Abstract
The rpoN gene encoding the transcription factor σ54 in Myxococcus xanthus has been cloned using a heterologous rpoN probe. The sequence of the cross-hybridizing DNA confirmed the existence of an ORF 1518 bp long that encodes a well conserved member of the σ54 family of σ factors. Low- as well as high-stringency hybridizations detected only a single rpoN gene in the M. xanthus chromosome. In other bacteria, σ54 is an alternative σ, and null mutants are viable. However, all attempts to construct a strain containing a null mutation in the M. xanthus rpoN have been unsuccessful. Partial diploids of rpoN+/rpoN null are viable. Recombination experiments with such partial diploids showed the impossibility of constructing, either by segregation or by transduction, a viable null haploid under any of a wide range of growth conditions. The product of the rpoN gene, σ54, therefore appears to be essential for growth in M. xanthus.
Keywords: transcription, σ factor, RNA polymerase, promoter, activator proteins
Promoter specificity in the eubacteria is directed by a σ factor, one component of the 5-subunit RNA polymerase holoenzyme (α2ββ′σ). The major or primary σ factor, called σ70 in Escherichia coli, is used in the transcription of many genes that are essential for vegetative growth of the organism. As a consequence, rpoD, which encodes σ70, is a vital gene for E. coli. In some organisms this single σ factor may be sufficient: both the fully sequenced 580-kbp genome of Mycoplasma genitalium, which may represent the minimal functional gene set of a self-replicating organism, as well as the genome of Mycoplasma pneumoniae appear to encode only a single σ factor, and it is like σ70 (1–3). However, in addition to their primary σ factor, many eubacteria have been found to express alternative σ factors that recognize alternative promoter sequences. The presence or activity of alternative σ factors is therefore important in determining which particular set of genes is expressed under a given set of conditions. The many alternative σ factors can be divided into two major families, the larger of which shows structural and functional similarity to the primary σ factor σ70. Many organisms produce several members of the σ70 family of alternative σ factors, each of which is often specialized to allow the cells to mount a coordinated physiological response, adapting the organism to particular growth conditions. Examples are the heat shock σ factor of E. coli, σ32, or the sporulation-specific σ factors of Bacillus subtilis (4). In addition to the primary σ factor σ70, the fully sequenced genome of Haemophilus influenzae Rd encodes two homologs of σE and one of σ32 (5). Despite their importance for the mounting of specialized metabolic responses, none of these alternative σ factors has been found essential for growth under standard laboratory conditions in any organism.
The second and smaller family of alternative σ factors, the σ54 family, has little sequence similarity to σ70 and is functionally distinct as well. In E. coli, σ54 represents at most 10% of the total stock of σ factors (6). Generally only one, but occasionally two, highly homologous genes encoding a σ54-type σ factor are found in an organism. As yet, no member of the σ54 family had been reported to be essential for growth. The σ54 family of σ factors is associated with the expression of specialized metabolic functions (for example, the utilization of alternative carbon or nitrogen sources), for nitrogen fixation, or for flagellar and pilus biosynthesis (7, 8). σ54-Dependent transcription is always positively regulated in response to signals from the cell’s environment or to a particular phase of the cell cycle. A change in the activity state of a positive regulatory protein, either by phosphorylation, direct protein–protein interaction, or direct effector activation, appears to be the regulatory mechanism (9). These specialized regulatory proteins, which bind to enhancer-like sequences upstream of the core σ54 promoter, are thus responsible for transducing the environmental or cell-cycle signals that activate transcription. When the required activator protein is either absent or inactive, the σ54-containing RNA polymerase holoenzyme, though able to bind a promoter as a closed complex, is unable to proceed to form an open complex and to initiate transcription (10, 11).
When starved for nutrients, Myxococcus xanthus undergoes a multicellular developmental program that takes roughly 1 day to complete. Within this program, new proteins are synthesized, genes are expressed at specific times, and extracellular signals are exchanged between cells, resulting in the aggregation of ≈100,000 cells into a mound and their differentiation into metabolically dormant spores within that mound (12–16). Promoters of the developmentally regulated 4521 and mbhA genes as well as the vegetatively expressed pilA gene do resemble in sequence the −24/−12 promoters recognized by σ54 in other bacteria (refs. 17 and 18; S. Wu, personal communication). Mutagenesis of the 4521 promoter region has shown that DNA base positions that are shared by the consensus σ54 and 4521 promoters are also essential for promoter activity (17). Moreover, both mbhA and 4521 require DNA upstream of the core promoter for transcription of the gene, providing for potential activator binding sites (18, 19).
To investigate the extent of σ54 function for gene expression during fruiting body development of M. xanthus, rpoN, the structural gene encoding σ54, was cloned and sequenced, and a gene knock-out was attempted. Unexpectedly, the rpoN gene appears to be essential for the growth of M. xanthus, even when all amino acids are provided in amounts sufficient to serve as carbon and energy sources for the tricarboxylic acid cycle and to support protein synthesis.
MATERIALS AND METHODS
Bacterial and Phage Growth and Transduction.
