Abstract
To elucidate the role of IgE-dependent mechanisms in inducing altered airway responsiveness in the atopic asthmatic state, the expression and actions of Fc receptor activation were examined in isolated rabbit tracheal smooth muscle (TSM) tissue and cultured cells passively sensitized with sera from atopic asthmatic patients or nonatopic/nonasthmatic (control) subjects. Relative to control tissues, the atopic asthmatic-sensitized TSM exhibited significantly increased maximal isometric contractility to acetylcholine (P < 0.01) and attenuated maximal relaxation responses and sensitivity (i.e., −log ED50) to isoproterenol (P < 0.005). These changes in agonist responsiveness in atopic sensitized TSM were ablated by pretreating the tissues with a blocking mAb to the low affinity receptor for IgE, FcɛRII (i.e., CD23) or by depleting the sensitizing serum of its immune complexes. Moreover, in complimentary experiments, exogenous administration of IgE immune complexes to naive TSM produced changes in agonist responsiveness that were qualitatively similar to those obtained in the atopic asthmatic-sensitized state. Extended studies further demonstrated that, in contrast to their respective controls, atopic asthmatic serum-sensitized human and rabbit TSM tissue and cultured cells exhibited markedly induced mRNA and cell surface expression of FcɛRII, whereas constitutive expression of the IgG receptor subtype, FcγRIII, was unaltered. Finally, the up-regulated mRNA expression of FcɛRII observed following exposure of TSM to atopic asthmatic serum or to exogenously administered IgE immune complexes was significantly inhibited by pretreating the tissues or cells with anti-CD23 mAb. Collectively, these observations provide evidence demonstrating that the altered agonist responsiveness in atopic asthmatic sensitized airway smooth muscle is largely attributed to IgE-mediated induction of the autologous expression and activation of FcɛRII receptors in the airway smooth muscle itself.
Bronchial asthma is characterized by airways inflammation, exaggerated airway reactivity to bronchoconstrictor agonists, and attenuated β-adrenoreceptor-mediated airway relaxation (1–3). In individuals with atopic asthma, mast cell activation has been implicated in mediating the immediate bronchoconstrictor response acutely following antigen inhalation, a process involving IgE-mediated activation of the high affinity IgE receptor (FcɛRI), leading to cellular degranulation and the release of various mast cell-derived mediators (4–6). The identification of Fc receptors on other cell types in the lung (e.g., mononuclear cells, eosinophils, and dendritic cells) suggests that, apart from mast cells per se, these other cell types may also serve to propagate the pro-inflammatory allergic pulmonary response, most likely via the orchestrated extended release of various cytokines (7–13). Accordingly, immune complex/Fc receptor interactions potentially underlie the progression of the airway inflammatory and bronchoconstrictor responses in asthma, wherein the immediate bronchoconstriction accompanying antigen exposure is followed by the development of the late phase asthmatic response involving various proinflammatory cells. Indeed, recent studies have demonstrated that expression of the inducible form of the low affinity IgE receptor (FcɛRII or CD23) is up-regulated on monocytes and alveolar macrophages (14), as well as on circulating B lymphocytes (15, 16) isolated from atopic asthmatic subjects.
In light of the above evidence, together with our recent observations demonstrating that exposure of isolated rabbit and human airway smooth muscle (ASM) to atopic asthmatic serum induces the autocrine release and action of specific cytokines [notably interleukin (IL)-1β] by the sensitized ASM cells (18), the present study examined whether ASM cells have the capacity to intrinsically express Fc receptors and whether activation of these receptors is altered in the atopic asthmatic sensitized state and contributes to changes in agonist responsiveness of the ASM tissue. The results provide evidence that ASM cells intrinsically express Fc receptors and that the induced altered responsiveness of atopic asthmatic sensitized ASM is largely attributed to its autologously up-regulated expression and activation of the FcɛRII receptor subtype. These findings identify a critical role for the ASM itself in autologously regulating IgE/CD23-coupled changes in airway reactivity that characterize the asthmatic state.
MATERIALS AND METHODS
Preparation and Sensitization of ASM Tissue.
New Zealand White rabbits were sacrificed with an overdose of pentobarbital (130 mg/kg), the tracheae were removed, divided into ring segments, and incubated for 24 hr at room temperature in either (i) human serum containing IgE >1,000 units/ml obtained from allergic patients with moderate to severe asthma and 4–5/6+ radioallergosorbent test (RAST) positive (specific IgE concentration of more than 17.5 Phadebas RAST units/ml) to Dermatophagoides pteronyssimus, Dermatophagoides farinae, and ragweed, and positive skin test to these antigens, or (ii) human serum from nonatopic nonasthmatic individuals with normal serum IgE levels (i.e., <70 units/ml) (19). In parallel experiments, tracheal smooth muscle (TSM) segments were incubated in either control serum or in atopic asthmatic serum that was (i) depleted of its Ig complexes by pretreatment with Sepharose Staph protein A (SpA), as described (20); (ii) coincubated with an anti-CD23 (40 μg/ml) blocking mAb; or (iii) coincubated with an anti-CD16 (2 μg/ml) blocking mAb. Finally, paired TSM segments were incubated for 24 hr in oxygenated DMEM, as described (21), containing either (i) human IgE [final bath concentration (FBC), 15 μg/ml], or (ii) human IgE–goat-anti-human IgE immune complexes (FBC, 15:5 μg/ml of IgE/anti-IgE).
