Abstract
Venous hypertension from failure of proper venous valve function in the veins of thelower extremities causes changes over time in the microcirculation of the skin of the distal extremity. These changes set the stage for the development of a chronic nonhealing ulceration, which typically occurs at the ankle. The mainstay of treatment has been conservative, with compression dressings and elevation of the extremity. However, results have been less than satisfactory because of delay in healing and high recurrence rates after successful healing. Elimination of the venous hypertension should be the goal of therapy using more recent minimally invasive surgical techniques, including ablation of incompetent truncal veins with laser or radiofrequency energy and use of ultrasound-guided foam sclerosant injections to close incompetent perforator veins that are frequently found in or near the ulcer bed. This approach will shorten ulcer healing time and reduce recurrence rates as well as patient suffering and expense.
Chronic venous insufficiency is the cause of ulcerations in 60% to 80% of patients with chronic leg ulcers. The prevalence of venous leg ulcers in the population over age 65 in the United Kingdom has been estimated to be 1% to 2% (1, 2). Probably more than 500,000 people in the USA suffer from these painful and debilitating ulcerations. While it is true that the peak prevalence occurs after age 60, many patients develop ulcers early in adulthood (3). The annual cost associated with the care of these ulcerations has been estimated to be £400 million in the United Kingdom and $1 billion in the USA (4–7). The annual cost of medical care for one of these difficult-to-heal lesions often runs into the thousands of dollars.
THE CAUSE OF CHRONIC VENOUS ULCERS
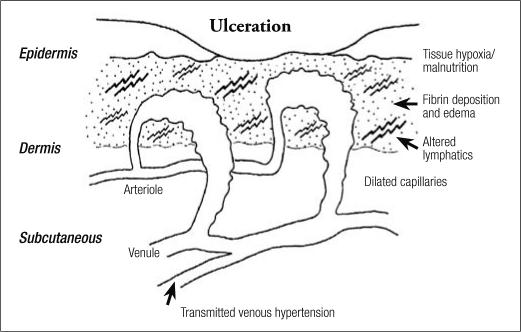
Elevated venous pressure is the underlying cause of the skin and tissue changes that lead to chronic ulceration (Figure 1). This elevated venous pressure is the result of failure of the venous valves in the deep or superficial veins or both. Two forces are at work in this venous hypertension: 1) the weight of the column of blood from the right atrium to the valveless inferior vena cava and from the iliac veins to the femoral vein and 2) the relatively high pressure generated by the contracting leg muscles that is transmitted through incompetent perforator veins of the leg. The elevated venous pressure, which is transmitted to the venules and capillaries of the skin and subcutaneous tissues, causes a series of clinical changes, including edema, lipodermatosclerosis or stasis dermatitis, and development of hyperpigmentation, hyperkeratosis, and atrophie blanche and ultimately a chronic nonhealing ulcer (9).
Figure 1.
The pathophysiology of venous ulcers. Reprinted from Venous Ulcers (8) with permission from Elsevier.
On a microscopic level, lymphatic vessels are enlarged (10) and capillaries are elongated and dilated (11), with the functional number of capillaries being reduced (10, 11). Capillaries often show occlusive microthrombi (12) and sludging of white blood cells (13). Plasma proteins and red blood cells leak into the interstitium, where hemosiderin and fibrin deposition occurs as well as fibrinoblastic activity (12). These microscopic changes result in the reduction and stagnation of blood flow and decreased oxygen levels at the capillary level in the preulcer skin, and increased flow and arteriovenous shunting in nearby tissues (11, 14). The protein-rich edema, acting as a diffusion barrier to oxygen transport, probably plays a major role in tissue hypoxia (14).
Some investigators have advanced the “cutaneous leukocyte trapping hypothesis” (15), which invokes white cell activation resulting in tissue damage from release of proteolytic enzymes and free radicals (16, 17). Others doubt the causative nature of these inflammatory responses and believe that the hemodynamic changes at the microvascular level are sufficient to explain the development of venous ulcerations (18). The end result is an area of skin in which capillaries are either missing or severely damaged and altered. In this setting, slight trauma or infection can lead to a nonhealing ulcer.
