Abstract
Epstein-Barr virus (EBV)–associated tumors developing in immunocompetent individuals present a challenge to immunotherapy, since they lack expression of immunodominant viral antigens. However, the tumors consistently express viral proteins including LMP2, which are immunologically “weak” but may nonetheless be targets for immune T cells. We previously showed that a majority of cytotoxic T lymphocytes (CTLs) reactivated using EBV-transformed B-lymphoblastoid cells lines (LCLs) contained minor populations of LMP2-specific T cells and homed to tumor sites. However, they did not produce remissions in patients with bulky disease. We have now used gene transfer into antigen-presenting cells (APCs) to augment the expression and immunogenicity of LMP2. These modified APCs increased the frequency of LMP2-specific CTLs by up to 100-fold compared with unmodified LCL-APCs. The LMP2-specific population expanded and persisted in vivo without adverse effects. Nine of 10 patients treated in remission of high-risk disease remain in remission, and 5 of 6 patients with active relapsed disease had a tumor response, which was complete in 4 and sustained for more than 9 months. It is therefore possible to generate immune responses to weak tumor antigens by ex vivo genetic modification of APCs and the CTLs so produced can have substantial antitumor activity. This study is registered at http://www.cancer.gov/clinicaltrials (protocol IDs: BCM-H-9936, NCT00062868, NCT00070226).
Introduction
Infused monoclonal antibodies enjoy significant success as cancer immunotherapies, but it has proved harder to exploit the equivalent promise of infused T lymphocytes.1–3 Many tumor-associated target antigens are functionally weak stimulators of the immune response.4 Consequently, preparation of tumor antigen–specific T cells for infusion requires specialized antigen-presenting cells (APCs). Since these APCs are required during both the induction and the amplification phase of the antitumor T-cell response, large numbers of APCs are necessary. This is problematic in many cancer patients whose APCs may be numerically and functionally defective, due to the disease or its treatment.5
Even when tumor-specific cytotoxic (CD8+) T cells are successfully prepared,6 lack of T helper (CD4+) cell function in vivo may lead to lack of in vivo expansion, a problem that is not overcome by infusion of large numbers of cells.6–8 Finally, although successful responses to adoptively transferred T cells have been intermittently observed in patients with relapsed or resistant cancer, the overall response rate has been low.6,7,9
For example, our previous experience using Epstein-Barr virus (EBV)–specific cytotoxic T lymphocytes (CTLs) for relapsed EBV-associated Hodgkin disease (HD) generated CTL lines with low frequencies of cells specific for the weak EBV-derived antigens expressed by the tumor cells (eg, LMP2) and produced responses only in a small proportion of patients, whose disease was limited.10,11
Of the potential CTL target antigens expressed in EBV-positive Hodgkin disease (HD) and non-Hodgkin lymphoma, EBNA-1 is not processed for presentation on class I antigens by tumor cells,12 while wild-type LMP1 is toxic when expressed in antigen-presenting cells.13 LMP2A, however, is consistently expressed on the malignant populations in Lymphoma tumors (eg, Hodgkin Reed-Sternberg cells), and its epitopes are conserved between viral strains and among lymphoma biopsy samples.14,15
To bias the EBV-specific CTL response to LMP2, we have replaced LCLs as APCs for the first stimulation, with dendritic cells (DCs) engineered to express LMP2 from an adenovirus (Ad) vector and used LCLs overexpressing LMP2 from the same Ad vector for subsequent stimulations.16,17 This approach expands polyclonal populations of both helper (CD4+) and cytotoxic (CD8+) T lymphocytes specific for this weak tumor antigen. After infusion into patients diagnosed with LMP2-positive lymphomas, LMP2-reactive T cells increased in number in the circulation and at tumor sites and produced sustained tumor responses.
Patients, materials, and methods
Patients and LMP status of the tumors
The protocol for the use of LMP2-specific CTLs as therapy for lymphoma was approved by the Food and Drug Administration, the Recombinant DNA Advisory Committee, and the Baylor College of Medicine Institutional Review Board and Institutional Biosafety Committees. Informed consent was obtained in accordance with the Declaration of Helsinki. Patients were eligible for this study if they had Epstein-Barr virus (EBV)–associated type II latency Hodgkin disease or non-Hodgkin lymphoma (NHL) detected by immunohistochemistry for LMP1 and/or in situ hybridization for EBER.11
Patients had either relapsed after receiving standard therapy (Table 1) or were considered high risk for relapse (Table 2) and received 2 infusions of T cells 2 weeks apart in the General Clinical Research Center (GCRC) at Texas Children's Hospital or The Methodist Hospital, where their vital signs were monitored before and immediately after each infusion. On level 1, patients received 2 doses of cells at 2 × 107/m2. At level 2, they received one dose of 2 × 107/m2 and a second dose of 108/m2, and at level 3, patients received one dose of 108/m2 and a second dose of 2 × 108/m2. If patients had a partial response or stable disease 8 weeks after CTL infusion they were eligible to receive additional CTLs, consisting of the same number of cells as their second injection. Total doses of CTLs received are shown in Tables 1-2. Complete blood counts were obtained, and electrolytes, and renal and liver function were evaluated at 2, 4, and 6 weeks after infusion and then at 3, 6, 9, and 12 months. Blood from patients was also monitored at regular intervals for EBV-DNA levels and immunologic assessment. Analysis of disease response to CTL therapy was performed using RECIST criteria.18
Table 1.
Patients with detectable lymphoma at time of T-cell infusion
| Patient ID | Age, y/sex | Disease, stage at diagnosis | No. of relapses | Most recent treatment (time before CTL infusion) | Total CTL dose, m−2 | Response to CTL infusion (survival mo) |
|---|---|---|---|---|---|---|
| 1 | 8/M | T-cell SCAEBV | 2 | Syngeneic SCT: BU/CY/ alemtuzumab (5 mo) | 4 × 107 | PR (> 36 mo) |
| 2* | 39/M | CD30+ B-cell NHL stage IVb | 5 (refractory disease for 2 y) | Anti-CD30 (11 mo) | 8 × 107 | CR (relapsed 10 mo, died 33 mo) |
| 3* | 30/F | Mixed cellularity HD stage IVb | 2 | ABVD (12 mo) rituximab (1 mo) | 8 × 107 | CR (> 32 mo) |
| 4* | 64/F | NK/T-cell NHL (nasal) | 3 | R-CHOP (1 mo) | 3.2 × 108 | CR (relapsed 9 mo, died 18 mo) |
| 5 | 17/M | Nodular sclerosing HD stage IIa | 3 | Autologous SCT-BEAM (3 mo) | 1.2 × 108 | NR (died 8 mo) |
| 6 | 15/F | Mixed cellularity HD stage IV | 0 (residual disease) | VP16/Dox/Rituximab (1 mo) | 3 × 108 | CR (> 13 mo) |
For all patients, there was no toxicity attributed to CTL infusion.