The passive sensitization protocol described above was also conducted on human TSM isolated 1 hr postmortem from a 53-year-old male who had no evidence of lung disease, as well as a surgically resected human bronchial smooth muscle segment obtained from a 72-year-old female with peripheral lung carcinoma. The ASM isolates were incubated in either control or atopic asthmatic serum in the absence or presence of an anti-CD23 antibody, and subsequently examined for Fc receptor expression (see below).
Preparation and Sensitization of ASM Cells.
ASM cells were isolated and subcultivated as described (22). For the sensitization protocol, cells were grown to confluence in Ham’s F-12 medium containing 20% fetal bovine serum. The culture medium was then replaced with Ham’s F-12 medium for 24 hr and subsequently changed to F-12 containing (i) 10% human serum containing >1,000 units/ml IgE obtained from allergic patients with moderate to severe asthma (as above), (ii) 10% human serum from nonatopic nonasthmatic (control) individuals (21), (iii) IgE immune complexes (15:5 μg/ml), or (iv) F-12 alone for the various time points.
Pharmacodynamic Studies.
After incubation, the rabbit airway segments were suspended in organ baths for measurement of isometric contractility, as described (23). Cholinergic contractility was assessed by cumulative administration of acetylcholine (ACh; 10−10 to 10−3 M). Thereafter, relaxation dose–response curves to isoproterenol (10−10–10−4 M) were conducted in tissues half-maximally contracted with ACh. The relaxant responses to isoproterenol were analyzed in terms of % maximal relaxation (Rmax) from the active cholinergic contraction, and sensitivity to the relaxing agent was determined as the negative logarithm of the dose of the relaxing agent producing 50% of Rmax (pD50) (i.e., geometric mean ED50 value).
Determination of Fc Receptor mRNA by Reverse Transcription–PCR (RT-PCR).
Total RNA was isolated from sensitized and control ASM cells and tissue as described (24). cDNA was synthesized by using 2.5 μg of total RNA isolated from the samples. The cDNA was primed with oligo(dT)12–18 and with random hexamer nucleotides (N6) in the absence of signal from the former method. The Fc receptor primers used for PCR were based on the published sequences of the human Fcγ -RI, -RII, -RIII, and FcɛRII genes (25, 26) and included the following primer sets: FcγRI: 5′ primer, 5′-ATGTGGTTCTTGACAACTCTGCTC-3′ and 3′ primer, 5′-ATGTCTGTCTTCTTGAAGGCTGGA-3′; FcγRIIA,C: 5′ primer, 5′-GACTCCATTCAGTGGTTCCA-3′; and 3′ primer: 5′-GTCAGCTGTTTCATAGTCATTG-3′; FcγRIIB: 5′ primer, 5′-GACTCCATTCAGTGGTTCCA-3′ and 3′ primer, 5′-CCCAACTTTGTCAGCCTCATC3′; FcγRIII: 5′ primer, 5′-AAGATCTCCCAAAGGCTGTG-3′ and 3′ primer, 5′-ATGGACTTCTAGCTGCACCG-3′; FcɛRII: 5′ primer, 5′-CGTCTCTCAAGTTTCCAAG-3′ and 3′ primer: 5′-GCACTTCCGTTGGAATTTG-3′.
Rabbit-specific α-actin primers and human specific ribosomal protein L7 (RPL7) primers, based on the published sequence of the rabbit α-actin (27) and human RPL7 genes (28), respectively, were used to control for the transcription level of each sample. The PCR number of cycles used were determined to be in the linear range of each PCR product. Equal aliquots of each PCR were then run on 1.2% agarose gels and transferred to a Zeta-probe membrane, and after capillary transfer, the DNA was immobilized by UV-crosslinking with a Stratalinker UV Crosslinker 2400. Prehybridization for 2–3 hr and hybridization for 20 hr, both at 42°C, were conducted as described (18). The FcγRI, -RII, -RIII, FcɛRII, RPL7, and α-actin DNA levels were assayed by Southern blot analysis with 32P-labeled probes, and fragments were subsequently sequenced for product confirmation. Of note, the 333-bp rabbit ASM CD23 RT-PCR products demonstrated ≈90% sequence homology with the human CD23 B-cell receptor gene.
Determination of Fc Receptor Expression in ASM Cells and Tissue.
Fc receptor cell surface expression was examined in cultured rabbit ASM cells by using a Coulter EPICS Elite flow cytometer. Fluorescence signals were accumulated as two parameter fluorescence histograms with both percent positive cells and mean channel fluorescence being recorded. Based on the results from the above Fc receptor mRNA expression studies, the targeted mAbs used for staining included the 3G8-FITC anti-FcγRIII (Medarex, Annandale, NJ) and the fluorescein isothiocyanate (FITC) mouse mAb to human CD23 (Caltag, South San Francisco, CA). The immortalized B cell line 8.1.6 was used as the positive control for the CD23 receptor expression assay. The cells were also stained with FITC- and phycoerthrin-conjugated mouse antibodies of the identical isotypes as the Fc receptor mAbs to measure background fluorescence. The antibody-stained cells were then evaluated by flow cytometry and analyzed by using the Elite Immuno 4 statistical software (Coulter).
