Abstract
Asthma is a chronic and sometimes fatal disease, which affects people of all ages throughout the world. Important hallmarks of asthma are airway inflammation and remodelling, with associated bronchial hyperresponsiveness and variable airflow obstruction. These features are orchestrated by cells of both the innate (eosinophils, neutrophils and mast cells) and the adaptive (TH2 T cells) immune system, in concert with structural airway cells. Chemokines are important for the recruitment of both immune and structural cells to the lung, and also for their microlocalisation within the lung tissue. Specific blockade of the responses elicited by chemokines and chemokine receptors responsible for the pathological migration of airway cells could therefore be of great therapeutic interest for the treatment of asthma.
Keywords: asthma, chemokines, chemokine receptor, lymphocyte, eosinophil, neutrophil, mast cell, remodelling
Introduction
The prevalence of asthma is increasing worldwide, particularly in young children, and is a significant cause of morbidity and mortality in all age groups (Braman, 2006). The word ‘asthma' originates from the Greek word ‘aazein', meaning panting and was clinically described as early as 500 BC in the Corpus Hippocraticum. However, asthma was not considered an inflammatory condition until the early twentieth century (Huber and Koessler, 1922; Becker, 1999).
Asthma pathophysiology
Asthma is a complex immunological and inflammatory disease characterized by the presence of airway inflammation, airway wall remodelling and bronchial hyperresponsiveness (BHR). Exactly how these three key features interact and whether they are dependent on each other for their expression remains unknown. The inflammatory response characteristically comprises activated T helper type 2 (TH2) lymphocytes, eosinophils and activated mast cells, features that are remarkably consistent among cases with apparently different etiology for example, atopic asthma versus non-atopic asthma (Humbert et al., 1996a, 1996b; Ying et al., 1997a). Although BHR appears to be exacerbated by the presence of airway inflammation, it remains once the inflammation is controlled, and is remarkably absent in eosinophilic bronchitis (Brightling et al., 1999). In addition to airway inflammation, a number of structural wall changes occur in asthma, generally described as ‘airway remodelling'. Features of airway remodelling include epithelial disruption, sub-basement membrane thickening and increased extracellular matrix deposition, goblet cell hyperplasia, increased vascularity, and smooth muscle cell hypertrophy and hyperplasia (Boxall et al., 2006).
There is continued debate about the most important cell type mediating the airway changes in asthma. However, analysis of the current evidence indicates that most if not all elements of the asthmatic airway are dysfunctional. There is clear evidence of epithelial dysfunction with failure of healing and overproduction of growth factors and proinflammatory cytokines (Holgate, 1998), glandular dysfunction with hyperplasia of mucus glands and mucus hypersecretion (Carroll et al., 2002), airway smooth muscle (ASM) dysfunction with resulting hypertrophy, hyperplasia, hyperresponsiveness and cytokine secretion (Ebina et al., 1990, 1993; Brightling et al., 2005a), and inflammatory cell activation with ‘overactive' mast cells (Bradding et al., 2006), T cells (Robinson et al., 1992), eosinophils (Bradding et al., 1994) and neutrophils (Carroll et al., 2002). The current cornerstone of asthma management is the use of inhaled steroids, which are highly efficacious in about 90% of patients because they inhibit many diverse pathological aspects of the dysfunctional airway (Barnes and Adcock, 2003). However, for about 10% of patients, these drugs are of poor efficacy for reasons which are not really understood. These patients are therefore difficult to treat, suffer great morbidity and use up a disproportionate fraction of healthcare resources (Wenzel, 2005). Novel potential treatments for the management of asthma, targeted at specific aspects of the inflammatory response, are emerging, but to date these have been disappointing. An example is the use of anti-interleukin (IL)-5, which specifically targets eosinophil infiltration in the airway. However, in spite of marked inhibition of eosinophilic inflammation, clinical asthma persists unabated (O'Byrne, 2006). There is therefore an unmet clinical need for novel modulators of inflammation and tissue remodelling with different mechanisms of action and/or adverse-effect profiles from existing drugs. Modulators of chemokines and their receptors have the potential to fill this gap.
Chemokines and their receptors
Chemokines represent a large family of 8–15 kDa chemotactic proteins expressed by many immune and non-immune cells, with an essential role in inflammatory reactions (Table 1). Although overall sequence homology between them is poor, they have a similar three-dimensional structure. Chemokines are derived from three or four ancestral genes and are, based on the position of two conserved cysteine residues, categorized into four subfamilies: the main -CC- and -CXC- groups and the less described -C- and -CX3C- groups (Zlotnik and Yoshie, 2000). Chemokines can also be classified according to function into the constitutive and the inducible chemokines. The constitutive group includes chemokines involved in the homeostatic trafficking, whereas the inducible chemokines are upregulated during inflammation. This is mediated mainly by cytokines produced at the site of inflammation, but also by endogenous molecules upregulated during infection and injury, for example, fibrinogen, elastase and defensins, which induce chemokine production through pattern recognition receptors (Smiley et al., 2001; Devaney et al., 2003).
Table 1.
Human chemokines and their receptors
| CC chemokines | Source | Receptor | Responding cell type |
|---|---|---|---|
| CCL1 (I-309) | Mo, T, MC | CCR8 | Mo, T |
| CCL2 (MCP-1) | Mo, L, Fib, End, Epi, Neut, MC, DC | CCR2 | Mo, DC, T, Neut |
| CCL3 (MIP-1α) | Mo, L, Neut, Eos, Fib, MC | CCR1, CCR5 | Mo, DC, Eos, T, NK |
| CCL4 (MIP-1β) | Mo, L, Neut, Eos, Fib, MC, Bas, NK | CCR5 | Mo, DC, T |
| CCL5 (RANTES) | Mo, T, Fib, MC | CCR5, CCR1, CCR3 | Mo, Eos, T, MC, NK, DC |
| CCL7 (MCP-3) | Mo, MC, Fib, End, Epi | CCR1, CCR2, CCR5 | Mo, DC, Eos, T, MC, NK, |
| CCL8 (MCP-2) | Mo, Fib | CCR2, CCR3, CCR5 | Mo, DC, Eos, T, NK |
| CCL11 (Eotaxin) | Epi ,End, Eos, MC | CCR3, CCR5, CXCR3 | Eos, T, MC |
| CCL13 (MCP-4) | Epi, DC | CCR1, CCR2, CCR3 | Mo, Eos, T, MC, NK, |
| CCL14 (HCC-1) | BM, SMC, Gut, Spleen, Liver | CCR1, CCR5 | Mo, DC, Eos, T, NK, MC |
| CCL15 (HCC-2) | Mo, DC, L, NK | CCR1, CCR3 | T, Mo, Eos, MC |
| CCL16 (HCC-4) | Mo | CCR1, CCR2, CCR5 | T , Mo, Eos, MC |
| CCL17 (TARC) | Mo, DC, Epi, SMC, Fib | CCR4 | T, DC, NK |
| CCL18 (PARC) | Mo, DC | CCR3 | T, MC, Eos |
| CCL19 (MIP-3β) | Neut, LN, Spleen, Thymus, Gut, MC | CCR7 | T, fibrocytes, DC |
| CCL20 (MIP-3α) | Liver, Lung, Thymus, Placenta, Appendix, Epidermis, Mo, T, Neut, End | CCR6 | DC, T |
| CCL21 (SLC) | End, LN | CCR7 | DC, T, B, NK, Fibrocytes |
| CCL22 (MDC) | Mo, Epi, DC, B, T, NK | CCR4 | T, CD, NK |
| CCL23 (MPIF-1) | DC, Mo, Lung, Liver | CCR1 | Mo, DC, Eos, T, NK |
| CCL24 (Eotaxin-2) | Mo, T, Lung, Liver, Spleen, Thymus | CCR3 | Eos, T, MC |
| CCL25 (TECK) | DC, Epi, End, Gut | CCR9 | T |
| CCL26 (Eotaxin-3) | End, Heart, Ovary | CCR3, CCR2 | Eos, MC, T |
| CCL27 (CTACK) | Placenta, Skin | CCR10 | T |
| CCL28 (MEC) | Epi, End | CCR10 | T |
| CXC chemokines | |||
| CXCL1 (GRO-α) | Mo, Neut, End, Fib, MC | CXCR2 | Neut, Mo |
| CXCL2 (GRO-β) | Mo, Neut, End, Fib, MC | CXCR2 | Neut, Mo |
| CXCL3 (GRO-γ) | Mo, Neut, End, Fib | CXCR2 | Neut, Mo |
| CXCL4 (PF-4) | Platelets | CXCR3B | End |
| CXCL5 (ENA-78) | End, Eos, MC | CXCR2 | Neut, Mo |
| CXCL6 (GCP-2) | Mo, End, Fib | CXCR1, CXCR2 | Neut, Mo, T |
| CXCL7 (NAP-2) | End | CXCR2 | Neut, Mo |
| CXCL8 (IL-8) | Mo, T, Fib, Epi End, Neut, Bas, NK, MC | CXCR1, CXCR2 | Neut, Mo, T |
| CXCL9 (MIG) | Mo, Neut | CXCR3, CCR3 | T, MC |
| CXCL10 (IP-10) | Mo, Neut, Fib, End | CXCR3, CCR3 | T, MC |
| CXCL11 (ITAC) | Mo, Neut | CXCR3,CXCR7, CCR3 | T, MC |
| CXCL12 (SDF-1) | End, Epi | CXCR4, CXCR7 | T, Mo, MC, NK, Eos, DC |
| CXCL13 (BLC) | Mo, End, DC | CXCR5 | T, B |
| CXCL14 (BRAK) | Mo, Fib, B | Not known | |
| CXCL16 (SR-PSOX) | Mo, DC, B | CXCR6 | T, NK |
| C chemokines | |||
| XCL1 (Lymphotactin) | T, NK, MC | XCR1 | T, NK |
| XCL2 (SMC-1β) | T, NK | XCR1 | T, NK |
| CXC chemokine | |||
| CX3CL1 (Fractalkine) | DC, End, T | CXC3CR1 | Mo, Neut, NK, T |
Bold indicates antagonistic binding of a chemokine receptor.
