Abstract
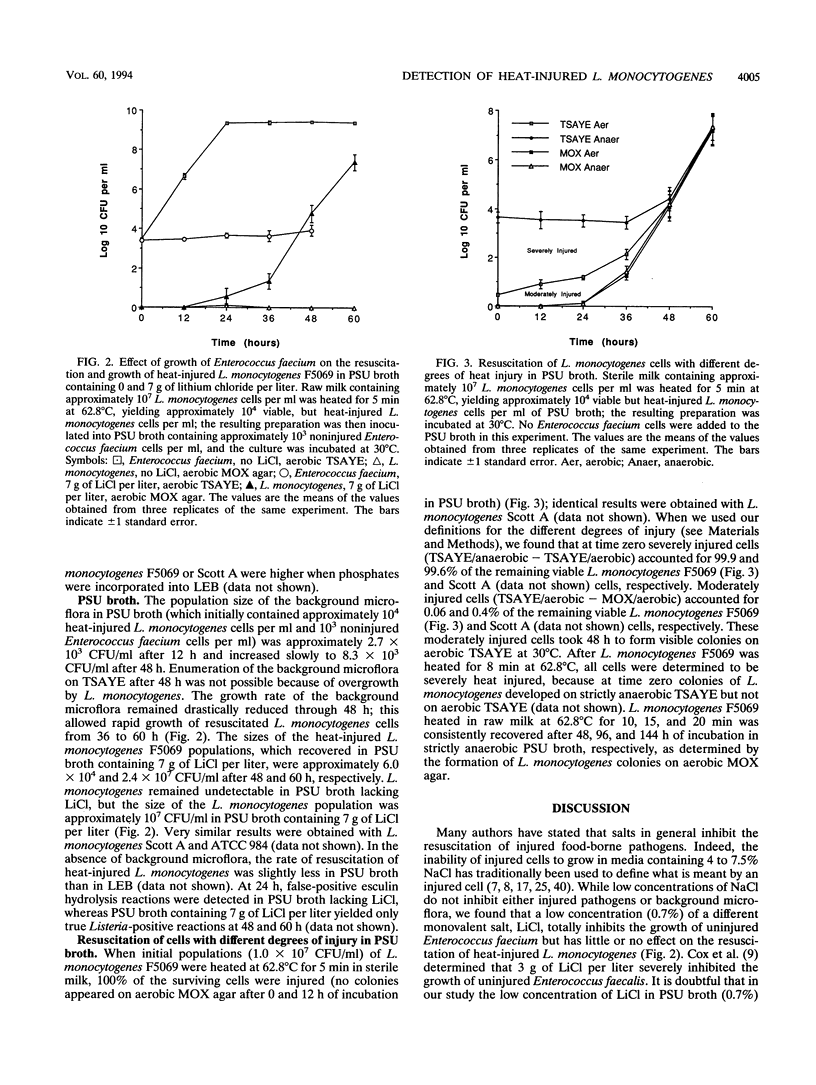
Heat-injured cells of Listeria monocytogenes were recovered from heated raw milk containing noninjured Enterococcus faecium by combining a simple method for obtaining strict anaerobiosis with a novel enrichment broth, Penn State University broth (PSU broth). Strictly anaerobic conditions were rapidly achieved by adding 0.5 g of filter-sterilized cysteine per liter to PSU broth and then purging the preparation with N2 gas. Little resuscitation or growth occurred in strictly anaerobic PSU broth without lithium chloride because of overgrowth by E. faecium. The growth of E. faecium decreased dramatically with increasing LiCl concentration; LiCl concentrations of 8 and 10 g/liter were completely bacteriostatic. The mechanism of inhibition by LiCl appeared to involve competition with the divalent cations Ca2+ and Mg2+. Heat-injured L. monocytogenes consistently recovered and grew rapidly in strictly anaerobic PSU broth containing 4, 6, or 7 g of LiCl per liter. The use of strictly anaerobic PSU broth containing 7 g of LiCl per liter permitted detection of severely heat-injured L. monocytogenes in one simple recovery-enrichment step by eliminating oxygen toxicity and inhibiting the growth of background microflora, without preventing the resuscitation and subsequent growth of heat-injured L. monocytogenes. L. monocytogenes heated in raw milk at 62.8 degrees C for 10, 15, and 20 min could be consistently recovered from strictly anaerobic PSU broth enrichment cultures at 30 degrees C after 48, 96, and 144 h, respectively, and hence, use of PSU broth may result in better recovery of both injured and noninjured cells from foods than currently used U.S. Department of Agriculture and Food and Drug Administration preenrichment procedures.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birch N. J. Letter: Lithium and magnesium-dependent enzymes. Lancet. 1974 Oct 19;2(7886):965–966. doi: 10.1016/s0140-6736(74)91187-8. [DOI] [PubMed] [Google Scholar]
- Budu-Amoako E., Toora S., Ablett R. F., Smith J. Evaluation of the ability of primary selective enrichment to resuscitate heat-injured and freeze-injured Listeria monocytogenes cells. Appl Environ Microbiol. 1992 Sep;58(9):3177–3179. doi: 10.1128/aem.58.9.3177-3179.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bunning V. K., Donnelly C. W., Peeler J. T., Briggs E. H., Bradshaw J. G., Crawford R. G., Beliveau C. M., Tierney J. T. Thermal inactivation of Listeria monocytogenes within bovine milk phagocytes. Appl Environ Microbiol. 1988 Feb;54(2):364–370. doi: 10.1128/aem.54.2.364-370.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busch S. V., Donnelly C. W. Development of a repair-enrichment broth for resuscitation of heat-injured Listeria monocytogenes and Listeria innocua. Appl Environ Microbiol. 1992 Jan;58(1):14–20. doi: 10.1128/aem.58.1.14-20.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark C. W., Witter L. D., Ordal Z. J. Thermal injury and recovery of Streptococcus faecalis. Appl Microbiol. 1968 Nov;16(11):1764–1769. doi: 10.1128/am.16.11.1764-1769.