting our results. The 9% difference in arterial oxygen tension between sea level and 1600 m has no effect on pregnancy outcome [18, 2], nor on placental structure at altitudes <2200 m [19]. The 24% difference in arterial PO2 at 3100 m measurably impacts placental morphology and function (reviewed in [4]). As the PO2 in the intervillous space near term is only ∼40 mmHg, or 6% O2 [20], at 3100 m intervillous PO2 could be reduced by an additional 2-3%. This yields a placental oxygen level that would be similar to the 3% oxygen used in some in vitro placental studies to mimic hypoxia and preeclampsia [21, 6]. That the high altitude placenta is hypoxic is consistent with our prior reports showing elevated HIF-1α, VEGF and sFlt-1 in the high altitude placenta but not at the lower altitude of 1600 m [22, 6, 23]. We show in Table 3 our previously published data from preeclamptics and controls at sea level. It is apparent that 1600 m is similar to sea level, and that preeclampsia and 3100 m parallel one another except in the measures of protein carbonylation and lipid peroxide.
Table 3.
Sea level and preeclamptic data compared to the present study (mean±SD)
| <10 m control |
<10 m preeclampsia |
1600 m control |
3100 m placental hypoxia |
|
|---|---|---|---|---|
| SOD (U/mg) | 2.5±0.6 | 2.0±0.5 [−20] | 2.0±0.7 | 1.4±0.6 [−30] |
| GPx (moles/min/mg) |
17.3±6.1 | 11.5±3.1 [−34] | 16.4±3.5 | 11.9±4.1 [−27] |
| THx(ng/mg) | 107.0±18.4 | 91.1±21.2 [−15] | 122.1±34.6 | 80.5±24.4 [−34] |
| THxRed (U/mg) | 19.2±9.8 | 13.0±5.7 [−48] | 16.5±7.8 | 8.9±5.4 [−46] |
| LPO (μmoles/mg) | 5.4±4.0 | 20.7±7.8 [+283] | 15.7±9.8 | 7.6±4.2 [−47] |
| PC (U/mg) | 209.7±82.6 | 248.1±97.7 [+18] | 122.1±34.6 | 80.5±24.4 [−34] |
Data from reference 10
Oxidative stress has been invoked as the link between placental insults (ischemiareperfusion, hypoxia, inflammation) and the increased apoptosis observed in preeclampsia (reviewed in [14, 15]. The rarity of cells positive for apoptosis in our studyis similar to what has been observed in normal pregnancy using similar markers and techniques [24], and lower than in preeclampsia [25]. Thus, at least when measured with IHC, high altitude placentas do not have increased apoptosis, consistent with reduced oxidative stress.
There is controversy over what are the best markers for measurement of oxidative stress. We chose to measure 4-HNE+MDA, protein carbonylation and nitration as endpoints because they reflect a broad range of cellular responses to excess ROS, and can be compared with a wide spectrum of tissue studies examining oxidative damage. There are data showing both increase [26, 27], and decrease in ROS with hypoxia [28], depending on tissue type and experimental design. While more direct markers such as ROS and NO production would be useful, they cannot be measured in these ex-vivo samples. The simplest explanation for our data is that ROS production is diminished and anti-oxidant enzymes are down-regulated, or, more importantly, not up-regulated in high-altitude placentas as a result of lower oxygen tension. This explanation is supported by in vivo experimental animal studies in which hypoxia did not change multiple parameters of oxidative status in the placenta, whereas they were increased in hyperoxia [29]. In contrast, in vitro studies show that hypoxia produces increased lipid peroxidation in human trophoblast [30] and increased trophoblast apoptosis that is reversible by caspase inhibition [31]. Differences between in vivo and in vitro data may well be due to the experimental conditions. In vitro studies obligatorily shift the placenta from a ∼6% oxygen environment to 21% room air for digestion and isolation of trophoblast, followed by re-entry into an extremely low (<1-2%) oxygen environment. This produces hypoxiare-oxygenation, which induces greater oxidative stress and apoptosis than hypoxia alone [32, 33].