Abbreviations: B, B cell; Bas, basophil; DC, dendritic cell; End, endothelial cell; Epi: epithelial cell; Eos, eosinophil; Fib, fibroblast; LN, lymph node; L, lymphocyte; MC, mast cell; Mo, monocyte/macrophage; Neut, neutrophil; NK, natural killer cell; T, T cell.
Most chemokines are secreted proteins, but there are two important exceptions: CX3CL1 (fractalkine) and CXCL16. These chemokines are expressed in a membrane-bound form at the end of a cleavable mucin stalk, which allows for dual functions. In the membrane-bound form, these chemokines function as leukocyte adhesion molecules, and when the chemokine is cleaved from its stalk by metalloproteases, they then function as soluble chemoattractants (Bazan et al., 1997; Matloubian et al., 2000).
To date, almost 50 chemokines have been identified, which exert their effects through over 20 distinct G protein-coupled seven-transmembrane receptors. Chemokine receptors fall into the subfamily of class A rhodopsin-like receptors and take up almost 5% of the human genome (Murphy, 1994). Chemokines bind with nanomolar affinity and most chemokine receptors can bind multiple ligands, for example, CCR3 can bind CCL5, CCL7, CCL8, CCL13, CCL11, CCL24 and CCL26. Many chemokines are capable of binding to more than one receptor, such as CCL5 that binds to CCR1, CCR3 and CCR5. A few chemokines and chemokine receptors demonstrate exclusive binding, such as CCL25/CCR9 and CXCL12/CXCR4 (Zimmermann et al., 2003). Recent reports show that chemokines can also act as antagonists for chemokine receptors as well as agonists. For example, CXCL10 is a natural antagonist of CCR3 (Loetscher and Clark-Lewis, 2001). In addition, Duffy antigen/receptor for chemokines (DARC) and D6 bind chemokines with high affinity without inducing any downstream signalling (Bonecchi et al., 2004). Instead these membrane-bound proteins function as decoy receptors, which trigger internalization or degradation of chemokines. The function of certain chemokine receptors (CCR2, CCR5 and CXCR4) can further be modulated by homo- and/or heterodimerization of receptors, a phenomenon that has effects on the binding of ligands and the types and magnitude of the intracellular signalling pathway (Mellado et al., 2001; Moriguchi et al., 2005; Springael et al., 2005).
Upon ligation of the chemokine to the extracellular portion of the chemokine receptor, the intracellular domain binds and activates the heterotrimeric G protein. In response, the Gαi subunit exchanges GDP for GTP, resulting in the dissociation of the heterotrimeric complex into the GαI- and the βγ-subunits. Downstream signalling triggers integrin activation, which enables firm adhesion of leukocytes to endothelial cells. Focal actin polymerization of the leading edge of the cell leads to forward extension. At the rear end of the cell, focal activation of myosin II and formation and contraction of myosin–actin complexes retracts the cell, allowing migration in the direction of the chemotactic gradient (Rot and von Andrian, 2004).
In the multistep model of leukocyte extravasation, chemokines are important not only in the activation of integrins, leading to firm adhesion and subsequent diapedesis, but also in directing leukocytes into different compartments within the tissue. The lung is a unique organ in that it is supplied with blood from both the pulmonary and the systemic circulations that deliver blood to the parenchyma and the airways respectively. Due to the low pressure of the pulmonary system, leukocyte rolling and firm adherence are not required for extravasation, and it is suggested that a weak chemotactic signal alone can mediate migration into the underlying tissue (Doerschuk et al., 2000).
T cells
As an integral part of the adaptive immune system, the T cell is often considered to play a crucial role in maintaining and regulating the allergic immune response in asthmatics. Depletion of T cells in some mouse models is associated with an inability to induce an ‘asthmatic' response. In humans, CD4+ T cells producing IL-4, IL-5 and IL-13 (putative TH2 cells) have been identified in bronchoalveolar lavage (BAL) and bronchial biopsies of both atopic and non-atopic asthmatic patients (Robinson et al., 1992; Brightling et al., 2002b). Lung T cells are mainly memory cells and are found principally in two compartments: the bronchial lamina propria and epithelium, and the peripheral alveolar and interstitial regions of the lung (Wardlaw et al., 2005).
Naive T cells leave the thymus expressing CCR7, essential for entering the high endothelial venules of lymph nodes, where the ligands CCL21 and CCL19 are produced. Naive T cells also express CXCR4, but the significance of this expression is unknown and CXCR4/CXCL12 are not necessary for entry into the lymph nodes (Weninger and von Andrian, 2003). Upon antigen presentation and activation, CD8+ T cells proliferate and differentiate into cytotoxic T cells, and CD4+ cells into either T helper type 1 (TH1) or TH2 effector cells, depending on the pathogenic insult. As a part of the differentiation into effector T cells, there is a drastic change in the chemokine receptor profile of the T cells, allowing them to migrate into the tertiary lymphoid tissues. It is thought that this migration is organ specific and that chemokines are important for providing a ‘homing signal' for the lymphocytes expressing the correct chemokine receptor. The chemokines involved in lymphocyte migration to the skin and gut are well defined (Morales et al., 1999; Kunkel et al., 2000), but it is not known whether there is specific T-cell homing to the lung. If this is the case, the T cells recovered from lung would be expected to express adhesion molecules and chemokine receptors different than those found in other tissues. Indeed, lung-derived T cells from both BAL and peripheral lung tissue express a pattern of chemokine receptors distinct from the small intestine and skin (CCR9−, CCR10− and CCR4LO), suggesting that there is a lung-specific pathway (Campbell et al., 2001). When comparing chemokine receptor expression in T cells from blood and lung, three chemokine receptors were upregulated on the lung-derived cells: CXCR3 and CCR5, which appear to be highly expressed on all tissue T cells, and CXCR6, which is greatly enriched in BAL T cells compared with blood, especially in patients with sarcoidosis or interstitial lung disease (Morgan et al., 2005a). High concentrations of the only ligand for CXCR6, CXCL16, is also found in BAL and is expressed in high amounts by alveolar macrophages, suggesting CXCR6 is a candidate lung homing receptor. However, there was no difference in the level of CXCR6 expression or CXCL16 production between normal subjects, patients with asthma, sarcoidosis or interstitial lung disease, suggesting that CXCR6 plays a role in the homeostatic trafficking of T cells to the lung rather than in the recruitment of T cells to the airways of asthmatics (Morgan et al., 2005a). Interestingly, although ASM expresses CXCL10, and CXCR3 is highly expressed on lung T cells, there is no infiltration of CD3+ cells in the smooth muscle bundle of asthmatic airways (Brightling et al., 2002a, 2005a).
In addition to tissue-specific migration, an inflammation-specific model has been proposed, which involves differential trafficking of TH1 and TH2 cells. Using either IL-12 or IL-4 to polarize naive T cells in vitro to a TH1 or TH2 phenotype, respectively, a distinct chemokine receptor expression pattern was demonstrated: the TH1 cells expressed CCR5 and CXCR3, whereas the TH2 cells expressed CCR3, CCR4 and CCR8 (Kim et al., 2001). CCR4-deficient mice develop less airway eosinophilia and BHR following antigen challenge (Schuh et al., 2002), but CCR8-deficient mice remain unaffected (Chung et al., 2003; Goya et al., 2003).
The physiological significance of CCR4 and CCR8 in humans is less certain and the studies investigating the chemokine receptor expression on TH2 and TH1 cells from human lung are somewhat contradictory. Using immunohistochemistry and reverse transcription-polymerase chain reaction (RT-PCR), Panina-Bordignon et al. (2001) reported that over 90% of T cells infiltrating the bronchial biopsies of allergen-challenged asthmatics produce IL-4 and express CCR4. They also reported that CCR8 was expressed on approximately 28% of infiltrating CCR4+ IL-4+ T cells, whereas no T cells were CCR3+. They also found that the two CCR4 ligands, CCL22 and CCL17, were expressed by airway epithelial cells and their expression was strongly upregulated after allergen challenge (Panina-Bordignon et al., 2001). In contrast, when analyzing the relationship between cytokine production and chemokine receptor expression on freshly isolated blood T cells using single-cell flow cytometry, our group found that CCR3 and CCR4 are preferentially expressed by IL-4-secreting T cells in the lung in both healthy subjects and patients with mild asthma (CCR8 not studied) (Morgan et al., 2005b). However, most lung T lymphocytes produced neither IL-4 nor interferon-γ (IFN-γ) after in vitro stimulation, and the ligands of CCR3 and CCR4 were not found in high amounts in BAL fluid. In addition, there was no polarization of the CXCR3+ CCR5+ T cells in terms of cytokine expression (Morgan et al., 2005b). Together, the results from these studies suggest that the distinct patterns of polarization seen in blood T cells do not apply in the physiological environment of the lung, and further work is needed to elucidate the mechanism by which TH2 cells are recruited to the asthmatic lung.
Invariant natural killer T cells
Invariant natural killer T cells (NKTs) are a subpopulation of T cells that express a conserved canonical T-cell receptor that recognizes the glycolipid α-galactosylceramide. In response to stimuli, NKT cells respond very rapidly by expressing cytokines and have the capacity to produce both IL-4 and IFN-γ. In some animal models, pulmonary NKT cells regulate development of airway eosinophilia, BHR and IgE production (Lisbonne et al., 2003). The role for NKT cells in human asthma has been debated. Akbari et al. (2006) claimed that 60 % of all CD4+ lung T cells in asthmatics were NKT cells and that these cells were absent in the lungs of normal subjects and patients with sarcoidosis (Akbari et al., 2006). However, these results have been severely criticised by several groups (Pham-Thi et al., 2006; Thomas et al., 2006), who suggest that the prevalence of these cells is nearer 1% of lung T cells. The extent to which NKT cells are involved in the pathogenesis of asthma is therefore unclear. Another study analyzing sputum from severe asthmatics found that NKT cells were increased as compared with normal controls and patients with mild asthma (Hamzaoui et al., 2006). Most peripheral blood NKT cells express receptors for chemokines, which mediate homing to extra-lymphoid tissue or sites of inflammation (CCR2, CCR1, CCR5, CCR6, CXCR3, CXCR4 and CXCR6), whereas few NKT cells express lymphoid tissue-homing chemokine receptors (CCR7 and CXCR5) (Kim et al., 2002; Motsinger et al., 2002). Sen et al. (2005) found that there were significantly more NKT cells in peripheral blood of patients with asthma compared with normal subjects. Interestingly, NKT cells from asthmatics drove T-cell expression of IL-4 and IL-13, whereas NKT cells derived from healthy donors drove IFN-γ expression. Whereas CXCR3 and CCR6 were expressed at equal levels by NKT cells from normal and asthmatic individuals, CCR9 was upregulated by the asthmatic NKT cells, which migrated to CCL25 in vitro. Furthermore, immunohistochemistry and RT-PCR of endobronchial biopsies identified more CCR9+ NKT cells in the submucosa of the asthmatic than in the normal subjects (Sen et al., 2005). CCR9 is widely recognized as a gut homing receptor, which is expressed on intraepithelial and lamina propria intestinal lymphocytes, and the authors suggest a role for CCR9 in NKT migration to the lung in asthma.
