Abstract
Aims
To determine the effect of grapefruit juice on omeprazole metabolism in vivo.
Methods
This was a randomized crossover study with a 2 week washout period. Omeprazole (20 mg) was taken orally by 13 healthy volunteers after an overnight fast with either grapefruit juice or water. Serial blood samples were obtained over 12 h and standardized meals were served 3 and 10 h after the administration of omeprazole. Plasma concentrations of omeprazole and its major metabolites, 5-hydroxyomeprazole and omeprazole sulphone, were determined by high performance liquid chromatography (h.p.l.c.).
Results
Mean area under the plasma concentration vs time curve (AUC) between 0 and 12 h for omeprazole sulphone was approximately 20% lower (P < 0.01) in the group receiving grapefruit juice. There was no significant difference in the mean AUC of 5-hydroxyomeprazole or omeprazole. The AUC ratio of omeprazole sulphone to omeprazole, an index of CYP3A4 activity, was decreased by 33% (P < 0.001) after administration of grapefruit juice whereas the AUC ratio of 5-hydroxyomeprazole to omeprazole, an index of CYP2C19 activity, did not differ between the two phases of the study. Although the time to peak concentration (tmax ) and terminal half-life (t1/2,z) of omeprazole and its two main metabolites were not altered, the peak concentration (Cmax ) of omeprazole sulphone was significantly reduced after administration of grapefruit juice.
Conclusion
Administration of grapefruit juice decreased the formation of omeprazole sulphone but not 5-hydroxyomeprazole. These results indicate that activities of CYP3A4, but not of CYP2C19, are inhibited by the simultaneous administration of grapefruit juice.
Keywords: CYP2C19, CYP3A4, grapefruit juice, interaction, intestinal P450, omeprazole, proton pump inhibitor
Introduction
Omeprazole, is a selective benzimidazole inhibitor of the H+/K+-ATPase proton pump in gastric parietal cells. It has been used widely for treatment of duodenal ulcer, refractory gastroesophageal reflux diseases, Zollinger–Ellison syndrome and other related hypersecretory conditions [1]. Omeprazole is oxidatively metabolized prior to excretion and the major metabolites in blood are omeprazole sulphone and 5-hydroxyomeprazole (Figure 1) [2]. The major urinary metabolites identified are 5-hydroxyomeprazole and its corresponding carboxylic acid. Neither omeprazole nor omeprazole sulphone are detected in urine. The metabolism of omeprazole to 5-hydroxyomeprazole cosegregates with 4′-hydroxylation of S-mephenytoin [3], indicating a role for the polymorphic CYP2C19 in this metabolic pathway. The rate of omeprazole hydroxylation has been used as an index for hepatic CYP2C19 activity in vivo [4, 5]. In vitro studies suggest that CYP3A4 is mainly responsible for omeprazole sulphoxidation [6]. Consistent with this, in vivo studies have shown that the formation of omeprazole sulphone was strongly inhibited by a CYP3A4 inhibitor, ketoconazole [7] and increased after prolonged treatment with a CYP3A4 inducer, carbamazepine [8]. Approximately 3% of Caucasian and 15–20% of Oriental populations are poor metabolisers (PMs) of S-mephenytoin or omeprazole hydroxylations. Omeprazole S-oxidation is thus the predominant metabolic pathway in the poor metaboliser phenotype. Moreover, 5-hydroxyomeprazole and omeprazole sulphone undergo further oxidation to a secondary metabolite, 5-hydroxyomeprazole sulphone, mediated by CYP3A4 and CYP2C19, respectively [9–11].
Figure 1.
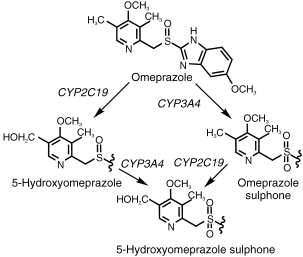
Oxidative metabolism of omeprazole in humans.