SCT indicates stem cell transplantation; BU, busulfan; CY, cyclophosphamide; PR, partial response; CR, complete response; ABVD, doxorubicin, bleomycin, vinblastine and dacarbazine; R-CHOP, rituximab plus cyclophosphamide, doxorubicin, vincristine and prednisone; BEAM, BCNU, etoposide, cytarabine and melphalan; NR, no response; VP-16, etoposide; and Dox, doxorubicin.
Patients who received 4 doses of CTLs.
Table 2.
Patients in remission of lymphoma at time of T-cell infusion
| UPN | Age, y/sex | Disease (stage at diagnosis) | No. of relapses | Most recent treatment (time before CTL infusion) | Total CTL dose, m−2 | Response to CTL infusion (survival mo) |
|---|---|---|---|---|---|---|
| 7 | 66/F | Diffuse large B-cell NHL stage IIa | 3 | Auto SCT-BEAM-R (3 mo) | 4 × 107 | Remains in remission (> 37 mo) |
| 8 | 57/F | Diffuse large B-cell NHL stage IVb | 2 | Auto SCT-BEAM-R (5 mo) | 4 × 107 | NR (died 22 mo) |
| 9 | 50/F | Mixed cellularity HD stage IIIa | 4 | Liposomal vincristine (6 mo) | 4 × 107 | Remains in remission (died secondary MDS 20 mo) |
| 10 | 25/F | Nodular sclerosing HD stage IIa | 1 | Auto SCT-BU/Mel/thiotepa (10 mo) | 1.2 × 108 | Remains in remission (> 24 mo) |
| 11 | 30/M | Nodular sclerosing HD stage IIb | 4 | ABVD (7 mo) | 1.2 × 108 | Remains in remission (> 23 mo) |
| 12 | 59/M | Nodular sclerosing HD stage IIb | 1 | Auto SCT-BEAM (4 mo) | 1.2 × 108 | Remains in remission (> 6 mo) |
| 13 | 51/M | NK/T-cell NHL (nasal) | 0 | RT (1 mo) | 1.2 × 108 | Remains in remission (6 mo) |
| 14 | 60/F | NK/T-cell NHL | 0 | COP (1 mo) | 1.2 × 108 | Remains in remission (3 mo) |
| 15 | 37/M | NK/T-cell NHL | 0 | CHOP and RT (3 mo) | 1.2 × 108 | Remains in remission (8 wk) |
| 16 | 7/M | Mixed cellularity HD stage Ia (after lung transplantation) | 0 | Rituximab (8 mo) | 3 × 108 | Remains in remission (> 18 mo) |
For all patients, there was no toxicity attributed to CTL infusion.
UPN indicates unique patient number; BEAM/BEAM-R, BCNU, etoposide, cytarabine, melphalan ± rituximab; MDS, myelodysplastic syndrome; SCT, stem cell transplantation; BU, busulfan; MEL, melphalan; ABVD, doxorubicin, bleomycin, vinblastine and dacarbazine; RT, radiotherapy; and COP, cyclophosphamide, vincristine and prednisone.
Generation of LMP2-specific CTLs and EBV-specific CTLs
Peripheral blood mononuclear cells (PBMCs, 5 × 106) from each patient were used for the establishment of an EBV-transformed B-lymphoblastoid cell line (LCL) for use as APCs.16 For DC production, PBMCs were plated for 2 hours, then nonadherent cells were cryopreserved and adherent cells were cultured with 800 U/mL granulocyte-macrophage colony-stimulating factor (GM-CSF; Immunex, Seattle, WA) and 500 U/mL IL-4 (R&D Systems, Minneapolis, MN) for 7 days. IL-4 and GM-CSF were again added on day 3. On day 5, immature DCs were harvested by vigorous pipetting, transduced with Ad5f35LMP2 vector, and treated with TNFα (R&D systems) and PGE1 (Merck, West Point, PA) for maturation. Before coculture with nonadherent PBMCs the DCs were gamma-irradiated (30 Gy). From day 10, responder T cells were restimulated weekly with irradiated LCLs transduced with the same LMP2 vector. At the time of final cryopreservation (30-50 days of culture), the patient-derived CTLs had an effector-memory phenotype (CD62L−, CCR7−, CD28−, CD45RA−, CD45R0+) and comprised a mean of 24.2% (range, 0.4%-94.2%) CD4+ and a mean of 71.5% (range, 6.62%-99.4%) CD8+ T cells. Less than 1% of cells expressed monocytoid or B-cell markers.10,19–22 The LMP2 specificity of the CTL lines was determined using HLA-peptide multimers, cytotoxicity assays, and IFN-γ enzyme-linked immunospot (ELISPOT) assays. In 10 of 16 CTL lines, LMP2-specific activity was confirmed using cytotoxicity16 and/or γ-IFN ELISPOT assays with overlapping LMP2 peptides.23 In the remaining 6 CTL lines, LMP2-specific activity could not be confirmed. In the case of 2 predominantly CD4+ HLA class II–restricted CTL lines, this may have been because the 15-mer overlapping peptide library did not detect class II epitopes. EBV-specific CTL lines were produced as previously described.24 Briefly, PBMCs were cocultured with autologous irradiated (40 Gy) LCLs at a responder-stimulator ratio of 40:1 for 9 days and then restimulated weekly with the autologous LCLs at a 4:1 ratio. The cultures were supplemented with 40 units/mL IL-2 from day 13.
Cytotoxicity assays
The cytotoxic specificity of each CTL line was analyzed in a standard 4-hour chromium51 release assay using effector-target ratios of 40:1, 20:1, 10:1, and 5:1.25 The LMP2-positive target cells tested were as follows: autologous LCLs; HLA class I– and II–mismatched LCLs; HLA-matched fibroblasts transduced with Ad5f35LMP2; or PHA blasts pulsed with the LMP2 pepmix (Jerini, Berlin, Germany). As additional controls, we used the following LMP2-negative target cells: autologous phytohemagglutinin-stimulated PBMCs (PHA blasts) alone or pulsed with irrelevant peptides from a cytomegalovirus (CMV) pp65 pepmix (Jerini), and HLA-matched fibroblasts transduced with Ad5f35GFP.