Standard culture conditions were employed for both E. coli (20) and M. xanthus (21) unless otherwise noted. Transducing stocks of phage Mx4 were prepared on plates as described (22, 23) and were stored at 4°C over chloroform. Recipient bacteria were transduced by adding 5–200 μl of phage stock to 400 μl of logarithmically growing recipient cells, allowing 15–30 min at room temperature for attachment, and plating the mixture with 2.5 ml casitone-Tris (CTT, a complex growth medium) soft agar onto CTT agar plates containing the selective antibiotic and other additions as noted. Plates were incubated at 33°C or 18°C for 5–7 days, and in cases of slow growth for several weeks.
General Methods.
Preparation and manipulation of chromosomal and plasmid DNA followed standard protocols (20, 22, 24). Electroporation of plasmid DNA into E. coli or M. xanthus has been described (25–27). Low-stringency Southern hybridization conditions included overnight hybridization in 30% formamide, 5× standard saline citrate (SSC), 5% dextran sulfate, 1% SDS at 42°C, and two 30 min washes in 5× standard saline phosphate/EDTA (SSPE) and 0.1% SDS at 42°C.
Cloning the rpoN Gene.
M. xanthus DK101 chromosomal DNA was digested with BamHI, and the resulting restriction fragments were separated on a 0.6% low-melting-temperature agarose gel. A piece of gel that contained restriction fragments of ≈6.5 kbp was excised and ligated to BamHI-digested and alkaline phosphatase-treated pBluescriptIIKS DNA. E. coli DH5α was electroporated with this ligation mixture, and the bacteria were plated on Luria–Bertani plates containing ampicillin and the chromogenic substrate 5-bromo-4-chloro-3-indolyl β-d-galactoside (X-Gal) to indicate which colonies carried recombinant plasmid. Altogether, 240 individual white colonies were picked onto fresh Luria–Bertani plus ampicillin plates. Liquid cultures were inoculated with pools of 10 individual colonies each, and plasmid DNA was prepared from the resulting 24 pools. Each plasmid pool was digested with BamHI, and the resulting restriction fragments were separated on an agarose gel and transferred onto a nylon membrane, and probed with Caulobacter crescentus rpoN DNA isolated from pYB101 (28). Six of the 24 pools gave a positive hybridization signal with the rpoN probe. Plasmid was prepared from single isolated colonies of two of the positive pools, digested with BamHI, blotted, and probed again as above. Three positive clones were identified, and two that had been isolated from different pools were analyzed further. These two plasmids yielded identical restriction patterns with several enzymes, and one plasmid was named pIMK200.
An NcoI fragment that was expected to contain a complete rpoN gene was similarly cloned from M. xanthus DK101 chromosomal DNA, except that positive clones were directly identified by colony hybridization (20) using a 1.6-kbp BamHI–SalI subfragment of pIMK200 (see Fig. 1). One of 194 screened colonies gave a positive signal, and the plasmid it contained was named pIMK206.
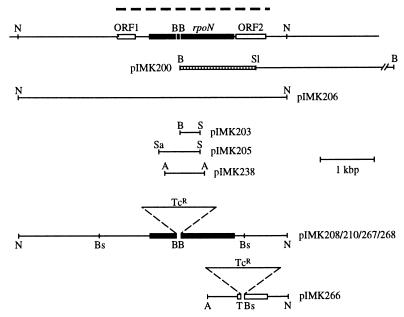
Figure 1.
Restriction map of the rpoN locus and derived plasmids. The top line shows a map of the chromosomal rpoN locus and its neighbors. The closed box drawn on this line denotes the extent of the predicted rpoN ORF, and the two open boxes show the location of ORF1 and ORF2 upstream and downstream of rpoN that are evident in the sequence. The heavy dashed line above the ORFs indicates the extent of the sequence deposited in GenBank. Restriction fragments that were used in this study are shown below the map of the chromosomal locus in the corresponding positions and with the plasmids that carry them. A BamHI–SalI subfragment of pIMK200 which was used as the probe for rpoN is indicated as a hatched box. The ORFs disrupted by insertion of a tetracycline resistance (TcR) gene in plasmids pIMK208/210/267/268 (deleting an 81-bp BamHI fragment) and pIMK266 (deleting a 64-bp Tth111I–BspEI fragment) are indicated by the closed and open boxes as noted above. A, ApaI; B, BamHI; Bs, BspEI; N, NcoI; S, SmaI; Sa, Sau3AI; Sl, SalI; T, Tth111I.
Sequencing and Sequence Analysis.
For sequencing M. xanthus rpoN, appropriate restriction fragments of pIMK200 and pIMK206 were subcloned into the pBluescriptII or pGEM5Zf(+) vectors. The rpoN gene and the surrounding DNA were sequenced on both strands by the dideoxy chain-termination method of Sanger et al. (29) using a Sequenase 2.0 kit (United States Biochemical). When subclones could not be obtained, specific oligonucleotides were synthesized to bridge occasional gaps in the sequence. Single-stranded DNA used for sequencing was prepared as described (30). The sequence was assembled with the geneworks program (IntelliGenetics) and analyzed with the help of the gcg package of programs of the University of Wisconsin Genetics Computer Group.
RESULTS AND DISCUSSION
Cloning the rpoN Gene from M. xanthus.