It is important to stress that other causes of leg ulceration must be considered before treatment is initiated. Arterial insufficiency must be ruled out before treatment, as compression bandaging is contraindicated in such cases. Diabetic foot ulcers, which are usually the result of neuropathy, may be combined with arterial insufficiency from distal small vessel arteriosclerotic occlusions. Decubitus ulcerations, infectious causes, and vasculitis should also be considered. Rare causes of leg ulcerations include pyoderma gangrenosum and primary and metastatic malignancies.
Venous ulcers are found in patients with superficial, deep, or perforator incompetence or a combination of two or all three. Duplex scanning has documented the importance of superficial venous reflux and the relatively infrequent finding of deep reflux in patients with venous ulcers (19–21). Labropoulos et al (22) studied 112 limbs with ulcers and distributed venous incompetence in superficial, deep, and perforator systems using color duplex imaging. They found that 64% of limbs had more than one system involved, 32% had only one system showing reflux, 6% had isolated deep system reflux, and 23% had isolated superficial vein incompetence. Deep system reflux was noted in 50% of patients with ulcers. Others have found superficial TenBrook et al venous reflux in 17% to 53% of patients with leg ulcers (19–21, 23, 24). Most deep venous reflux in limbs disappears when the superficial reflux is eliminated (25).
The importance of incompetent perforators has been em by several investigators including Labropoulos (26), who found them present in 48.8% of patients with skin changes and in 60.8% of patients with ulceration. Others (27, 28) have documented a similar distribution. The presence of incompetent calf perforators occurring with either superficial or deep system reflux has been reported in 73% of limbs with venous ulceration (29). The perforating veins are usually located near the ulcerations and transmit elevated venous pressure directly to the small vessels of the skin and subcutaneous tissues (11, 18).
TREATMENT
Hippocrates believed that venous ulcers were important (36) as sites for release of “evil humours” from the body, and as such they should not be allowed to heal. If an ulcer healed, he advocated reopening the wound (30). In the 14th century, bandaging and compression were found to relieve much of the discomfort of ulceration and to promote healing (31). Conservative treatment has come to include elevation, local treatment, and compression therapy. Until recently this was considered the mainstay of treatment, and its success in achieving ulcer healing has been documented (32). Ulcer recurrence has been a nagging problem when only conservative treatment has been employed (33, 34). Slow healing and the significant recurrence rate of ulcers given conservative treatment reflect the failure to treat the underlying causative venous hypertension.
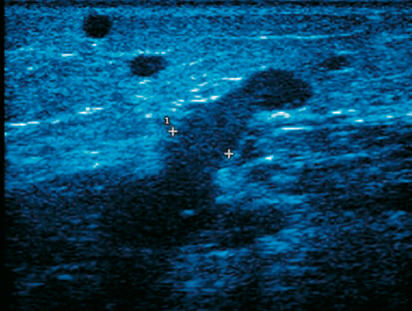
The diagnosis and surgical treatment of superficial venous reflux have been advanced by the availability of duplex ultrasound scanning, now the gold standard for accurate evaluation of the highest point of valve failure and extent of reflux (Figure 2).. Ultrasound has also become invaluable as a tool in the newer minimally invasive procedures for ablation of refluxing superficial truncal veins of the lower extremity. Using ultrasound, surgeons can delineate and treat all of the sources of high venous pressure. Complete eradication of all sources of venous hypertension is further facilitated by endovenous ablation using either radiofrequency or laser energy and by ultrasound-guided foam chemical ablation either as sole therapy or in combination. Armed with ultrasound technology and what are probably the most effective methods of eradicating the venous hypertension, vein surgeons are capable of optimal diagnosis and surgical treatment of venous reflux. Thus it appears reasonable to compare the results of modern surgical treatments with conservative noninterventional therapy. Comparison of ulcer healing rates, length of time to complete healing, interval of time to ulcer recurrence, and ulcer recurrence rates are indicated.
Figure 2.