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford R. G., Beliveau C. M., Peeler J. T., Donnelly C. W., Bunning V. K. Comparative recovery of uninjured and heat-injured Listeria monocytogenes cells from bovine milk. Appl Environ Microbiol. 1989 Jun;55(6):1490–1494. doi: 10.1128/aem.55.6.1490-1494.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallmier A. W., Martin S. E. Catalase and superoxide dismutase activities after heat injury of Listeria monocytogenes. Appl Environ Microbiol. 1988 Feb;54(2):581–582. doi: 10.1128/aem.54.2.581-582.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doyle M. P., Glass K. A., Beery J. T., Garcia G. A., Pollard D. J., Schultz R. D. Survival of Listeria monocytogenes in milk during high-temperature, short-time pasteurization. Appl Environ Microbiol. 1987 Jul;53(7):1433–1438. doi: 10.1128/aem.53.7.1433-1438.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farr S. B., D'Ari R., Touati D. Oxygen-dependent mutagenesis in Escherichia coli lacking superoxide dismutase. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8268–8272. doi: 10.1073/pnas.83.21.8268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez Garayzabal J. F., Dominguez Rodriguez L., Vazquez Boland J. A., Rodriguez Ferri E. F., Briones Dieste V., Blanco Cancelo J. L., Suarez Fernandez G. Survival of Listeria monocytogenes in raw milk treated in a pilot plant size pasteurizer. J Appl Bacteriol. 1987 Dec;63(6):533–537. doi: 10.1111/j.1365-2672.1987.tb02723.x. [DOI] [PubMed] [Google Scholar]
- Fleming D. W., Cochi S. L., MacDonald K. L., Brondum J., Hayes P. S., Plikaytis B. D., Holmes M. B., Audurier A., Broome C. V., Reingold A. L. Pasteurized milk as a vehicle of infection in an outbreak of listeriosis. N Engl J Med. 1985 Feb 14;312(7):404–407. doi: 10.1056/NEJM198502143120704. [DOI] [PubMed] [Google Scholar]
- Hayes P. S., Feeley J. C., Graves L. M., Ajello G. W., Fleming D. W. Isolation of Listeria monocytogenes from raw milk. Appl Environ Microbiol. 1986 Feb;51(2):438–440. doi: 10.1128/aem.51.2.438-440.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iandolo J. J., Ordal Z. J. Repair of thermal injury of Staphylococcus aureus. J Bacteriol. 1966 Jan;91(1):134–142. doi: 10.1128/jb.91.1.134-142.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imlay J. A., Chin S. M., Linn S. Toxic DNA damage by hydrogen peroxide through the Fenton reaction in vivo and in vitro. Science. 1988 Apr 29;240(4852):640–642. doi: 10.1126/science.2834821. [DOI] [PubMed] [Google Scholar]
- Imlay J. A., Fridovich I. Assay of metabolic superoxide production in Escherichia coli. J Biol Chem. 1991 Apr 15;266(11):6957–6965. [PubMed] [Google Scholar]
- Knabel S. J., Walker H. W., Hartman P. A., Mendonca A. F. Effects of growth temperature and strictly anaerobic recovery on the survival of Listeria monocytogenes during pasteurization. Appl Environ Microbiol. 1990 Feb;56(2):370–376. doi: 10.1128/aem.56.2.370-376.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovett J. Isolation and identification of Listeria monocytogenes in dairy products. J Assoc Off Anal Chem. 1988 May-Jun;71(3):658–660. [PubMed] [Google Scholar]