To search for a M. xanthus homolog of the rpoN gene, which would encode a σ factor of the σ54 family, the rpoN gene from C. crescentus—a similarly high G+C organism—was used as a heterologous probe. A Southern blot of M. xanthus genomic DNA that had been digested with XhoI, EcoRI, SalI, PstI, or BamHI was probed with a 1.6-kbp EcoRI–BamHI fragment containing the entire C. crescentus rpoN coding region (28). In all restriction digests, this probe detected a clearly hybridizing fragment of M. xanthus chromosomal DNA, indicating that M. xanthus contained a homolog of the C. crescentus rpoN gene. One of the hybridizing restriction fragments of M. xanthus chromosomal DNA, a 6.5-kbp BamHI fragment, was cloned to yield pIMK200 (details in Materials and Methods; map in Fig. 1). Subsequent hybridization and sequence analysis showed that pIMK200 contains most but not all of the DNA encoding σ54. To find a useful restriction enzyme for cloning the entire rpoN locus, M. xanthus genomic DNA was digested with a panel of restriction enzymes and probed with a 1.4-kbp BamHI–SalI subfragment of pIMK200 indicated in Fig. 1. Integration of the restriction maps of pIMK200 and the genomic rpoN locus implied that an ≈5-kbp NcoI restriction fragment should contain the entire rpoN gene. This NcoI fragment was cloned to yield pIMK206 (Fig. 1).
Low stringency hybridizations of a homologous rpoN probe consisting of the BamHI–SalI fragment of pIMK200 to M. xanthus genomic DNA digested with a panel of restriction enzymes did not reveal any additional hybridizing fragments beyond those expected from the restriction map of the rpoN locus itself (data not shown). This indicates that only one homolog of σ54 is encoded in the M. xanthus genome. The position of this unique rpoN gene on the physical map of the M. xanthus chromosome was determined by hybridizing a DNA fragment internal to rpoN to a Southern blot of chromosomal DNA digested with the rarely cutting restriction enzymes AseI and SpeI and separated by clamped homogeneous electric field (CHEF; Bio-Rad) gel electrophoresis. Hybridization to the AseI fragments M or N and SpeI fragments U′ or U" of the M. xanthus genome map (31, 32) was detected (data not shown). Fragments M and N as well as U′ and U" are of similar size and difficult to resolve from each other even on CHEF electrophoresis gels, but they are all located in the upper right quadrant of the whole genome map as conventionally drawn (33), thus localizing rpoN to that quadrant.
Sequence of M. xanthus rpoN.
Both pIMK200 and pIMK206 were subcloned to facilitate sequencing of the rpoN gene and its upstream and downstream regions. The combined sequence has been deposited in GenBank under accession number U81516U81516. Because M. xanthus DNA contains 67.5% G+C base pairs (34), which is close to the theoretical maximum, third positions in its codons and codon usage are strongly biased and can be used to identify ORFs (35, 36). Both codon usage (33, 37) and third-position G+C bias indicated the presence of one incomplete and two complete ORFs in the same orientation in the sequence. The largest and complete ORF in the region is similar in predicted amino acid sequence to other σ54 proteins. There are three potential ATG translational start codons in this reading frame following an upstream stop codon. Based on the lack of a ribosome binding site, third-position G+C bias, codon usage, and the lack of sequence similarity, the first ATG codon appeared unlikely to define the start of the rpoN coding sequence. Two more ATG codons, separated from each other by only one codon, occur in the same reading frame 51 bases downstream from the first ATG and could be used for the initiation of translation. A potential ribosome binding site (GGAG) is located 10 bp upstream of the first of these two ATGs. Tentatively, this more upstream ATG codon is adopted as the start codon, as both the Azorhizobium caulinodans and Acinetobacter calcoaceticus rpoN genes also begin with two potential ATG codons, separated by one codon, in the same reading frame (38, 39). With this assignment, the predicted rpoN coding sequence extends for 1518 bp; it has the potential to encode a 505-amino acid polypeptide, predicting an unmodified molecular weight of 56.9 kDa (Fig. 2A).
Figure 2.
Sequence of M. xanthus RpoN. (A) Short fragment of nucleotide sequence (lowercase letters), which includes the putative ribosome binding site (boldface letters) and the proposed ATG start codon (underlined). The deduced RpoN amino acid sequence is shown below the nucleotide sequence; amino acids are abbreviated using the standard single-letter code, and the stop codon is represented by an asterisk. (B) Proposed domain structure of M. xanthus RpoN: Q-rich domain, amino acids 1–40; acidic domain, amino acids 40–139; helix-turn-helix motif, amino acids 395–412; RpoN box, amino acids 481–493.
When the predicted amino acid sequence of the M. xanthus rpoN gene was aligned with that of rpoN genes from other organisms, few differences were observed (Fig. 2B). The elements that are normally highly conserved, such as the so-called RPON box at the C-terminal end of the protein (40), were present in the M. xanthus sequence. In addition, a conserved glutamine-rich domain is present at the amino terminus of RpoN, followed by a nonconserved, but generally acidic, domain that is of variable length, and in the case of M. xanthus extends from residues 40 to 139. A proposed helix-turn-helix motif is also found in the M. xanthus sequence, extending from positions 395 to 412.
Deletion of rpoN.