An ultrasound of an incompetent perforator vein.
TenBrook et al (35) reviewed 20 studies reported between 1991 and 2003, including 1 randomized trial and 19 case series, in all involving 1140 treated limbs, 526 with stasis ulcers. All had perforator vein interruption using subfascial endoscopic perforator surgery (SEPS) with or without superficial vein ligation or stripping procedures. All patients also had conservative management, including compression and wound care. Ulcers healed in 88% of limbs treated; 40% were healed by 30 days and 64% by 60 days. The ulcer recurrence rate was 13% with a mean time to recurrence of 21 months in the 16 studies, with a mean follow-up of 29 months. Analysis of the data in the studies that looked at the presence or absence of residual perforator veins revealed that postoperative incompetent perforators were a significant risk factor for ulcer nonhealing and recurrence. The presence of deep venous system incompetence did not appear to be a significant risk factor for recurrence of the ulceration. Van Gent (36), in addition to finding that surgical treatment improved the results over conservative treatment, found that surgical treatment of incompetent superficial and perforator veins increased the rate of ulcer healing and decreased recurrences. Others have emphasized the same findings and have recommended surgical treatment of patients with venous ulcers (37, 38).
Obermayer (39) reported a series of surgically treated patients in which healing occurred in 87% and ulcer recurrence was 5% at 5 years. Patients with truncal vein incompetence received vein stripping and ligation, and incompetent perforator veins were abolished by open ligation or SEPS. He stressed that ulcer patients should have venous surgery regardless of the ulcer's duration even if conservative treatment has continued for years. Cabrera et al (40) used ultrasound-guided foam sclerosant injections of refluxing superficial and perforator veins to treat 151 venous ulcers previously treated with conservative therapy and achieved complete healing in 86% of patients within 6 months. Others have reported limited success in closing incompetent truncal and perforator veins of ulcer patients with ultrasound-guided foam sclerosant injections (41).
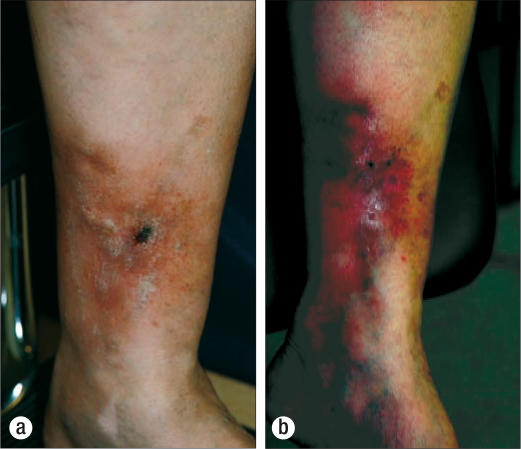
In the last year, my associate and I at Southwest Vein Institute have treated eight chronic venous stasis ulcers. In all cases, minimally invasive endovenous laser ablation of truncal refluxing veins or ultrasound-guided foam sclerosant injections of refluxing veins and perforator veins or a combination of both modalities have been used. To date, six patients have demonstrated ulcer healing 26 days to 3 months after treatment (Figure 3). One patient's ulcer failed to heal and was shown on biopsy to have basal cell carcinoma in the ulcer bed (Figure 4). He underwent excision and skin grafting. One patient developed a recurrence and had repeat ultrasound-guided sclerotherapy with subsequent healing.
Figure 3.
(a) Chronic venous stasis ulcer treated by endovascular laser ablation of an incompetent great saphenous vein and ultrasound-guided injections of foam sclerosant into veins joining incompetent perforator veins in close proximity to the ulceration. (b) The same leg 26 days after treatment, showing healing of the ulceration.
Figure 4.