Systemic oxidative stress is associated with an altered balance between reactive oxygen species (ROS) and nitric oxide; several possible explanations for our findings derive from this. Excess superoxide anion, largely mitochondrial in origin, is most commonly implicated as the source of increased ROS in preeclampsia [34, 35, 14]. Of relevance to our findings in light of the increased nitration of trophoblast is that NO also serves as an effective scavenger of superoxide (forming peroxynitrite) and of reactive lipid radicals (e.g. peroxyl). Thus an alternative explanation is that NO may be acting as a superoxide sink in the high-altitude placenta. This explanation is attractive as increased NO scavenging combined with low SOD activity would contribute to lower hydrogen peroxide and hence the reduced lipid peroxidation seen in the high altitude placenta. Alternatively, or in addition, it may be that superoxide production is similar at low versus high altitude, but the relative decrease we report in SOD, combined with a faster reaction rate of NO and superoxide relative to SOD [36] channels more of the superoxide towards the formation of peroxynitrite instead of hydrogen peroxide (Figure 5). If there were increased peroxynitrite production in the high altitude placenta, it could inhibit SOD (Mn) though not SOD (Cu/Zn) activity [37, 38] and inactivate glutathione peroxidase [39]. This mechanism would be consistent with our data and would further escalate NO scavenging of superoxides (Figure 5). Uterine vascular eNOS and NO are increased by hypoxia [40], as are NO metabolites in the circulation of experimental animals [41] and humans with preeclampsia [42]. There is increased eNOS in the syncytiotrophoblast and endothelium in preeclamptic placentas [43, 44] and in in vitro placental hypoxia [45]. Thus stimulated NO production may be elevated in both preeclampsia and in chronic hypoxia, and account for the parallel between our data on placental hypoxia and the increased nitrotyrosine staining seen in preeclampsia.
In conclusion, our findings show no link between in vivo chronic placental hypoxia, oxidative stress and increased apoptosis. Chronic hypoxia does appear to contribute to increased nitrative stress. It is possible that there is preferential diversion of ROS to interact with NO to form peroxynitrites, and hence increased syncytiotrophoblast nitration, but this does not translate into increased apoptosis. These data suggest that oxidation as opposed to nitration may be the more important determinant of increased apoptosis in the preeclamptic placenta. Greater nitration is not inherently linked to increased apoptosis in our model, thus it is unlikely that increased nitrative stress in preeclampsia reflects a pathophysiological process unique to preeclampsia. It may instead be secondary to placental hypoxia, to generalized reduction in anti-oxidant defenses, or to metabolic derangement as in gestational diabetes [46, 47]. Further investigation is necessary to determine the balance between NO and ROS under hypoxic conditions in the placenta and the role of reactive nitrative species in modulating ROS production. We also speculate that under conditions of chronic hypoxia, if anti-oxidant defenses are insufficiently induced, additional oxidative insults, e.g. short periods of ischemia, may render the system more vulnerable and this may contribute to the well-known increased susceptibility to preeclampsia at high altitude.
ACKNOWLEDGEMENTS
