Abstract
Active efflux of antimicrobial substances is likely to be an important bacterial defense against inhibitory host factors inherent to different body sites. Two well-characterized multidrug resistance efflux systems (MtrCDE and FarAB-MtrE) exist in Neisseria gonorrhoeae, a bacterial pathogen of the human genital mucosae. In vitro studies suggest that the MtrCDE and FarAB-MtrE efflux systems protect the gonococcus from hydrophobic antimicrobial substances that are likely to be present on mucosal surfaces. Here we report that a functional MtrCDE efflux system, but not a functional FarAB-MtrE system, enhances experimental gonococcal genital tract infection in female mice. Specifically, the recovery of mtrD and mtrE mutants, but not a farB mutant, from mice inoculated with mutant or wild-type gonococci was reduced compared with that of the wild-type strain. Competitive-infection experiments confirmed the survival disadvantage of MtrCDE-deficient gonococci. This report is the first direct evidence that a multidrug resistance efflux system enhances survival of a bacterial pathogen in the genital tract. Additionally, experiments using ovariectomized mice showed that MtrCDE-deficient gonococci were more rapidly cleared from mice that were capable of secreting gonadal hormones. MtrCDE-deficient gonococci were more sensitive to nonphysiological concentrations of progesterone in vitro than were wild-type or FarAB-MtrE-deficient gonococci. These results suggest that progesterone may play an inhibitory role in vivo. However, hormonally regulated factors rather than progesterone itself may be responsible for the more rapid clearance of mtr-deficient gonococci from intact mice.
Bacterial colonization of mucosal surfaces is challenged by components of the host innate immune response, including hydrophobic, membrane-damaging compounds such as bile salts, fatty acids, and antibacterial peptides. Gram-negative bacteria have evolved elaborate active efflux systems, which together with the low permeability of the outer membrane confer inherent resistance to these antimicrobial substances. One class of active efflux systems, the multidrug resistance pumps, is remarkable in the capacity to recognize structurally dissimilar substrates, including diverse antibiotics (34, 35). Two such systems, namely, the mtrCDE-encoded (6, 12, 13, 36) and farAB-encoded (22) efflux systems, have been well characterized in Neisseria gonorrhoeae. Like other multidrug resistance pumps, the mtrCDE- and farAB-encoded efflux systems are composed of three components that function together to capture the substrate in the inner membrane and transport it through the periplasm and out to the external milieu via an outer membrane pore (34, 49). A third efflux system that utilizes a transporter homologous to NorM of Vibrio parahaemolyticus was recently identified in N. gonorrhoeae and Neisseria meningitidis (37).
The gonococcal MtrCDE (“mtr” stands for multiple transferable resistance) system is encoded by an operon consisting of three genes, mtrC, mtrD, and mtrE (11), and is homologous to the AcrAB-TolC and AcrEF-TolC systems of Escherichia coli and the MexAB-OprM system of Pseudomonas aeruginosa (34, 35, 49). The mtrC gene encodes a periplasmic accessory lipoprotein (or membrane fusion protein) (MtrC), which links the inner and outer membrane components (13). mtrD encodes an energy-dependent transporter protein (MtrD) that transverses the inner membrane and belongs to the resistance-nodulation-division (RND) family of transporter proteins (12). mtrE encodes an exporter protein (MtrE) that forms a channel in the outer membrane (6). MtrR, a transcriptional repressor of the mtrCDE operon, is encoded upstream of the mtrC gene and is divergently transcribed with respect to the mtrCDE operon (13, 23, 36). The gonococcal farAB-encoded efflux system (“far” stands for fatty acid resistance) has homology with the emrAB- and vceAB-encoded pumps of E. coli and Vibrio cholerae, respectively (22, 49). In this system, the FarA and FarB proteins serve as a periplasmic accessory protein and a cytoplasmic transporter, respectively. FarB belongs to the major facilitator superfamily of transporter proteins. It has been hypothesized that the MtrE protein serves as the outer membrane channel for the farAB-encoded system, based on the demonstration that mutations in mtrE knock out both the mtrCDE- and farAB-encoded efflux systems (22). Sharing of an outer membrane exporter by more than one efflux pump has been described for other gram-negative multidrug transport systems (49).
Research on bacterial multidrug efflux systems has been motivated in part by concern over the continual emergence of antibiotic-resistant strains. For example, overexpression of the MtrCDE efflux system confers high-level resistance to lipophilic antibiotics such as erythromycin (13, 36) and azithromycin (48) and is required for high-level, chromosomal-mediated penicillin resistance (46). The evolution of multidrug resistance pumps surely predates the clinical use of antibiotics, however, and it is therefore likely that these systems also protect bacteria from host substances present in the various niches in which they reside. N. gonorrhoeae most commonly infects the lower urogenital tract, namely, the urethra of men and the endocervix and/or urethra in females of reproductive age. Rectal, pharyngeal, and conjunctival mucosae are also frequently infected (14). Evidence that the MtrCDE and FarAB efflux systems protect N. gonorrhoeae from host innate mucosal defenses has thus far been indirect. Substrates exported by the gonococcal MtrCDE and FarAB-MtrE systems that are relevant to rectal mucosa include bile salts and long chain fatty acids, respectively (6, 12, 22). Active efflux of antimicrobial substances present on urogenital mucosa, such as fatty acids, protegrins, and LL-37, occurs via the MtrCDE system (12, 13, 41). Here we tested genetically defined mutants in a mouse infection model to directly assess the contribution of the MtrCDE and FarAB-MtrE efflux systems to the survival of N. gonorrhoeae in the female lower genital tract.