A null mutant of rpoN was sought to investigate the function of σ54. Such a mutant might be expected to exhibit a defect in social motility, since vegetative expression of the pilin structural gene, pilA, may be σ54 dependent (S. Wu, personal communication). pilA null mutants are viable but lack pili (41). In addition, it has been suggested that at least two genes regulated by fruiting body development, mbhA (42) and 4521 (17), have σ54 promoters. Mutations in the conserved −24 and −12 regions or a 1-base shortening of the 4521 promoter decreased expression from a β-galactosidase transcriptional reporter by 87% or more (17). However, neither mbhA nor 4521 is essential for development, since their null mutants develop normally or are only slightly delayed (12, 18). If σ54 is involved in the expression of other developmentally regulated genes in addition to mbhA and 4521, some of them may be essential for particular steps in the developmental process. The nature of those steps would be revealed in the developmental phenotype of an rpoN null mutant.
Several different approaches were used to construct the desired knock-out of rpoN and are summarized in Table 1. First, we attempted to disrupt rpoN by seeking the integration by homologous recombination of plasmids carrying an internal fragment of the rpoN gene (Fig. 3A). Two different internal fragments were used, a 384-bp BamHI–SmaI (pIMK203) and the considerably larger 779-bp Sau3AI–SmaI (pIMK205) restriction fragment (Fig. 1). pIMK203 and pIMK205, conferring KmR, were electroporated into M. xanthus DK101 in 6 and 15 experiments, respectively, using a total of 7 independent preparations of electroporation recipient cells. Although 6 of the 7 cell preparations accepted and integrated other (control) plasmids successfully by homologous recombination, none of these 21 experiments yielded kanamycin-resistant colonies containing the plasmids integrated at the rpoN locus (Table 1).
Table 1.
Knock-out of rpoN
| Type of experiment | Plasmids | Successes/no. of experimental trials |
|---|---|---|
| Integration of internal fragment | pIMK203 | Recovery of electroporants: 0/6 |
| pIMK205 | Recovery of electroporants: 0/15 | |
| Resolution of tandem duplications | pIMK208 | Double recombinants: 0/100 |
| pIMK210 | Loss of wt allele: 0/26; 0/3050 | |
| pIMK267/268 | Double recombinants: 0/39 | |
| Loss of wt allele: 0/438 | ||
| Transduction of null allele | pIMK238 | 0/232; 0/6216* |
wt, wild type.
*More data in Table 2.
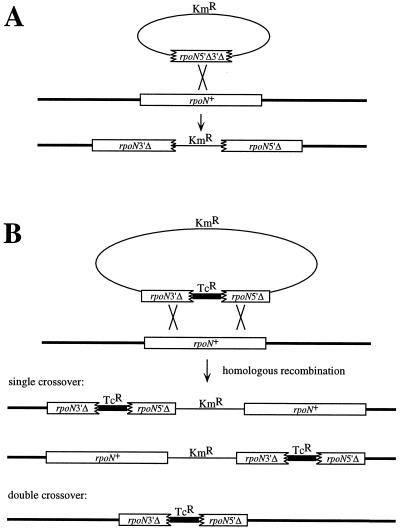
Figure 3.
Disruption of rpoN. (A) Integration of an internal fragment. A plasmid containing kanamycin resistance (KmR) as the selectable marker and an internal fragment of the rpoN gene (rpoN5′Δ 3′Δ, shown as a box with both ends jagged) can integrate by homologous recombination (denoted by an X) into the chromosomal wild-type rpoN locus, resulting in two partially deleted copies of rpoN (rpoN3′Δ and rpoN5′Δ, shown as boxes with a jagged 3′ end and a jagged 5′ end, respectively) separated by the plasmid carrying the KmR marker. (B) Gene replacement. A plasmid containing KmR as the selectable marker is shown as a thin line; it also contains the rpoN gene disrupted by a TcR gene (heavy line) and by loss of 81 bp. Integration into the chromosomal locus by a single crossover homologous recombination can create two different types of tandem duplications with one intact and one disrupted copy of rpoN. A double crossover will replace the wild-type rpoN allele with the TcR-marked null allele and will have lost the KmR marker carried on the plasmid.
An alternative strategy was tested to interrupt the rpoN gene. A gene encoding TcR was inserted between two BamHI sites within the rpoN coding region (Fig. 1). This insertion both disrupted rpoN and deleted 81 bp from the interior of the gene. The 6.4-kbp NcoI fragment containing the rpoN gene now disrupted by a TcR gene was cloned into pBGS18 (43), pDelta-1 (Stratagene), or pBJ113 (ref. 44; B. Julien, personal communication), resulting in pIMK208, pIMK210, and pIMK267/268, respectively, and electroporated into M. xanthus with selection for TcR. None of these plasmids are autonomous; they can therefore only be maintained in M. xanthus if they integrate by homologous recombination, becoming part of the rpoN chromosomal locus. Three types of TcR integrants are expected as diagrammed in Fig. 3B. The two types of single crossover recombinants (recombination to the left or to the right of TcR) retain the KmR that is carried by the vector, while the double crossover recombinant loses KmR; thus single and double crossover events are readily distinguished by their drug resistance. pIMK208 electroporants selected for TcR were tested for KmR. All the ≈100 isolates tested retained KmR, indicating that only single crossover recombinants had survived. In these recombinants a complete pIMK208 should have integrated into the chromosome and created a tandem duplication with one null and one wild-type allele of rpoN (Fig. 3B). This was confirmed by Southern blots of restriction digests of DNA prepared from three independent recombinants. No strains were obtained (0/100) that were double crossover recombinants; such strains would have their wild-type chromosomal rpoN allele replaced by the plasmid-borne gene disrupted by the TcR cassette, and would completely lack σ54 (Table 1). Parallel results were obtained when pIMK210 was electroporated into M. xanthus.
