Abstract
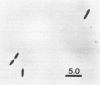
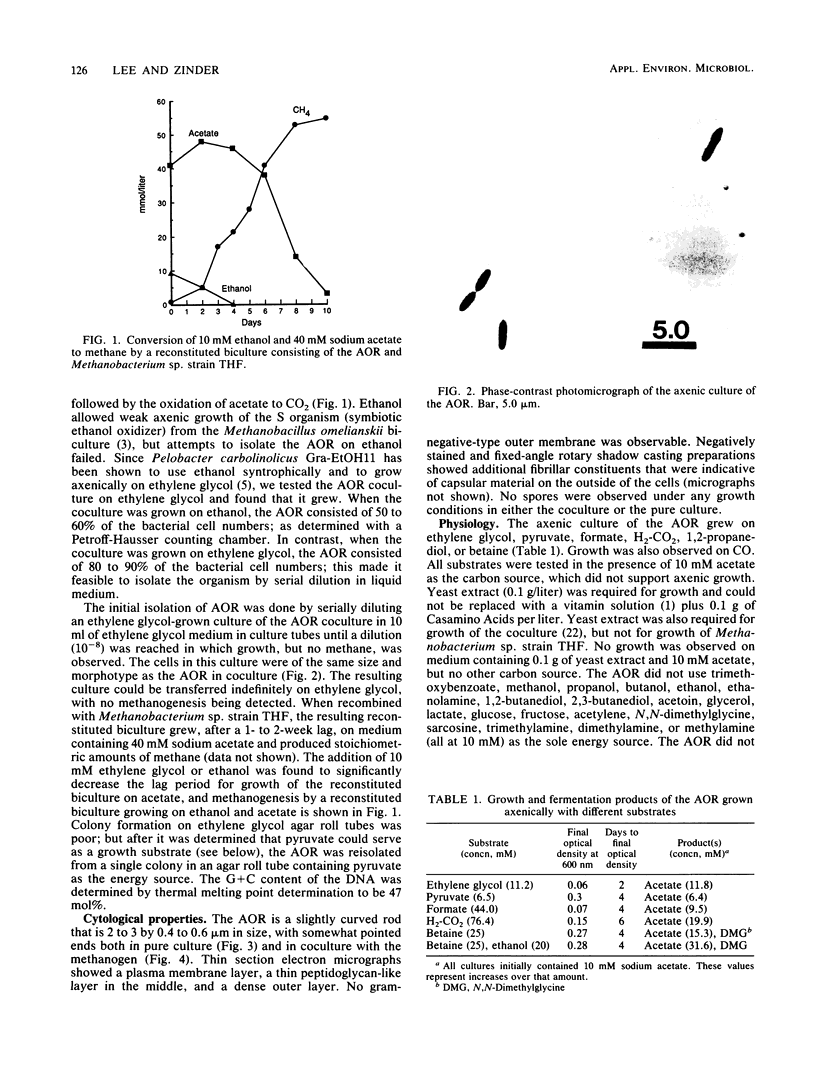
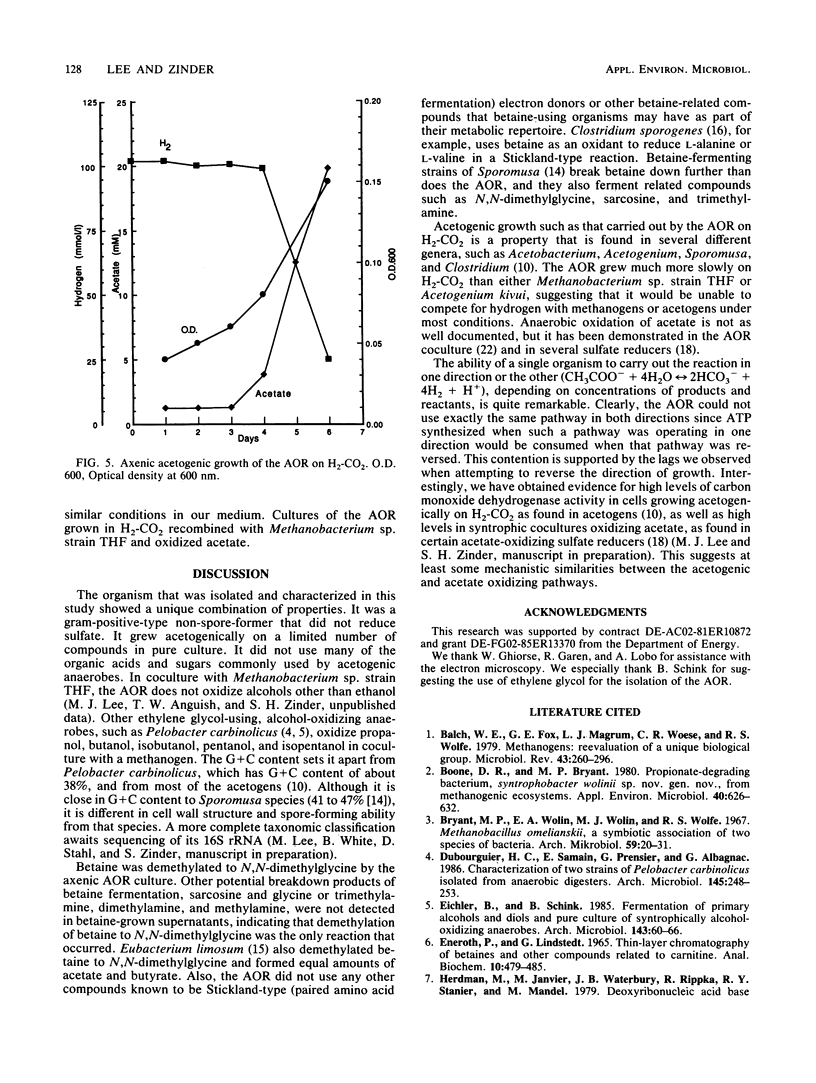
We previously described a thermophilic (60°C), syntrophic, two-membered culture which converted acetate to methane via a two-step mechanism in which acetate was oxidized to H2 and CO2. While the hydrogenotrophic methanogen Methanobacterium sp. strain THF in the biculture was readily isolated, we were unable to find a substrate that was suitable for isolation of the acetate-oxidizing member of the biculture. In this study, we found that the biculture grew on ethylene glycol, and an acetate-oxidizing, rod-shaped bacterium (AOR) was isolated from the biculture by dilution into medium containing ethylene glycol as the growth substrate. When the axenic culture of the AOR was recombined with a pure culture of Methanobacterium sp. strain THF, the reconstituted biculture grew on acetate and converted it to CH4. The AOR used ethylene glycol, 1,2-propanediol, formate, pyruvate, glycine-betaine, and H2-CO2 as growth substrates. Acetate was the major fermentation product detected from these substrates, except for 1,2-propanediol, which was converted to 1-propanol and propionate. N,N-Dimethylglycine was also formed from glycine-betaine. Acetate was formed in stoichiometric amounts during growth on H2-CO2, demonstrating that the AOR is an acetogen. This reaction, which was carried out by the pure culture of the AOR in the presence of high partial pressures of H2, was the reverse of the acetate oxidation reaction carried out by the AOR when hydrogen partial pressures were kept low by coculturing it with Methanobacterium sp. strain THF. The DNA base composition of the AOR was 47 mol% guanine plus cytosine, and no cytochromes were detected.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balch W. E., Fox G. E., Magrum L. J., Woese C. R., Wolfe R. S. Methanogens: reevaluation of a unique biological group. Microbiol Rev. 1979 Jun;43(2):260–296. doi: 10.1128/mr.43.2.260-296.