MATERIALS AND METHODS
Bacterial strains.
Neisseria gonorrhoeae strain FA19 and isogenic mutants KH14 (mtrD::Kmr), RD1 (mtrE::aphA-3), and EL1 (farB::Kmr) have been described previously (6, 12, 22). Mutants KH14 and RD1 lack functional MtrCDE and FarAB-MtrE efflux systems due to the shared requirement for the MtrE outer membrane component by these pumps. Mutant EL1 lacks a functional FarAB-MtrE efflux pump; the MtrCDE efflux system in EL1 is not affected (22). For this study, a streptomycin-resistant derivative of each efflux pump mutant was constructed in the same wild-type background as follows. Briefly, 109 CFU of N. gonorrhoeae strain FA19 were inoculated onto GC agar containing 100 μg of streptomycin/ml. A streptomycin-resistant derivative (FA19-Sm) was isolated and then transformed with chromosomal DNA from mutant strains KH14, RD1, and EL1 (9). Strains KH14-Sm, RD1-Sm, and EL1-Sm were checked by PCR for the presence of the correct insertion mutation in each respective efflux pump gene. To ensure that any differences seen during murine infection were not due to differences in sensitivity to the antibiotics that were used to treat the mice, the sensitivities of wild-type and mutant strains to streptomycin and trimethoprim were determined by standard agar dilution assay. No differences in sensitivities to these antibiotics were detected for any of the strains. The bacterial culture media and incubation conditions have been described previously (16); streptomycin (100 μg/ml) and kanamycin (50 μg/ml) were used as antibiotic selection when indicated.
In vitro inhibition assays.
The sensitivity of wild-type and efflux pump-deficient gonococci to progesterone (4-pregnene-3,20-dione) or 17-β estradiol (Sigma Biochem, St. Louis, Mo.) was tested using a standard agar dilution assay. The surface of GC agar, containing twofold-decreasing concentrations of each agent to be tested, was inoculated with 105 CFU of each strain in a 10-μl volume; plates were incubated without spreading the inoculum. The MIC was defined as the lowest concentration of agent that inhibited growth of the organism after overnight incubation. Bacterial sensitivity to progesterone was further tested by comparing the plating efficiencies of wild-type and mutant N. gonorrhoeae on GC agar containing 0 to 40 μg of progesterone/ml dissolved in dimethyl sulfoxide (DMSO). The concentration of DMSO present in all test agar or GC broth (see below) was 0.2%. To study the effect of progesterone on wild-type versus mutant gonococci with respect to growth rate, late-log-phase cultures of wild-type FA19-Sm and mutant strains RD-1-Sm and EL1-Sm were combined to create 1:1 ratios of wild-type to mutant gonococci. Fifty milliliters of supplemented GC broth containing 5, 10, or 20 μg of progesterone/ml dissolved in DMSO or DMSO only was inoculated with ca. 5 × 107 CFU of each mixed suspension and incubated at 37°C with aeration. The absorbency of each culture at 600 nm was measured over time, and samples corresponding to lag phase and early, mid-, and late-logarithmic phase after overnight incubation were serially diluted and cultured on GC agar (total number of gonococci recovered) or GC with kanamycin (number of mutants recovered).
Experimental genital tract infection.
Intact or ovariectomized female BALB/c mice (4 to 6 weeks old) (National Cancer Institute, Bethesda, Md.) were treated with 17-β estradiol and antibiotics to promote susceptibility to N. gonorrhoeae as described previously (17). For single infections, groups of seven or eight mice were inoculated intravaginally with 106 CFU of piliated wild-type or mutant N. gonorrhoeae. Preparation of the inocula and culturing of vaginal mucus were done as described previously (17). For competitive infection experiments, equal volumes of saline suspensions containing FA19-Sm gonococci and similar numbers of viable KH14-Sm, RD1-Sm, or EL1-Sm gonococci were combined prior to inoculation of mice. The frequency of mutant gonococci (Kmr CFU) in each inoculum and among vaginal isolates was determined by culturing equal volumes of diluted inoculum and vaginal swab suspensions on GC agar containing streptomycin (total gonococci) and GC agar containing streptomycin and kanamycin (mutant gonococci) and dividing the number of Kmr CFU by the total number of gonococci recovered. For all mouse infection experiments, the limit of detection was 4 CFU per 100 μl of vaginal swab suspension; in all calculations, this value was used for time points at which no bacteria were recovered. Animal experiments were conducted in the laboratory animal facility at the Uniformed Services University, which is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care under a protocol that was approved by the University's Institutional Animal Care and Use Committee.
Measurement of serum progesterone levels.
Blood (150 μl) was collected via periorbital bleed from uninfected estradiol-treated mice on days 5, 7, 9, and 11 after implantation of the estradiol pellet, and the concentration of progesterone in the serum fraction was determined using an indirect enzyme-linked immunoassay kit (Diagnostic Systems Laboratories, Inc., Webster, Tex.) according to the manufacturer's instructions. Serum was collected from groups of seven mice at each time point, and samples were not collected from the same mouse for any two consecutive time points.