Dendritic cells
Dendritic cells play a potentially important role in asthma through their ability to process and subsequently present antigen to T cells. They form an intricate network in the airway epithelium and lamina propria of the airways, where they exist in an immature state capable of internalizing foreign antigens but unable to activate naive T cells. Once antigen has been captured, airway myeloid dendritic cells mature and migrate to the regional lymph nodes, where they present processed antigen to T cells, preferentially inducing TH2 responses. In mice, this process of migration to the lymph node is dependent on the activation of CCR7. Thus, on repeated exposure to aerosolized house dust mite, severe combined immunodeficiency (SCID) mice reconstituted with human peripheral blood mononuclear cells and pulsed with dendritic cells from allergic patients developed airway eosinophilia, an increase in IL-4 and IL-5 production in the lung lavage fluid and increased IgE production compared with that in mice primed with unpulsed dendritic cells. All these effects were reduced following in vivo neutralization of the CCR7 ligand CCL21 (Hammad et al., 2002). Furthermore, the indirect upregulation of CCR7 in mice lacking the transcription factor Runx3 is associated with enhanced migration of alveolar dendritic cells to the mediastinal lymph nodes, and the development of elevated serum IgE, and BHR. These features are blocked by anti-CCR7 antibodies (Fainaru et al., 2005). Interestingly, in plt mice, which lack the ability to generate both CCL19 and CCL21, there is attenuation of airway eosinophilia and BHR in the short term after ovalbumin challenge, and also failure for the inflammation to resolve in the longer term (Yamashita et al., 2006). Targeting CCR7 therefore has the potential to do more harm than good.
Eosinophils
Airway eosinophilia is a common feature of asthma. About 80% of asthmatic subjects have an increased number of eosinophils in BAL, endobronchial biopsies and induced sputum compared with normal controls. Sputum eosinophilia correlates with asthma severity and treatment directed at normalization of the sputum eosinophil count reduces asthma exacerbations and admissions without the need for additional anti-inflammatory treatment (Green et al., 2002a). Eosinophils are end-stage granulocytes that derive from the bone marrow under the influence of IL-3, IL-5 and granulocyte-macrophage colony-stimulating factor (GM-CSF) (Rothenberg, 1998). Under baseline conditions, they leave the bone marrow and migrate to the gastrointestinal tract, where they reside within the lamina propria (Mishra et al., 1999). In asthma and other inflammatory conditions, however, eosinophils migrate to extraintestinal tissues such as the lung, where they have been thought to affect the airways in a number of ways (Wardlaw, 1999). In vitro, toxic granule proteins such as major basic protein, eosinophilic cationic protein and eosinophil peroxidases have a direct cytotoxic effect on the respiratory epithelium, enhance ASM responsiveness and trigger mast cell degranulation (Gundel et al., 1991; Piliponsky et al., 2001). Eosinophils also secrete a number of proinflammatory interleukins, including IL-2, IL, 4, IL-5, IL-10, IL-12 and IL-13, which enhance the TH2 response, and the profibrotic cytokine transforming growth factor-β (TGF-β), which is involved in airway remodelling (Martin et al., 1996; Ohno et al., 1996).
IL-5 is a key cytokine for the growth, activation and survival of eosinophils (Sanderson, 1992; Dewson et al., 2001), and together with IL-4 and IL-13, regulates the trafficking of these cells into sites of inflammation (Sher et al., 1990; Horie et al., 1997). IL-4 and IL-13 control eosinophil trafficking directly by upregulating adhesion molecules on endothelial cells, particularly vascular cell adhesion molecule 1 (Schleimer et al., 1992; Ying et al., 1997b) or by inducing chemokine expression by airway epithelial cells (Wardlaw, 2001). However, as discussed above, the ability of anti-IL-5 to markedly deplete eosinophils but have no effect on asthma symptoms or experimental allergen challenge means there is doubt as to their importance in asthma pathophysiology.
Several studies have shown the role of the CCR3 binding chemokines in eosinophil migration to the lung, particularly CCL5 and the eotaxins CCL11, CCL24 and CCL26. CCR3 is the chemokine receptor, which is most highly expressed by eosinophils (Ponath et al., 1996), and CCR3 expression is relatively abundant on these cells. CCL11, CCL5 and CCR3 mRNA and protein have been detected in bronchial biopsies and BAL from asthmatic patients, and the number of CCR3 mRNA-positive cells in the bronchial mucosa correlated with airway eosinophilia and BHR (Ying et al., 1997c). CCL26, which is upregulated in bronchial epithelial cells after stimulation with IL-4, is important for eosinophil transepithelial migration in vitro (Cuvelier and Patel, 2001; Yuan et al., 2006). Various mouse models have been used to further elucidate the specific role of CCR3 in eosinophil migration and airway physiology. Results of two studies using CCR3 knockout mice demonstrated reduced eosinophil recruitment, but were contradictory with respect to the development of BHR and seemed to depend on the allergen sensitization protocol. Humbles et al. (2002), who used a systemic ovalbumin (OVA)/alum sensitization followed by respiratory OVA challenge to mimic the asthmatic response, found a reduction in lung eosinophils in the CCR3−/− mice. The eosinophils appeared to be able to migrate through the endothelial cells, but not the endothelial basement membrane, suggesting that in this model, CCR3 binding chemokines are not essential for rolling, adhesion and transmigration through endothelium, but important for migration into the lung parenchyma (Humbles et al., 2002). Perhaps surprisingly, this reduction in eosinophil recruitment was accompanied by a marked increase in BHR. In contrast, Ma et al. (2002), using epicutaneous OVA sensitization, were able to significantly reduce BAL eosinophils, with a parallel decrease in BHR (Ma et al., 2002). A proportion of human donors have CCR1+ eosinophils and thus respond to CCL3. CCL3 has also been detected in the lungs of asthmatics (Sabroe et al., 1999; Phillips et al., 2003), and an in vivo blockade of murine CCL3 using monoclonal antibodies resulted in reduced airway eosinophilia and BHR in the early allergic response (Campbell et al., 1998).
Lukacs (2001) proposed a mechanism for murine eosinophil migration in response to chemokines, where migration from blood into the lung tissue is regulated by CCL3, CCL7 and CCL22, produced by macrophages in the lung interstitium (Lukacs, 2001). Once in the lung, a gradient of CCL5 and CCL11 is responsible for guiding the eosinophils to the airway epithelium. CCL11 is also an important eosinophil degranulator (Fujisawa et al., 2000) and it would be logical if eosinophils did not degranulate before having reached their final destination. This mechanism of recruitment is supported by data from another mouse model demonstrating that initial eosinophil localization was dependent on CCL3, whereas secondary recruitment and degranulation was CCL11 dependent and CCL3 independent (Campbell et al., 1998). The relevance of these observations in human asthma needs to be further clarified.
The role for CCR6 in human eosinophil recruitment is uncertain. Sullivan et al. (1999), using RT-PCR and polyclonal anti-human CCR6 serum to detect receptor expression, declare that 20% of blood eosinophils from asthmatic patients express low levels of CCR6, and that these eosinophils respond to CCL20 (Sullivan et al., 1999). Other more recent studies using flow cytometry and monoclonal antibodies found CCR6 to be undetectable in BAL and blood (Liu et al., 2003).
Neutrophils
Several clinical studies have observed a correlation between airway neutrophilia and asthma severity, as measured by forced expiratory volume in 1 second (FEV1), and some investigators have suggested that a predominantly neutrophilic recruitment pattern may be a feature of a subgroup of severe asthmatics (Sur et al., 1993; Fahy et al., 1995; Pavord et al., 1999). There are several reasons why neutrophils could be important in severe asthma: activated neutrophils release tissue damaging enzymes such as neutrophil elastase, reactive oxygen compounds, cytokines and lipid mediators. Neutrophil elastase acts as a secretagogue for goblets cells and may thus contribute to the prominent mucus hypersecretion in asthma. The importance of neutrophils in severe asthma is further evidenced by postmortem investigations of patients with sudden-onset fatal asthma, which demonstrated absence of eosinophils and relatively more neutrophils in the airway submucosa (Sur et al., 1993) and airway mucosal glands (Carroll et al., 2002).
Sputum neutrophilia combined with normal numbers of eosinophils is a characteristic of non-atopic asthmatics, who have an impaired response to steroid treatment (Green et al., 2002b). This supports the idea already proposed by Wenzel et al. (1999) that neutrophilic asthma represents a distinct inflammatory phenotype. By thoroughly characterizing severe asthmatics, the authors distinguished between the non-eosinophilic and the eosinophilic severe asthmatics, which both had high levels of neutrophils in their airways. The non-eosinophilic airways exhibited a lower degree of sub-basement membrane thickening, less infiltration of mast cells, lymphocytes and macrophages (Wenzel et al., 1999).
Neutrophils express two main chemokine receptors: CXCR1 and CXCR2, and it is thought that CXCR2 is the major receptor involved in recruitment (White et al., 1998), whereas CXCR1 is important in neutrophil activation (Chuntharapai and Kim, 1995; Sabroe et al., 1997). CXCL8, which is known to be expressed by activated airway epithelium, fibroblasts, macrophages, mast cells and by the neutrophils themselves, binds both receptors and is a potent chemoattractant for neutrophils. When analyzing the tracheal aspirates of acute severe asthmatics, Ordonez et al. (2000) detected high amounts of both neutrophils and CXCL8, and that the levels of CXCL8 correlated significantly and positively with the length of the acute asthma exacerbation (Ordonez et al., 2000). Gibson et al. (2001) confirmed these results when studying sputum from persistent asthmatics of all severities, where they found that levels of CXCL8 correlated with neutrophil numbers and was upregulated in non-eosinophilic asthmatics (Gibson et al., 2001).