An immunofluorescence detection assay was also used to examine for FcɛRII surface receptor expression in rabbit ASM cells and tissue following 24-hr treatment with human control vs. atopic asthmatic serum, as described above. After fixation serial 3- to 5-μm TSM sections were prepared and mounted on poly-l-lysine coated slides, and subsequently labeled overnight with primary mouse anti-human CD23 (FcɛRII) antibody. In control sections, the primary antibody was replaced by Igs of the same isotype following the manufacture’s protocol (mouse IgG1 negative control). After subsequent repeat washing, FITC-labeled F(ab′)2 goat anti-mouse IgE (Fcɛ) fragments were used as secondary antibody and incubated for 1 hr in 1:500–1,000 dilutions in PBS containing 0.5% BSA. The slides then were examined by using a fluorescent microscope.
Statistical Analysis.
Unless otherwise indicated, results are expressed as mean ± SE. Statistical analysis was performed by means of two-tailed paired Student’s t test. P values < 0.05 were considered significant.
Reagents.
The FcγRI, -RII, -RIII, FcɛRII, RPL7, and rabbit α-actin primers were obtained from Integrated DNA Technologies (Coralville, IA). ACh and isoproterenol hydrochloride were obtained from Sigma. The purified human IgE and IgG, and the goat-anti-human IgE and anti-human IgG antibodies were purchased from Biodesign International (Kennebunkport, ME). The purified rat IgE (rat myeloma) was purchased from Chemicon. The 3G8-FITC anti-FcγRIII and FITC-mouse mAb to the human CD23 receptors used in flow cytometric studies were purchased from Medarex and Caltag, respectively. The anti-CD23 blocking mAb (mAb135) was the gift of M. Sarfati (Montreal). The FcɛRII (CD23) antibody and F(ab′)2-FITC fragments used in the immunofluoresence studies were purchased from Serotec. The immortalized B cell line 8.1.6 was provided by E. Mellins (University of Pennsylvania). The FcγRI, -RII, and -RIII cDNA probes and U937 cells were provided by S. McKenzie and D. Herrick (University of Pennsylvania). The human tissue was provided by the Cooperative Human Tissue Network (University of Pennsylvania), which is funded by the National Cancer Institute.
RESULTS
Role of Fc Receptors in Altered Responsiveness of Atopic Asthmatic Sensitized ASM.
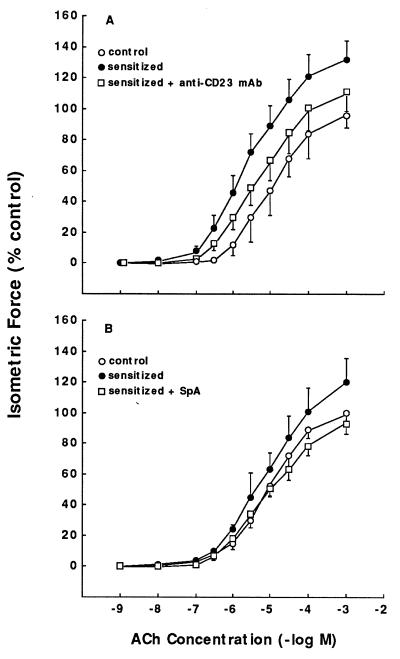
We recently demonstrated that passive sensitization of isolated naive ASM tissue with human atopic asthmatic serum induces changes in the tissue’s agonist-mediated constrictor and relaxant responsiveness that phenotypically resemble the pro-asthmatic state (19). To examine whether these effects of atopic asthmatic serum are mediated, at least in part, by the presence of elevated levels of IgE in the sensitizing serum (see Materials and Methods), constrictor and relaxation responses were separately examined in TSM segments that were treated with human control or atopic asthmatic serum in the absence and presence of blockade of specific Fc receptors, or following depletion of immune complexes in the sensitizing serum by treatment with SpA (20). As shown in Fig. 1, relative to tissues incubated with control serum (○), the maximal constrictor (Tmax) responses to ACh were significantly enhanced in TSM passively sensitized with atopic asthmatic serum (•). Accordingly, the mean ± SE Tmax values amounted to 121.7 ± 5.3 and 146.0 ± 15.5 g/g TSM in the control and sensitized tissues, respectively (P < 0.01). The induced augmented constrictor responses to ACh, however, were largely prevented in atopic serum-sensitized tissues that were pretreated with an anti-CD23 blocking mAb (anti-CD23 mAb) (Fig. 1A, □) or when the sensitizing serum was depleted of its immune complexes by pretreatment with SpA (Fig. 1B, □).
Figure 1.
(A) Comparison of contractile dose–response relationships to ACh in paired control serum-treated (○) and atopic asthmatic serum-treated TSM segments in the absence (•) and presence (□) of anti-CD23 mAb. (B) Comparison of contractile dose-response relationships to ACR in control serum-treated (○) and atopic asthmatic serum-treated TSM in the absence (•) and presence (□) of SpA. Data represent mean ± SE values from six paired tissue samples.