Funding was provided by the National Science Foundation BCS 0309142, NIH HD 42737, American Heart Association Colorado Affiliate CWGB 27 (the Honorable and Harry and Mrs. Florence Ruston Award) & American Heart Association Grant-in-Aid 96-014220 to Dr. Zamudio. Additional funding was provided by NIH HD 46982 to Dr. Illsley. The authors thank Polly Lee and Martha Tissot van Patot for help with patient recruitment and sample collection. Dr. Lisa Zwerdlinger of the Leadville Medical Center is gratefully acknowledged for patient referrals and support of the research effort. We thank the nursing staff in the Labor and Delivery sections of the following hospitals: St. Vincent's Hospital, Leadville, Colorado, University Hospital, St. Joseph's Hospital, St. Luke's Presbyterian Hospital, Rose Medical Center, Swedish Hospital and St. Anthony's Hospital, Denver, Colorado for their help and support in placental collections.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Mahfouz AAR, El-Aid MM, Alakija W, Al-Erian RAG. Altitude and socio-biological determinants of pregnancy-associated hypertension. Int J. Obstet Gynecol. 1994;44:135–138. doi: 10.1016/0020-7292(94)90067-1. [DOI] [PubMed] [Google Scholar]
- 2.Palmer SK, Moore LG, Young D, Cregger B, Berman JC, Zamudio S. Altered blood pressure course during normal pregnancy and increased preeclampsia at high altitude (3100 meters) in Colorado. Am J Obstet Gynecol. 1999;180:1161–8. doi: 10.1016/s0002-9378(99)70611-3. [DOI] [PubMed] [Google Scholar]
- 3.Keyes LE, Armaza JF, Niermeyer S, Vargas E, Young DA, Moore LG. Intrauterine growth restriction, preeclampsia, and intrauterine mortality at high altitude in Bolivia. Pediatr Res. 2003;54:20–5. doi: 10.1203/01.PDR.0000069846.64389.DC. [DOI] [PubMed] [Google Scholar]
- 4.Zamudio S. The placenta at high altitude. High Alt Med Biol. 2003;4:171–91. doi: 10.1089/152702903322022785. [DOI] [PubMed] [Google Scholar]
- 5.Zamudio S, Palmer SK, Droma T, Stamm E, Coffin C, Moore LG. Effect of altitude on uterine artery blood flow during normal pregnancy. J. Appl. Physiol. 1995;79:7–14. doi: 10.1152/jappl.1995.79.1.7. [DOI] [PubMed] [Google Scholar]
- 6.Soleymanlou N, Jurisica I, Nevo O, Ietta F, Zhang X, Zamudio S, Post M, Caniggia I. Molecular evidence of placental hypoxia in preeclampsia. J Clin Endocrinol Metab. 2005;90:4299–308. doi: 10.1210/jc.2005-0078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hubel CA, Roberts JM, Taylor RN, Musci TJ, Rogers GM, McLaughlin MK. Lipid peroxidation in pregnancy: new perspectives on preeclampsia. Am J Obstet Gynecol. 1989;161:1025–34. doi: 10.1016/0002-9378(89)90778-3. [DOI] [PubMed] [Google Scholar]
- 8.Fattman CL, Schaefer LM, Oury TD. Extracellular superoxide dismutase in biology and medicine. Free Radic Biol Med. 2003;35:236–56. doi: 10.1016/s0891-5849(03)00275-2. [DOI] [PubMed] [Google Scholar]
- 9.Sen CK. Cellular thiols and redox-regulated signal transduction. Curr Top Cell Regul. 2000;36:1–30. doi: 10.1016/s0070-2137(01)80001-7. [DOI] [PubMed] [Google Scholar]
- 10.Vanderlelie J, Venardos K, Clifton VL, Gude NM, Clarke FM, Perkins AV. Increased biological oxidation and reduced anti-oxidant enzyme activity in preeclamptic placentae. Placenta. 2005;26:53–8. doi: 10.1016/j.placenta.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 11.Shibata E, Ejima K, Nanri H, Toki N, Koyama C, Ikeda M, Kashimura M. Enhanced protein levels of protein thiol/disulphide oxidoreductases in placentae from preeclamptic subjects. Placenta. 2001;22:566–72. doi: 10.1053/plac.2001.0693. [DOI] [PubMed] [Google Scholar]
- 12.Magalhaes J, Ascensao A, Marques F, Soares JM, Ferreira R, Neuparth MJ, Duarte JA. Effect of a high-altitude expedition to a Himalayan peak (Pumori, 7,161 m) on plasma and erythrocyte antioxidant profile. Eur J Appl Physiol. 2005;93:726–32. doi: 10.1007/s00421-004-1222-2. [DOI] [PubMed] [Google Scholar]
- 13.Jefferson JA, Simoni J, Escudero E, Hurtado ME, Swenson ER, Wesson DE, Schreiner GF, Schoene RB, Johnson RJ, Hurtado A. Increased oxidative stress following acute and chronic high altitude exposure. High Alt Med Biol. 2004;5:61–9. doi: 10.1089/152702904322963690. [DOI] [PubMed] [Google Scholar]
- 14.Myatt L, Cui X. Oxidative stress in the placenta. Histochem Cell Biol. 2004;122:369–82. doi: 10.1007/s00418-004-0677-x. [DOI] [PubMed] [Google Scholar]
- 15.Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science. 2005;308:1592–4. doi: 10.1126/science.1111726. [DOI] [PubMed] [Google Scholar]