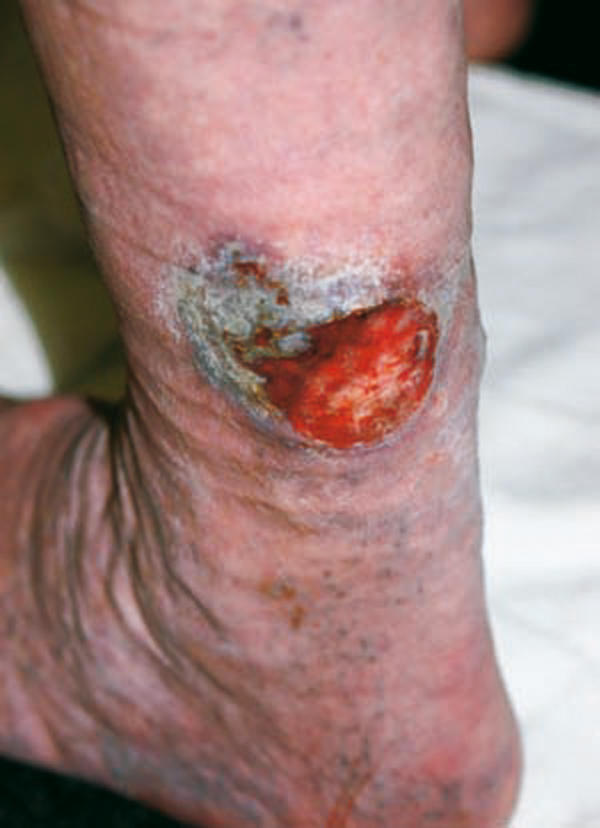
The ulceration failed to heal after endovascular laser ablation of incompetent great and small saphenous veins and ultra-sound-guided injections of foam sclerosant solutions into incompetent veins and perforator veins in the vicinity of the ulceration. A biopsy showed basal cell carcinoma.
DISCUSSION
Now surgeons are armed with the necessary tools and treatment techniques to predictably heal chronic venous ulcers in a relatively rapid manner and with the expectation of a low rate of recurrence. The minimally invasive ablation treatments easily performed under local anesthesia using radiofrequency or laser energy are successful at stopping superficial reflux of incompetent truncal veins. The recovery is rapid, with most patients returning to work and usual activities in 1 to 2 days.
If by duplex ultrasound scanning the direction of venous blood flow in perforator veins with leg muscle contraction is from deep to superficial and if one or more perforator veins are found in or near the area of advanced skin changes or ulceration, prudence would dictate that all incompetent perforator veins be interrupted or ablated. The ulcer recurrence rate in some series is probably high because of missed and untreated incompetent perforators. The early enthusiasm for treatment of incompetent perforators with SEPS has been tempered by failures that have provoked reexamination of the procedure and its deficiencies. Perforators located in the retromalleolar and the inframalleolar spaces are inaccessible to the SEPS approach. Stacey et al reported that 50% of incompetent perforators are within 10 cm of the sole of the foot and are missed at SEPS even when identified preoperatively by ultrasound (42).
Based on the success of foam sclerosant delivered by ultrasound-guided injection directly into the superficial veins overlying the perforators, this approach should be included in the treatment of any incompetent perforator vein found with careful routine ultrasound study of every ulcer patient. The use of foam sclerosant appears to be safe and effective and can easily be done in the office setting, whereas SEPS requires general or spinal anesthesia and surgical wounds that impair patients’ rapid return to usual activities. Additionally, the location of some perforators is inaccessible with SEPS, while ultrasound-guided injections can reach any location on the lower extremity. Careful attention to the detection and treatment of perforator veins near venous ulcers achieves excellent ulcer healing rates and shorter healing time with lower recurrence rates.
CONCLUSION
No good study has yet been published using the minimally invasive ablation techniques and ultrasound-guided sclerosant foam injections in the treatment of chronic venous ulcers. Neither is there one comparing these treatment methods and so-called conservative therapy. Nevertheless, evidence that surgical intervention increases the rate of ulcer healing and decreases the rate of recurrence and the excellent experience with the minimally invasive ablation techniques and ultrasound-guided foam sclerosant injections make a strong case for the early referral of venous ulcer patients for such treatment. Our limited experience with a small number of patients supports this approach. Early surgical treatment will likely decrease patient suffering and significantly decrease the length and overall cost of the illness.