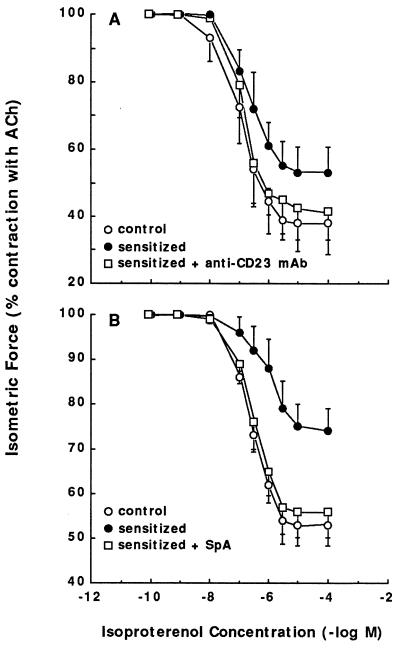
In separate studies, administration of the β-adrenergic receptor agonist isoproterenol elicited cumulative dose-dependent relaxation of the TSM precontracted with ACh (≈40% Tmax) (Fig. 2). Relative to control TSM, the maximal relaxation responses (Rmax) and sensitivities (pD50; i.e., −log ED50) to isoproterenol were significantly attenuated in the atopic asthmatic serum-sensitized TSM, wherein the mean Rmax values amounted to 26.0 ± 5.1% vs. 47.0 ± 4.7% in the control serum-treated TSM (P < 0.005), with corresponding pD50 values averaging 5.82 ± 0.16 and 6.59 ± 0.11 −log M, respectively (P < 0.005). The attenuated isoproterenol-induced relaxation responses, however, were ablated in atopic serum-sensitized TSM that were pretreated with anti-CD23 mAb (Fig. 2A) or when the sensitizing serum was initially depleted of its immune complexes with SpA (Fig. 2B).
Figure 2.
(A) Comparison of relaxation dose-relationships to isoproterenol in paired control serum-treated (○) and atopic asthmatic serum-treated TSM segments in the absence (•) and presence (□) of anti-CD23 mAb. (B) Comparison of relaxation responses to isoproterenol in control (○) and atopic asthmatic serum-treated TSM in the absence (•) and presence (□) of SpA. Data are mean ± SE values from eight paired tissue samples.
In contrast to the above observations, in control and serum-treated tissues, neither anti-CD23 mAb nor pretreatment of the control serum with SpA affected the tissues’ subsequent contractility to ACh or relaxation responsiveness to isoproterenol (data not shown). Moreover, contrasting the above inhibitory effects of anti-CD23 mAb in atopic sensitized TSM, exposure of the sensitized tissues to a blocking mAb to the FcγRIII receptor, which is also expressed in ASM (see below), had no appreciable effect on the tissues’ altered responsiveness to ACh or to isoproterenol (data not shown).
Effects of IgE and IgE Immune Complexes on ASM Responsiveness.
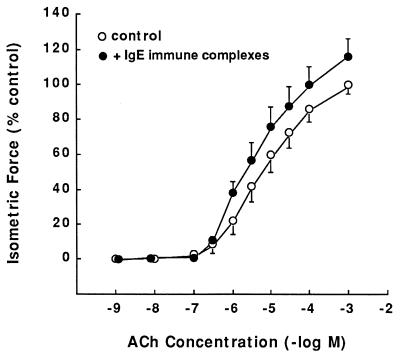
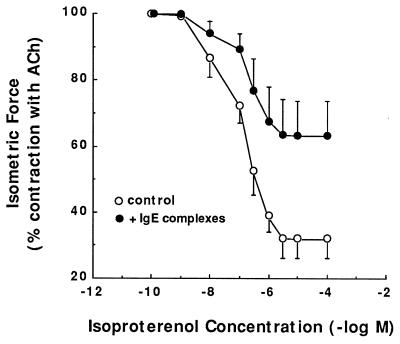
In light of the above observations suggesting a role for IgE immune complex/FcɛRII receptor interaction in mediating altered ASM responsiveness in the atopic asthmatic sensitized state, to further substantiate this mechanism we examined whether the sensitizing effects of the atopic asthmatic serum could be simulated by treatment of naive TSM with either exogenous IgE or IgE immune complexes. Constrictor and relaxant dose–response relationships to ACh and isoproterenol, respectively, were separately compared in TSM exposed for 24 hr to vehicle alone (control) or varying concentrations of human IgE or IgE immune complexes (see Materials and Methods). Relative to their respective controls, the maximal constrictor responses to ACh were significantly (P < 0.05) enhanced in tissues treated with IgE immune complexes (i.e., 15:5 μg/ml of IgE/anti-IgE), wherein the mean ± Tmax responses amounted to 108.8 ± 10.2 vs. 94.2 ± 5.1 g/g TSM wt in the control tissues (Fig. 3). Comparably, the subsequent relaxation responses to isoproterenol in ACh-precontracted TSM were markedly reduced in the IgE immune complex-treated vs. control tissues (Fig. 4). Qualitatively, similar results were obtained in tissues treated with human IgE alone, although the changes in ACh-induced contraction and isoproterenol-induced relaxation were quantitatively less pronounced than those obtained in IgE immune complex-treated TSM (data not shown). Moreover, in contrast to the above effects of human IgE and IgE immune complexes, we found that treatment of rabbit ASM with rat IgE, which reportedly does not interact with CD23 (47), neither heightened the tissues’ constrictor responses to ACh nor attenuated their relaxation responses to isoproterenol (data not shown).
Figure 3.