Immunophenotyping
CTL lines were stained with CD3, CD4, CD8, CD16, CD56, TCRαβ, TCRγδ, CD19, CD28, CD62L, CCR7, CD45RA, and CD45RO (Becton Dickinson, San Jose, CA). For each sample, 10 000 cells were analyzed by fluorescence-activated cell sorting (FACS) Calibur using Cell Quest software (Becton Dickinson).
Detection of EBV-DNA in PBMCs by quantitative real-time PCR
DNA was isolated from 3 to 5 × 106 PBMCs using an anion exchange column (Qiagen, Valencia, CA). DNA (500 ng) was analyzed by EBV EBER-specific quantitative real-time polymerase chain reaction (Q-PCR) as previously described to quantitate EBV genome copy number per microgram.26
LMP2 multimers and peptides
To detect LMP2-specific T cells in the CTL lines and PBMCs, we used the soluble HLA-peptide tetramers prepared by the Baylor College of Medicine Tetramer Core Facility or pentamers Proimmune (Springfield, VA) (together termed multimers). Multimers were HLA-A*0201-CLGGLLTMV, HLA-A*0201-FLYALALLL, HLA-A*0201-LLWTLVVLL, HLA-A*1101-SSCSSCPLSKI, HLA-A*2301-PYLFWLAAI, HLA-A24-PYLFWLAAI, HLA-A24-TYGPVFMSL, HLA-A*0201-CLGGLLTMV, HLA-A*0201-FLYALALLL, HLA-A*1101-SSCSSCPLSKI, HLA-A24-PYLFWLAAI, HLA-A24-TYGPVFMSL, HLA B*2705-RPPIFIRRL, B*2705-RRIYDLIEL, and HLA-B*3501-MGSLEMVPM. Peptides were synthesized by the Baylor College of Medicine Protein Core Facility or by Genemed Synthesis (South San Francisco, CA). Tetramer staining of CTLs (5 × 105) or PBMCs (1 × 106) is previously described.10 For pentamer staining, CTLs or PBMCs were incubated with unlabeled pentamer followed by Pro5 Flurotag (PE; Proimmune) according to the manufacturer's instructions. For each sample, 100 000 cells were analyzed by FACS.
Panels of 15-mer peptides (overlapping by 11 amino acids) covering the entire amino acid sequence of LMP2 from the prototype EBV strain B95-8 were synthesized as previously described.16,27 Twenty-three peptide pools comprising 2 to 12 15-mer peptides were prepared, so that each 15-mer peptide was represented in 2 pools.16,28 This LMP2 peptide library was designed to identify all possible HLA class I restricted epitopes, which have a length of 9 to 11 amino acids, however, it is not ideal for the identification of HLA class II restricted epitopes, which have lengths of 13 to 17 amino acids.29
Enzyme-linked immunospot (ELISPOT) assay
ELISPOT analysis was used to determine the frequency and function of T cells secreting IFN-γ in response to LMP2 pepmix (Jerini), which contains all 15-mer peptides of LMP2 in one pool, or to LCLs.10 ELISPOT assays were performed on the CTL lines and on PBMCs before and after CTL infusion. In addition, to define the CD4 and CD8 restricted LMP2-specific activity, we pulsed OKT3 blasts with either LMP2 pepmix or CMV pepmix17 and used them as antigen-presenting cells to stimulate either CD8+ or CD4+ T cells sorted from CTL lines using flow cytometry. To reduce interassay variability, patient PBMC samples were cryopreserved and batched for ELISPOT analysis after more than 6 weeks of follow-up. PBMCs stimulated with CMV pepmix and staphylococcal enterotoxin B (1 μg/mL; Sigma-Aldrich, St Louis, MO) served as controls. Spot-forming cells (SFCs) and input cell numbers were plotted after plates were evaluated by Zellnet Consulting (New York, NY).
Statistical analysis
Descriptive statistics were calculated to summarize SFCs for LMP2- or EBV-specific T cells at preinfusion and postinfusion time points as well as the changes in SFCs from before infusion. Assessments of data normality on these outcomes were performed and data were log-transformed if indicated. Comparisons between preinfusion and postinfusion LMP2-specific and EBV (LCL)–specific T cell SFCs were performed using paired t tests on log-transformed data. Comparison of repeated measurements of LMP2 and EBV SFC levels over time between patients with versus without LMP2 sensitivity in their CTL lines was performed using random coefficient models. P values less than .05 were considered statistically significantly different.
Results
Patient characteristics
All patients had EBV-positive lymphoma. Eight had Hodgkin disease (4 nodular sclerosing Hodgkin disease and 4 mixed cellularity) and 8 had non-Hodgkin lymphoma (2 diffuse large B cell, 1 anaplastic large cell, 1 T-cell severe chronic active EBV infection (SCAEBV), and 4 nasal natural killer [NK]/T-cell NHL). Their ages ranged from 7 to 66 years (median, 30 years) and their initial disease presentation ranged from stage IA to IVB. Samples for CTL generation were collected from patients either at diagnosis (n = 2) or after a first or subsequent relapse (n = 14). Before CTL infusion, the 16 patients had lymphocyte counts ranging from 333.9/mm3 to 3494.4/mm3 (median, 850 cells/mm3). Lymphopenia as defined as a peripheral blood lymphocyte count of less than 1000/mm3 was observed in 9 of 16 patients.
CTLs are LMP2 specific and polyclonal
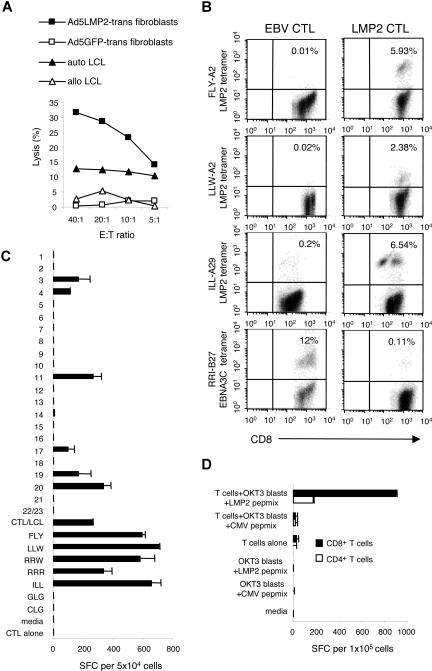
The specificity of the LMP2-CTL lines was determined using HLA-peptide multimers when available. Specific function was determined, using cytotoxicity assays and IFNγ ELISPOT assays. The cytolytic activity of the responder cells was tested against a panel of 51Cr-labeled autologous and allogeneic target cells. Ten of the 16 lines infused had demonstrable LMP2-specific activity. LMP2-specific CTLs generated from a patient representative of these 12 patients killed autologous LCLs and HLA-matched fibroblasts only if transduced with Ad5f35LMP2A (30% at an E/T ratio of 20:1 [Figure 1A]), whereas the LCL-activated EBV-specific CTL line demonstrated no LMP2-specific activity (data not shown). Killing was not due to adenovirus-directed CTLs, since fibroblasts infected with recombinant adenovirus encoding GFP were not recognized. There was no cytotoxic activity against HLA-mismatched LCLs (Figure 1A) or untransduced fibroblasts (data not shown).