Statistical methods.
A two-tailed t test was used to evaluate the difference in durations of recovery in single-infection experiments and recovery of RD1-Sm gonococci from intact versus ovariectomized mice at each time point. A repeated analysis of variance was used to evaluate differences in the number of viable wild-type and mutant N. gonorrhoeae recovered from mice over time (GraphPad Prism; GraphPad Software, Inc., San Diego, Calif.). For competitive-infection experiments, the Kruskal-Wallis test was utilized to measure overall significance and the Mann-Whitney test was used to determine the significance of pairwise comparisons (SPSS, Inc., Chicago, Ill.).
RESULTS
Survival of gonococcal efflux pump mutants in the murine genital tract.
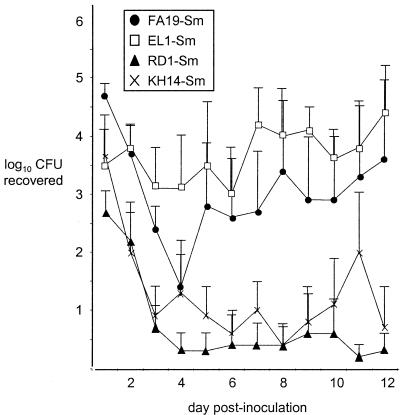
Although N. gonorrhoeae is a human-specific pathogen, experimental gonococcal genital tract infection can be established in female mice through exogenous administration of 17-β estradiol (16). In this model, high numbers of gonococci are recovered from the lower genital tract following intravaginal inoculation for an average of 12 days in a 14-day period (and for as long as 40 days) (16, 17). To assess the requirement of the MtrCDE and FarAB-MtrE efflux systems for gonococcal genital tract infection, we compared the recovery of mutants KH14-Sm (mtrD::Kmr) (12), RD1-Sm (mtrE::aphA-3) (6), and EL1-Sm (farB::Kmr) (22) from the lower genital tract of estradiol-treated mice with that of the wild-type parent strain FA19-Sm. We observed no reproducible significant difference in the mean durations of recovery of any strain from mice inoculated with 106 CFU of wild-type (9.0 days; range, 3 to 12 days), RD1-Sm (3.4 days, range, 0 to 12 days), KH14-Sm (5.0 days; range, 0 to 12 days), or EL1-Sm (12 days; no range) gonococci. However, the average number of gonococci recovered each day was significantly lower in mice inoculated with mtrD and mtrE mutants KH14-Sm and RD1-Sm than that in mice inoculated with the wild-type strain (Fig. 1). In contrast, similar numbers of wild-type FA19-Sm and farB mutant EL1-Sm gonococci were recovered from mice throughout the course of infection. No in vivo reversion of the mutations was detected by in vitro testing of vaginal isolates from mice infected with any of the efflux pump-deficient mutants (data not shown).
FIG. 1.
Recovery of wild-type and efflux pump-deficient N. gonorrhoeae from estradiol-treated mice. Results are expressed as the average number of CFU (expressed as log10) recovered from vaginal swab suspensions over a 12-day period. P = 0.012 and 0.024 for KH14-Sm and RD1-Sm, respectively, compared with the wild-type strain. The experiment was performed twice, and the results were similar.
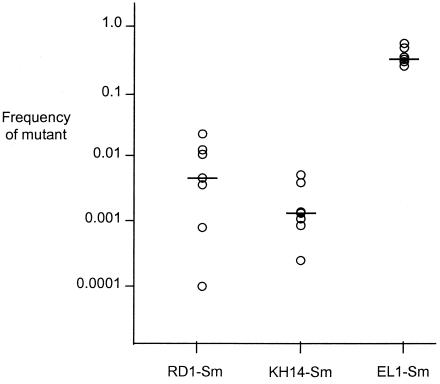
To minimize the potential influence of animal variability on survival of the mutant strains, we inoculated mice with mixed suspensions containing 1:1 ratios of wild-type FA19 and KH14-Sm, RD1-Sm, or EL1-Sm gonococci. The recovery of RD1-Sm and KH14-Sm gonococci, but not EL1-Sm gonococci, was dramatically reduced compared with that of the wild-type strain in all mice tested within 2 days postinoculation. No RD1-Sm or KH14-Sm gonococci were recovered from any mouse within 4 to 5 days following inoculation. The frequency of each mutant within the total population of gonococci recovered on day 4 postinoculation is shown in Fig. 2. No difference in the growth rates of wild-type and mutant gonococci cultured independently in broth or in mixed broth cultures was detected (data not shown). We conclude from these data that the wild-type strain has a competitive advantage over mtr mutants RD1-Sm and KH14-Sm in the lower genital tract of female mice but that mutation in farB does not attenuate survival.
FIG. 2.
Recovery of efflux pump-deficient gonococci relative to that of the wild-type strain in competitive infections. The frequency of the mutant in the total population of gonococci recovered for each mouse in each experimental group on day 4 postinoculation is shown on the y axis. The frequency of mutant CFU within the inocula ranged from 0.35 to 0.59. The medians are represented by horizontal bars. P = 0.003 for RD1-Sm and KH14-Sm when compared with the recovery of EL1-Sm (as determined by the Mann-Whitney test). The experiment was performed three times using four to seven mice per group, and the results were similar.
Sensitivity of MtrCDE-deficient gonococci to gonadal steroids.