References
- 1.Rejan TJ. The epidemiology of leg ulcers. In: Westerhof W, editor. Leg Ulcers: Diagnosis and Treatment. Amsterdam: Elsevier Science Publishers BV; 1993. pp. 19–27. [Google Scholar]
- 2.Baker SR, Stacey MC, Jopp-McKay AG, Hoskin SE, Thompson PJ. Epidemiology of chronic venous ulcers. Br J Surg. 1991;78(7):864–867. doi: 10.1002/bjs.1800780729. [DOI] [PubMed] [Google Scholar]
- 3.Callam MJ, Harper DR, Dale JJ, Ruckley CV. Chronic ulcer of the leg: clinical history. Br Med J (Clin Res Ed) 1987;294(6584):1389–1391. doi: 10.1136/bmj.294.6584.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Margolis DJ, Bilker W, Santanna J, Baumgarten M. Venous leg ulcer: incidence and prevalence in the elderly. J Am Acad Dermatol. 2002;46(3):381–386. doi: 10.1067/mjd.2002.121739. [DOI] [PubMed] [Google Scholar]
- 5.Phillips TJ. Chronic cutaneous ulcers: etiology and epidemiology. J Invest Dermatol. 1994;102(6):38S–41S. doi: 10.1111/1523-1747.ep12388556. [DOI] [PubMed] [Google Scholar]
- 6.Miller OF, 3rd, Phillips TJ. Leg ulcers. J Am Acad Dermatol. 2000;43(1 Pt 1):91–95. doi: 10.1067/mjd.2000.105505. [DOI] [PubMed] [Google Scholar]
- 7.Ruckley CV. Socioeconomic impact of chronic venous insufficiency and leg ulcers. Angiology. 1997;48(1):67–69. doi: 10.1177/000331979704800111. [DOI] [PubMed] [Google Scholar]
- 8.Mayberry JC, Moneta GL, Taylor LM, Jr, Porter JM. Nonoperative treatment of venous statis ulcers. In: Bergan JJ, Yao JS, editors. Venous Disorders. Philadelphia: Saunders; 1991. p. 382. [Google Scholar]
- 9.Valencia IC, Falabella A, Kirsner RS, Eaglstein WH. Chronic venous insufficiency and venous leg ulceration. J Am Acad Dermatol. 2001;44(3):401–421. doi: 10.1067/mjd.2001.111633. [DOI] [PubMed] [Google Scholar]
- 10.Partsch H. Investigations on the pathogenesis of venous leg ulcers. Acta Chir Scand Suppl. 1988;544:25–29. [PubMed] [Google Scholar]
- 11.Junger M, Steins A, Hahn M, Hafner HM. Microcirculatory dysfunction in chronic venous insufficiency (CVI) Microcirculation. 2000;7(6 Pt 2):S3–S12. [PubMed] [Google Scholar]
- 12.Leu HJ. Morphology of chronic venous insufficiency—light and electron microscopic examinations. Vasa. 1991;20(4):330–342. [PubMed] [Google Scholar]
- 13.Thomas PR, Nash GB, Dormandy JA. White cell accumulation in dependent legs of patients with venous hypertension: a possible mechanism for trophic changes in the skin. Br Med J (Clin Res Ed) 1988;296(6638):1693–1695. doi: 10.1136/bmj.296.6638.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mani R, White JE, Barrett DF, Weaver PW. Tissue oxygenation, venous ulcers and fibrin cuffs. J R Soc Med. 1989;82(6):345–346. doi: 10.1177/014107688908200610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Coleridge Smith P, Scurr J. Current views on the pathogenesis of venous ulceration. In: Bergan JJ, Yao JS, editors. Venous Disorders. Philadelphia: Saunders; 1991. pp. 46–51. [Google Scholar]
- 16.Wilkinson LS, Bunker C, Edwards JC, Scurr JH, Smith PD. Leukocytes: their role in the etiopathogenesis of skin damage in venous disease. J Vasc Surg. 1993;17(4):669–775. [PubMed] [Google Scholar]