Figure 1.
LMP2-specific CTL lines derived from lymphoma patients contained functional LMP2-specific T-cell populations. This figure shows the LMP2-specific activity in a CTL line generated from a patient with relapsed lymphoma. The patient is representative of the 10 patients whose CTL lines recognized LMP2. To demonstrate cytolytic specificity of patient CTL lines in vitro, percentage specific 51Cr release was determined 6 hours after coincubation with HLA-matched fibroblasts transduced with Ad5LMP2 (■), or Ad5GFP (□), autologous LCLs (▴), and allogeneic LCLs (▵). The LMP2-specific CTL line from a representative patient shown in panel B showed killing of autologous LCLs and HLA-matched fibroblasts only if they were transduced with Ad5LMP2 (30% at an E/T ratio of 20:1). Killing was not due to adenovirus-directed CTLs, since fibroblasts infected with recombinant adenovirus encoding GFP were not recognized. There was no killing of HLA-mismatched LCLs, (A) or nontransduced fibroblasts (data not shown). Panel B shows the frequency of LMP2-specific T cells in the CTL line generated from the same patient (HLA A2;29/B13;27). CTLs were costained with PE-conjugated multimers CD8-FITC and CD3-PerCP. Multimers were as follows: LMP2-HLA-A*0201-LLW, HLA-A*0201-FLYALALLL, HLA A29-ILL, and EBNA 3C-HLA-B27-RRI. When compared with the EBV-LCL–activated CTL line, the LMP2-activated CTL line showed an increase in the frequency of T cells reactive with all 3 LMP2 tetramers, but decreased frequency in the EBNA3C tetramer reactivity (B). To assess the breadth and function of the LMP2-specific response, the CTLs were coincubated with overlapping LMP2 peptides and IFN-γ release in response to specific 15-mer and 8-mer peptides was measured in an ELISPOT assay. (C) This patient's polyclonal CTL line was then sorted for CD4+ and CD8+ T cells. Subsequently, recognition of LMP2 by these separated CD4+ and CD8+ T cells was determined in an IFN-γ ELISPOT assay using OKT3 blasts pulsed with either LMP2pepmix or CMVpepmix as the APCs (D). Error bars are SD.
This same patient's HLA type was A2;29/B13;27. Using HLA A2, A29, and B27 multimers we demonstrated that CTLs generated using Ad5f35LMP2-transduced APCs (LMP2-CTL) were markedly enriched for T cells specific for LMP2 epitopes compared with the EBV-CTL line generated from the same patient using nontransduced LCLs alone. Further, the frequency of T cells specific for the HLA B27-restricted non–tumor-associated immunodominant EBV epitope RRIYDLIEL (RRI) in EBNA 3C was markedly decreased in the LMP2-CTL line (0.11%) compared with the population detectable in the EBV-CTL line (12%) (Figure 1B).
Figure 1C illustrates that LMP2-specific lines can secrete IFNγ in response to several different LMP2 peptides, representing discrete epitopes on the LMP2 antigen. Furthermore, using OKT3 blasts expressing LMP2 peptides as APCs we were able to demonstrate that both the CD4+ and CD8+ T cells present in the polyclonal CTL lines have LMP2 specificity (Figure 1D). Hence the lines infused, though reactive to LMP2 are multispecific and are not restricted to predetermined peptide epitopes of LMP2, thereby reducing the probability of tumor-immune escape due to outgrowth of a single-epitope loss variant.
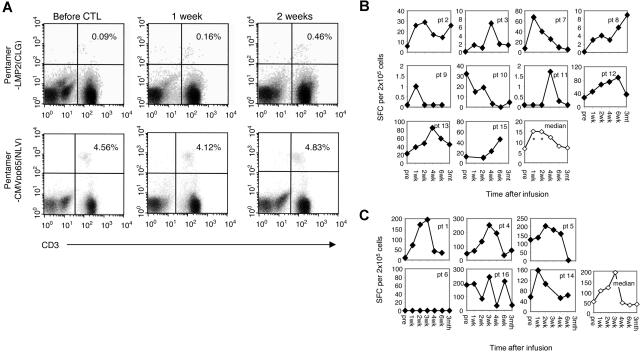
Adoptively transferred LMP2-specific CTL lines expand in vivo
Sixteen patients received total numbers of CTLs ranging from 4 × 107/m2 to 3.2 × 108/m2. Multimer and ELISPOT assays were performed to measure the frequency of LMP2-specific CTLs in peripheral blood before and after infusion. During the follow-up period, LMP2-specific T cells increased up to 5-fold following infusion of the first dose of T cells as measured by multimer assay (Figure 2A). Antigen responsiveness based on ELISPOT analysis also showed a significant increase in cells that secreted IFNγ in response to LMP2 pepmix. In the 10 patients with a measurable LMP2-specific response in their CTL line, there was a significant increase in LMP2-specific T cells in the peripheral blood 1 week after infusion (mean difference from before infusion = 1.4 log units, P = .018) and 2 weeks after infusion (mean difference from before infusion = 1.0 log units, P = .037) and a similar outcome was also seen at 4 weeks (mean difference from before infusion = 1.3 log units, P = .051) (Figure 2B). In the remaining 6 patients for whom LMP2 specificity could not be quantified, we observed a similar pattern of increase in peripheral blood T cells responding to autologous EBV-LCLs, which present all 8 latency-associated EBV proteins as well as early lytic cycle proteins. This trend did not, however, reach statistical significance in this smaller group (mean difference from before = 0.57, 0.69, 1.05 log units at 1, 2, and 3 weeks, respectively) (Figure 2C). There was a significant difference in LMP2 CTL expansion over time between those patients with versus without LMP2 specificity in their CTL lines (P = .006). The elevation in LMP2- and LCL-specific T cells in the peripheral blood persisted for more 3 months following infusion. There was no corresponding rise in T cells with specificity to CMV antigens (P > .05) (data not shown). No relationship between the number of CTLs infused and the degree of in vivo expansion or final frequency of LMP2-reactive cells in peripheral blood was detected.
Figure 2.