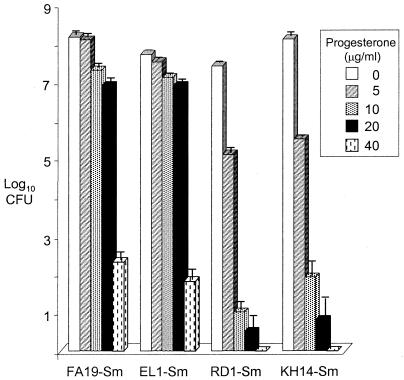
Lysko and Morse reported an association between the gonococcal mtr locus and resistance to the inhibitory effects of progesterone (24). Wild-type gonococci were used in this study, and the genetic basis of the mtr phenotype (resistance to hydrophobic dyes, detergents, and multiple antibiotics) was not defined at the time of this earlier study. Based on the results of this previous study, we tested the progesterone sensitivities of wild-type FA19 and each of the efflux pump-deficient mutants. Plating efficiencies on agar with increasing concentrations of progesterone showed that RD1-Sm and KH14-Sm gonococci were markedly more susceptible to progesterone than either the parent strain FA19-Sm or the farB mutant EL1-Sm at all concentrations tested. All strains were highly sensitive to 40 μg of progesterone/ml (Fig. 3). The progesterone MIC for each strain, as determined by standard agar dilution, was consistent with the plating efficiencies (FA19 and EL1, >20 μg/ml; RD-1 and KH14, 5 μg/ml). None of the mutants was sensitive to 17-β estradiol (data not shown).
FIG. 3.
Plating efficiency of efflux pump-deficient N. gonorrhoeae on agar containing progesterone. GC agar contained 0, 5, 10, 20, and 40 μg of progesterone/ml. The experiment was performed twice, and the results were similar.
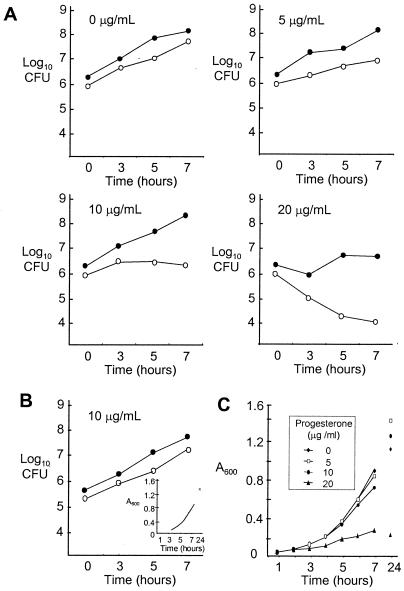
The increased sensitivities of the mtrD and mtrE mutants, but not the farB mutant, to progesterone is only indirect evidence that progesterone is pumped out by the MtrCDE efflux pump system. Efflux of progesterone through this system is reasonable, however, based on its hydrophobic nature and the demonstration that progesterone, like other inhibitory substrates, binds the gonococcal inner membrane (24, 31, 32). Inhibition of N. gonorrhoeae by progesterone was previously shown to be bacteriostatic and to occur via the inhibition of respiration (32). To further examine the inhibitory effect of progesterone on growth of the MtrCDE-deficient mutants, mixed broth cultures of wild-type and RD-1 or EL-1 mutant gonococci were grown in the presence of 0 to 20 μg of progesterone/ml (Fig. 4). RD1-Sm gonococci grew more slowly than the wild-type strain in the presence of 5 μg of progesterone/ml, and no detectable growth of this mutant occurred in broth containing 10 μg of progesterone/ml. Growth of both FA19-Sm and RD1-Sm was inhibited by 20 μg of progesterone/ml, with the mutant being inhibited more than the parental strain. Inhibition of MtrCDE-deficient N. gonorrhoeae by progesterone appeared to be bacteriostatic in nature (Fig. 4C). In contrast, the growth rate of EL1-Sm gonococci in mixed cultures was similar to that of the wild-type strain in the presence of 5 μg (data not shown) and 10 μg (Fig. 4B) of progesterone/ml.
FIG. 4.
Inhibition of Mtr-deficient N. gonorrhoeae by progesterone in mixed broth cultures. Mutants RD1-Sm and EL1-Sm were cocultured with wild-type FA19-Sm in GC broth containing 0 to 20 μg of progesterone/ml. (A) Recovery of bacteria on solid agar without antibiotic selection (closed circles, total CFU) versus that on agar containing kanamycin (open circles, mutant CFU) from FA19-Sm-RD1-Sm mixed cultures containing progesterone at the concentrations indicated on each graph. (B) Recovery of mutant EL1-Sm (open circles) when cocultured with FA19-Sm in the presence of 10 μg of progesterone/ml (closed circles, total CFU) and the change in absorbency of this culture over time (inset). (C) Total bacterial growth within FA19-Sm-RD1-Sm mixed cultures in the presence of increasing concentrations of progesterone as measured by change in absorbency at 600 nm. The experiments were performed twice, and the results were similar.
Delayed attenuation of MtrCDE-deficient gonococci in ovariectomized mice.