DNA from one TcR KmR isolate (DK9600) which had integrated pIMK210 by single crossover, as indicated by its retention of KmR, was digested with restriction enzymes and Southern blotted, confirming the integration of pIMK210 by single crossover of the second type (Fig. 3B). The absence of double crossover recombinants might indicate that the second crossover which would resolve the partial duplication and remove the vector sequences from the chromosome is for some reason difficult and thus of low frequency. Extended opportunities for the second (resolving) recombination were provided by propagation of this duplication strain (DK9600) for 20–40 generations without antibiotic, conditions that are nonselective. Without antibiotic selection, homologous recombination could occur between the two rpoN alleles at any stage of this extended growth, excising one of those copies and the plasmid, with its KmR, integrated between them. After plating on CTT agar without either antibiotic, single colonies were scored for resistance to tetracycline and kanamycin. Among 3050 single colony isolates tested, 26 (0.85%) had lost both KmR and TcR and had therefore lost the mutant copy of rpoN and the vector plasmid. None of the colonies (0/3050) had lost KmR while retaining TcR (Table 1). The combination of kanamycin sensitivity and TcR would, of course, have indicated a loss of the rpoN wild-type allele together with the vector sequences, leaving only the null rpoN allele in single copy.
To increase the sensitivity of this type of segregation experiment even further, two new plasmids, pIMK267 and pIMK268, were constructed. These plasmids contain the full rpoN gene disrupted by TcR as in pIMK208/210, but cloned in pBJ113, which is a derivative of pUC118 containing KmR and galK as the selectable and counterselectable markers, respectively (ref. 44; B. Julien, personal communication). The presence of galK, which encodes galactokinase, renders M. xanthus cells sensitive to galactose in the medium, presumably because the product of galactokinase action on galactose, galactose-1-phosphate, is toxic. pIMK267 and pIMK268 were electroporated into DK101 selecting for TcR. Of 39 electroporants that were tested by transferring onto fresh plates containing a single antibiotic, all were both tetracycline- and kanamycin-resistant, indicating that a single crossover had occurred in all cases. Restriction digests and Southern blots of DNA prepared from six independent recombinants confirmed that integration had occurred by single crossover, in one case upstream and in five cases downstream of the tetracycline cassette insertion. Four of these strains were then grown without selection for ≈20 generations to allow for recombination to occur, and plated onto CTT agar containing 1% galactose. Single galactose-resistant colonies, which should have lost the rpoN tandem duplication, leaving either the wild-type or mutant rpoN allele behind in single copy, were tested for resistance to kanamycin and tetracycline. Of 443 galactose-resistant colonies tested, 438 had lost both KmR and TcR, retaining the rpoN wild-type allele. Five colonies were resistant to both antibiotics, indicating the presence of both rpoN alleles. These doubly resistant strains may have arisen by mutation of the galK gene. Of the galactose-resistant single colony isolates that were obtained, none (0/438) retained TcR alone (Table 1). Again, no haploid segregants with only the rpoN null allele were obtained.
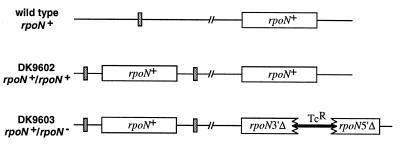
The total absence of recombinants carrying only a disrupted rpoN allele implied either that rpoN function is essential for survival, or that the rpoN locus must be intact for some reason related to chromosomal structure in the vicinity of rpoN. To discriminate between these possibilities, DK9602, a merodiploid strain, was constructed into which auxiliary wild-type copies of rpoN were integrated at the Mx8 prophage attachment site (Fig. 4). Such a strain should be viable regardless of whether function or structure is required. If rpoN function is required, it should then be possible to construct a chromosomal rpoN null allele because rpoN function is provided from the Mx8 prophage attachment site. If rpoN+ structure is required in its normal chromosomal locus, that copy is also present in DK9602. However, in this latter case a functional copy of rpoN at the Mx8 prophage attachment site would not suffice for viability. Plasmid pIMK238 was constructed that contained a 726-bp ApaI restriction fragment internal to the rpoN gene inserted in the EcoRI site of pACYC184 (45) (Fig. 1). Using DK9602 as a recipient, it was possible to integrate pIMK238 by homologous recombination into the chromosomal rpoN gene, disrupting that locus and creating a partial tandem duplication as drawn in Fig. 3A, but tagged by the TcR of the vector pACYC184 instead of KmR. The location of the integration event in the resulting strain DK9603 was confirmed by Southern blots of restriction digests of DNA prepared from DK9603, verifying the structure shown in Fig. 4. The isolation and evident viability of DK9603 rules out the possibility of a structural requirement for the intact rpoN locus and is consistent with a requirement for rpoN function.
Figure 4.