The frequency of LMP2-specific T cells increased after infusion of polyclonal LMP2-specific CTLs. LMP2 multimer analysis was also used to compare the frequency of LMP2-specific CTLs before and after infusion. The average number of LMP2 and CMV multimer-reactive cells per 106 CD8+ T cells are shown for 1 patient before and after infusion (A). This patient was representative of the 10 patients who received CTLs where specific LMP2 epitopes had been characterized. In these 10 patients in whom the HLA-restricted LMP2 peptide(s) were available, peripheral blood T cells were incubated with LMP2pepmix, and the number of IFN-γ spot-forming cells per 2 × 105 mononuclear cells was measured (B). In the 6 patients in whom LMP2 peptides were not detected, peripheral blood T cells were incubated with autologous LCLs, and the number of IFN-γ spot-forming cells per 2 × 105 mononuclear cells was measured (C).
Outcome
No short- or long-term toxicities were observed after CTL infusion. Of the 10 patients treated in remission of high-risk disease, 9 have remained in complete remission for up to 37 months after CTL infusion (Table 2). The tenth patient progressed within 8 weeks of receiving CTLs.
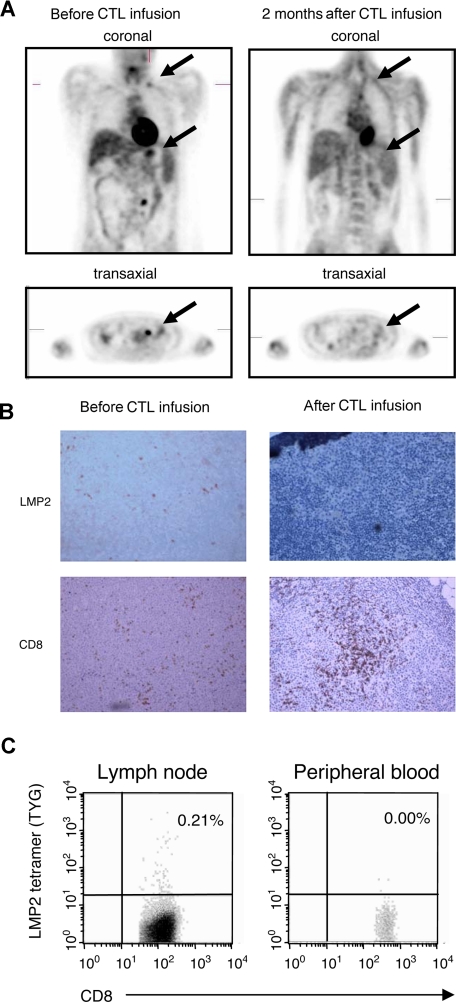
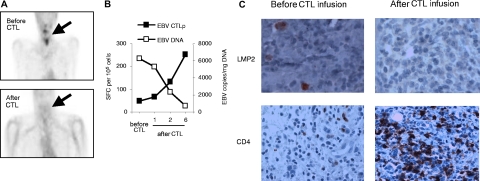
Of the 6 patients with relapsed disease that was refractory to standard treatment (Table 1), 4 had complete clinical responses and one had a very good partial response, all as defined by RECIST criteria.18 Reponses were seen in both Hodgkin and non-Hodgkin lymphomas, and in patients receiving multiple doses of CTLs at either 2 × 107/m2 per dose or 1 × 108/m2 per dose or 2 × 108/m2 per dose (total dose range, 8 × 107/m2 to ≥ 3 × 108/m2 CTLs). Of the 4 complete responders, the first patient (pt 3) had Hodgkin disease involving the bone marrow that had proved resistant to multiple courses of chemotherapy and now remains in complete remission 34 months after CTL infusion. The second patient (pt 6) had residual Hodgkin disease evident on imaging after chemotherapy that resolved 8 weeks after receiving CTLs. She remains in complete remission (CR) at 15 months after CTL infusion. Figure 3A shows imaging data from a third responding patient (pt 2) who had refractory NHL with multiple disease sites that cleared after CTL infusion. This patient also had a follow-up needle biopsy of a previously involved cervical node (Figure 3B), which showed no residual LMP2+ tumor cells, but a heavy infiltrate of CD8+ T cells. Tetramer staining (Figure 3C) showed many of these cells were specific for an LMP2 epitope strongly represented in the line. The fourth patient (pt 4) had a relapsed NK/T non-Hodgkin lymphoma whose resolution is shown in Figure 4A together with an associated increase in LMP2-specific CTL activity and corresponding fall in the level of his tumor marker EBV-DNA (Figure 4B). A follow-up nasopharyngeal tissue biopsy (Figure 4C) revealed no residual LMP2+ cells and a marked increase in infiltrating CD4+ T cells, consistent with the predominant phenotype of this patient's LMP2-specific T-cell line.
Figure 3.
Induction of complete clinical response and LMP2-specific T-cell accumulation at tumor site after CTL infusion. A positron emission tomography (PET) scan demonstrating abnormal fluorodeoxyglucose (FDG) uptake in supraclavicular and para-aortic lymph nodes was observed before CTL infusion in a patient with NHL (pt 2). The follow-up scan 8 weeks after CTL infusion is reported as normal (A). Using immunohistochemistry, CD8+ infiltrating T cells were seen in lymph node biopsy after CTL infusion, which corresponded to a clearance of LMP2+ tumor cells (B). In addition, the percentage CD8+/LMP2 tetramer+ T cells in the lymph node and peripheral blood were compared after CTL infusion by flow cytometry (C). Images were acquired with an Olympus BX41 microscope (OlympusAmerica, Center Valley, PA) with a Plan Achromat 10×/0.25 NA oil objective lens (Olympus, Tokyo, Japan). Cells were stained with hematoxylin (Mayer)-eosin and also with CD8 monoclonal antibody (Dako, Carpinteria, CA) and LMP2 antibody (gift of Dr Friedrich A. Grässer, Institut für Mikrobiologie und Hygiene Abteilung Virologie, Homburg/Saar, Germany) and were used in immunoperoxidase protocol. Images were photographed with an Olympus Q-Color 5 digital color camera using FireWire technology and processed with Adobe Photoshop Elements 2.0 imaging software (Adobe Systems, San Jose, CA). Original magnification, ×10.
Figure 4.