Co-intake of grapefruit juice has been shown to increase the oral bioavailability of several CYP3A4-related drugs including dihydropyridine-type calcium channel blockers [12–14], ethinyloestradiol [15], cyclosporin [16], midazolam [17], triazolam [18] and terfenadine [19]. These interactions are apparently caused by components specific to grapefruit juice, because no such interactions occur with other beverages. Among the components, furanocoumarins, particularly the dimers GF-I-1 and GF-I-4, have been shown by us to be the most potent inhibitors of CYP3A4 in grapefruit juice [20]. In addition, these two furanocoumarin dimers as well as furanocoumarin monomers, bergamottin and 6′,7′-dihydroxybergamottin inhibit CYP2C19-mediated omeprazole 5-hydroxylation in vitro (unpublished observations). Although the inhibitory effects of grapefruit juice on CYP3A4 substrates are well documented, its effect on other cytochromes P450 (P450s) in humans remains unclear. Drugs affected by grapefruit juice share some common features such as low oral bioavailability, interaction occurring only with orally administered drugs and no change in the elimination rates of the drug [21]. Previous in vivo studies showed that elimination half-lives of caffeine and coumarin were extended by coingestion of grapefruit juice. These reactions are considered to be mediated mainly by CYP1A2 and CYP2A6, respectively [22–24]. Since omeprazole undergoes two different metabolic pathways in vivo, i.e. CYP2C19-mediated 5-hydroxylation and CYP3A4-mediated S-oxidation, the effect of grapefruit juice on the activities of these P450s in vivo has been investigated in the present study.
Methods
Subjects
Thirteen healthy subjects (11 men and 2 women) participated in this study. Subjects were healthy as defined by medical history, physical examination and routine laboratory analysis. Four of them were cigarette smokers. All subjects were informed both verbally and in writing about the experimental procedures and the purpose of the study. Each subject gave written informed consent before the study. The subjects were asked to refrain from any medications, alcoholic beverages, citrus fruits and citrus juices for at least one week before and throughout the study period. The study was approved by the Ethics Committee of KhonKaen University (KhonKaen, Thailand).
Study protocol
After an overnight fast, the subjects took 20 mg omeprazole (Losec,® Astra-Hässle) with either 300 ml grapefruit juice (Tropicana,® regular strength, Kirin, Tokyo, Japan) or water in a randomized, crossover study (2 week washout period). Blood samples (8–10 ml) were collected through a venous catheter before and 1, 2, 3, 4, 6, 8, 10 and 12 h after the drug administration. Plasma was separated after centrifugation at 3000 g for 15 min and stored at −20° C until analysis. Meals were served 3 and 10 h after administration of omeprazole. Similar meals were served throughout the two study periods.
Determination of omeprazole and its metabolites
Plasma concentrations of omeprazole and its major metabolites, 5-hydroxyomeprazole and omeprazole sulphone, were determined by h.p.l.c. as described previously [25]. An internal standard (phenacetin, 4 μg), sodium chloride (0.5 g) and 0.5 m sodium potassium phosphate buffer (0.5 ml, pH 8.0) were added into a screw-capped tube containing 1 ml plasma. Extraction with dichloromethane/butanol mixture (99:1 v/v, 5 ml) was performed by rotary-mixing for 30 min. The organic phase after centrifugation at 3000 rev min−1 for 10 min was transferred to a glass tube and then evaporated to dryness under nitrogen. Aliquots (50 μl) of the residue dissolved in mobile phase (200 μl) were injected on to the column. The metabolites were separated with a CAPCELL PAK C18 SG 120 column (25 cm×4.6 mm i.d., 5 μm particle size; Shiseido, Tokyo, Japan) using a mixture of acetonitrile: 50 mm phosphate buffer (20:80, pH 8.5) at flow rate of 1.5 ml min−1. Metabolites were detected by their absorbance at 302 nm. Under the chromatographic conditions, 5-hydroxyomeprazole, phenacetin, omeprazole sulphone and omeprazole were eluted at the retention time of 6.6, 9.1, 11.0 and 18.8 min, respectively. Amounts were calculated from standard curves for omeprazole and its metabolites constructed in the concentration range 25–1000 ng ml−1.
Pharmacokinetic calculations
The time to peak concentration (tmax ) and the peak concentration (Cmax ) of omeprazole and its two metabolites, 5-hydroxyomeprazole and omeprazole sulphone, were noted directly. The area under the plasma concentration vs time curve (AUC) from 0 to 12 h was calculated by using the linear trapezoidal rule. The elimination rate constant (λz ) was estimated by least-squares regression analysis of the terminal phase of the plasma concentration-time curve. The half-life (t1/2,z) was calculated as follows
Statistical analysis
Values for pharmacokinetic variables are reported as mean±s.d.. Data were analysed by the nonparametric Wilcoxon Rank Sum Test using SAS software (6.12). Differences were regarded statistically significant when P values were <0.05.