- 16.Clifton VL, Vanderlelie J, Perkins AV. Increased anti-oxidant enzyme activity and biological oxidation in placentae of pregnancies complicated by maternal asthma. Placenta. 2005;26:773–9. doi: 10.1016/j.placenta.2004.10.018. [DOI] [PubMed] [Google Scholar]
- 17.Lessey BA, Castelbaum AJ, Sawin SW, Buck CA, Schinnar R, Bilker W, Strom BL. Aberrant integrin expression in the endometrium of women with endometriosis. J Clin Endocrinol Metab. 1994;79:643–9. doi: 10.1210/jcem.79.2.7519194. [DOI] [PubMed] [Google Scholar]
- 18.Yip R. Altitude and birth weight. J Pediatr. 1987;111:869–76. doi: 10.1016/s0022-3476(87)80209-3. [DOI] [PubMed] [Google Scholar]
- 19.Reshetnikova OS, Burton GJ, Milovanov AP. Effects of hypobaric hypoxia on the fetoplacental unit: the morphometric diffusing capacity of the villous membrane at high altitude. American Journal of Obstetrics & Gynecology. 1994;171:1560–5. doi: 10.1016/0002-9378(94)90402-2. [DOI] [PubMed] [Google Scholar]
- 20.Soothill PW, Nicolaides KH, Rodeck CH, Campbell S. Effect of gestational age on fetal and intervillous blood gas and acid-base values in human pregnancy. Fetal Ther. 1986;1:168–75. doi: 10.1159/000262264. [DOI] [PubMed] [Google Scholar]
- 21.Caniggia I, Winter J, Lye SJ, Post M. Oxygen and placental development during the first trimester: implications for the pathophysiology of pre-eclampsia. Placenta. 2000;21(Suppl A):S25–30. doi: 10.1053/plac.1999.0522. [DOI] [PubMed] [Google Scholar]
- 22.Caniggia I, Wu YY, Zamudio S. Overexpression of HIF-1 alpha in placentas from high altitude pregnancies. Placenta. 2002;23:A49. [Google Scholar]
- 23.Nevo O, Soleymanlou N, Wu Y, Xu J, Kingdom J, Many A, Zamudio S, Caniggia I. Increased Expression of sFlt-1 in In Vivo and In Vitro Models of Human Placental Hypoxia is Mediated by HIF-1. Am J Physiol Regul Integr Comp Physiol. 2006 doi: 10.1152/ajpregu.00794.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Austgulen R, Chedwick L, Vogt Isaksen C, Vatten L, Craven C. Trophoblast apoptosis in human placenta at term as detected by expression of a cytokeratin 18 degradation product of caspase. Arch Pathol Lab Med. 2002;126:1480–6. doi: 10.5858/2002-126-1480-TAIHPA. [DOI] [PubMed] [Google Scholar]
- 25.Huppertz B, Kadyrov M, Kingdom JC. Apoptosis and its role in the trophoblast. Am J Obstet Gynecol. 2006 doi: 10.1016/j.ajog.2005.07.039. [DOI] [PubMed] [Google Scholar]
- 26.Guzy RD, Schumacker PT. Oxygen sensing by mitochondria at complex III: the paradox of increased reactive oxygen species during hypoxia. Experimental Physiology. 2006;91:807–19. doi: 10.1113/expphysiol.2006.033506. [DOI] [PubMed] [Google Scholar]
- 27.Zuo L, Clanton TL. Reactive oxygen species formation in the transition to hypoxia in skeletal muscle. Am J Physiol Cell Physiol. 2005;289:C207–16. doi: 10.1152/ajpcell.00449.2004. [DOI] [PubMed] [Google Scholar]
- 28.Hoffman DL, Salter JD, Brookes PS. The response of mitochondrial reactive oxygen species generation to steady-state oxygen tension: implications for hypoxic cell signaling. Am J Physiol Heart Circ Physiol. 2006 doi: 10.1152/ajpheart.00699.2006. [DOI] [PubMed] [Google Scholar]
- 29.Mover-Lev H, Ar A. Changes in enzymatic antioxidant activity in pregnant rats exposed to hyperoxia or hypoxia. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol. 1997;118:353–9. doi: 10.1016/s0742-8413(97)00156-4. [DOI] [PubMed] [Google Scholar]
- 30.Li H, Gu B, Zhang Y, Lewis DF, Wang Y. Hypoxia-induced increase in soluble Flt-1 production correlates with enhanced oxidative stress in trophoblast cells from the human placenta. Placenta. 2005;26:210–7. doi: 10.1016/j.placenta.2004.05.004. [DOI] [PubMed] [Google Scholar]
- 31.Levy R, Smith SD, Chandler K, Sadovsky Y, Nelson DM. Apoptosis in human cultured trophoblasts is enhanced by hypoxia and diminished by epidermal growth factor. American Journal of Physiology - Cell Physiology. 2000;278:C982–8. doi: 10.1152/ajpcell.2000.278.5.C982. [DOI] [PubMed] [Google Scholar]