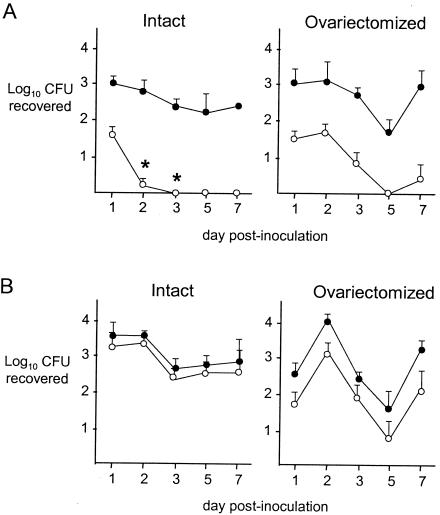
The majority of progesterone in mammalian females is produced by the corpus luteum, a specialized region of the ovaries that develops from the preovulatory follicles (8). Although exogenous administration of estradiol disrupts the normal estrous cycle, estradiol-treated mice are still capable of progesterone secretion. To explore the environment of the genital tract in the absence of ovarian hormones or their effects, we compared the relative survival rates of wild-type and efflux pump-deficient N. gonorrhoeae in intact versus ovariectomized mice. As before, mutant RD1-Sm, but not the farB mutant EL1-Sm, displayed a marked survival disadvantage compared with the wild-type strain in intact mice following inoculation with defined mixtures of mutant and wild-type gonococci. A decrease in the relative recovery rate of RD1-Sm, but not EL1-Sm, gonococci also occurred in ovariectomized mice (Fig. 5), a result that suggests the presence of inhibitory substrates in the genital tract that are independent of ovarian function. Interestingly, however, the decrease in RD1-Sm gonococci was reproducibly more pronounced in intact mice than in ovariectomized mice early in infection (Fig. 5A). The more rapid attenuation of mutant RD1-Sm in intact mice suggests that in the absence of the MtrCDE efflux system, gonococci are more susceptible to gonadal steroids or their downstream effects in vivo.
FIG. 5.
Recovery of efflux pump-deficient gonococci relative to that of the wild-type strain in competitive-infection experiments using intact versus ovariectomized mice. Results are expressed as the total number of CFU (closed circles) and the number of Kmr CFU (mutant) (open circles) recovered from mice inoculated with mixed suspensions containing a 1:1 ratio of wild-type FA19 to mutant RD1-Sm (A) or mutant EL1-Sm (B). Asterisks indicate time points at which significant differences in the recovery of RD1-Sm gonococci were detected in intact versus ovariectomized mice (P = 0.012 and 0.013 on days 2 and 3, respectively). The experiment was performed twice with six to nine mice per group, and the results were similar.
To further assess the possibility that progesterone contributed to the more rapid clearance of MtrCDE-deficient gonococci in intact mice, we determined the average serum progesterone levels in peripheral blood from intact mice at selected time points within 1 week after estradiol treatment. Levels ranged from 4.2 to 9.4 ng/ml, which are similar to preovulatory levels of progesterone in cycling mice (38) and ca. 1,000-fold lower than the concentration of progesterone required to inhibit the mtr mutants in vitro.
DISCUSSION
The mtr locus was initially identified in studies on the genetic basis of resistance to multiple antibiotics in N. gonorrhoeae (10, 25, 39, 43). Further definition of the substrate repertoire of this efflux system was facilitated by the subsequent cloning of the mtr locus and the construction of genetically defined mutants. Here we present data that support the role of the MtrCDE, but not the FarAB-MtrE, efflux system in survival of N. gonorrhoeae in the lower genital tract of female mice. This conclusion is based on comparisons between two mutants that are deficient in both the MtrCDE and FarAB-MtrE efflux systems and a mutant that lacks only the FarAB-MtrE efflux system. The FarAB-MtrE efflux-deficient mutant behaved similarly to the wild-type strain in terms of both duration of infection and colonization load. In contrast, mutants that lacked both efflux systems were attenuated, as evidenced by a reduced colonization load within 5 days postinoculation in single-organism infections and a strong survival disadvantage compared with the wild-type strain in competitive-infection experiments. It is possible that a loss of both efflux systems is required for attenuation in vivo. However, the known substrate repertoire of the MtrCDE efflux system is more consistent with the type of inhibitory factors that might be present in the lower genital tract than is that of the FarAB-MtrE system. Unfortunately, previous attempts to create a mutant that was deficient only in the MtrCDE system by introducing a nonpolar mutation into mtrC were unsuccessful, perhaps because continued production of MtrE may be toxic. We do not know the identity of the factor(s) responsible for inhibition of the mtr mutants in the lower genital tract of mice. We did not observe a temporal association between loss of the mtr mutants and an infiltration of neutrophils into the vaginal lumen (data not shown). The presence of fatty acids and/or the production of antimicrobial peptides by epithelial cells in response to infection may play a role (7). Specifically, one might hypothesize that a murine analogue to the human peptide LL37 may select against MtrCDE efflux pump-deficient gonococci in vivo, since this host factor is likely to be present on urogenital mucosae (41).