Comparison of airway constrictor responses to ACh in isolated paired TSM segments in the absence (○) and presence (•) of IgE immune complexes (see Materials and Methods).
Figure 4.
Comparison of airway relaxation responses to isoproterenol in paired TSM segments in the absence (○) and presence (•) of human IgE immune complexes (see Materials and Methods).
Fc Receptor Expression in Atopic Asthmatic Sensitized ASM.
Given the above pharmacodynamic results, we next examined whether cultured ASM cells and isolated ASM tissue express Fc receptors and whether the expression of these receptors is modulated in the atopic asthmatic sensitized state. The latter was examined by using RT-PCR and Fc receptor-specific primers for the different Fcγ and Fcɛ receptor subtypes, and the signals for the Fcγ, Fcɛ and α-actin PCR products were quantitated on a PhosphorImager.
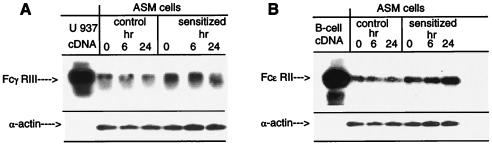
In contrast to the lack of detectable mRNA expression of FcγRI and FcγRII, ASM cells expressed mRNA for the FcγRIII receptor (Fig. 5A). In parallel with the unaltered constitutive expression of α-actin, the FcγRIII signal did not temporally vary in cells treated for up to 24 hr with either control or atopic sensitized serum, and there were no significant differences in FcγRIII expression between control and sensitized cells. Expression of the high affinity IgE receptor, FcɛRI, was also undetectable in the ASM cells. On the other hand, as shown in Fig. 5B, mRNA expression of the low affinity IgE receptor, FcɛRII (i.e., CD23) was present in both the control and atopic asthmatic sensitized cells. Moreover, in contrast to control serum-treated ASM cells wherein the mRNA signal did not systematically vary with time, the intensity of the FcɛRII signal progressively increased at 6 and 24 hr in the atopic asthmatic serum-sensitized cells.
Figure 5.
Comparison of expression of FcγRIII (A) and FcɛRII (CD23) (B) receptor mRNAs by using RT-PCR in rabbit ASM cells after 0, 6-, and 24-hr treatment with 10% control serum and 10% atopic asthmatic sensitizing serum. Expression of α-actin was used to control for loading. cDNA from activated U937 cells (i.e., for FcγRIII) and the immortalized human B-cell line 8.1.6 (i.e., for FcɛRII) were used as positive controls. Note in contrast to FcγRIII, expression of FcɛRII is significantly up-regulated at 24 hr (i.e., >2-fold) after treatment with atopic asthmatic serum.
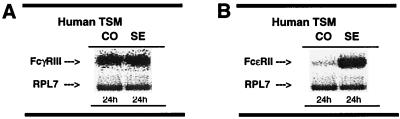
Similar results were obtained in comparable experiments conducted on isolated human TSM tissue wherein adjacent alternating sections were exposed for 24 hr to either control or atopic asthmatic serum. The Southern blots were then prepared using the above 32P-labeled human-specific FcγRIII, FcɛRII, and RPL7 (157-bp) probes. As depicted in Fig. 6, the signals for FcγRIII and RPL7 were similar in the control (CO) and atopic asthmatic-sensitized (SE) tissue samples (Fig. 6A). In contrast, FcɛRII expression, although detectable in the control samples, was markedly induced in the atopic asthmatic serum-treated tissues (Fig. 6B), whereas the RPL7 signal was unaffected.
Figure 6.
Representative Southern blots probed with full-length FcγRIII (A), FcɛRII (B), and 157-bp RPL7 human cDNA probes. Paired human ASM samples were incubated with control (CO) or atopic asthmatic (SE) serum for 24 hr. cDNA was transcribed from total RNA primed with oligo(dT). PCR products were amplified by using human-specific FcγRIII, FcɛRII, and RPL7 primers, and then probed. Note the marked induction of FcɛRII expression in the atopic asthmatic serum-sensitized sample (i.e., >7.5-fold), whereas expression of FcγRIII or RPL7 was similar in both samples.
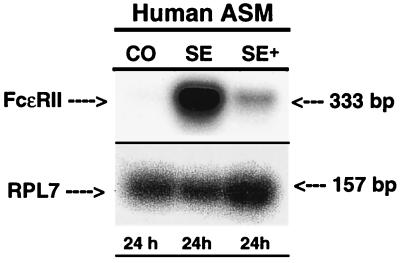
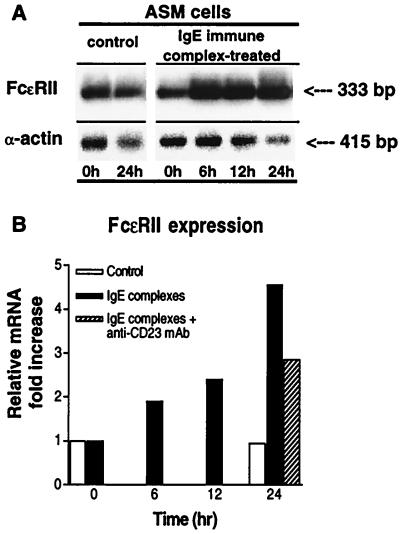
In light of the above observations, we next examined whether our observed up-regulation of FcɛRII expression in the atopic asthmatic sensitized state is attributed to activation of the FcɛRII receptor. As shown in Fig. 7, by using ASM tissue sections isolated from the same human lung specimen we found that, whereas the tissue samples exposed to atopic asthmatic serum (SE) displayed markedly induced FcɛRII expression relative to control (CO) serum-treated samples, the up-regulation of FcɛRII expression was largely inhibited by pretreatment of the atopic serum-sensitized tissues with anti-CD23 mAb (SE+). Moreover, in separate complimentary experiments examining the effects of exogenous administration of IgE immune complexes on FcɛRII expression in cultured ASM cells, we found that treatment of the cells with IgE immune complexes induced progressive enhancement of FcɛRII mRNA expression (Fig. 8A), resulting in a near 5-fold increase in FcɛRII expression at 24 hr (Fig. 8B). In addition, in concert with the above ASM tissue studies, pretreatment of cells with anti-CD23 mAb significantly inhibited (i.e., by ≈40%) the magnitude of IgE immune complex-induced FcɛRII expression at 24 hr (Fig. 8B).
