Abstract
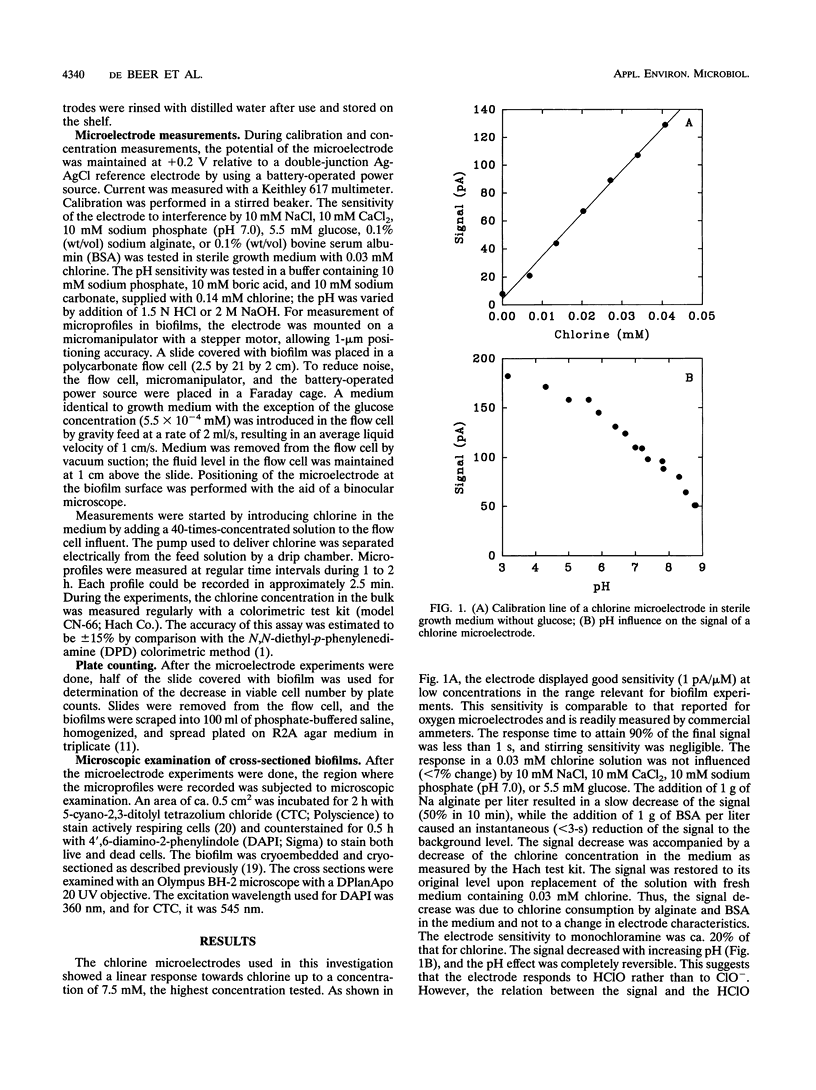
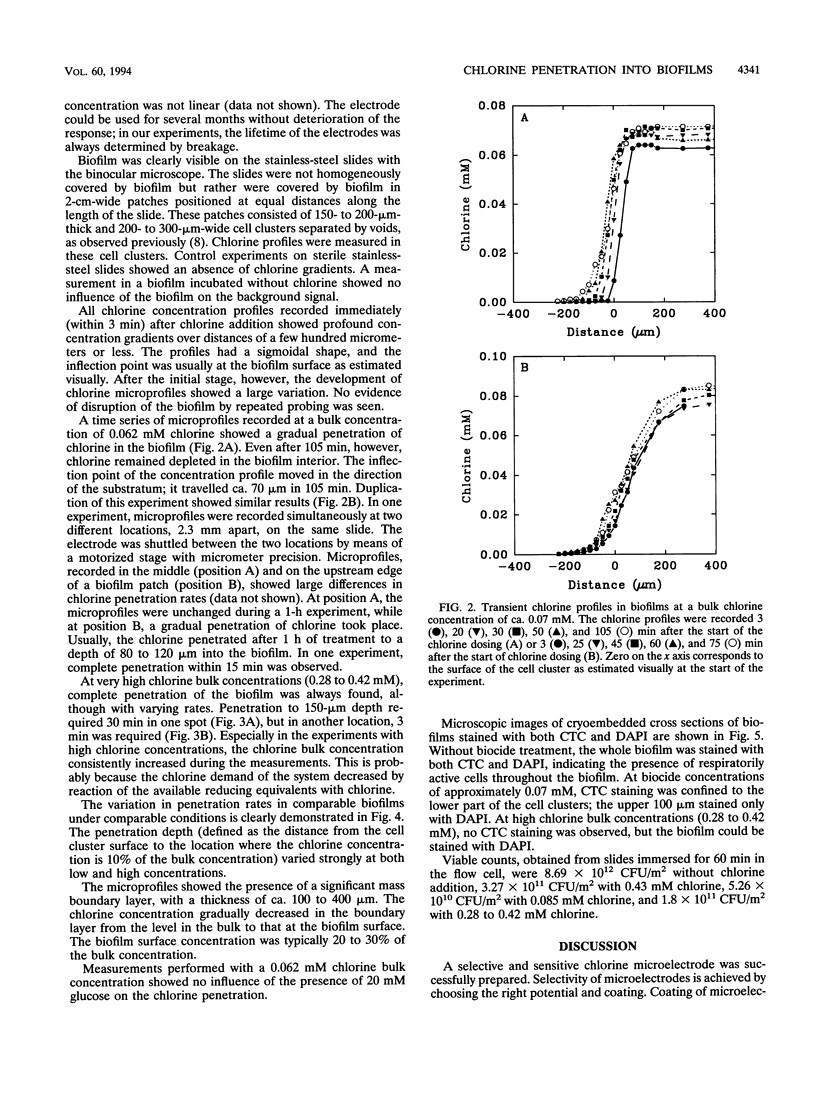
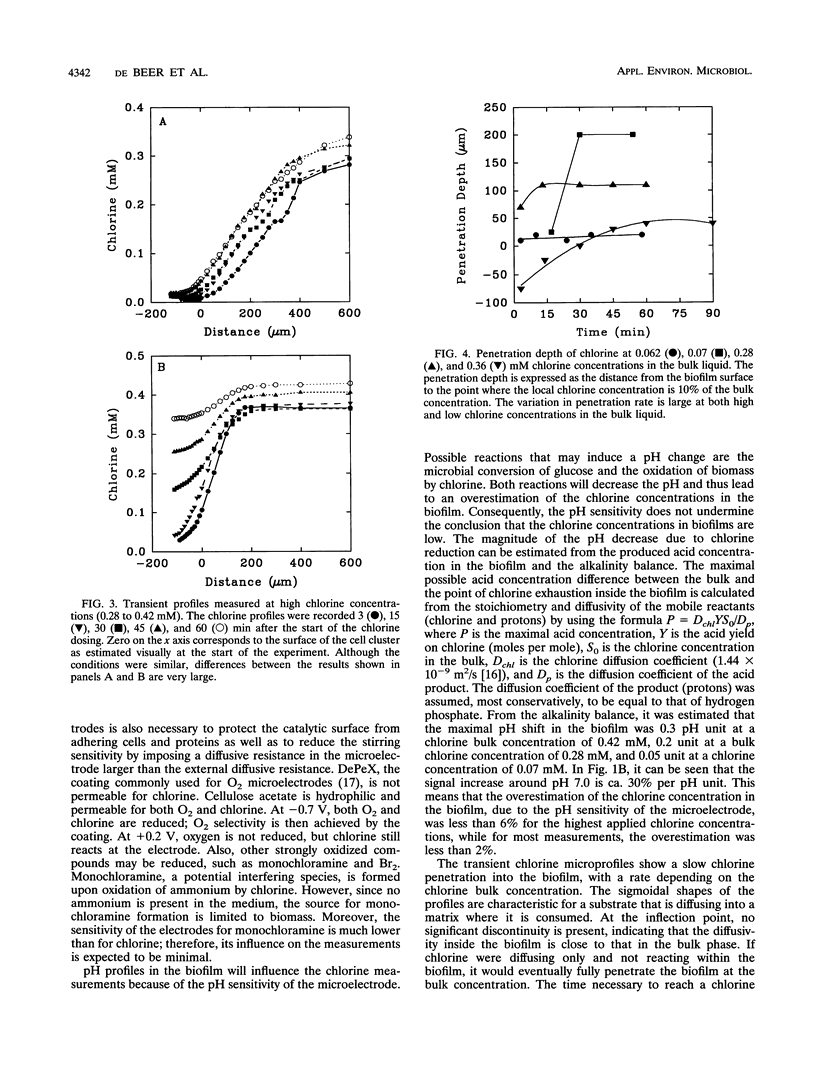
Transient chlorine concentration profiles were measured in biofilms during disinfection by use of a microelectrode developed for this investigation. The electrode had a tip diameter of ca. 10 microm and was sensitive to chlorine in the micromolar range. The biofilms contained Pseudomonas aeruginosa and Klebsiella pneumoniae. Chlorine concentrations measured in biofilms were typically only 20% or less of the concentration in the bulk liquid. Complete equilibration with the bulk liquid did not occur during the incubation time of 1 to 2 h. The penetration depth of chlorine into the biofilm and rate of penetration varied depending on the measurement location, reflecting heterogeneity in the distribution of biomass and in local hydrodynamics. The shape of the chlorine profiles, the long equilibration times, and the dependence on the bulk chlorine concentration showed that the penetration was a function of simultaneous reaction and diffusion of chlorine in the biofilm matrix. Frozen cross sections of biofilms, stained with a redox dye and a DNA stain, showed that the area of chlorine penetration overlapped with nonrespiring zones near the biofilm-bulk fluid interface. These data indicate that the limited penetration of chlorine into the biofilm matrix is likely to be an important factor influencing the reduced efficacy of this biocide against biofilms as compared with its action against planktonic cells.