Prior to this report, the strongest evidence supporting the role of the MtrCDE efflux system in protecting the gonococcus from host innate mucosal defenses was the isolation of gonococci with increased resistance to bile salts, fatty acids, and fecal lipids from rectal cultures (29, 33), the majority of which were due to mutations affecting the mtrR repressor promoter region and/or structural gene (13, 40, 47). This indirect evidence that active efflux of inhibitory host substances promotes gonococcal survival during rectal infection is consistent with recent reports that efflux pump mutants of Vibrio cholerae (1, 30), Salmonella enterica serovar Typhimurium (21), and Escherichia coli K1 (26) display reduced survival in the gastrointestinal tract, most likely due to an increased sensitivity to bile salts (1, 21). Recently, a gene predicted to encode a protein belonging to the AcrA-AcrE family was identified in N. meningitidis by signature-tagged mutagenesis as being important for survival during systemic infection of infant rats. Competition experiments with the wild-type strain showed only modest attenuation for this mutant (44). This result, together with the dramatic attenuation of the mtr mutants in the murine lower genital tract, suggests that the gonococcal MtrCDE system may not be as critical a defense against systemic host factors as it is at mucosal sites.
Although gonococci that lack the MtrCDE efflux system were markedly attenuated in both intact and ovariectomized mice, the slower decline of mtrE mutant RD1-Sm in ovariectomized mice suggests that the mtrCDE-encoded pump may have evolved in part to escape inhibitory factors of ovarian origin. The increased progesterone sensitivity of MtrCDE-deficient gonococci suggests that progesterone itself may play an inhibitory role in vivo. It is not known whether N. gonorrhoeae directly encounters inhibitory concentrations of progesterone during mucosal infection. Normal human plasma progesterone levels fluctuate from 0.5 to 1.5 ng/ml (preovulation) to 10 to 20 ng/ml postovulation (4), which are lower than that required to inhibit gonococci in vitro. Higher progesterone levels may be present locally, however, and other physiological factors within the host might increase gonococcal sensitivity to progesterone.
The possibility that other substrates of the MtrCDE system may undergo hormonal regulation is intriguing. Hormonal regulation of immunological factors (5) as well as adherence receptors (27) can play a role in host-pathogen interactions, and the influence of reproductive hormones on susceptibility to infection has been described for several pathogens of the urogenital tract (2, 19, 42, 45). With regard to gonococcal infection, an association between N. gonorrhoeae culture rates and the phase of the menstrual cycle of infected women suggests that inhibitory host factors may be present during the secretory phase of the female reproductive cycle (15, 18, 20, 28). The fact that short-term gonococcal vaginal colonization of mice can be achieved during the proestrus but not the postovulatory stages of the estrous cycle (3) also supports the hypothesis that the secretory stage of the mammalian reproductive cycle is inhospitable to N. gonorrhoeae. The relationship between gonadal hormones and specific host defenses that challenge the survival of N. gonorrhoeae during natural and experimental infection has yet to be elucidated.
Acknowledgments
We thank Afrin Begum for technical assistance, Cara Olsen for statistical consultation, and E.-H. Lee for providing strain EL1.
This work was supported by NIH grants AI-42053 (A.E.J.) and A-21150-16 (W.M.S.). W.M.S. was supported by a Senior Research Career Scientist award from the VA Medical Research Service.
Editor: J. N. Weiser
REFERENCES
- 1.Bina, J. E., and J. J. Mekalanos. 2001. Vibrio cholerae tolC is required for bile resistance and colonization. Infect. Immun. 69:4681-4685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brabin, L. 2002. Interactions of the female hormonal environment, susceptibility to viral infection, and disease progression. AIDS Patient Care STDs 16:211-221. [DOI] [PubMed] [Google Scholar]
- 3.Braude, A. I., L. B. Corbeil, S. Levine, J. Ito, and J. A. McCutchan. 1978. Possible influence of cyclic menstrual changes on resistance to the gonococcus, p. 328-337. In G. F. Brooks, E. C. Gotschlich, K. K. Holmes, W. D. Sawyer, and F. E. Young (ed.), Immunobiology of Neisseria gonorrhoeae. American Society for Microbiology, Washington, D.C.
- 4.Chattoraj, S. C. 1976. Endocrine function, p. 699-823. In N. W. Tietz (ed.), Fundamentals of clinical chemistry. W. B. Saunders Company, Philadelphia, Pa.
- 5.Critchley, H. O. D., R. W. Kelly, R. M. Brenner, and D. T. Baird. 2001. The endocrinology of menstruation—a role for the immune system. Clin. Endocrinol. 55:701-710. [DOI] [PubMed] [Google Scholar]
- 6.Delahay, R. M., B. D. Robertson, J. T. Balthazar, W. M. Shafer, and C. A. Ison. 1997. Involvement of the gonococcal MtrE protein in the resistance of Neisseria gonorrhoeae to toxic hydrophobic agents. Microbiology 143:2127-2133. [DOI] [PubMed] [Google Scholar]
- 7.Devine, D. A., and R. E. W. Hancock. 2002. Cationic peptides: distribution and mechanisms of resistance. Curr. Pharm. Des. 8:703-724. [DOI] [PubMed] [Google Scholar]
- 8.Freeman, M. E. 1994. The neuroendocrine control of the ovarian cycle of the rat, p. 613-658. In E. Knobil, J. D. Neill, G. S. Greenwald, C. L. Markert, and D. W. Pfaff (ed.), The physiology of reproduction. Raven Press, New York, N.Y.