Figure 7.
Comparison of expression of FcɛRII mRNA in human bronchial smooth muscle tissue after 24-hr treatment with control (CO) or atopic asthmatic serum in the absence (SE) and presence (SE+) of anti-CD23 mAb. The products of the RT-PCRs with 2.5 μg of total RNA and human-specific primers for the FcɛRII receptor mRNA are shown. Expression of RPL7 was used to control for gel loading. Note that the anti-CD23 mAb significantly attenuated the induction of FcɛRII mRNA expression at 24 hr in atopic asthmatic-serum sensitized ASM.
Figure 8.
(A) Comparison of expression of FcɛRII in rabbit ASM cells after 0 and 24-hr treatment with media alone (control) or with IgE immune complexes for 0, 6, 12, and 24 hr. The products of RT-PCRs with 2.5 μg of total RNA and human-specific primers for the FcɛRII receptor mRNA are shown. mRNA expression of α-actin is also shown for comparison. (B) Corresponding time-dependent changes in FcɛRII/α-actin ratio, expressed as fold increase above baseline (i.e., time 0) in control (open bars) and IgE immune complex-treated cells (filled bars). Note the progressive induction of FcɛRII mRNA expression up to 24 hr after treatment with IgE immune complexes, and inhibition of FcɛRII expression at 24 hr in the presence of anti-CD23 mAb (hatched bar).
Expression of FcγRIII and FcɛRII Cell Surface Proteins in Sensitized ASM.
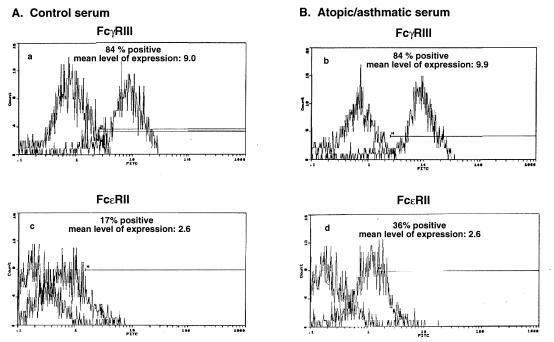
Given the above findings, we next examined whether ASM cells express FcγRIII and FcɛRII receptor proteins on their cell surface, examined by flow cytometry (see Materials and Methods). As shown in Fig. 9, ASM cells expressed surface protein for both the low-affinity FcγRIII (Fig. 9A) and FcɛRII (Fig. 9B) receptors. In contrast to unaltered FcγRIII receptor expression after exposure of the cells for 24 hr to control or atopic asthmatic serum (Fig. 9A), cell surface expression of the FcɛRII receptor was increased by >2-fold in the atopic asthmatic serum-treated vs. control serum-treated cells (Fig. 9B). In additional comparative studies, we also examined for altered FcɛRII receptor expression in sensitized ASM cells and tissue by immunofluorescence staining (see Materials and Methods). In parallel with the above flow cytometric analysis, we found that surface immunofluorescence staining for the CD23 receptor protein was notably enhanced in the atopic asthmatic serum-treated cells and tissue (data not shown).
Figure 9.
Flow cytometric analysis of FcγRIII and FcɛRII surface expression in rabbit ASM cells. Cells treated for 24 hr with either 10% control serum or 10% atopic asthmatic serum were stained with FITC-conjugated human mAbs specific for the low-affinity FcγRIII (A) and FcɛRII (CD23) (B) receptors. Activated B-cells (8.1.6) were used as a positive control for the CD23 receptor. The level of nonspecific background staining was measured in both the control- and atopic asthmatic- serum-treated cells by staining with FITC-conjugated isotype control antibodies. Note that the rabbit ASM cells express surface protein for both FcγRIII and FcɛRII receptors. In contrast to FcγRIII receptor expression, which is unaltered in the presence of atopic asthmatic serum, expression of the FcɛRII receptor is increased by >2-fold (i.e., from 17 to 36% in the presence of atopic asthmatic serum.