Induction of complete clinical response and T-cell accumulation at tumor site after CTL infusion. A PET scan demonstrating abnormal FDG uptake in the nasopharyngeal region was observed before CTL infusion in another patient (pt 4) with NK/T-cell lymphoma. The follow-up scan 8 weeks after CTL infusion was reported as normal (A). EBV DNA levels in this patient's (pt 4) PBMCs measured by quantitative real-time PCR before and after CTL infusion were plotted against the EBV(LCL)-specific T-cell response after infusion as measured by IFN-γ secretion in ELISPOT assay (B). Using immunohistochemistry, CD4+ infiltrating T cells were seen in a lymph node biopsy after CTL infusion, and corresponded to a clearance of LMP2-positive tumor cells in patient 4 (C). Images were acquired with an Olympus BX41 microscope, with a Plan Achromat 40×/0.65 NA oil objective lens (Olympus). Cells were stained with hematoxylin (Mayer)-eosin and CD4 monoclonal antibody (Dako) and LMP2 antibody (gift of Dr Friedrich A. Grässer) were used in immunoperoxidase protocol. Images were photographed and processed as in Figure 3B. Original magnification, ×40.
Discussion
Efforts to exploit the promise of T lymphocytes for the targeted therapy of cancer have been hindered by the limited immunogenicity of most tumor-associated antigens. They have also been impaired by lack of subsequent in vivo expansion and homing of tumor-specific T cells, since substantial numbers of effector cells are required to control established human tumors, which are often bulky and widely dispersed.
We have used cytotoxic T lymphocytes (CTLs) targeting the subdominant tumor-associated viral antigen LMP2 to treat patients with EBV-associated Hodgkin and non-Hodgkin lymphoma. Our results show how the genetic modification of APCs so that they overexpress LMP2 allows them to amplify an in vitro T-cell response against this weak lymphoma-associated antigen, expanding both CD4+ (helper) (Figure 1D) and CD8+ (cytotoxic) T cells directed to an array of LMP2 epitopes (Figure 1C). Infusion of these T cells was followed by a substantial in vivo increase in the frequency of tumor-reactive T cells, so that as few as 80 million infused T cells were able to produce tumor responses in 5 of 6 patients with measurable lymphoma.
In our previous clinical studies, we treated patients with relapsed EBV-positive Hodgkin disease (HD) using EBV-CTLs in which only a small component of the line was specific for the weak EBV-derived antigens expressed by the tumor cells: only 2 of 11 patients had a complete clinical response, and both had only limited disease.10 By genetically modifying the antigen-presenting cells used to stimulate the cytotoxic T cells (CTLs), we have been able to direct the response away from the immunodominant EBV antigens (EBNA 3A, 3B, 3C) that are absent on the malignant cells,25,30 and toward the weak tumor-associated antigen LMP2.
LMP2 is an EBV-encoded latency protein expressed in up to 40% of all lymphomas.31 It is apparent that EBV-associated lymphomas arising in the immunocompetent host lack expression of viral proteins that dominate the human T-cell response following exposure to the virus.32–34 Most EBV-positive lymphomas arising in the immunocompetent host are characterized as viral latency pattern II, and express the EBV antigens EBNA-1, LMP1, and LMP2. We chose LMP2 as the target antigen for the current trial since EBNA-1 is processed and presented poorly to classical CD8+ T cells35–37 and LMP1 shows significant sequence variability between viral strains.38 By contrast, LMP2 is consistently expressed, and its epitopes are conserved.39–41
To express whole LMP2 protein in APCs, we used an adenovirus vector, Ad5f35, encoding LMP2 as a transgene. This choice allows APCs and T cells to “self-select” the processed peptides they present and react with, so that generation of LMP2-specific T cells is not restricted to patients whose HLA polymorphisms are appropriately matched to the individual peptides used. The use of full-length antigen may have 2 additional benefits. First, T cells may recognize multiple epitopes within the protein as observed in multimer and ELISPOT assays. This decreases the chances of tumor escape due to emergence of epitope loss variants within the malignant population.42,43 While such events cannot be entirely avoided, they are likely to be reduced in frequency if the immune response is directed to multiple distinct regions of a target molecule rather than to a single peptide epitope. Second, combinations of both CD4 and CD8 epitopes that recruit both CD4+ and CD8+ T lymphocytes can be presented, increasing the likelihood that both types of effector cells will be generated, favoring the subsequent sustained expansion of transferred cells and eradication of established lymphoma. Although we do not know if the in vivo expansion we observed was directly attributable to the mix of CD4+ and CD8+ LMP2-specific T cells we infused, clinical effects were observed with as few as 80 million infused cells, and the increase in the frequency ofT cells in peripheral blood implied significant in vivo expansion had occurred. Although these increases in LMP2-specific T cells were observed without prior lymphodepletion, the majority of the patients were lymphopenic at the time of infusion and this may have contributed to subsequent in vivo proliferation.
How essential is our technique of LMP2-CTL enrichment for the antitumor activity we observed? Although we could not confirm that amplification of LMP2 specificity had occurred in 6 lines (2 that were exclusively CD4+) there is consistent corroborative evidence from the remaining 10 of 16 patients to support the value of enrichment. In these individuals, infusion of LMP2-enriched lines produced an increase in the number of LMP2 antigen-binding and tumor antigen–reactive cells not only in the circulation, but also at sites of tumor. Moreover, this accumulation occurred reciprocally to the decline in LMP2+ tumor cells.
Although our study is not randomized, these lymphomas do not undergo spontaneous complete remission, so that the clinical responses we observed are likely causally related to the antitumor activity of the CTLs we infused. Of note, benefits were obtained in the absence of the severe morbidity and mortality that are an inevitable accompaniment to conventional salvage chemoradiation.
Our technique using genetically modified APCs that overexpress weak antigens may have utility for preparing effector cells that target other “weak” tumor-associated antigens, since it favors expansion of T cells present at low frequencies, and with low antigen affinity (Figure 1A). This current study, however, modulates the response to a weak viral-derived tumor antigen and it is possible that it will be less effective for tumor antigens that are of host origin. Even if such a limitation applies, this approach should nonetheless prove valuable for the targeted treatment of other viral-associated malignancies of the head and neck, the cervix, and the hematopoietic system. Furthermore, the clinical outcome we report, taken in conjunction with successful studies in which the T lymphocytes themselves are genetically modified to express tumor-specific receptors,9 demonstrates that the convergence of cellular immunotherapy and gene transfer can be of clinical value.
Acknowledgments
This work was supported in part by National Institutes of Health grants PO1 CA94237 and a Specialized Center of Research Award from the Leukemia Lymphoma Society. H.E.H. was also supported by a Doris Duke Distinguished Clinical Scientist Award. C.M.B. was also supported by the Kimmel Translational Science Award and an award from the Gillson Longenbaugh Foundation. S.G. was supported by a translational research award from the Leukemia Lymphoma Society and a Doris Duke Clinical Scientist Development Award. The clinical trial was also supported in part by Baylor College of Medicine, Texas Children's Hospital, and the Methodist Hospital, Houston, TX. The LMP2 vector was provided by a grant from the National Gene Vector Laboratories (NIH-NCRR U42 RR16578).