- 9.Gunn, J. S., and D. C. Stein. 1996. Use of a non-selective transformation technique to construct a multiply restriction/modification-deficient mutant of Neisseria gonorrhoeae. Mol. Gen. Genet. 251:509-517. [DOI] [PubMed] [Google Scholar]
- 10.Guymon, L. F., and P. F. Sparling. 1975. Altered crystal violet permeability and lytic behavior in antibiotic-resistant and -sensitive mutants of Neisseria gonorrhoeae. J. Bacteriol. 124:757-763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hagman, K. E., and W. M. Shafer. 1995. Transcriptional control of the mtr efflux system of Neisseria gonorrhoeae. J. Bacteriol. 177:4162-4165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hagman, K. E., C. E. Lucas, J. T. Balthazar, L. Snyder, M. Nilles, R. C. Judd, and W. M. Shafer. 1997. The MtrD protein of Neisseria gonorrhoeae is a member of the resistance/nodulation/division protein family constituting part of an efflux system. Microbiology 143:2117-2125. [DOI] [PubMed] [Google Scholar]
- 13.Hagman, K. E., W. Pan., B. G. Spratt, J. T. Balthazar, R. C. Judd, and W. M. Shafer. 1995. Resistance of Neisseria gonorrhoeae to antimicrobial hydrophobic agents is modulated by the mtrRCDE efflux system. Microbiology 141:611-622. [DOI] [PubMed] [Google Scholar]
- 14.Hook, E. W., and H. H. Handsfield. 1999. Gonococcal infections in the adult, p. 451-472. In K. K. Holmes, P. A. Mardh, F. Sparling, S. M. Lemon, W. E. Stamm, P. Piot, and J. N. Wasserheit (ed.), Sexually transmitted diseases, 3rd ed. McGraw-Hill Companies, Inc., New York, N.Y.
- 15.James, J. F., and J. Swanson. 1978. Color/opacity colonial variants of Neisseria gonorrhoeae and their relationship to the menstrual cycle, p. 338-343. In G. F. Brooks, E. C. Gotschlich, K. K. Holmes, W. D. Sawyer, and F. E. Young (ed.), Immunobiology of Neisseria gonorrhoeae. American Society for Microbiology, Washington, D.C.
- 16.Jerse, A. E. 1999. Experimental gonococcal genital tract infection and opacity protein expression in estradiol-treated mice. Infect. Immun. 67:5699-5708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jerse, A. E., E. T. Crow, A. N. Bordner, I. Rahman, C. N. Cornelissen, T. R. Moench, and K. Mehrazar. 2002. Growth of Neisseria gonorrhoeae in the genital tract of female mice does not require the gonococcal transferrin or hemoglobin receptors and may be enhanced by commensal lactobacilli. Infect. Immun. 70:2549-2558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Johnson, D. W., K. K. Holmes, P. A. Kvale, C. W. Halverson, and W. P. Hirsch. 1969. An evaluation of gonorrhea case findings in the chronically infected female. Am. J. Epidemiol. 90:438-448. [DOI] [PubMed] [Google Scholar]
- 19.Kaushic, C., A. A. Ashkar, L. A. Reid, and K. L. Rosenthal. 2003. Progesterone increases susceptibility and decreases immune responses to genital herpes infection. J. Virol. 77:4558-4565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Koch, M. L. 1947. A study of cervical cultures taken in cases of acute gonorrhea with special reference to the phases of the menstrual cycle. Am. J. Obstet. Gynecol. 54:861-866. [DOI] [PubMed] [Google Scholar]
- 21.Lacroix, J. F., A. Cloeckaert, O. Grepinet, C. Pinault, M. Y. Popoff, H. Waxin, and P. Pardon. 1996. Salmonella typhimurium acrB-like gene: identification and role in resistance to biliary salts and detergents and in murine infection. FEMS Microbiol. Lett. 135:161-167. [DOI] [PubMed] [Google Scholar]
- 22.Lee, E. H., and W. M. Shafer. 1999. The farAB-encoded efflux pump mediates resistance of gonococci to long-chained antibacterial fatty acids. Mol. Microbiol. 33:839-845. [DOI] [PubMed] [Google Scholar]
- 23.Lucas, C. E., J. T. Balthazar, K. E. Hagman, and W. M. Shafer. 1997. The MtrR repressor binds the DNA sequence between the mtrR and mtrC genes of Neisseria gonorrhoeae. J. Bacteriol. 179:4123-4128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lysko, P. G., and S. A. Morse. 1981. Neisseria gonorrhoeae cell envelope: permeability to hydrophobic molecules. J. Bacteriol. 145:946-952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Maier, T. W., L. Zubryzycki, and M. B. Coyle. 1975. Genetic analysis of drug resistance in Neisseria gonorrhoeae: identification and linkage relationships of loci controlling drug resistance. Antimicrob. Agents Chemother. 7:676-681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martindale, J., D. Stoud, E. R. Moxon, and C. M. Tang. 2000. Genetic analysis of Escherichia coli K1 gastrointestinal colonization. Mol. Microbiol. 37:1293-1305. [DOI] [PubMed] [Google Scholar]
- 27.Maslow, A. S., C. H. Davis, J. Choong, and P. B. Wyrick. 1988. Estrogen enhances attachment of Chlamydia trachomatis to human endometrial epithelial cells in vitro. Am. J. Obstet. Gynecol. 159:1006-1014. [DOI] [PubMed] [Google Scholar]