Recently, different antagonists of CXCR2 have been developed to prevent neutrophil accumulation; in particular, a selective non-peptide antagonist of CXCR2 has been shown to exhibit significant anti-inflammatory effects in acute and chronic models of arthritis in rabbits (Podolin et al., 2002). However, in a recent study, Pignatti et al. (2005) show a downregulation of CXCR1 and CXCR2 in airway neutrophils, suggesting that activation of the neutrophils with CXCL8 in the airways would downregulate the receptors once the cells were recruited to the lung (Pignatti et al., 2005). This phenomenon might render antagonistic therapies aimed at CXCL8 receptors redundant. CXCL8 expression is regulated by various cytokines such as tumor necrosis factor-α (TNF-α) and the relatively recently discovered IL-17, expressed by CD4+ cells. Stimulation of a human bronchial epithelial cell line with IL-17 upregulated expression of CXCL8, and a recent study found that sputum IL-17 was associated with neutrophilia in severe asthma (Sun et al., 2005).
Mast cells
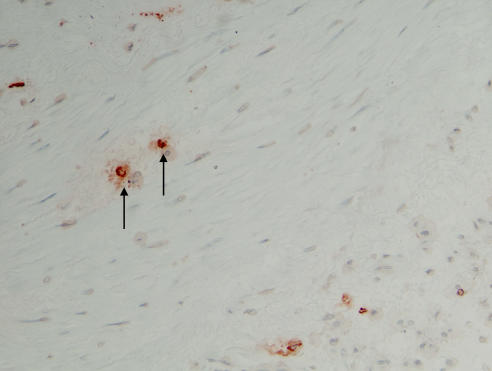
Mast cells play a significant role in the pathophysiology of asthma through their ability to release a plethora of autacoid mediators, proteases and cytokines in response to their activation by both allergen and other non-immunological stimuli (Bradding et al., 2006). Importantly, mast cells are present in a chronically ‘activated' secretory state within the asthmatic bronchial mucosa, with evidence of ongoing mediator release and cytokine synthesis (Wenzel et al., 1988; Broide et al., 1991; Bradding et al., 1994; Ying et al., 1997a). Mast cells present in the BAL fluid of asthmatic subjects exhibit increased spontaneous and IgE-dependent mediator release, and strong correlations have been observed between the severity of BHR and mast cell numbers, histamine concentrations and constitutive histamine release in BAL fluid (Flint et al., 1985; Casale et al., 1987; Wardlaw et al., 1988). Mast cells are resident in normal airways, and their number in the lamina propria is similar in both normal subjects and patients with asthma. However, in asthma, mast cells infiltrate three key structures: ASM (Figure 1), the airway submucosal glands and the airway epithelium (Bradding et al., 1994; Carroll et al., 2002; Brightling et al., 2002a). Since mast cells in the asthmatic airway are activated and their mediators have profound effects on these structural elements, this redistribution of mast cells is likely to be of great importance for the development and propagation of this disease. This concept is supported by the observation that the number and activation state of mast cells in the submucosal glands correlate strongly with the extent of mucus plugging, whereas the number of mast cells in the ASM correlates with the severity of BHR. Furthermore, in eosinophilic bronchitis, the mucosal immunopathology is virtually identical when compared with asthma, in terms of cellular infiltrates and TH2 cytokine expression, but mast cells are not found in the ASM in eosinophilic bronchitis (Brightling et al., 2002a). This further suggests that the mast cell myositis in asthma is a key requirement for the expression of BHR and variable airflow obstruction.
Figure 1.
Bronchial biopsy specimen from a patient with asthma showing mast cell infiltration of airway smooth muscle.
Human lungs mast cells (HLMC) express numerous chemokine receptors. In particular, CCR3, CXCR1, CXCR3 and CXCR4 are highly expressed by ex vivo HLMC, and the respective ligands for these receptors CCL11, CXCL8, CXCL10 and CXCL12 mediate lung mast cell chemotaxis (Brightling et al., 2005a, b). It is not clear whether the mast cell infiltration of ASM in asthma involves the recruitment of progenitors, which then differentiate locally, or the local migration of resident differentiated cells. However, it is well recognized that mature differentiated cells have the ability to migrate within the airway wall. Interestingly, we found that both CXCR3 and CCR3 expression were increased on ex vivo HLMC compared with human bone marrow-derived mast cells, suggesting that they may be important for the migration of mature differentiated cells (Brightling et al., 2005b). In bronchial biopsies from patients with asthma, approximately 50% of mast cells expressed CXCR3 compared with nearly 100% of mast cells within the asthmatic ASM. In contrast, CCR3 was almost absent on ASM mast cells (Brightling et al., 2005b). We also found that there was increased expression of the CXCR3 ligand CXCL10 in asthmatic ASM compared with that from normal subjects in both biopsies and when activated in vitro (Brightling et al., 2005a). Taken together, these observations suggest that CXCR3 and CXCL10 play a crucial role in determining the distribution of mast cells within the lung, especially migration to the ASM in asthma.
These experiments also showed that CXCR1 and CXCR4 contribute to HLMC chemotaxis induced by asthmatic ASM-conditioned media, but CCR3 did not in spite of there being high concentrations of its ligand CCL11 present (Brightling et al., 2005a). This may be because CXCL10 is a natural antagonist of CCR3 (Loetscher and Clark-Lewis, 2001). However, when asthmatic ASM is activated by TH2 cytokines, CCL11 contributes to the HLMC migration, which occurs in response to ASM-conditioned medium. There also seems to be an added level of complexity in that normal ASM appears to secrete an inhibitor of HLMC migration (Sutcliffe et al., 2006). Romagnani et al. (1999) suggested that CCR3 was involved in the homing of tryptase–chymase double positive cells (MCTC), which they found to be the predominant mast cell phenotype expressing this receptor (Romagnani et al., 1999). They also showed that CCL11 and CCL5 mediated mast cell migration through CCR3, thereby suggesting that the CCR3/CCL11/CCL5 pathway is important in mast cell migration in connective tissues (Romagnani et al., 1999). However, MCTC mast cells within ASM rarely express this receptor (Brightling et al., 2005a). Furthermore, Humbles et al. (2002) surprisingly found increased baseline numbers of submucosal mast cells in the trachea and large bronchi of CCR3 knockout mice. Furthermore, following antigen challenge, intraepithelial mast cell numbers increased significantly more in the CCR3-deficient mice compared with normal littermates (Humbles et al., 2002). The mechanism behind this is unclear but indicate a multifaceted and poorly understood role for CCR3 in mast cell migration in the airway.
Juremalm et al. (2000) showed that CCL5, a recognized agonist for CCR1, also appeared to induce cord blood mast cell chemotaxis through CCR4, whereas the recognized ligands CCL17 and CCL22 did not (Juremalm et al., 2000). They also demonstrated that both CCL17 and CCL22 significantly inhibited the migration toward CCL5 by 54 and 74%, respectively. In contrast, CCL2, CCL3, CXCL10 and CXC12 did not affect CCL5-induced migration. The authors concluded that CCL17 and CCL22 act as antagonists for CCL5-mediated migration induced through CCR4. The role this has in determining the specificity of the mast cell migratory response in asthmatic airways requires further work, particularly as expression of CCR4 on HLMC is relatively low.
Structural lung cells
As described above, structural lung cells such as airway epithelium, smooth muscle and fibroblasts are important sources of chemokines, and contribute to the recruitment of leukocytes both to and within the lung. However, structural cells also express certain chemokine receptors and can therefore be activated by chemokines produced by inflammatory cells infiltrating the lung, or in an autocrine manner, which may potentially contribute to airway remodelling.
One of the main features of airway remodelling is increased ASM mass (Kay, 1996). Mathematical models have suggested that an increase in ASM volume is enough to induce BHR through local mechanical effects alone (Macklem, 1996). ASM mass correlates with asthma severity and is thought to be the result of hypertrophy and/or hyperplasia (Ebina et al., 1993; Benayoun et al., 2003; Woodruff et al., 2004). In an ex vivo study of asthmatic ASM, increased proliferation has been suggested to be the main cause of hyperplasia in asthmatics (Johnson et al., 2001), but several studies have failed to show that this is the case in vivo (Benayoun et al., 2003; Woodruff et al., 2004). Another explanation is that smooth muscle progenitors, either located within the airway or recruited from peripheral blood, such as CD34+ collagen I+ fibrocytes, are recruited to the ASM and differentiate into ASM. Chemokines might therefore be involved in this migration. This theory is supported by a recent study from our group demonstrating the functional expression of CCR7 on ASM and fibroblasts from both asthmatic and normal donors (Kaur et al., 2006). In the bronchial mucosa, one of the ligands for CCR7, CCL19, was expressed by blood vessels, but it was also expressed by mast cells within the asthmatic ASM bundles, and in severe asthma by the ASM itself. In vitro, CCL19 expression by ASM was increased in response to injury as shown by a wound healing assay. Furthermore, peripheral blood-derived fibrocytes have been shown to express CCR3, CCR5, CCR7 and CXCR4, but only CCR7 and CXCR4 have been proven to be functional. It is therefore plausible that these cells are recruited to the airway through CCL19+ blood vessels in the lung and then recruited to the ASM bundle by mast cell- and ASM-derived CCL19. Coupled with the observation that the number of mast cells in the ASM bundle is increased in asthma, these results focus attention on a potential mechanism by which mast cell-derived CCL19 binds to and activates CCR7+ ASM, myofibroblasts and/or fibroblasts, and thereby contributes to the increased ASM mass in asthmatic airways (Kaur et al., 2006).