We thank V. Torrano for research coordination, T. Gotsolva and A. Durrett for expert technical assistance, and T. Lopez and staff in the Good Manufacturing Practice (GMP) facilities for assisting in CTL preparation and quality assurance.
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: C.M.B. participated in the development of the clinical study, was a co–principal investigator on the clinical trial, cared for the enrolled patients, manufactured CTL lines, performed the characterization of these CTL lines, performed many of the immune reconstitution studies, and contributed to the writing of the paper; S.G. participated in the development of the clinical study and cared for some of the enrolled patients; A.M.L. participated in the generation and administration of the CTLs; H.W. and M.W. provided statistical support; K.C.S. participated in the development of the clinical study and performed characterization of CTL lines; G.C. participated in the clinical study by caring for some of the enrolled lymphoma patients; M.K. performed the characterization of CTL lines and performed the immune reconstitution studies; M.H.H. supervised CTL preparation and quality assurance; C.-C.C. and M.V.G. reviewed tumor samples for EBV positivity; A.P.G. performed quality assurance on all CTL lines prior to clinical use; M.K.B. participated in the development of the clinical study and contributed to the writing of the paper; C.M.R. developed the clinical study and was a co–principal investigator and contributed to the writing of the paper; H.E.H. developed the clinical study and was a co–principal investigator on the clinical trial, cared for some of the enrolled patients, and contributed to the writing of the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Catherine Bollard, Center for Cell and Gene Therapy, Baylor College of Medicine, 6621 Fannin St, MC 3–3320, Houston, TX 77030; e-mail: cmbollar@txccc.org.
References
- 1.Reichert JM, Rosensweig CJ, Faden LB, Dewitz MC. Monoclonal antibody successes in the clinic. Nat Biotechnol. 2005;23:1073–1078. doi: 10.1038/nbt0905-1073. [DOI] [PubMed] [Google Scholar]
- 2.Leen AM, Rooney CM, Foster AE. Improving T cell therapy for cancer. Annu Rev Immunol. 2007;25:243–265. doi: 10.1146/annurev.immunol.25.022106.141527. [DOI] [PubMed] [Google Scholar]
- 3.Gattinoni L, Powell DJ, Jr, Rosenberg SA, Restifo NP. Adoptive immunotherapy for cancer: building on success. Nat Rev Immunol. 2006;6:383–393. doi: 10.1038/nri1842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baumgaertner P, Rufer N, Devevre E, et al. Ex vivo detectable human CD8 T-cell responses to cancer-testis antigens. Cancer Res. 2006;66:1912–1916. doi: 10.1158/0008-5472.CAN-05-3793. [DOI] [PubMed] [Google Scholar]
- 5.Poppema S. Immunobiology and pathophysiology of hodgkin lymphomas. Hematology (Am Soc Hematol Educ Program) 2005:231–238. doi: 10.1182/asheducation-2005.1.231. [DOI] [PubMed] [Google Scholar]
- 6.Yee C, Thompson JA, Byrd D, et al. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci U S A. 2002;99:16168–16173. doi: 10.1073/pnas.242600099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dudley ME, Rosenberg SA. Adoptive-cell-transfer therapy for the treatment of patients with cancer. Nat Rev Cancer. 2003;3:666–675. doi: 10.1038/nrc1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Riddell SR, Greenberg PD. Principles for adoptive T cell therapy of human viral diseases. Annu Rev Immunol. 1995;13:545–586. doi: 10.1146/annurev.iy.13.040195.002553. [DOI] [PubMed] [Google Scholar]
- 9.Morgan RA, Dudley ME, Wunderlich JR, et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science. 2006;314:126–129. doi: 10.1126/science.1129003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bollard CM, Aguilar L, Straathof KC, et al. Cytotoxic T lymphocyte therapy for Epstein-Barr virus+ Hodgkin's disease. J Exp Med. 2004;200:1623–1633. doi: 10.1084/jem.20040890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Roskrow MA, Suzuki N, Gan Y-J, et al. EBV-specific cytotoxic T lymphocytes for the treatment of patients with EBV positive relapsed Hodgkin's disease. Blood. 1998;91:2925–2934. [PubMed] [Google Scholar]
- 12.Levitskaya J, Coram M, Levitsky V, et al. Inhibition of antigen processing by the internal repeat region of the Epstein-Barr virus nuclear antigen-1. Nature. 1995;375:685–688. doi: 10.1038/375685a0. [DOI] [PubMed] [Google Scholar]
- 13.Gottschalk S, Edwards OL, Sili U, et al. Generating CTL against the subdominant Epstein-Barr virus LMP1 antigen for the adoptive Immunotherapy of EBV-associated malignancies. Blood. 2003;101:1905–1912. doi: 10.1182/blood-2002-05-1514. [DOI] [PubMed] [Google Scholar]
- 14.Niedobitek G, Kremmer E, Herbst H, et al. Immunohistochemical detection of the Epstein-Barr virus-encoded latent membrane protein 2A in Hodgkin's disease and infectious mononucleosis. Blood. 1997;90:1664–1672. [PubMed] [Google Scholar]
- 15.Berger C, Rothenberger S, Bachmann E, et al. Sequence polymorphisms between latent membrane proteins LMP1 and LMP2A do not correlate in EBV-associated reactive and malignant lympho-proliferations. Int J Cancer. 1999;81:371–375. doi: 10.1002/(sici)1097-0215(19990505)81:3<371::aid-ijc10>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 16.Bollard CM, Straathof KC, Huls MH, et al. The generation and characterization of LMP2-specific CTLs for use as adoptive transfer from patients with relapsed EBV-positive Hodgkin disease. J Immunother. 2004;27:317–327. doi: 10.1097/00002371-200407000-00008. [DOI] [PubMed] [Google Scholar]
- 17.Leen AM, Myers GD, Sili U, et al. Monoculture-derived T lymphocytes specific for multiple viruses expand and produce clinically relevant effects in immunocompromised individuals. Nat Med. 2006;12:1160–1166. doi: 10.1038/nm1475. [DOI] [PubMed] [Google Scholar]
- 18.Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors: European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–216. doi: 10.1093/jnci/92.3.205. [DOI] [PubMed] [Google Scholar]