Chromosomal organization of a haploid and two rpoN partial diploids. The wild-type strain, DK101, contains a single, wild-type copy of rpoN (rpoN+) and an undisturbed Mx8 prophage attachment site (Mx8 att, stippled box). DK9602 has wild-type copies of rpoN inserted at the Mx8 att site, and DK9603, a derivative of DK9602, has a disrupted chromosomal rpoN allele in addition to the wild-type rpoN copies at Mx8 att.
In continued pursuit of a way to delete rpoN, a lysate of the generalized transducing phage Mx4 (46) prepared on strain DK9603 was used to transduce TcR into a haploid rpoN+ recipient (DK101) and into the merodiploid strain DK9602, which is protected from the effects of loss of chromosomal rpoN function by the integrated copies of rpoN at the Mx8 prophage attachment site. Recovery of tetracycline-resistant transductants would indicate the replacement of the chromosomal rpoN wild-type allele with the tandem 3′ and 5′ deletion alleles. In parallel experiments with the very same stock of transducing phage, several thousand tetracycline-resistant colonies were recovered when DK9602 (protected from loss of rpoN function) was the recipient, while none were recovered when DK101 (unprotected) was the recipient (Table 2). This transducing phage stock was titrated on both the recipient strains DK101 and DK9602. The Mx4 stock grown on DK9603 infected DK101 at 6 × 109 plaque-forming units (pfu)/ml and DK9602 at 4.5 × 109 pfu/ml, showing that the transducing phage lysate was able to infect both strains with approximately equal efficiency. Because plaque forming and transducing phage particles have the same protein coats, differential resistance of DK101 and DK9602 cannot account for differences in transduction frequency. Transduction mixtures were also plated on CTT containing yeast extract and CTT supplemented with glutamine or tyrosine in case rpoN were required for biosynthesis of glutamine, tyrosine, or another essential water soluble metabolite. Still no tetracycline-resistant transductants of DK101 were obtained (Table 2). In case σ54 were required for transcription of genes that are necessary for growth at 32°C, transductions of DK101 and DK9602 were performed at 18°C. Under those conditions, transductants appear after 2–3 weeks of incubation; again no transductants of the rpoN null allele into DK101 were isolated (Table 2).
Table 2.
Transduction of rpoN alleles
| Growth conditions* | No. of TcR transductants (rpoN null allele) into DK9602 | No. of TcR transductants (rpoN null allele) into DK101 | No. of KmR transductants (rpoN wild-type allele) into DK101 |
|---|---|---|---|
| CTT | 587 | 0 | 165 |
| CTT | 2444 | 0 | ND |
| CTT + 0.2% yeast extract | 283 | 0 | ND |
| CTT + 0.1% glutamine | 842 | 0 | ND |
| CTT + 0.1% tyrosine | 1962 | 0 | ND |
| CTT, 18°C | 98 | 0 | 67 |
| Total no. of transductants | 6216 | 0 | 232 |
ND, not determined.
*All experiments at 32°C unless otherwise noted.
Disruption of ORF2.
ORF2 begins 63 bp downstream of the end of the rpoN coding region, and it is oriented in the same direction as rpoN. If ORF2 is cotranscribed with rpoN and ORF2 provides a vital function, then disruptions of rpoN may be ORF2-negative due to a polar effect. It was therefore important to test whether ORF2 can be disrupted, and if so, what the phenotype of such a disruption strain might be. A disrupted copy of ORF2, which contained a TcR cassette inserted between the Tth111I and BspEI sites within ORF2 (Fig. 1), deleting 64 bp, was constructed. The plasmid bearing this construct, pIMK266, contained both the galK gene of E. coli as a counterselectable marker and KmR as a selectable marker. pIMK266 was electroporated into M. xanthus DK101 selecting for TcR. Of 27 electroporants that were tested, 7 (26%) appeared to have undergone double recombination as judged by kanamycin sensitivity. This double recombination replaced the wild-type ORF2 with a disrupted copy. The chromosomal structure of four single recombinants and two double recombinants was confirmed by Southern blot analysis. The evident viability of the ORF2 disruption strains implies that ORF2 is not essential for growth. Nor can it be argued that an unknown gene downstream of ORF2 in the same transcription unit is essential for growth, since the disruption of ORF2 should have been polar on such a gene. It follows, then, that the failure of rpoN disruption strains to survive should be attributed to the lack of rpoN function.
Why Might σ54 Be Vital?
The lack of success in the repeated attempts for isolating an rpoN null mutant strain of M. xanthus was unanticipated. Where it has been tested, an intact rpoN gene only appears to be essential for growth under specific nutrient limiting conditions; for example, rpoN mutants of P. aeruginosa are glutamine auxotrophs (47). Growth on media rich in amino acids and carbon has not been reported to require rpoN in any organism other than M. xanthus; the Δ(rpoN-npr)::kan mutant of E. coli is viable on Luria–Bertani medium, for example (48). However, in M. xanthus, rich media such as CTT do not appear to support growth of the organism in the absence of a functional rpoN gene. Even when CTT medium is supplemented with yeast extract, glutamine or tyrosine, or when the growth temperature is lowered to 18°C, an rpoN null mutant does not appear to be viable. It is currently not known for which specific function(s) rpoN may be required. In C. crescentus, expression of rpoN itself is cell cycle-regulated, and rpoN null mutants have a well defined, though not lethal defect in cell division (28). Perhaps expression of some essential cell cycle gene(s) depends on σ54 in M. xanthus.