- 32.Hung TH, Skepper JN, Burton GJ. In vitro ischemia-reperfusion injury in term human placenta as a model for oxidative stress in pathological pregnancies. Am J Pathol. 2001;159:1031–43. doi: 10.1016/S0002-9440(10)61778-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hung TH, Skepper JN, Charnock-Jones DS, Burton GJ. Hypoxia-reoxygenation: a potent inducer of apoptotic changes in the human placenta and possible etiological factor in preeclampsia. Circ Res. 2002;90:1274–81. doi: 10.1161/01.res.0000024411.22110.aa. [DOI] [PubMed] [Google Scholar]
- 34.Wang Y, Walsh SW. Placental mitochondria as a source of oxidative stress in preeclampsia. Placenta. 1998;19:581–6. doi: 10.1016/s0143-4004(98)90018-2. [DOI] [PubMed] [Google Scholar]
- 35.Sikkema JM, van Rijn BB, Franx A, Bruinse HW, de Roos R, Stroes ES, van Faassen EE. Placental superoxide is increased in pre-eclampsia. Placenta. 2001;22:304–8. doi: 10.1053/plac.2001.0629. [DOI] [PubMed] [Google Scholar]
- 36.Huie RE, Padmaja S. The reaction of no with superoxide. Free Radic Res Commun. 1993;18:195–9. doi: 10.3109/10715769309145868. [DOI] [PubMed] [Google Scholar]
- 37.MacMillan-Crow LA, Crow JP, Kerby JD, Beckman JS, Thompson JA. Nitration and inactivation of manganese superoxide dismutase in chronic rejection of human renal allografts. Proc Natl Acad Sci U S A. 1996;93:11853–8. doi: 10.1073/pnas.93.21.11853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van der Loo B, Bachschmid M, Skepper JN, Labugger R, Schildknecht S, Hahn R, Mussig E, Gygi D, Luscher TF. Age-associated cellular relocation of Sod 1 as a self-defense is a futile mechanism to prevent vascular aging. Biochem Biophys Res Commun. 2006;344:972–80. doi: 10.1016/j.bbrc.2006.03.224. [DOI] [PubMed] [Google Scholar]
- 39.Padmaja S, Squadrito GL, Pryor WA. Inactivation of glutathione peroxidase by peroxynitrite. Arch Biochem Biophys. 1998;349:1–6. doi: 10.1006/abbi.1997.0407. [DOI] [PubMed] [Google Scholar]
- 40.Xiao D, Bird IM, Magness RR, Longo LD, Zhang L. Upregulation of eNOS in pregnant ovine uterine arteries by chronic hypoxia. Am J Physiol Heart Circ Physiol. 2001;280:H812–20. doi: 10.1152/ajpheart.2001.280.2.H812. [DOI] [PubMed] [Google Scholar]
- 41.Zhang L, Xiao D, Bouslough DB. Long-term high-altitude hypoxia increases plasma nitrate levels in pregnant ewes and their fetuses. American Journal of Obstetrics & Gynecology. 1998;179:1594–8. doi: 10.1016/s0002-9378(98)70031-6. [DOI] [PubMed] [Google Scholar]
- 42.Norris LA, Higgins JR, Darling MR, Walshe JJ, Bonnar J. Nitric oxide in the uteroplacental, fetoplacental, and peripheral circulations in preeclampsia. Obstetrics & Gynecology. 1999;93:958–63. doi: 10.1016/s0029-7844(99)00007-1. [DOI] [PubMed] [Google Scholar]
- 43.Myatt L, Rosenfield RB, Eis AL, Brockman DE, Greer I, Lyall F. Nitrotyrosine residues in placenta. Evidence of peroxynitrite formation and action. Hypertension. 1996;28:488–93. doi: 10.1161/01.hyp.28.3.488. [DOI] [PubMed] [Google Scholar]
- 44.Nasiell J, Nisell H, Blanck A, Lunell NO, Faxen M. Placental expression of endothelial constitutive nitric oxide synthase mRNA in pregnancy complicated by preeclampsia. Acta Obstetricia et Gynecologica Scandinavica. 1998;77:492–6. [PubMed] [Google Scholar]
- 45.Seligman SP, Nishiwaki T, Kadner SS, Dancis J, Finlay TH. Hypoxia stimulates ecNOS mRNA expression by differentiated human trophoblasts. Ann N Y Acad Sci. 1997;828:180–7. doi: 10.1111/j.1749-6632.1997.tb48538.x. [DOI] [PubMed] [Google Scholar]
- 46.Lyall F, Gibson JL, Greer IA, Brockman DE, Eis AL, Myatt L. Increased nitrotyrosine in the diabetic placenta: evidence for oxidative stress. Diabetes Care. 1998;21:1753–8. doi: 10.2337/diacare.21.10.1753. [DOI] [PubMed] [Google Scholar]
- 47.Coughlan MT, Vervaart PP, Permezel M, Georgiou HM, Rice GE. Altered placental oxidative stress status in gestational diabetes mellitus. Placenta. 2004;25:78–84. doi: 10.1016/S0143-4004(03)00183-8. [DOI] [PubMed] [Google Scholar]