- 19.Roskrow MA, Rooney CM, Heslop HE, et al. Administration of neomycin resistance gene marked EBV specific cytotoxic T-lymphocytes to patients with relapsed EBV-positive Hodgkin disease. Hum Gene Ther. 1998;9:1237–1250. doi: 10.1089/hum.1998.9.8-1237. [DOI] [PubMed] [Google Scholar]
- 20.Kaech SM, Tan JT, Wherry EJ, et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat Immunol. 2003;4:1191–1198. doi: 10.1038/ni1009. [DOI] [PubMed] [Google Scholar]
- 21.Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 22.Lanzavecchia A, Sallusto F. Progressive differentiation and selection of the fittest in the immune response. Nat Rev Immunol. 2002;2:982–987. doi: 10.1038/nri959. [DOI] [PubMed] [Google Scholar]
- 23.Straathof KC, Leen AM, Buza EL, et al. Characterization of latent membrane protein 2 specificity in CTL lines from patients with EBV-positive nasopharyngeal carcinoma and lymphoma. J Immunol. 2005;175:4137–4147. doi: 10.4049/jimmunol.175.6.4137. [DOI] [PubMed] [Google Scholar]
- 24.Smith CA, Ng CYC, Heslop HE, et al. Production of genetically modified EBV-specific cytotoxic T cells for adoptive transfer to patients at high risk of EBV-associated lymphoproliferative disease. J Hematother. 1995;4:73–79. doi: 10.1089/scd.1.1995.4.73. [DOI] [PubMed] [Google Scholar]
- 25.Rooney CM, Smith CA, Ng CYC, et al. Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood. 1998;92:1549–1555. [PubMed] [Google Scholar]
- 26.Wagner HJ, Cheng YC, Huls MH, et al. Prompt versus preemptive intervention for EBV lymphoproliferative disease. Blood. 2004;103:3979–3981. doi: 10.1182/blood-2003-12-4287. [DOI] [PubMed] [Google Scholar]
- 27.Meij P, Leen A, Rickinson AB, et al. Identification and prevalence of CD8(+) T-cell responses directed against Epstein-Barr virus-encoded latent membrane protein 1 and latent membrane protein 2. Int J Cancer. 2002;99:93–99. doi: 10.1002/ijc.10309. [DOI] [PubMed] [Google Scholar]
- 28.Kern F, Faulhaber N, Frommel C, et al. Analysis of CD8 T cell reactivity to cytomegalovirus using protein-spanning pools of overlapping pentadecapeptides. Eur J Immunol. 2000;30:1676–1682. doi: 10.1002/1521-4141(200006)30:6<1676::AID-IMMU1676>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 29.Chang ST, Ghosh D, Kirschner DE, Linderman JJ. Peptide length-based prediction of peptide-MHC class II binding. Bioinformatics. 2006;22:2761–2767. doi: 10.1093/bioinformatics/btl479. [DOI] [PubMed] [Google Scholar]
- 30.Steven NM, Annels NE, Kumar A, et al. Immediate early and early lytic cycle proteins are frequent targets of the Epstein-Barr virus-induced cytotoxic T cell response. J Exp Med. 1997;185:1605–1617. doi: 10.1084/jem.185.9.1605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ambinder RF, Lemas MV, Moore S, et al. Epstein-Barr virus and lymphoma. Cancer Treat Res. 1999;99:27–45. [PubMed] [Google Scholar]
- 32.Pallesen G, Hamilton-Dutoit SJ, Rowe M, Young LS. Expression of Epstein-Barr virus latent gene products in tumour cells of Hodgkin's disease. Lancet. 1991;337:320–322. doi: 10.1016/0140-6736(91)90943-j. [DOI] [PubMed] [Google Scholar]
- 33.Weiss LM, Movahed LA, Warnke RA, Sklar J. Detection of Epstein-Barr viral genomes in Reed-Sternberg cells of Hodgkin's disease. N Engl J Med. 1989;320:502–506. doi: 10.1056/NEJM198902233200806. [DOI] [PubMed] [Google Scholar]
- 34.Thorley-Lawson DA, Gross A. Persistence of the Epstein-Barr virus and the origins of associated lymphomas. N Engl J Med. 2004;350:1328–1337. doi: 10.1056/NEJMra032015. [DOI] [PubMed] [Google Scholar]
- 35.Voo KS, Fu T, Wang HY, et al. Evidence for the presentation of major histocompatibility complex class i-restricted Epstein-Barr virus nuclear antigen 1 peptides to CD8+ T lymphocytes. J Exp Med. 2004;199:459–470. doi: 10.1084/jem.20031219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nikiforow S, Bottomly K, Miller G. CD4+ T-cell effectors inhibit Epstein-Barr virus-induced B-cell proliferation. J Virol. 2001;75:3740–3752. doi: 10.1128/JVI.75.8.3740-3752.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Levitskaya J, Sharipo A, Leonchiks A, Ciechanover A, Masucci MG. Inhibition of ubiquitin/proteasome-dependent protein degradation by the Gly-Ala repeat domain of the Epstein-Barr virus nuclear antigen 1. Proc Natl Acad Sci U S A. 1997;94:12616–12621. doi: 10.1073/pnas.94.23.12616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Khanim F, Yao QY, Niedobitek G, et al. Analysis of Epstein-Barr virus gene polymorphisms in normal donors and in virus-associated tumors from different geographic locations. Blood. 1996;88:3491–3501. [PubMed] [Google Scholar]
- 39.Murray PG, Constandinou CM, Crocker J, Young LS, Ambinder RF. Analysis of major histocompatibility complex class I, TAP expression, and LMP2 epitope sequence in Epstein-Barr virus-positive Hodgkin's disease. Blood. 1998;92:2477–2483. [PubMed] [Google Scholar]
- 40.Busson P, Edwards RH, Tursz T, Raab-Traub N. Sequence polymorphism in the Epstein-Barr virus latent membrane protein (LMP)-2 gene. J Gen Virol. 1995;76(pt 1):139–145. doi: 10.1099/0022-1317-76-1-139. [DOI] [PubMed] [Google Scholar]
- 41.Lee SP, Thomas WA, Murray RJ, et al. HLA A2 1-restricted cytotoxic T cells recognizing a range of Epstein-Barr virus isolates through a defined epitope in latent membrane protein LMP2. J Virol. 1993;67:7428–7435. doi: 10.1128/jvi.67.12.7428-7435.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gottschalk S, Ng CYC, Smith CA, et al. An Epstein-Barr virus deletion mutant that causes fatal lymphoproliferative disease unresponsive to virus-specific T cell therapy. Blood. 2001;97:835–843. doi: 10.1182/blood.v97.4.835. [DOI] [PubMed] [Google Scholar]
- 43.Khong HT, Restifo NP. Natural selection of tumor variants in the generation of “tumor escape” phenotypes. Nat Immunol. 2002;3:999–1005. doi: 10.1038/ni1102-999. [DOI] [PMC free article] [PubMed] [Google Scholar]