It has not yet been formally shown that the M. xanthus homolog of rpoD, which should encode the major vegetative σ factor σ70, is indeed essential for viability. A single point mutant in rpoD has been described, but no systematic attempts at constructing a null allele have been published (49, 50). It is thus not clear that the rpoD gene product indeed serves as the primary σ factor in M. xanthus, although this appears to be the case in all other bacteria where such information is available. The possibility that the rpoN gene product serves this function in M. xanthus can therefore not yet be ruled out.
Transcription initiation by σ54 requires the presence of an activator protein wherever it has been tested. In M. xanthus, a general search for σ54-dependent activator proteins by PCR yielded 13 potential activator proteins, 1 of which is the previously identified pilR (41, 51). pilR regulates pilA, but knock-outs of pilA, R, or S are all viable (41). Given the necessity for rpoN+, one or a combination of several of the other activator proteins may be found essential for viability of M. xanthus.
Acknowledgments
We thank E. Wickstrom for providing the clamped homogeneous electric field (CHEF) blot of M. xanthus chromosomal DNA and S. Inouye for providing galK plasmids. This work was supported by Public Health Service Grant GM23441 to D.K.
ABBREVIATIONS
- CTT
casitone-Tris
- TcR
tetracycline resistance
- KmR
kanamycin resistance
Footnotes
References
- 1.Fraser C M, Gocayne J D, White O, Adams M D, Clayton R A, et al. Science. 1995;270:397–403. doi: 10.1126/science.270.5235.397. [DOI] [PubMed] [Google Scholar]
- 2.Ouzounis C, Casari G, Valencia A, Sander C. Mol Microbiol. 1996;20:898–890. doi: 10.1111/j.1365-2958.1996.tb02529.x. [DOI] [PubMed] [Google Scholar]
- 3.Himmelreich R, Hilbert H, Plagens H, Pirkl E, Li B-C, Herrmann R. Nucleic Acids Res. 1996;24:4420–4449. doi: 10.1093/nar/24.22.4420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gross C A, Lonetto M, Losick R. In: Transcriptional Regulation. McKnight S L, Yamamoto K R, editors. Plainview, NY: Cold Spring Harbor Lab. Press; 1992. pp. 129–176. [Google Scholar]
- 5.Fleischmann R D, Adams M D, White O, Clayton R A, Kirkness E F, et al. Science. 1995;269:496–512. doi: 10.1126/science.7542800. [DOI] [PubMed] [Google Scholar]
- 6.Jishage M, Iwata A, Ueda S, Ishihama A. J Bacteriol. 1996;178:5447–5451. doi: 10.1128/jb.178.18.5447-5451.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kustu S, Santero E, Keener J, Popham D, Weiss D. Microbiol Rev. 1989;53:367–376. doi: 10.1128/mr.53.3.367-376.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Merrick M J. Mol Microbiol. 1993;10:903–909. doi: 10.1111/j.1365-2958.1993.tb00961.x. [DOI] [PubMed] [Google Scholar]
- 9.Shingler V. Mol Microbiol. 1996;19:409–416. doi: 10.1046/j.1365-2958.1996.388920.x. [DOI] [PubMed] [Google Scholar]
- 10.Sasse-Dwight S, Gralla J D. Proc Natl Acad Sci USA. 1988;85:8934–8938. doi: 10.1073/pnas.85.23.8934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Popham D L, Szeto D, Keener J, Kustu S. Science. 1989;243:629–635. doi: 10.1126/science.2563595. [DOI] [PubMed] [Google Scholar]
- 12.Kroos L, Kuspa A, Kaiser D. Dev Biol. 1986;117:252–266. doi: 10.1016/0012-1606(86)90368-4. [DOI] [PubMed] [Google Scholar]
- 13.Kroos, L. R. (1986) Dissertation (Stanford Univ., Stanford, CA).
- 14.Hagen D C, Bretscher A P, Kaiser D. Dev Biol. 1978;64:284–296. doi: 10.1016/0012-1606(78)90079-9. [DOI] [PubMed] [Google Scholar]
- 15.LaRossa R, Kuner J, Hagen D, Manoil C, Kaiser D. J Bacteriol. 1983;153:1394–1404. doi: 10.1128/jb.153.3.1394-1404.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kuspa A, Kroos L, Kaiser D. Dev Biol. 1986;117:267–276. doi: 10.1016/0012-1606(86)90369-6. [DOI] [PubMed] [Google Scholar]
- 17.Keseler I M, Kaiser D. J Bacteriol. 1995;177:4638–4644. doi: 10.1128/jb.177.16.4638-4644.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Romeo J M, Zusman D R. J Bacteriol. 1987;169:3801–3808. doi: 10.1128/jb.169.8.3801-3808.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gulati P, Xu D, Kaplan H B. J Bacteriol. 1995;177:4645–4651. doi: 10.1128/jb.177.16.4645-4651.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sambrook J, Fritsch E F, Maniatis T. Molecular Cloning: A Laboratory Manual. 2nd Ed. Plainview, NY: Cold Spring Harbor Lab. Press; 1989. [Google Scholar]
- 21.Hodgkin J, Kaiser D. Proc Natl Acad Sci USA. 1977;74:2938–2942. doi: 10.1073/pnas.74.7.2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Avery L, Kaiser D. Mol Gen Genet. 1983;191:99–109. doi: 10.1007/BF00330896. [DOI] [PubMed] [Google Scholar]
- 23.Keseler, I. M. (1996) Dissertation (Stanford Univ., Stanford, CA).