Another characteristic of airway remodelling is subepithelial collagen deposition, which is a result of deposition of collagen I, III and V, fibronectin and tenascin in the lamina reticularis (Roche et al., 1989). The cells responsible for producing these proteins are thought to be myofibroblasts positioned directly beneath the airway epithelium (Brewster et al., 1990). After in vitro stimulation with the fibrogenic cytokines endothelin-1 (ET-1) and TGF-β1, which are produced in the airways of asthmatic patients, peripheral blood fibrocytes differentiate into myofibroblast like cells, suggesting that fibrocytes are potential myofibroblast precursors (Schmidt et al., 2003). Fibrocytes located underneath the epithelial layer were also found in biopsies from asthmatics patients. The recruitment of fibrocytes to the epithelium is therefore important for the development of airway remodelling in asthma. In addition to CCR7, CXCR4 is functionally expressed by fibrocytes and its ligand, CXC12, is produced by bronchial epithelium, and has been proven to attract human peripheral blood fibrocytes (Phillips et al., 2004; Kaur et al., 2006). Taken together, this suggests that CXCL12 is involved in the migration of fibrocytes toward the epithelium, thus contributing to the increase in subepithelial and luminal myofibroblasts observed in asthma.
Future perspectives
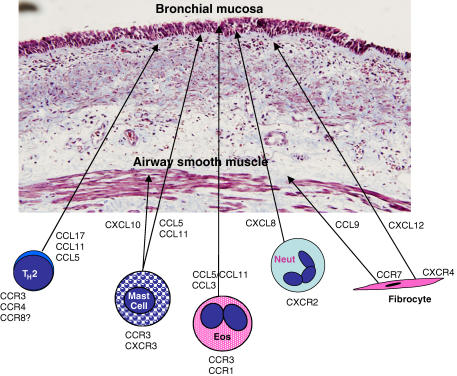
The discovery of chemokines as important regulators of the immune system has led to intense research of over the last 20 years. Since chemokines are involved in the abnormal migration patterns of leukocytes and structural cells within the lung, they make attractive drug targets. However, designing novel pharmacological agents targeting chemokines and their receptors as treatments for asthma raises a number of questions. Firstly, inhibition of which chemokines/chemokine receptors would be most effective in order to obtain a significant reduction in asthma symptoms? Secondly, how could selective inhibition be achieved? The situation is complicated by the fact that numerous chemokines are upregulated in airway inflammation and that chemokines bind several receptors leading to marked redundancy. Inhibition of chemokine receptor activity can be achieved either by using modified chemokines or small-molecule chemokine receptor antagonists, which function as antagonists, or by using neutralizing antibodies against the receptor directly. CCR3 is a potential target, since this chemokine receptor has proven to be involved in migration of eosinophils, certain TH2 cells and possibly airway mast cells. A recent mouse study used two types of small-molecule antagonists to block CCR3, which significantly and selectively reduced eosinophil infiltration to the lung of mice (Das et al., 2006). The CXCL10/CXCR3 axis also looks interesting as it appears to target mast cells to the ASM in asthma. Very few chemokine receptor antagonists have been developed for humans to date, but there is great interest from the pharmaceutical industry in this area, and further antagonists are likely to emerge in the future. A schematic summary of chemokines potentially involved in distribution of cells in the asthmatic airway is depicted in Figure 2. Lastly, bearing in mind the obvious redundancy in the chemokine network, more general approaches aimed at inhibiting cell migration in asthma might ultimately prove more fruitful. For example, HLMC express the K+ channel KCa3.1 (Duffy et al., 2001, 2004) and blockade of this inhibits HLMC migration to a number of diverse chemoattractants (Cruse et al., 2006).
Figure 2.
Chemokines and chemokine receptors with potential as therapeutic targets for the treatment of asthma.
Abbreviations
- ASM
airway smooth muscle
- BAL
bronchoalveolar lavage
- BHR
bronchial hyperresponsiveness
- FEV1
forced expiratory volume in 1 second
- HLMC
human lung mast cell
- IL
interleukin
- NKT cell
natural killer T cell
- TH1
T helper type 1 cell
- TH2 cell
T helper type 2 cell
- TGF-β
transforming growth factor-β
Conflict of interest
The authors state no conflict of interest.
References
- Akbari O, Faul JL, Hoyte EG, Berry GJ, Wahlstrom J, Kronenberg M, et al. CD4+ invariant T-cell-receptor+natural killer T cells in bronchial asthma. N Engl J Med. 2006;354:1117–1129. doi: 10.1056/NEJMoa053614. [DOI] [PubMed] [Google Scholar]
- Barnes PJ, Adcock IM. How do corticosteroids work in asthma. Ann Intern Med. 2003;139 5 Part 1:359–370. doi: 10.7326/0003-4819-139-5_part_1-200309020-00012. [DOI] [PubMed] [Google Scholar]
- Bazan JF, Bacon KB, Hardiman G, Wang W, Soo K, Rossi D, et al. A new class of membrane-bound chemokine with a CX3C motif. Nature: 1997;385:640. doi: 10.1038/385640a0. [DOI] [PubMed] [Google Scholar]
- Becker EL. Elements of the history of our present concepts of anaphylaxis, hay fever and asthma. Clin Exp Allergy. 1999;29:875. doi: 10.1046/j.1365-2222.1999.00531.x. [DOI] [PubMed] [Google Scholar]
- Benayoun L, Druilhe A, Dombret MC, Aubier M, Pretolani M. Airway structural alterations selectively associated with severe asthma. Am J Respir Crit Care Med. 2003;167:1360–1368. doi: 10.1164/rccm.200209-1030OC. [DOI] [PubMed] [Google Scholar]
- Bonecchi R, Locati M, Galliera E, Vulcano M, Sironi M, Fra AM, et al. Differential recognition and scavenging of native and truncated macrophage-derived chemokine (macrophage-derived chemokine/CC chemokine ligand 22) by the D6 decoy receptor. J Immunol. 2004;172:4972–4976. doi: 10.4049/jimmunol.172.8.4972. [DOI] [PubMed] [Google Scholar]
- Boxall C, Holgate ST, Davies DE. The contribution of transforming growth factor-beta and epidermal growth factor signalling to airway remodelling in chronic asthma. Eur Respir J. 2006;27:208–229. doi: 10.1183/09031936.06.00130004. [DOI] [PubMed] [Google Scholar]
- Bradding P, Roberts JA, Britten KM, Montefort S, Djukanovic R, Mueller R, et al. Interleukin-4, -5, and -6 and tumor necrosis factor-alpha in normal and asthmatic airways: evidence for the human mast cell as a source of these cytokines. Am J Respir Cell Mol Biol. 1994;10:471–480. doi: 10.1165/ajrcmb.10.5.8179909. [DOI] [PubMed] [Google Scholar]
- Bradding P, Walls AF, Holgate ST. The role of the mast cell in the pathophysiology of asthma. J Allergy Clin Immunol. 2006;117:1277–1284. doi: 10.1016/j.jaci.2006.02.039. [DOI] [PubMed] [Google Scholar]
- Braman SS. The global burden of asthma. Chest. 2006;130:4S–12. doi: 10.1378/chest.130.1_suppl.4S. [DOI] [PubMed] [Google Scholar]
- Brewster CE, Howarth PH, Djukanovic R, Wilson J, Holgate ST, Roche WR. Myofibroblasts and subepithelial fibrosis in bronchial asthma. Am J Respir Cell Mol Biol. 1990;3:507–511. doi: 10.1165/ajrcmb/3.5.507. [DOI] [PubMed] [Google Scholar]
- Brightling C, Ammit AJ, Kaur D, Black JL, Wardlaw AJ, Hughes JM, et al. The CXCL10/CXCR3 axis mediates human lung mast cell migration to asthmatic airway smooth muscle. Am J Respir Crit Care Med. 2005a;171:1103–1108. doi: 10.1164/rccm.200409-1220OC. [DOI] [PubMed] [Google Scholar]
- Brightling C, Bradding P, Symon FA, Holgate ST, Wardlaw AJ, Pavord ID. Mast cell infiltration of airway smooth muscle in asthma. N Engl J Med. 2002a;346:1699–1705. doi: 10.1056/NEJMoa012705. [DOI] [PubMed] [Google Scholar]
- Brightling CE, Kaur D, Berger P, Morgan AJ, Wardlaw AJ, Bradding P. Differential expression of CCR3 and CXCR3 by human lung and bone marrow-derived mast cells: implications for tissue mast cell migration. J Leukoc Biol. 2005b;77:759–766. doi: 10.1189/jlb.0904511. [DOI] [PubMed] [Google Scholar]
- Brightling CE, Symon FA, Birring SS, Bradding P, Pavord ID, Wardlaw AJ. TH2 cytokine expression in bronchoalveolar lavage fluid T lymphocytes and bronchial submucosa is a feature of asthma and eosinophilic bronchitis. J Allergy Clin Immunol. 2002b;110:899–905. doi: 10.1067/mai.2002.129698. [DOI] [PubMed] [Google Scholar]
- Brightling CE, Ward R, Goh KL, Wardlaw AJ, Pavord ID. Eosinophilic bronchitis is an important cause of chronic cough. Am J Respir Crit Care Med. 1999;160:406–410. doi: 10.1164/ajrccm.160.2.9810100. [DOI] [PubMed] [Google Scholar]
- Broide DH, Gleich GJ, Cuomo AJ, Coburn DA, Federman EC, Schwartz LB, et al. Evidence of ongoing mast cell and eosinophil degranulation in symptomatic asthma airway. J Allergy Clin Immunol. 1991;88:637–648. doi: 10.1016/0091-6749(91)90158-k. [DOI] [PubMed] [Google Scholar]
- Campbell EM, Kunkel SL, Strieter RM, Lukacs NW. Temporal role of chemokines in a murine model of cockroach allergen-induced airway hyperreactivity and eosinophilia. J Immunol. 1998;161:7047–7053. [PubMed] [Google Scholar]
- Campbell JJ, Brightling CE, Symon FA, Qin S, Murphy KE, Hodge M, et al. Expression of chemokine receptors by lung T cells from normal and asthmatic subjects. J Immunol. 2001;166:2842–2848. doi: 10.4049/jimmunol.166.4.2842. [DOI] [PubMed] [Google Scholar]