DISCUSSION
Allergic asthma is characterized by increased airway reactivity to bronchoconstrictor agonists, impaired β-adrenoreceptor-mediated airway relaxation, and inflammation of the airways (11, 12, 29). The altered airway responsiveness in asthmatic individuals has been associated with elevations in total and antigen-specific serum IgE (30, 31). Moreover, genetic analyses of family members with allergic asthma have demonstrated a significant correlation between altered airway responsiveness and serum IgE levels (32, 33). Although the mechanistic basis for this association remains to be identified, it has long been known that IgE can initiate immediate hypersensitivity reactions following allergen exposure by triggering the degranulation of mast cells via activation of the high affinity IgE receptor, FcɛRI (4–6). Progression of this initial IgE/FcɛRI-coupled mast cell response into a subsequent late phase asthmatic response has been largely attributed to the orchestrated extended release of various cytokines by infiltrating pro-inflammatory cells (5, 7, 10, 11). However, more recent studies have demonstrated that expression of the inducible form of the low affinity IgE receptor (FcɛRII) is up-regulated on monocytes and lung alveolar macrophages (14, 17) as well as on circulating B lymphocytes (15, 16) isolated from atopic asthmatic individuals. Thus, these studies would suggest that, apart from mast cells per se, IgE/Fcɛ receptor interactions involving other pro-inflammatory cell types may also contribute to the presentation of allergic asthma.
Although the above conventional thinking implicates both mast cells and other pro-inflammatory cells in the pathogenesis of asthma, the present study addressed the hypothesis that the resident ASM itself also plays an etiologic role in the pathobiology of the disease. The results provided compelling evidence demonstrating that ASM cells intrinsically express the low affinity receptor for IgE (FcɛRII) and that the induced altered responsiveness of ASM in the atopic asthmatic-sensitized state is largely attributed to its autologously up-regulated expression and activation of FcɛRII.
Our observed changes in responsiveness in the atopic-sensitized tissues closely mimicked the pertubations in airway function that characterize the in vivo asthmatic condition, including exaggerated bronchoconstrictor responsiveness to contractile agonists and impaired airway relaxation to β-adrenoreceptor stimulation (18, 19). In previously addressing the mechanistic link between exposure of naive airway tissue to atopic asthmatic serum and its resultant altered responsiveness, and by using the same experimental model described herein, we found that the latter effect is largely mediated by the induced autologous expression and autocrine action of the cytokine IL-1β in atopic-sensitized ASM (18). Moreover, the mechanism of action of IL-1β in eliciting the altered responsiveness in sensitized tissues was attributed to its induced up-regulated expression of Gi protein, specifically Giα2 and Giα3, which inhibit intracellular cAMP accumulation (21).
These findings, when coupled to the results of the present study, suggest that the sequence of events leading to cytokine-induced changes in ASM responsiveness in the sensitized state is initiated by IgE-coupled FcɛRII receptor activation involving the ASM itself. Indeed, in support of this concept, the present observations demonstrated that either depletion of serum immune complexes or pretreatment with a specific anti-CD23 blocking mAb largely prevented the changes in ASM responsiveness induced by exposure of the tissues to atopic asthmatic serum (Figs. 1 and 2). Furthermore, in complimentary studies, exposure of naive ASM tissue to human IgE immune complexes produced changes in ASM responsiveness (Figs. 3 and 4), which closely resembled those observed in the atopic-sensitized state. Thus, at least with respect to our experimental model, IgE-coupled Fc receptor activation principally involving the FcɛRII receptor in ASM itself appears to be fundamentally responsible for producing the pertubations in airway responsiveness that characterize the pro-asthmatic state.
In concert with the above pharmacodynamic results, our extended observations demonstrated the presence of constitutive FcɛRII and FcγRIII receptor mRNA and cell surface expression, both in cultured ASM cells and human ASM tissue (Figs. 5 and 6). Furthermore, in contrast to unaltered FcγRIII expression, FcɛRII was markedly enhanced in ASM exposed to the sensitizing atopic serum (Figs. 5 and 6). In considering these results, it is relevant to note that a number of studies have reported a direct correlation between serum IgE levels and FcɛRII and -RI expression (34–36). Although these reports raised the notion of a possible common mechanism regulating the production of both IgE and its receptors, more recent evidence based on studies with different cell lines suggests that IgE itself may enhance FcɛRI (37, 38) or FcɛRII expression (39–41). The latter concept is supported by our present observations, wherein we observed that the induced expression of FcɛRII in ASM treated with atopic asthmatic serum was largely inhibited in the presence of an anti-CD23 blocking antibody (Fig. 7). Moreover, in extended support of the concept of IgE-dependent modulation of FcɛRII expression, we also found that exogenously administered IgE immune complexes elicited an increase in FcɛRII expression (Fig. 8A) and, further, that this effect was significantly inhibited by pretreatment with anti-CD23 mAb (Fig. 8B). Thus, our findings are consistent with the presence of a positive feedback system in ASM wherein IgE up-regulates the expression of its own low affinity receptor. This notion is in general agreement with previous studies which reported the presence of IgE-dependent Fcɛ receptor induction in other cell types (37–41).