Results
The plasma concentration-time curves of omeprazole and its metabolites in control and grapefruit juice experiments are shown separated as EM and PM (Figure 2). Analyses of the pharmacokinetic data for omeprazole indicate that 2 out of 13 subjects enrolled in the present study are PMs of CYP2C19. The AUC ratio of omeprazole to 5-hydroxyomeprazole in the control phase for these two PMs were 14.1 and 26.8 compared with 2.61±1.50 (range 0.7–5.8) for the extensive metabolisers. The mean AUC of omeprazole in the control phase for these two PMs (14233 nmol l−1 h) were about 5 fold greater than that observed with EMs (2873 nmol l−1 h, P < 0.05). Half-lives for omeprazole, 5-hydroxyomeprazole and omeprazole sulphone in the control phase were 0.9±0.2, 0.9±0.3 and 2.9±1.6 h for EMs and 2.5±0.4, 1.7±0.2 and 13.6±3.5 h for PMs, respectively. Since there were no marked differences in the effect of grapefruit juice on the pharmacokinetic parameters of omeprazole and its metabolites between PMs and EMs, the data from all 13 subjects were combined.
Figure 2.
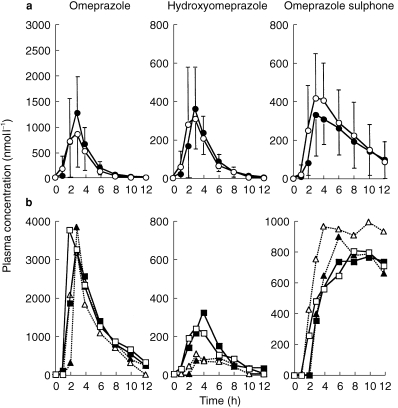
Plasma concentration profiles of omeprazole and its metabolites in CYP2C19 extensive (EM) and poor metabolizers (PM). The mean concentration in EM are shown in (a), and individual concentrations for PM are shown in (b). gf; grapefruit juice-treated. ○ EM controls,  EM gf, □ PMI control, ▪ PMI gf, ▵ PMR control, ▴ PM2 gf.
EM gf, □ PMI control, ▪ PMI gf, ▵ PMR control, ▴ PM2 gf.
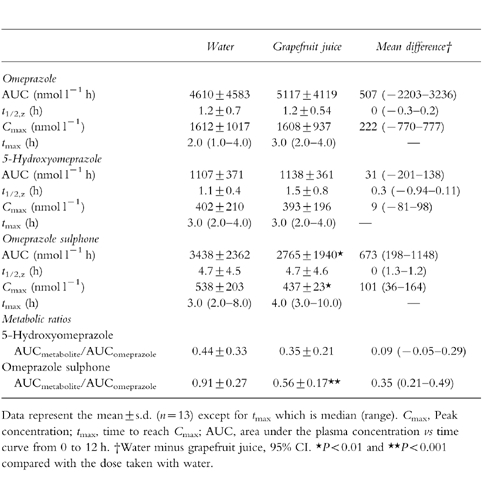
Pharmacokinetic parameters of both phases of the study are described in Table 1. Plasma Cmax and tmax of omeprazole and 5-hydroxyomeprazole were not significantly different whereas the Cmax value for omeprazole sulphone was significantly lower (P < 0.01) in the presence of grapefruit juice compared with the control. The AUC of omeprazole sulphone was decreased significantly(P < 0.01) from 3348±2362 nmol l−1 h in the control group to 2765±1940 nmol l−1 h in the grapefruit juice-treated group. Therefore, the AUC ratio of omeprazole sulphone to omeprazole was decreased significantly (P < 0.001) in the group receiving grapefruit juice. There was no change in the AUC of omeprazole or 5-hydroxyomeprazole or in the AUC ratio of 5-hydroxyomeprazole to omeprazole in the presence of grapefruit juice.
Table 1.
Pharmacokinetic parameters of omeprazole and its metabolites after intake of 20 mg omeprazole together with 300 ml of either water or grapefruit juice.
Discussion
In the present study, grapefruit juice has been shown to inhibit formation of omeprazole sulphone but not 5-hydroxyomeprazole. Additionally, half-lives of omeprazole sulphone and 5-hydroxyomeprazole were not extended by intake of grapefruit juice. Since in preliminary experiments, human microsomal CYP2C19 activity as well as CYP3A4 activity was clearly inhibited by the addition of a grapefruit extract or the major inhibitory components, furanocoumarins, the present results suggest that the inhibitory effect of grapefruit juice is likely confined to gastrointestinal mucosa, therefore more greatly affecting CYP3A4, but not liver-specific CYP2C19.