- 17.Pappas PJ, Fallek SR, Garcia A, Araki CT, Back TL, Duran WN, Hobson RW., 2nd Role of leukocyte activation in patients with venous stasis ulcers. J Surg Res. 1995;59(5):553–559. doi: 10.1006/jsre.1995.1205. [DOI] [PubMed] [Google Scholar]
- 18.Mekkes JR, Loots MA, Van Der Wal AC, Bos JD. Causes, investigation and treatment of leg ulceration. Br J Dermatol. 2003;148(3):388–401. doi: 10.1046/j.1365-2133.2003.05222.x. [DOI] [PubMed] [Google Scholar]
- 19.Shami SK, Sarin S, Cheatle TR, Scurr JH, Smith PD. Venous ulcers and the superficial venous system. J Vasc Surg. 1993;17(3):487–490. [PubMed] [Google Scholar]
- 20.Hanrahan LM, Araki CT, Rodriguez AA, Kechejian GJ, LaMorte WW, Menzoian JO. Distribution of valvular incompetence in patients with venous stasis ulceration. J Vasc Surg. 1991;13(6):805–811. [PubMed] [Google Scholar]
- 21.van Rij AM, Solomon C, Christie R. Anatomic and physiologic characteristics of venous ulceration. J Vasc Surg. 1994;20(5):759–764. doi: 10.1016/s0741-5214(94)70163-6. [DOI] [PubMed] [Google Scholar]
- 22.Labropoulos N, Leon M, Geroulakos G, Volteas N, Chan P, Nicolaides AN. Venous hemodynamic abnormalities in patients with leg ulceration. Am J Surg. 1995;169(6):572–574. doi: 10.1016/s0002-9610(99)80223-2. [DOI] [PubMed] [Google Scholar]
- 23.Sethia KK, Darke SG. Long saphenous incompetence as a cause of venous ulceration. Br J Surg. 1984;71(10):754–755. doi: 10.1002/bjs.1800711006. [DOI] [PubMed] [Google Scholar]
- 24.Nelzen O, Bergqvist D, Lindhagen A. Leg ulcer etiology—a cross sectional population study. J Vasc Surg. 1991;14(4):557–564. [PubMed] [Google Scholar]
- 25.Gloviczki P, Bergan JJ, Rhodes JM, Canton LG, Harmsen S, Ilstrup DM, the North American Study Group Mid-term results of endoscopic perforator vein interruption for chronic venous insufficiency: lessons learned from the North American subfascial endoscopic perforator surgery registry. J Vasc Surg. 1999;29(3):489–502. doi: 10.1016/s0741-5214(99)70278-8. [DOI] [PubMed] [Google Scholar]
- 26.Labropoulos N, Delis K, Nicolaides AN, Leon M, Ramaswami G. The role of the distribution and anatomic extent of reflux in the development of signs and symptoms in chronic venous insufficiency. J Vasc Surg. 1996;23(3):504–510. doi: 10.1016/s0741-5214(96)80018-8. [DOI] [PubMed] [Google Scholar]
- 27.Lees TA, Lambert D. Patterns of venous reflux in limbs with skin changes associated with chronic venous insufficiency. Br J Surg. 1993;80(6):725–728. doi: 10.1002/bjs.1800800617. [DOI] [PubMed] [Google Scholar]
- 28.Delis KT, Ibegbuna V, Nicolaides AN, Lauro A, Hafez H. Prevalence and distribution of incompetent perforating veins in chronic venous insufficiency. J Vasc Surg. 1998;28(5):815–825. doi: 10.1016/s0741-5214(98)70056-4. [DOI] [PubMed] [Google Scholar]
- 29.Stuart WP, Adam DJ, Bradbury AW, Ruckley CV. Subfascial endoscopic perforator surgery is associated with significantly less morbidity and shorter hospital stay than open operation (Linton's procedure) Br J Surg. 1997;84(10):1364–1365. doi: 10.1111/j.1365-2168.1997.00600.x. [DOI] [PubMed] [Google Scholar]