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boone D. R., Bryant M. P. Propionate-Degrading Bacterium, Syntrophobacter wolinii sp. nov. gen. nov., from Methanogenic Ecosystems. Appl Environ Microbiol. 1980 Sep;40(3):626–632. doi: 10.1128/aem.40.3.626-632.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryant M. P., Wolin E. A., Wolin M. J., Wolfe R. S. Methanobacillus omelianskii, a symbiotic association of two species of bacteria. Arch Mikrobiol. 1967;59(1):20–31. doi: 10.1007/BF00406313. [DOI] [PubMed] [Google Scholar]
- Ljungdahl L. G. The autotrophic pathway of acetate synthesis in acetogenic bacteria. Annu Rev Microbiol. 1986;40:415–450. doi: 10.1146/annurev.mi.40.100186.002215. [DOI] [PubMed] [Google Scholar]
- McInerney M. J., Bryant M. P., Hespell R. B., Costerton J. W. Syntrophomonas wolfei gen. nov. sp. nov., an Anaerobic, Syntrophic, Fatty Acid-Oxidizing Bacterium. Appl Environ Microbiol. 1981 Apr;41(4):1029–1039. doi: 10.1128/aem.41.4.1029-1039.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller E., Fahlbusch K., Walther R., Gottschalk G. Formation of N,N-Dimethylglycine, Acetic Acid, and Butyric Acid from Betaine by Eubacterium limosum. Appl Environ Microbiol. 1981 Sep;42(3):439–445. doi: 10.1128/aem.42.3.439-445.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naumann E., Hippe H., Gottschalk G. Betaine: New Oxidant in the Stickland Reaction and Methanogenesis from Betaine and l-Alanine by a Clostridium sporogenes-Methanosarcina barkeri Coculture. Appl Environ Microbiol. 1983 Feb;45(2):474–483. doi: 10.1128/aem.45.2.474-483.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RYTER A., KELLENBERGER E., BIRCHANDERSEN A., MAALOE O. Etude au microscope électronique de plasmas contenant de l'acide désoxyribonucliéique. I. Les nucléoides des bactéries en croissance active. Z Naturforsch B. 1958 Sep;13B(9):597–605. [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]