- McCarthy S. A. Pathogenicity of nonstressed, heat-stressed, and resuscitated Listeria monocytogenes 1A1 cells. Appl Environ Microbiol. 1991 Aug;57(8):2389–2391. doi: 10.1128/aem.57.8.2389-2391.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClain D., Lee W. H. Development of USDA-FSIS method for isolation of Listeria monocytogenes from raw meat and poultry. J Assoc Off Anal Chem. 1988 May-Jun;71(3):660–664. [PubMed] [Google Scholar]
- McCord J. M., Keele B. B., Jr, Fridovich I. An enzyme-based theory of obligate anaerobiosis: the physiological function of superoxide dismutase. Proc Natl Acad Sci U S A. 1971 May;68(5):1024–1027. doi: 10.1073/pnas.68.5.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niederhauser C., Candrian U., Höfelein C., Jermini M., Bühler H. P., Lüthy J. Use of polymerase chain reaction for detection of Listeria monocytogenes in food. Appl Environ Microbiol. 1992 May;58(5):1564–1568. doi: 10.1128/aem.58.5.1564-1568.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ninet B., Bannerman E., Bille J. Assessment of the Accuprobe Listeria monocytogenes culture identification reagent kit for rapid colony confirmation and its application in various enrichment broths. Appl Environ Microbiol. 1992 Dec;58(12):4055–4059. doi: 10.1128/aem.58.12.4055-4059.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sogin S. J., Ordal Z. J. Regeneration of ribosomes and ribosomal ribonucleic acid during repair of thermal injury to Staphylococcus. J Bacteriol. 1967 Oct;94(4):1082–1087. doi: 10.1128/jb.94.4.1082-1087.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler D. D. Role of superoxide radicals in the lipid peroxidation of intracellular membranes. FEBS Lett. 1975 Mar 1;51(1):180–183. doi: 10.1016/0014-5793(75)80882-9. [DOI] [PubMed] [Google Scholar]
- Umeda K., Shiota S., Futai M., Tsuchiya T. Inhibitory effect of Li+ on cell growth and pyruvate kinase activity of Escherichia coli. J Bacteriol. 1984 Nov;160(2):812–814. doi: 10.1128/jb.160.2.812-814.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R. F., Cao W. W., Johnson M. G. 16S rRNA-based probes and polymerase chain reaction method to detect Listeria monocytogenes cells added to foods. Appl Environ Microbiol. 1992 Sep;58(9):2827–2831. doi: 10.1128/aem.58.9.2827-2831.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warburton D. W., Farber J. M., Armstrong A., Caldeira R., Hunt T., Messier S., Plante R., Tiwari N. P., Vinet J. A comparative study of the 'FDA' and 'USDA' methods for the detection of Listeria monocytogenes in foods. Int J Food Microbiol. 1991 Jun;13(2):105–117. doi: 10.1016/0168-1605(91)90053-r. [DOI] [PubMed] [Google Scholar]
- van Netten P., Perales I., van de Moosdijk A., Curtis G. D., Mossel D. A. Liquid and solid selective differential media for the detection and enumeration of L. monocytogenes and other Listeria spp. Int J Food Microbiol. 1989 Jul;8(4):299–316. doi: 10.1016/0168-1605(89)90001-9. [DOI] [PubMed] [Google Scholar]