- 28.McCormack, W. M., G. H. Reynolds, and the Cooperative Study Group. 1982. Effect of menstrual cycle and method of contraception recovery of Neisseria gonorrhoeae. JAMA 247:1292-1294. [PubMed] [Google Scholar]
- 29.McFarland, L., T. A. Mietzner, J. S. Knapp, E. Sandstrom, K. K. Holmes, and S. A. Morse. 1983. Gonococcal sensitivity to fecal lipids can be mediated by an Mtr-independent mechanism. J. Clin. Microbiol. 18:121-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Merrell, D. S., D. L. Hava, and A. Camilli. 2002. Identification of novel factors involved in colonization and acid tolerance of Vibrio cholerae. Mol. Microbiol. 43:1471-1491. [DOI] [PubMed] [Google Scholar]
- 31.Miller, R. D., and S. A. Morse. 1977. Binding of progesterone to Neisseria gonorrhoeae and other gram-negative bacteria. Infect. Immun. 16:115-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Morse, S. A., and T. J. Fitzgerald. 1974. Effect of progesterone on Neisseria gonorrhoeae. Infect. Immun. 10:1370-1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morse, S. A., P. G. Lysko, L. McFarland, J. S. Knapp, E. Sandstrom, C. Critchlow, and K. K. Holmes. 1982. Gonococcal strains from homosexual men have outer membranes with reduced permeability to hydrophobic molecules. Infect. Immun. 37:432-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nikaido, H. 1998. Antibiotic resistance caused by gram-negative multidrug efflux pumps. Clin. Infect. Dis. 27:S32-S41. [DOI] [PubMed] [Google Scholar]
- 35.Nikaido, H. 1996. Multidrug efflux pumps of gram-negative bacteria. J. Bacteriol. 178:5853-5859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pan, W., and B. G. Spratt. 1994. Regulation of the permeability of the gonococcal cell envelope by the mtr system. Mol. Microbiol. 11:769-775. [DOI] [PubMed] [Google Scholar]
- 37.Rouquette-Loughlin, C., S. A. Dunham, M. Kuhn, J. T. Balthazar, and W. M. Shafer. 2003. The NorM efflux pump of Neisseria gonorrhoeae and Neisseria meningitidis recognizes antimicrobial cationic compounds. J. Bacteriol. 185:1101-1106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ryan, K. D., and N. B. Schwartz. 1980. Changes in serum hormone levels associated with male-induced ovulation in group-housed adult female mice. Endocrinology 106:959-966. [DOI] [PubMed] [Google Scholar]
- 39.Sarubbi, F. A., E. Blackman, and P. F. Sparling. 1974. Genetic mapping of linked antibiotic resistance loci in Neisseria gonorrhoeae. J. Bacteriol. 120:1284-1292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shafer, W. M., J. T. Balthazar, K. E. Hagman, and S. A. Morse. 1995. Missense mutations that alter the DNA-binding domain of the MtrR protein occur frequently in rectal isolates of Neisseria gonorrhoeae that are resistant to faecal lipids. Microbiology 141:907-911. [DOI] [PubMed] [Google Scholar]
- 41.Shafer, W. M., X.-D. Qu, A. J. Waring, and R. I. Lehrer. 1998. Modulation of Neisseria gonorrhoeae susceptibility to vertebrate antibacterial peptides due to a member of the resistance/nodulation/division efflux pump family. Proc. Natl. Acad. Sci. USA 95:1829-1833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sonnex, C. 1998. Influence of ovarian hormones on urogenital infection. Sex. Transm. Infect. 74:11-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sparling, P. F., F. A. Sarubbi, and E. Blackman. 1975. Inheritance of low-level resistance to penicillin, tetracycline, and chloramphenicol in Neisseria gonorrhoeae. J. Bacteriol. 124:740-749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sun, Y.-H., S. Bakshi, R. Chlamers, and C. M. Tang. 2000. Functional genomics of Neisseria meningitidis pathogenesis. Nat. Med. 6:1269-1273. [DOI] [PubMed] [Google Scholar]
- 45.Taylor-Robinson, D., and P. M. Furr. 1993. Models of infection due to mycoplasmas, including Mycoplasma fermentans, in the genital tract and other sites in mice. Clin. Infect. Dis. 17:S280-S282. [DOI] [PubMed] [Google Scholar]
- 46.Veal, W. F., and W. M. Shafer. 2003. Identification of a cell envelope protein (MtrF) involved in hydrophobic antimicrobial resistance in Neisseria gonorrhoeae. J. Antimicrob. Chemother. 51:27-37. [DOI] [PubMed] [Google Scholar]
- 47.Xia, M., W. L. H. Whittington, W. M. Shafer, and K. K. Holmes. 2000. Gonorrhea among men who have sex with men: outbreak caused by a single genotype of erythromycin-resistant Neisseria gonorrhoeae with a single-base pair deletion in the mtrR promoter region. J. Infect. Dis. 181:2080-2082. [DOI] [PubMed] [Google Scholar]
- 48.Zarantonelli, L., G. Borthagaray, E.-H. Lee, and W. M. Shafer. 1999. Decreased azithromycin susceptibility of Neisseria gonorrhoeae due to mtrR mutations. Antimicrob. Agents Chemother. 43:2468-2472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zgurskaya, H. I., and H. Nikaido. 2000. Multidrug resistance mechanisms: drug efflux across two membranes. Mol. Microbiol. 37:219-225. [DOI] [PubMed] [Google Scholar]