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Costerton J. W., Cheng K. J., Geesey G. G., Ladd T. I., Nickel J. C., Dasgupta M., Marrie T. J. Bacterial biofilms in nature and disease. Annu Rev Microbiol. 1987;41:435–464. doi: 10.1146/annurev.mi.41.100187.002251. [DOI] [PubMed] [Google Scholar]
- LeChevallier M. W., Cawthon C. D., Lee R. G. Factors promoting survival of bacteria in chlorinated water supplies. Appl Environ Microbiol. 1988 Mar;54(3):649–654. doi: 10.1128/aem.54.3.649-654.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeChevallier M. W., Cawthon C. D., Lee R. G. Inactivation of biofilm bacteria. Appl Environ Microbiol. 1988 Oct;54(10):2492–2499. doi: 10.1128/aem.54.10.2492-2499.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu F. P., McFeters G. A. Rapid in situ assessment of physiological activities in bacterial biofilms using fluorescent probes. J Microbiol Methods. 1994;20:1–10. doi: 10.1016/0167-7012(94)90058-2. [DOI] [PubMed] [Google Scholar]
- de Beer D., van den Heuvel J. C., Ottengraf S. P. Microelectrode measurements of the activity distribution in nitrifying bacterial aggregates. Appl Environ Microbiol. 1993 Feb;59(2):573–579. doi: 10.1128/aem.59.2.573-579.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]