- 24.Holmes D S, Quigley M. Anal Biochem. 1981;114:193–197. doi: 10.1016/0003-2697(81)90473-5. [DOI] [PubMed] [Google Scholar]
- 25.Smith M, Jessee J, Landers T, Jordan J. Focus. 1990;12:38–40. [Google Scholar]
- 26.Kalman L V, Cheng Y L, Kaiser D. J Bacteriol. 1994;176:1434–1442. doi: 10.1128/jb.176.5.1434-1442.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kashefi K, Hartzell P L. Mol Microbiol. 1995;15:483–494. doi: 10.1111/j.1365-2958.1995.tb02262.x. [DOI] [PubMed] [Google Scholar]
- 28.Brun Y V, Shapiro L. Genes Dev. 1992;6:2395–2408. doi: 10.1101/gad.6.12a.2395. [DOI] [PubMed] [Google Scholar]
- 29.Sanger F, Nicklen S, Coulson A R. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vieira J, Messing J. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- 31.Chen H, Keseler I M, Shimkets L J. J Bacteriol. 1990;172:4206–4213. doi: 10.1128/jb.172.8.4206-4213.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen H, Kuspa A, Keseler I M, Shimkets L J. J Bacteriol. 1991;173:2109–2115. doi: 10.1128/jb.173.6.2109-2115.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shimkets L J. In: Myxobacteria II. Dworkin M, Kaiser D, editors. Washington, DC: Am. Soc. for Microbiol.; 1993. [Google Scholar]
- 34.Mesbah M, Premachandran U, Whitman W B. J Syst Bacteriol. 1989;39:159–167. [Google Scholar]
- 35.Bibb M J, Findlay P R, Johnson M W. Gene. 1984;30:157–166. doi: 10.1016/0378-1119(84)90116-1. [DOI] [PubMed] [Google Scholar]
- 36.Gribskov M, Devereux J, Burgess R R. Nucleic Acids Res. 1984;12:539–549. doi: 10.1093/nar/12.1part2.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hagen T J, Shimkets L J. J Bacteriol. 1990;172:15–23. doi: 10.1128/jb.172.1.15-23.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stigter J, Schneider M, de Bruijn F J. Mol Plant–Microbe Interact. 1993;6:238–252. doi: 10.1094/mpmi-6-238. [DOI] [PubMed] [Google Scholar]
- 39.Ehrt S, Ornston L N, Hillen W. J Bacteriol. 1994;176:3493–3499. doi: 10.1128/jb.176.12.3493-3499.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Slooten J C, Cervantes E, Broughton W J, Wong C H, Stanley J. J Bacteriol. 1990;172:5563–5574. doi: 10.1128/jb.172.10.5563-5574.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wu S S, Kaiser D. Mol Microbiol. 1995;18:547–558. doi: 10.1111/j.1365-2958.1995.mmi_18030547.x. [DOI] [PubMed] [Google Scholar]
- 42.Romeo J M, Zusman D R. J Bacteriol. 1991;173:2969–2976. doi: 10.1128/jb.173.9.2969-2976.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Spratt B G, Hedge P J, te Heesen S, Edelman A, Broome-Smith J K. Gene. 1986;41:337–342. doi: 10.1016/0378-1119(86)90117-4. [DOI] [PubMed] [Google Scholar]
- 44.Ueki T, Inouye S, Inouye M. Gene. 1996;183:153–157. doi: 10.1016/s0378-1119(96)00546-x. [DOI] [PubMed] [Google Scholar]
- 45.Chang A C Y, Cohen S N. J Bacteriol. 1978;134:1141–1156. doi: 10.1128/jb.134.3.1141-1156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Campos J M, Geisselsoder J, Zusman D R. J Mol Biol. 1978;119:167–178. doi: 10.1016/0022-2836(78)90431-x. [DOI] [PubMed] [Google Scholar]
- 47.Totten P A, Lara J C, Lory S. J Bacteriol. 1990;172:389–396. doi: 10.1128/jb.172.1.389-396.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Powell B S, Court D L, Inada T, Nakamura Y, Michotey V, Cui X, Reizer A, Saier M H, Jr, Reizer J. J Biol Chem. 1995;270:4822–4839. doi: 10.1074/jbc.270.9.4822. [DOI] [PubMed] [Google Scholar]
- 49.Inouye S. J Bacteriol. 1990;172:80–85. doi: 10.1128/jb.172.1.80-85.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Davis J M, Mayor J, Plamann L. Mol Microbiol. 1995;18:943–952. doi: 10.1111/j.1365-2958.1995.18050943.x. [DOI] [PubMed] [Google Scholar]
- 51.Kaufman R I, Nixon B T. J Bacteriol. 1996;178:3967–3970. doi: 10.1128/jb.178.13.3967-3970.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]