- Carroll NG, Mutavdzic S, James AL. Increased mast cells and neutrophils in submucosal mucous glands and mucus plugging in patients with asthma. Thorax. 2002;57:677–682. doi: 10.1136/thorax.57.8.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casale TB, Wood D, Richerson HB, Trapp S, Metzger WJ, Zavala D, et al. Elevated bronchoalveolar lavage fluid histamine levels in allergic asthmatics are associated with methacholine bronchial hyperresponsiveness. J Clin Invest. 1987;79:1197–1203. doi: 10.1172/JCI112937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung CD, Kuo F, Kumer J, Motani AS, Lawrence CE, Henderson WR, Jr, et al. CCR8 is not essential for the development of inflammation in a mouse model of allergic airway disease. J Immunol. 2003;170:581–587. doi: 10.4049/jimmunol.170.1.581. [DOI] [PubMed] [Google Scholar]
- Chuntharapai A, Kim KJ. Regulation of the expression of IL-8 receptor A/B by IL-8: possible functions of each receptor. J Immunol. 1995;155:2587–2594. [PubMed] [Google Scholar]
- Cruse G, Duffy SM, Brightling CE, Bradding P. Functional KCa3.1 K+ channels are required for human lung mast cell migration. Thorax. 2006;61:880–885. doi: 10.1136/thx.2006.060319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuvelier SL, Patel KD. Shear-dependent eosinophil transmigration on interleukin 4-stimulated endothelial cells: a role for endothelium-associated eotaxin-3. J Exp Med. 2001;194:1699–1709. doi: 10.1084/jem.194.12.1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das AM, Vaddi KG, Solomon KA, Krauthauser C, Jiang X, McIntyre KW, et al. Selective inhibition of eosinophil influx into the lung by small molecule CC chemokine receptor 3 antagonists in mouse models of allergic inflammation. J Pharmacol Exp Ther. 2006;318:411–417. doi: 10.1124/jpet.105.099812. [DOI] [PubMed] [Google Scholar]
- Devaney JM, Greene CM, Taggart CC, Carroll TP, O'Neill SJ, McElvaney NG. Neutrophil elastase up-regulates interleukin-8 via toll-like receptor 4. FEBS Lett. 2003;544:129–132. doi: 10.1016/s0014-5793(03)00482-4. [DOI] [PubMed] [Google Scholar]
- Dewson G, Cohen GM, Wardlaw AJ. Interleukin-5 inhibits translocation of Bax to the mitochondria, cytochrome c release, and activation of caspases in human eosinophils. Blood. 2001;98:2239–2247. doi: 10.1182/blood.v98.7.2239. [DOI] [PubMed] [Google Scholar]
- Doerschuk CM, Tasaka S, Wang Q. CD11/CD18-dependent and -independent neutrophil emigration in the lungs: how do neutrophils know which route to take. Am J Respir Cell Mol Biol. 2000;23:133–136. doi: 10.1165/ajrcmb.23.2.f193. [DOI] [PubMed] [Google Scholar]
- Duffy SM, Lawley WJ, Conley EC, Bradding P. Resting and activation-dependent ion channels in human mast cells. J Immunol. 2001;167:4261–4270. doi: 10.4049/jimmunol.167.8.4261. [DOI] [PubMed] [Google Scholar]
- Duffy SM, Berger P, Cruse G, Yang W, Bolton SJ, Bradding P. The K+ channel IKCa1 potentiates Ca2+ influx and degranulation in human lung mast cells. J Allergey Clin Immunol. 2004;114:66–72. doi: 10.1016/j.jaci.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Ebina M, Takahashi T, Chiba T, Motomiya M. Cellular hypertrophy and hyperplasia of airway smooth muscles underlying bronchial asthma. A 3-D morphometric study. Am Rev Respir Dis. 1993;148:720–726. doi: 10.1164/ajrccm/148.3.720. [DOI] [PubMed] [Google Scholar]
- Ebina M, Yaegashi H, Chiba R, Takahashi T, Motomiya M, Tanemura M. Hyperreactive site in the airway tree of asthmatic patients revealed by thickening of bronchial muscles. A morphometric study. Am Rev Respir Dis. 1990;141 5 Part 1:1327–1332. doi: 10.1164/ajrccm/141.5_Pt_1.1327. [DOI] [PubMed] [Google Scholar]
- Fahy JV, Kim KW, Liu J, Boushey HA. Prominent neutrophilic inflammation in sputum from subjects with asthma exacerbation. J Allergy Clin Immunol. 1995;95:843–852. doi: 10.1016/s0091-6749(95)70128-1. [DOI] [PubMed] [Google Scholar]
- Fainaru O, Shseyov D, Hantisteanu S, Groner Y. Accelerated chemokine receptor 7-mediated dendritic cell migration in Runx3 knockout mice and the spontaneous development of asthma-like disease. Proc Natl Acad Sci USA. 2005;102:10598–10603. doi: 10.1073/pnas.0504787102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint KC, Leung KB, Hudspith BN, Brostoff J, Pearce FL, Johnson NM. Bronchoalveolar mast cells in extrinsic asthma: a mechanism for the initiation of antigen specific bronchoconstriction. BMJ. 1985;291:923–926. doi: 10.1136/bmj.291.6500.923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujisawa T, Kato Y, Nagase H, Atsuta J, Terada A, Iguchi K, et al. Chemokines induce eosinophil degranulation through CCR-3. J Allergy Clin Immunol. 2000;106:507–513. doi: 10.1067/mai.2000.108311. [DOI] [PubMed] [Google Scholar]
- Gibson PG, Simpson JL, Saltos N. Heterogeneity of airway inflammation in persistent asthma: evidence of neutrophilic inflammation and increased sputum interleukin-8. Chest. 2001;119:1329–1336. doi: 10.1378/chest.119.5.1329. [DOI] [PubMed] [Google Scholar]
- Goya I, Villares R, Zaballos A, Gutierrez J, Kremer L, Gonzalo JA, et al. Absence of CCR8 does not impair the response to ovalbumin-induced allergic airway disease. J Immunol. 2003;170:2138–2146. doi: 10.4049/jimmunol.170.4.2138. [DOI] [PubMed] [Google Scholar]
- Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Bradding P, et al. Asthma exacerbations and sputum eosinophil counts: a randomised controlled trial. Lancet. 2002a;360:1715–1721. doi: 10.1016/S0140-6736(02)11679-5. [DOI] [PubMed] [Google Scholar]
- Green RH, Brightling CE, Woltmann G, Parker D, Wardlaw AJ, Pavord ID. Analysis of induced sputum in adults with asthma: identification of subgroup with isolated sputum neutrophilia and poor response to inhaled corticosteroids. Thorax. 2002b;57:875–879. doi: 10.1136/thorax.57.10.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gundel RH, Letts LG, Gleich GJ. Human eosinophil major basic protein induces airway constriction and airway hyperresponsiveness in primates. J Clin Invest. 1991;87:1470–1473. doi: 10.1172/JCI115155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammad H, Lambrecht BN, Pochard P, Gosset P, Marquillies P, Tonnel AB, et al. Monocyte-derived dendritic cells induce a house dust mite-specific Th2 allergic inflammation in the lung of humanized SCID mice: involvement of CCR7. J Immunol. 2002;169:1524–1534. doi: 10.4049/jimmunol.169.3.1524. [DOI] [PubMed] [Google Scholar]
- Hamzaoui A, Rouhou SC, Grairi H, Abid H, Ammar J, Chelbi H, et al. NKT cells in the induced sputum of severe asthmatics. Mediators Inflamm. 2006;2006:71214. doi: 10.1155/MI/2006/71214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holgate ST. The inflammation-repair cycle in asthma: the pivotal role of the airway epithelium. Clin Exp Allergy. 1998;28 Suppl 5:97–103. doi: 10.1046/j.1365-2222.1998.028s5097.x. [DOI] [PubMed] [Google Scholar]
- Horie S, Okubo Y, Hossain M, Sato E, Nomura H, Koyama S, et al. Interleukin-13 but not interleukin-4 prolongs eosinophil survival and induces eosinophil chemotaxis. Intern Med. 1997;36:179–185. doi: 10.2169/internalmedicine.36.179. [DOI] [PubMed] [Google Scholar]
- Huber HL, Koessler KK. The pathology of bronchial asthma. Arch Intern Med. 1922;30:689–695. [Google Scholar]
- Humbert M, Durham SR, Ying S, Kimmitt P, Barkans J, Assoufi B, et al. IL-4 and IL-5 mRNA and protein in bronchial biopsies from patients with atopic and nonatopic asthma: evidence against ‘intrinsic' asthma being a distinct immunopathologic entity. Am J Respir Crit Care Med. 1996a;154:1497–1504. doi: 10.1164/ajrccm.154.5.8912771. [DOI] [PubMed] [Google Scholar]
- Humbert M, Grant JA, Taborda-Barata L, Durham SR, Pfister R, Menz G, et al. High-affinity IgE receptor (FcepsilonRI)-bearing cells in bronchial biopsies from atopic and nonatopic asthma. Am J Respir Crit Care Med. 1996b;153 6 Part 1:1931–1937. doi: 10.1164/ajrccm.153.6.8665058. [DOI] [PubMed] [Google Scholar]
- Humbles AA, Lu B, Friend DS, Okinaga S, Lora J, Al-Garawi A, et al. The murine CCR3 receptor regulates both the role of eosinophils and mast cells in allergen-induced airway inflammation and hyperresponsiveness. Proc Natl Acad Sci USA. 2002;99:1479–1484. doi: 10.1073/pnas.261462598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson PR, Roth M, Tamm M, Hughes M, Ge Q, King G, et al. Airway smooth muscle cell proliferation is increased in asthma. Am J Respir Crit Care Med. 2001;164:474–477. doi: 10.1164/ajrccm.164.3.2010109. [DOI] [PubMed] [Google Scholar]