Although the metabolic pathways of omeprazole in vivo are rather complex, cytochrome P450 isoenzymes responsible for these pathways are now well characterized [2]. The polymorphic CYP2C19 is the major metabolic enzyme responsible for the formation of 5-hydroxyomeprazole whereas the formation of omeprazole sulphone is highly dependent on CYP3A4. Omeprazole sulphone and 5-hydroxyomeprazole undergo further metabolism to the common secondary metabolite, 5-hydroxyomeprazole sulphone. CYP2C19 is largely responsible for the hydroxylation of omeprazole sulphone whereas CYP3A4 is mainly involved in the sulphoxidation of 5-hydroxyomeprazole [9–11]. The pharmacokinetic data observed in our subjects are consistent with previously reports that the mean half-life of omeprazole in EMs was 0.9±0.2 h (0.7–1.2 h) whereas the mean half-life of this drug in PMs was 2.5±0.4 h (2.2–2.7 h) [9, 11]. The primary metabolite, 5-hydroxyomeprazole showed a longer apparent half-life in the PM than in the EM group. The mean AUC ratios of omeprazole to 5-hydroxyomeprazole were markedly higher in PMs compared with those calculated from EMs. Omeprazole sulphone was eliminated from plasma at a much lower rate in the PM than in the EM group. The mean half-life of omeprazole sulphone in the PM group was greater than that calculated from the EM group (2.9±1.6 h for EMs and 13.6±3.5 h for PMs). These findings support the idea that the metabolism of omeprazole sulphone is mediated by the polymorphic CYP2C19.
Plasma AUC of omeprazole sulphone decreased by approximately 20% after grapefruit juice intake without changing the half-life of this metabolite, indicating that the formation but not the elimination of omeprazole sulphone is inhibited by grapefruit juice. These data support the idea that CYP3A4, but not CYP2C19, is inhibited by grapefruit juice. Consistently, the AUC ratio of omeprazole sulphone to omeprazole, a proposed index of in vivo CYP3A4 activity, was decreased by about 33% in the grapefruit juice treatment group. The magnitude of inhibition of grapefruit juice on the formation of omeprazole sulphone observed in the present study was less than that reported with multiple-doses of ketoconazole [7]. The greater increase in the AUC of omeprazole after ketoconazole treatment was noted in PMs compared with the EMs. Ketoconazole is a potent CYP3A4 inhibitor, which acts in both liver and small intestine. Although only two PMs were enrolled in the present study, there was no significant difference in the effect of grapefruit juice on the formations of 5-hydroxyomeprazole and omeprazole sulphone between PMs and EMs. Results from in vitro studies suggests that the monomers [26] and the dimers [20, unpublished observations] of furanocoumarins in grapefruit juice are potent mechanism-based inhibitors of CYP3A4. These compounds are likely to be trapped in intestine and not released to the systemic circulation and thus affect only the first-pass metabolism of omeprazole in the gastrointestinal tract. Furthermore, plasma half-life of 5-hydroxyomeprazole which is mainly mediated by hepatic CYP3A4 was not significantly different between the two phases of the study. Consistent with these findings, intake of grapefruit juice has been shown to alter bioavailability after oral but not intravenous administration of several drugs including felodipine, midazolam and cyclosporin [16, 17, 27]. Moreover, it has been reported recently that ingestion of grapefruit juice for 6 days results in a marked decreased in enterocyte CYP3A4 immunoreactive protein concentration with no change on hepatic CYP3A4 activity as determined by the erythromycin breath test [28]. Reduction in intestinal CYP3A4 concentration was rapid, since it occurred within 4 h of after grapefruit juice intake [26].
Acknowledgments
The support of the Tokyo Biochemical Research Foundation (TBRF) and the Japan Health Sciences Foundation are gratefully acknowledged. We would like to thanks Dr Kjell Andersson and Dr Tommy Andersson, Astra, Möndal, Sweden, for kindly providing omeprazole and its metabolites and the Astra, Co. Ltd, Thailand for supplying omeprazole capsules (Losec® ).