- 30.Chadwick J, Mann WN. The Medical Works of Hippocrates. Oxford: Blackwell; 1950. [Google Scholar]
- 31.de Mondeville H. Chirurgie de Maitre Henri Mondeville Composee de 1306 a 1320. Paris NE: trans. Alcan; 1893. [Google Scholar]
- 32.Cullum N, Nelson EA, Fletcher AW, Sheldon TA. Compression for venous leg ulcers. Cochrane Database Syst Rev. 2001;(2):CD000265. doi: 10.1002/14651858.CD000265. [DOI] [PubMed] [Google Scholar]
- 33.Nelzen O. How can we improve outcomes for leg ulcer patients? In: Ruckley CV, Fowkes FGR, Bradburg A, editors. Venous Disease: Epidemiology, Management and Delivery of Care. Berlin: Springer; 1999. pp. 246–253. [Google Scholar]
- 34.Monk BE, Sarkany I. Outcome of treatment of venous stasis ulcers. Clin Exp Dermatol. 1982;7(4):397–400. doi: 10.1111/j.1365-2230.1982.tb02447.x. [DOI] [PubMed] [Google Scholar]
- 35.TenBrook JA, Jr, Iafrati MD, O'donnell TF, Jr, Wolf MP, Hoffman SN, Pauker SG, Lau J, Wong JB. Systematic review of outcomes after surgical management of venous disease incorporating subfascial endoscopic perforator surgery. J Vasc Surg. 2004;39(3):583–589. doi: 10.1016/j.jvs.2003.09.017. [DOI] [PubMed] [Google Scholar]
- 36.van Gent WB, Hop WC, van Praag MC, Mackaay AJ, de Boer EM, Wittens CH. Conservative versus surgical treatment of venous leg ulcers: a prospective, randomized, multicenter trial. J Vasc Surg. 2006;44(3):563–571. doi: 10.1016/j.jvs.2006.04.053. [DOI] [PubMed] [Google Scholar]
- 37.Zamboni P, Cisno C, Marchetti F, Mazza P, Fogato L, Carandina S, De Palma M, Liboni A. Minimally invasive surgical management of primary venous ulcers vs. compression treatment: a randomized clinical trial. Eur J Vasc Endovasc Surg. 2003;25(4):313–318. doi: 10.1053/ejvs.2002.1871. [DOI] [PubMed] [Google Scholar]
- 38.Danielsson G, Arfvidsson B, Eklof B, Kistner RL, Masuda EM, Satoc DT. Reflux from thigh to calf, the major pathology in chronic venous ulcer disease: surgery indicated in the majority of patients. Vasc Endovascular Surg. 2004;38(3):209–219. doi: 10.1177/153857440403800303. [DOI] [PubMed] [Google Scholar]
- 39.Obermayer A, Gostl K, Walli G, Benesch T. Chronic venous leg ulcers benefit from surgery: long-term results from 173 legs. J Vasc Surg. 2006;44(3):572–579. doi: 10.1016/j.jvs.2006.05.039. [DOI] [PubMed] [Google Scholar]
- 40.Cabrera J, Redondo P, Becerra A, Garrido C, Cabrera J, Jr, Garcia-Olmedo MA, Sierra A, Lloret P, Martinez-Gonzalez MA. Ultrasound-guided injection of polidocanol microfoam in the management of venous leg ulcers. Arch Dermatol. 2004;140(6):667–673. doi: 10.1001/archderm.140.6.667. [DOI] [PubMed] [Google Scholar]
- 41.Hertzman PA, Owens R. Rapid healing of chronic venous ulcers following ultrasound-guided foam sclerotherapy. Phlebology. 2007;22(1):34–39. doi: 10.1258/026835507779700662. [DOI] [PubMed] [Google Scholar]
- 42.Stacey MC, Burnand KG, Layer GT, Pattison M. Calf pump function in patients with healed venous ulcers is not improved by surgery to the communicating veins or by elastic stockings. Br J Surg. 1988;75(5):436–439. doi: 10.1002/bjs.1800750514. [DOI] [PubMed] [Google Scholar]