- Juremalm M, Hjertson M, Olsson N, Harvima I, Nilsson K, Nilsson G. The chemokine receptor CXCR4 is expressed within the mast cell lineage and its ligand stromal cell-derived factor-1alpha acts as a mast cell chemotaxin. Eur J Immunol. 2000;30:3614–3622. doi: 10.1002/1521-4141(200012)30:12<3614::AID-IMMU3614>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- Kaur D, Saunders R, Berger P, Siddiqui S, Woodman L, Wardlaw A, et al. Airway smooth muscle and mast cell-derived CCL19 mediate airway smooth muscle migration in asthma. Am J Respir Crit Care Med. 2006;174:1179–1188. doi: 10.1164/rccm.200603-394OC. [DOI] [PubMed] [Google Scholar]
- Kay AB.Pathology of mild, severe, and fatal asthma Am J Respir Crit Care Med 1996154S66–S69.(August) [DOI] [PubMed] [Google Scholar]
- Kim C, Rott L, Kunkel EJ, Genovese MC, Andrew DP, Wu L, et al. Rules of chemokine receptor association with T cell polarization in vivo. J Clin Invest. 2001;108:1331–1339. doi: 10.1172/JCI13543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim CH, Johnston B, Butcher EC. Trafficking machinery of NKT cells: shared and differential chemokine receptor expression among V alpha 24(+)V beta 11(+) NKT cell subsets with distinct cytokine-producing capacity. Blood. 2002;100:11–16. doi: 10.1182/blood-2001-12-0196. [DOI] [PubMed] [Google Scholar]
- Kunkel EJ, Campbell JJ, Haraldsen G, Pan J, Boisvert J, Roberts AI, et al. Lymphocyte CC chemokine receptor 9 and epithelial thymus-expressed chemokine (TECK) expression distinguish the small intestinal immune compartment: epithelial expression of tissue-specific chemokines as an organizing principle in regional immunity. J Exp Med. 2000;192:761–768. doi: 10.1084/jem.192.5.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisbonne M, Diem S, de Castro Keller A, Lefort J, Araujo LM, Hachem P, et al. Cutting edge: invariant V alpha 14 NKT cells are required for allergen-induced airway inflammation and hyperreactivity in an experimental asthma model. J Immunol. 2003;171:1637–1641. doi: 10.4049/jimmunol.171.4.1637. [DOI] [PubMed] [Google Scholar]
- Liu LY, Jarjour NN, Busse WW, Kelly EA. Chemokine receptor expression on human eosinophils from peripheral blood and bronchoalveolar lavage fluid after segmental antigen challenge. J Allergy Clin Immunol. 2003;112:556–562. doi: 10.1016/s0091-6749(03)01798-6. [DOI] [PubMed] [Google Scholar]
- Loetscher P, Clark-Lewis I. Agonistic and antagonistic activities of chemokines. J Leuk Biol. 2001;69:881–884. [PubMed] [Google Scholar]
- Lukacs NW. Role of chemokines in the pathogenesis of asthma. Nat Rev Immunol. 2001;1:108–116. doi: 10.1038/35100503. [DOI] [PubMed] [Google Scholar]
- Ma W, Bryce PJ, Humbles AA, Laouini D, Yalcindag A, Alenius H, et al. CCR3 is essential for skin eosinophilia and airway hyperresponsiveness in a murine model of allergic skin inflammation. J Clin Invest. 2002;109:621–628. doi: 10.1172/JCI14097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macklem PT. A theoretical analysis of the effect of airway smooth muscle load on airway narrowing. Am J Respir Crit Care Med. 1996;153:83–89. doi: 10.1164/ajrccm.153.1.8542167. [DOI] [PubMed] [Google Scholar]
- Martin LB, Kita H, Leiferman KM, Gleich GJ. Eosinophils in allergy: role in disease, degranulation, and cytokines. Int Arch Allergy Immunol. 1996;109:207–215. doi: 10.1159/000237239. [DOI] [PubMed] [Google Scholar]
- Matloubian M, David A, Engel S, Ryan JE, Cyster JG. A transmembrane CXC chemokine is a ligand for HIV-coreceptor Bonzo. Nat Immunol. 2000;1:298–304. doi: 10.1038/79738. [DOI] [PubMed] [Google Scholar]
- Mellado M, Vila-Coro AJ, Martinez C, Rodriguez-Frade JM. Receptor dimerization: a key step in chemokine signaling. Cell Mol Biol (Noisy-le-grand) 2001;47:575–582. [PubMed] [Google Scholar]
- Mishra A, Hogan SP, Lee JJ, Foster PS, Rothenberg ME. Fundamental signals that regulate eosinophil homing to the gastrointestinal tract. J Clin Invest. 1999;103:1719–1727. doi: 10.1172/JCI6560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morales J, Homey B, Vicari AP, Hudak S, Oldham E, Hedrick J, et al. CTACK, a skin-associated chemokine that preferentially attracts skin-homing memory T cells. Proc Natl Acad Sci USA. 1999;96:14470–14475. doi: 10.1073/pnas.96.25.14470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan AJ, Guillen C, Symon FA, Huynh TT, Berry MA, Entwisle JJ, et al. Expression of CXCR6 and its ligand CXCL16 in the lung in health and disease. Clin Exp Allergy. 2005a;35:1572–1580. doi: 10.1111/j.1365-2222.2005.02383.x. [DOI] [PubMed] [Google Scholar]
- Morgan AJ, Symon FA, Berry MA, Pavord ID, Corrigan CJ, Wardlaw AJ. IL-4-expressing bronchoalveolar T cells from asthmatic and healthy subjects preferentially express CCR 3 and CCR 4. J Allergy Clin Immunol. 2005b;116:594–600. doi: 10.1016/j.jaci.2005.03.052. [DOI] [PubMed] [Google Scholar]
- Moriguchi M, Hissong BD, Gadina M, Yamaoka K, Tiffany HL, Murphy PM, et al. CXCL12 signaling is independent of Jak2 and Jak3. J Biol Chem. 2005;280:17408–17414. doi: 10.1074/jbc.M414219200. [DOI] [PubMed] [Google Scholar]
- Motsinger A, Haas DW, Stanic AK, Van Kaer L, Joyce S, Unutmaz D. CD1d-restricted human natural killer T cells are highly susceptible to human immunodeficiency virus 1 infection. J Exp Med. 2002;195:869–879. doi: 10.1084/jem.20011712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy PM. The molecular biology of leukocyte chemoattractant receptors. Annu Rev Immunol. 1994;12:593–633. doi: 10.1146/annurev.iy.12.040194.003113. [DOI] [PubMed] [Google Scholar]
- O'Byrne PM. Cytokines or their antagonists for the treatment of asthma. Chest. 2006;130:244–250. doi: 10.1378/chest.130.1.244. [DOI] [PubMed] [Google Scholar]
- Ohno I, Nitta Y, Yamauchi K, Hoshi H, Honma M, Woolley K, et al. Transforming growth factor beta 1 (TGF beta 1) gene expression by eosinophils in asthmatic airway inflammation. Am J Respir Cell Mol Biol. 1996;15:404–409. doi: 10.1165/ajrcmb.15.3.8810646. [DOI] [PubMed] [Google Scholar]
- Ordonez CL, Shaughnessy TE, Matthay MA, Fahy JV. Increased neutrophil numbers and IL-8 levels in airway secretions in acute severe asthma: clinical and biologic significance. Am J Respir Crit Care Med. 2000;161 4 Part 1:1185–1190. doi: 10.1164/ajrccm.161.4.9812061. [DOI] [PubMed] [Google Scholar]
- Panina-Bordignon P, Papi A, Mariani M, Di Lucia P, Casoni G, Bellettato C, et al. The C-C chemokine receptors CCR4 and CCR8 identify airway T cells of allergen-challenged atopic asthmatics. J Clin Invest. 2001;107:1357–1364. doi: 10.1172/JCI12655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavord ID, Brightling CE, Woltmann G, Wardlaw AJ. Non-eosinophilic corticosteroid unresponsive asthma. Lancet. 1999;353:2213–2214. doi: 10.1016/S0140-6736(99)01813-9. [DOI] [PubMed] [Google Scholar]
- Pham-Thi N, de Blic J, Leite-de-Moraes MC. Invariant natural killer T cells in bronchial asthma. N Engl J Med. 2006;354:2613n–2616n. [PubMed] [Google Scholar]
- Phillips RJ, Burdick MD, Hong K, Lutz MA, Murray LA, Xue YY, et al. Circulating fibrocytes traffic to the lungs in response to CXCL12 and mediate fibrosis. J Clin Invest. 2004;114:438–446. doi: 10.1172/JCI20997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phillips RM, Stubbs VE, Henson MR, Williams TJ, Pease JE, Sabroe I. Variations in eosinophil chemokine responses: an investigation of CCR1 and CCR3 function, expression in atopy, and identification of a functional CCR1 promoter. J Immunol. 2003;170:6190–6201. doi: 10.4049/jimmunol.170.12.6190. [DOI] [PubMed] [Google Scholar]
- Pignatti P, Moscato G, Casarini S, Delmastro M, Poppa M, Brunetti G, et al. Downmodulation of CXCL8/IL-8 receptors on neutrophils after recruitment in the airways. J Allergy Clin Immunol. 2005;115:88–94. doi: 10.1016/j.jaci.2004.08.048. [DOI] [PubMed] [Google Scholar]
- Piliponsky AM, Pickholtz D, Gleich GJ, Levi-Schaffer F. Human eosinophils induce histamine release from antigen-activated rat peritoneal mast cells: a possible role for mast cells in late-phase allergic reactions. J Allergy Clin Immunol. 2001;107:993–1000. doi: 10.1067/mai.2001.114656. [DOI] [PubMed] [Google Scholar]
- Podolin PL, Bolognese BJ, Foley JJ, Schmidt DB, Buckley PT, Widdowson KL, et al. A potent and selective nonpeptide antagonist of CXCR2 inhibits acute and chronic models of arthritis in the rabbit. J Immunol. 2002;169:6435–6444. doi: 10.4049/jimmunol.169.11.6435. [DOI] [PubMed] [Google Scholar]
- Ponath PD, Qin S, Post TW, Wang J, Wu L, Gerard NP, et al. Molecular cloning and characterization of a human eotaxin receptor expressed selectively on eosinophils. J Exp Med. 1996;183:2437–2448. doi: 10.1084/jem.183.6.2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson DS, Hamid Q, Ying S, Tsicopoulos A, Barkans J, Bentley AM, et al. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med. 1992;326:298–304. doi: 10.1056/NEJM199201303260504. [DOI] [PubMed] [Google Scholar]