References
- 1.Walan A. Clinical experience with omeprazole. assessment of efficacy and safety. J Gastroenterol Hepatol. 1989;4:27–33. [PubMed] [Google Scholar]
- 2.Birkett DJ, Andersson T, Miners JO. Assays of omeprazole metabolism as a substrate probe for human CYP isoforms. In: Johnson EF E F, Waterman MR, editors. Method Enzymol Cytochrome P450, Part B. Vol. 272. San Diego: Academic Press; 1996. pp. 132–139. [DOI] [PubMed] [Google Scholar]
- 3.Andersson T, Regardh CG, Dahl-Puustinen ML, Bertilsson L. Slow omeprazole metabolizers are also poor S-mephenytoin hydroxylators. Ther Drug Monit. 1990;12:415–416. doi: 10.1097/00007691-199007000-00020. [DOI] [PubMed] [Google Scholar]
- 4.Balian JD, Sukhova N, Harris JW, et al . The hydroxylation of omeprazole correlates with s-mephenytoin metabolism: a population study. Clin Pharmacol Ther. 1995;57:662–669. doi: 10.1016/0009-9236(95)90229-5. [DOI] [PubMed] [Google Scholar]
- 5.Chang M, Tybring G, Dahl ML, et al . Interphenotype differences in disposition and effect on gastrin levels of omeprazole—suitability of omeprazole as a probe for CYP2C19. Br J Clin Pharmacol. 1995;39:511–518. doi: 10.1111/j.1365-2125.1995.tb04488.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Andersson T, Miners JO, Veronese ME, et al . Identification of human liver cytochrome P450 isoforms mediating omeprazole metabolism. Br J Clin Pharmacol. 1993;36:521–530. doi: 10.1111/j.1365-2125.1993.tb00410.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Böttiger Y, Tybring G, Götharson E, Bertilsson L. Inhibition of the sulfoxidation of omeprazole by ketoconazole in poor and extensive metabolizers of S-mephenytoin. Clin Pharmacol Ther. 1997;62:384–391. doi: 10.1016/S0009-9236(97)90116-8. [DOI] [PubMed] [Google Scholar]
- 8.Bertilsson L, Tybring G, Widen J, Chang M, Tomson T. Carbamazepine treatment induces the CYP3A4 catalysed sulphoxidation of omeprazole, but has no or less effect on hydroxylation via CYP2C19. Br J Clin Pharmacol. 1997;44:186–189. doi: 10.1046/j.1365-2125.1997.00630.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sohn DR, Kobayashi K, Chiba K, et al. Disposition kinetics and metabolism of omeprazole in extensive and poor metabolizers of S-mephenytoin 4′-hydroxylation recruited from an oriental population. J Pharmacol Exp Ther. 1992;262:1195–1202. [PubMed] [Google Scholar]
- 10.Andersson T, Miners JO, Veronese ME, Birkett DJ. Identification of human liver cytochrome P450 isoforms mediating secondary omeprazole metabolism. Br J Clin Pharmacol. 1994;37:597–604. doi: 10.1111/j.1365-2125.1994.tb04310.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ieiri I, Kubota T, Urae A, et al. Pharmacokinetics of omeprazole (a substrate of CYP2C19) and comparison with two mutant alleles, CYPP. 2C19m1 in exon 5 and CYPP. 2C19m2 in exon 4, in japanese subjects. Clin Pharmacol Ther. 1996;59:647–653. doi: 10.1016/S0009-9236(96)90004-1. [DOI] [PubMed] [Google Scholar]
- 12.Bailey D, Edger B, Spence JD, Munoz C, Arnold JMO. Felodipine and nifedipine interactions with grapefruit juice. Clin Pharmacol Ther. 1990;47:180. [Google Scholar]
- 13.Soons PA, Vogels BA, Roosemalen MC, et al. Grapefruit juice and cimetidine inhibit stereoselective metabolism of nitrendipine in humans. Clin Pharmacol Ther. 1991;50:394–403. doi: 10.1038/clpt.1991.156. [DOI] [PubMed] [Google Scholar]
- 14.Bailey DG, Arnold JM, Munoz C, Spence JD. Grapefruit juice—felodipine interaction. Mechanism, predictability, and effect of naringin. Clin Pharmacol Ther. 1993;53:637–642. doi: 10.1038/clpt.1993.84. [DOI] [PubMed] [Google Scholar]
- 15.Schubert W, Cullberg G, Edgar B, Hedner T. Inhibition of 17β-estradiol metabolism by grapefruit juice in ovariectomized women. Maturitas. 1994;20:155–163. doi: 10.1016/0378-5122(94)90012-4. [DOI] [PubMed] [Google Scholar]
- 16.Ducharme MP, Warbasse LH, Edwards DJ. Disposition of intravenous and oral cyclosporine after administration with grapefruit juice. Clin Pharmacol Ther. 1995;57:485–491. doi: 10.1016/0009-9236(95)90032-2. [DOI] [PubMed] [Google Scholar]
- 17.Kupferschmidt HH, Ha HR, Ziegler WH, Meier PJ, Krahenbuhl S. Interaction between grapefruit juice and midazolam in humans. Clin Pharmacol Ther. 1995;58:20–28. doi: 10.1016/0009-9236(95)90068-3. [DOI] [PubMed] [Google Scholar]
- 18.Hukkinen SK, Varhe A, Olkkola KT, Neuvonen PJ. Plasma concentrations of triazolam are increased by concomitant ingestion of grapefruit juice. Clin Pharmacol Ther. 1995;58:127–131. doi: 10.1016/0009-9236(95)90188-4. [DOI] [PubMed] [Google Scholar]
- 19.Benton RE, Honig PK, Zamani K, Cantilena LR, Woosley RL. Grapefruit juice alters terfenadine pharmacokinetics resulting in prolongation of repolarization on the electrocardiogram. Clin Pharmacol Ther. 1996;59:383–388. doi: 10.1016/S0009-9236(96)90105-8. [DOI] [PubMed] [Google Scholar]
- 20.Fukuda K, Ohta T, Oshima Y, et al. Specific CYP3A4 inhibitors in grapefruit juice: furocoumarin dimers as components of drug interaction. Pharmacogenetics. 1997;7:391–396. doi: 10.1097/00008571-199710000-00008. [DOI] [PubMed] [Google Scholar]
- 21.Bailey DG, Malcolm J, Arnold O, Spence JD. Grapefruit juice–drug interactions. Br J Clin Pharmacol. 1998;46:101–110. doi: 10.1046/j.1365-2125.1998.00764.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fuhr U, Klittich K, Staib AH. Inhibitory effect of grapefruit juice and its bitter principal, naringenin, on CYP1A2 dependent metabolism of caffeine in man. Br J Clin Pharmacol. 1993;35:431–436. doi: 10.1111/j.1365-2125.1993.tb04162.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Merkel U, Sigusch H, Hoffmann A. Grapefruit juice inhibits 7-hydroxylation of coumarin in healthy volunteers. Eur J Clin Pharmacol. 1994;46:175–177. doi: 10.1007/BF00199885. [DOI] [PubMed] [Google Scholar]
- 24.Runkel M, Bourian M, Tegtmeier M, Legrum W. The character of inhibition of the metabolism of 1, 2-benzopyrone (coumarin) by grapefruit juice in human. Eur J Clin Pharmacol. 1997;53:265–269. doi: 10.1007/s002280050374. [DOI] [PubMed] [Google Scholar]
- 25.Kobayashi K, Chiba K, Sohn DR, Kato Y, Ishizaki T. Simultaneous determination of omeprazole and its metabolites in plasma and urine by reversed-phase high-performance liquid chromatography with an alkaline-resistant polymer-coated C18 column. J Chromatogr. 1992;579:299–305. doi: 10.1016/0378-4347(92)80395-7. [DOI] [PubMed] [Google Scholar]
- 26.Schmiedlin-ren P, Edwards DJ, Fitzsimmons ME, et al . Mechanisms of enhanced oral availability of CYP3A4 substrates by grapefruit constituents. Decreased enterocyte CYP3A4 concentration and mechanism-based inactivation by furanocoumarins. Drug Metab Dispos. 1997;25:1228–1233. [PubMed] [Google Scholar]
- 27.Lundahl J, Regardh CG, Edgar B, Johnsson G. Effects of grapefruit juice ingestion—pharmacokinetics and haemodynamics of intravenously and orally administered felodipine in healthy men. Eur J Clin Pharmacol. 1997;52:139–145. doi: 10.1007/s002280050263. [DOI] [PubMed] [Google Scholar]
- 28.Lown KS, Bailey DG, Fontana RJ, et al. Grapefruit juice increases felodipine oral availability in humans by decreasing intestinal CYP3A protein expression. J Clin Invest. 1997;99:2545–2553. doi: 10.1172/JCI119439. [DOI] [PMC free article] [PubMed] [Google Scholar]