- Roche WR, Beasley R, Williams JH, Holgate ST. Subepithelial fibrosis in the bronchi of asthmatics. Lancet. 1989;1:520–524. doi: 10.1016/s0140-6736(89)90067-6. [DOI] [PubMed] [Google Scholar]
- Romagnani P, de Paulis A, Beltrame C, Annunziato F, Dente V, Maggi E, et al. Tryptase–chymase double-positive human mast cells express the eotaxin receptor CCR3 and are attracted by CCR3-binding chemokines. Am J Pathol. 1999;155:1195–1204. doi: 10.1016/S0002-9440(10)65222-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rot A, von Andrian UH. Chemokines in innate and adaptive host defense: basic chemokinese grammar for immune cells. Annu Rev Immunol. 2004;22:891–928. doi: 10.1146/annurev.immunol.22.012703.104543. [DOI] [PubMed] [Google Scholar]
- Rothenberg ME. Eosinophilia. N Engl J Med. 1998;338:1592–1600. doi: 10.1056/NEJM199805283382206. [DOI] [PubMed] [Google Scholar]
- Sabroe I, Hartnell A, Jopling LA, Bel S, Ponath PD, Pease JE, et al. Differential regulation of eosinophil chemokine signaling via CCR3 and non-CCR3 pathways. J Immunol. 1999;162:2946–2955. [PubMed] [Google Scholar]
- Sabroe I, Williams TJ, Hebert CA, Collins PD. Chemoattractant cross-desensitization of the human neutrophil IL-8 receptor involves receptor internalization and differential receptor subtype regulation. J Immunol. 1997;158:1361–1369. [PubMed] [Google Scholar]
- Sanderson CJ. Interleukin-5, eosinophils, and disease. Blood. 1992;79:3101–3109. [PubMed] [Google Scholar]
- Schleimer RP, Sterbinsky SA, Kaiser J, Bickel CA, Klunk DA, Tomioka K, et al. IL-4 induces adherence of human eosinophils and basophils but not neutrophils to endothelium. Association with expression of VCAM-1. J Immunol. 1992;148:1086–1092. [PubMed] [Google Scholar]
- Schmidt M, Sun G, Stacey MA, Mori L, Mattoli S. Identification of circulating fibrocytes as precursors of bronchial myofibroblasts in asthma. J Immunol. 2003;171:380–389. doi: 10.4049/jimmunol.171.1.380. [DOI] [PubMed] [Google Scholar]
- Schuh JM, Power CA, Proudfoot AE, Kunkel SL, Lukacs NW, Hogaboam CM. Airway hyperresponsiveness, but not airway remodeling, is attenuated during chronic pulmonary allergic responses to Aspergillus in CCR4−/− mice. FASEB J. 2002;16:1313–1315. doi: 10.1096/fj.02-0193fje. [DOI] [PubMed] [Google Scholar]
- Sen Y, Yongyi B, Yuling H, Luokun X, Li H, Jie X, et al. V alpha 24-invariant NKT cells from patients with allergic asthma express CCR9 at high frequency and induce Th2 bias of CD3+ T cells upon CD226 engagement. J Immunol. 2005;175:4914–4926. doi: 10.4049/jimmunol.175.8.4914. [DOI] [PubMed] [Google Scholar]
- Sher A, Coffman RL, Hieny S, Cheever AW. Ablation of eosinophil and IgE responses with anti-IL-5 or anti-IL-4 antibodies fails to affect immunity against Schistosoma mansoni in the mouse. J Immunol. 1990;145:3911–3916. [PubMed] [Google Scholar]
- Smiley ST, King JA, Hancock WW. Fibrinogen stimulates macrophage chemokine secretion through toll-like receptor 4. J Immunol. 2001;167:2887–2894. doi: 10.4049/jimmunol.167.5.2887. [DOI] [PubMed] [Google Scholar]
- Springael JY, Urizar E, Parmentier M. Dimerization of chemokine receptors and its functional consequences. Cytokine Growth Factor Rev. 2005;16:611–623. doi: 10.1016/j.cytogfr.2005.05.005. [DOI] [PubMed] [Google Scholar]
- Sullivan SK, McGrath DA, Liao F, Boehme SA, Farber JM, Bacon KB. MIP-3alpha induces human eosinophil migration and activation of the mitogen-activated protein kinases (p42/p44 MAPK) J Leukoc Biol. 1999;66:674–682. doi: 10.1002/jlb.66.4.674. [DOI] [PubMed] [Google Scholar]
- Sun YC, Zhou QT, Yao WZ. Sputum interleukin-17 is increased and associated with airway neutrophilia in patients with severe asthma. Chin Med J (England) 2005;118:953–956. [PubMed] [Google Scholar]
- Sur S, Crotty TB, Kephart GM, Hyma BA, Colby TV, Reed CE, et al. Sudden-onset fatal asthma. A distinct entity with few eosinophils and relatively more neutrophils in the airway submucosa. Am Rev Respir Dis. 1993;148:713–719. doi: 10.1164/ajrccm/148.3.713. [DOI] [PubMed] [Google Scholar]
- Sutcliffe A, Kaur D, Page S, Woodman L, Armour CL, Baraket M, et al. Mast cell migration to Th2 stimulated airway smooth muscle from asthmatics. Thorax. 2006;61:657–662. doi: 10.1136/thx.2005.056770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas SY, Lilly CM, Luster AD. Invariant natural killer T cells in bronchial asthma. N Engl J Med. 2006;354:2613–2616. doi: 10.1056/NEJMc066189. [DOI] [PubMed] [Google Scholar]
- Wardlaw AJ. Molecular basis for selective eosinophil trafficking in asthma: a multistep paradigm. J Allergy Clin Immunol. 1999;104:917–926. doi: 10.1016/s0091-6749(99)70069-2. [DOI] [PubMed] [Google Scholar]
- Wardlaw AJ. Eosinophil trafficking in asthma. Clin Med. 2001;1:214–218. doi: 10.7861/clinmedicine.1-3-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wardlaw AJ, Dunnette S, Gleich GJ, Collins JV, Kay AB. Eosinophils and mast cells in bronchoalveolar lavage in subjects with mild asthma. Relationship to bronchial hyperreactivity. Am Rev Respir Dis. 1988;137:62–69. doi: 10.1164/ajrccm/137.1.62. [DOI] [PubMed] [Google Scholar]
- Wardlaw AJ, Guillen C, Morgan A. Mechanisms of T cell migration to the lung. Clin Exp Allergy. 2005;35:4–7. doi: 10.1111/j.1365-2222.2005.02139.x. [DOI] [PubMed] [Google Scholar]
- Weninger W, von Andrian UH. Chemokine regulation of naive T cell traffic in health and disease. Semin Immunol. 2003;15:257–270. doi: 10.1016/j.smim.2003.08.007. [DOI] [PubMed] [Google Scholar]
- Wenzel S. Severe asthma in adults. Am J Respir Crit Care Med. 2005;172:149–160. doi: 10.1164/rccm.200409-1181PP. [DOI] [PubMed] [Google Scholar]
- Wenzel SE, Fowler AA, Schwartz LB. Activation of pulmonary mast cells by bronchoalveolar allergen challenge. In vivo release of histamine and tryptase in atopic subjects with and without asthma. Am Rev Respir Dis. 1988;137:1002–1008. doi: 10.1164/ajrccm/137.5.1002. [DOI] [PubMed] [Google Scholar]
- Wenzel SE, Schwartz LB, Langmack EL, Halliday JL, Trudeau JB, Gibbs RL, et al. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am J Respir Crit Care Med. 1999;160:1001–1008. doi: 10.1164/ajrccm.160.3.9812110. [DOI] [PubMed] [Google Scholar]
- White JR, Lee JM, Young PR, Hertzberg RP, Jurewicz AJ, Chaikin MA, et al. Identification of a potent, selective non-peptide CXCR2 antagonist that inhibits interleukin-8-induced neutrophil migration. J Biol Chem. 1998;273:10095–10098. doi: 10.1074/jbc.273.17.10095. [DOI] [PubMed] [Google Scholar]
- Woodruff PG, Dolganov GM, Ferrando RE, Donnelly S, Hays SR, Solberg OD, et al. Hyperplasia of smooth muscle in mild to moderate asthma without changes in cell size or gene expression. Am J Respir Crit Care Med. 2004;169:1001–1006. doi: 10.1164/rccm.200311-1529OC. [DOI] [PubMed] [Google Scholar]
- Yamashita N, Tashimo H, Matsuo Y, Ishida H, Yoshiura K, Sato K, et al. Role of CCL21 and CCL19 in allergic inflammation in the ovalbumin-specific murine asthmatic model. J Allergy Clin Immunol. 2006;117:1040–1046. doi: 10.1016/j.jaci.2006.01.009. [DOI] [PubMed] [Google Scholar]
- Ying S, Humbert M, Barkans J, Corrigan CJ, Pfister R, Menz G, et al. Expression of IL-4 and IL-5 mRNA and protein product by CD4+ and CD8+ T cells, eosinophils, and mast cells in bronchial biopsies obtained from atopic and nonatopic (intrinsic) asthmatics. J Immunol. 1997a;158:3539–3544. [PubMed] [Google Scholar]
- Ying S, Meng Q, Barata LT, Robinson DS, Durham SR, Kay AB. Associations between IL-13 and IL-4 (mRNA and protein), vascular cell adhesion molecule-1 expression, and the infiltration of eosinophils, macrophages, and T cells in allergen-induced late-phase cutaneous reactions in atopic subjects. J Immunol. 1997b;158:5050–5057. [PubMed] [Google Scholar]
- Ying S, Robinson DS, Meng Q, Rottman J, Kennedy R, Ringler DJ, et al. Enhanced expression of eotaxin and CCR3 mRNA and protein in atopic asthma. Association with airway hyperresponsiveness and predominant co-localization of eotaxin mRNA to bronchial epithelial and endothelial cells. Eur J Immunol. 1997c;27:3507–3516. doi: 10.1002/eji.1830271252. [DOI] [PubMed] [Google Scholar]
- Yuan Q, Campanella GS, Colvin RA, Hamilos DL, Jones KJ, Mathew A, et al. Membrane-bound eotaxin-3 mediates eosinophil transepithelial migration in IL-4-stimulated epithelial cells. Eur J Immunol. 2006;36:2700–2714. doi: 10.1002/eji.200636112. [DOI] [PubMed] [Google Scholar]
- Zimmermann N, Hershey GK, Foster PS, Rothenberg ME.Chemokines in asthma: cooperative interaction between chemokines and IL-13 J Allergy Clin Immunol 2003111227–242.quiz 243 [DOI] [PubMed] [Google Scholar]
- Zlotnik A, Yoshie O. Chemokines: a new classification system and their role in immunity. Immunity. 2000;12:121–127. doi: 10.1016/s1074-7613(00)80165-x. [DOI] [PubMed] [Google Scholar]