In recent years, the complexity of transmembrane signaling via the FcɛRII receptor has been partially unraveled (42–45). Accordingly, in B-lymphocytes, a network involving antigen/IgE immune complex molecules coupled to the FcɛRII receptor apparently serves to facilitate antigen presentation to antigen-specific CD4+ (helper) T-lymphocytes, a process resulting in the expression of the CD4+/Th2 phenotype. The latter is associated with the release of such cytokines as IL-4 and IL-5 which are involved in B-lymphocyte switching to IgE production and eosinophil accumulation, respectively (44), effects that have been recently documented in the lungs of immunized mice after inhaled antigen challenge (46). In light of this, the results of the present study raise the consideration that, apart from inducing changes in airway responsiveness, up-regulated FcɛRII expression and activation in ASM cells may also play a role in modulating local airway immune responses. Indeed, this compelling concept is in part supported by our recent observation that ASM cells are autologously induced to release IL-1β in the atopic asthmatic-sensitized state (18), a finding that, together with our present observations, suggests the presence of FcɛRII-coupled local stimulation of cytokine release and signaling.
In conclusion, the present study identified the presence and up-regulated expression of FcɛRII receptors in atopic asthmatic-sensitized ASM, and further demonstrated that the latter phenomenon is associated with the induction of altered airway responsiveness in the atopic asthmatic-sensitized state. Given the important immunological and proinflammatory responses attributed to FcɛRII receptor activation, these findings identify a potentially important role and mechanism for the resident ASM in autologously inducing its own IgE-mediated state of altered responsiveness contributing to the atopic asthmatic condition.
Acknowledgments
We thank P. Gonzales-Serrano, J. Grunstein, S. Chang, and S. Ling for expert technical assistance; Donald Campbell for the use of the Flow-Cytometric Core Facility; and M. Brown for typing the manuscript. H.H. is a Parker B. Francis Fellow in Pulmonary Research. This work was supported in part by National Heart, Lung, and Blood Institute Grant HL-31467, and by an Institutional Developmental Fund Award from The Joseph Stokes, Jr., Research Institute of the Children’s Hospital of Philadelphia.
ABBREVIATIONS
- TSM
tracheal smooth muscle
- ASM
airway smooth muscle
- IL
interleukin
- ACh
acetylcholine
- RT-PCR
reverse transcription–PCR
- FITC
fluorescein isothiocyanate
- SpA
Sepharose Staph protein A
References
- 1.Bai T R. Am Rev Respir Dis. 1990;141:552–557. doi: 10.1164/ajrccm/141.3.552. [DOI] [PubMed] [Google Scholar]
- 2.Goldie R G, Spina D, Henry P J, Lulich K M, Paterson J W. Br J Clin Pharmacol. 1986;22:669–676. doi: 10.1111/j.1365-2125.1986.tb02956.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McFadden E R., Jr Am J Respir Crit Care Med. 1994;150:523–526. doi: 10.1164/ajrccm/150.5_Pt_2.S23. [DOI] [PubMed] [Google Scholar]
- 4.Metzger H. Immunol Rev. 1992;125:37–48. doi: 10.1111/j.1600-065x.1992.tb00624.x. [DOI] [PubMed] [Google Scholar]
- 5.Beaven M A, Metzger H. Immunol Today. 1993;14:222–226. doi: 10.1016/0167-5699(93)90167-j. [DOI] [PubMed] [Google Scholar]
- 6.Galli S J. N Engl J Med. 1993;328:257–265. doi: 10.1056/NEJM199301283280408. [DOI] [PubMed] [Google Scholar]
- 7.Walker C, Bode E, Boer L, Hansel T T, Blaser K, Virchow J C. Am Rev Respir Dis. 1992;146:109–115. doi: 10.1164/ajrccm/146.1.109. [DOI] [PubMed] [Google Scholar]
- 8.Watson M L, Smith D, Bourne A D, Thompson R C, Westwick J. Am J Respir Cell Mol Biol. 1993;8:365–369. doi: 10.1165/ajrcmb/8.4.365. [DOI] [PubMed] [Google Scholar]
- 9.Capron M, Spiegelberg H L, Prin L, Bennich H, Butterworth A E, Pierce R J, Ouaissi M A, Capron A. J Immunol. 1984;132:462–468. [PubMed] [Google Scholar]
- 10.Beasley R, Roche W R, Roberts J A, Holgate S T. Am Rev Respir Dis. 1989;139:806–817. doi: 10.1164/ajrccm/139.3.806. [DOI] [PubMed] [Google Scholar]
- 11.Litchfield T M, Lee T H. J Asthma. 1992;29:181–191. doi: 10.3109/02770909209099026. [DOI] [PubMed] [Google Scholar]
- 12.Barnes P J, Chung K F, Page C P. Pharmacol Rev. 1988;40:49–84. [PubMed] [Google Scholar]
- 13.Borish L, Mascali J J, Rosenwasser L J. J Immunol. 1991;146:63–67. [PubMed] [Google Scholar]
- 14.Williams J, Johnson S, Mascali J J, Smith H, Rosenwasser L J, Borish L. J Immunol. 1992;149:2823–2829. [PubMed] [Google Scholar]
- 15.Gagro A, Rabatic S, Trescec A, Dekaris D, Medar-Lasic M. Int Arch Allergy Immunol. 1993;101:203–208. doi: 10.1159/000236520. [DOI] [PubMed] [Google Scholar]
- 16.Rabatic S, Gagro A, Medar-Lasic M. Exp Immunol. 1993;94:337–340. doi: 10.1111/j.1365-2249.1993.tb03454.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Joseph M, Tonnel A B, Torpier G, Capron A, Arnoux B, Benveniste J. J Clin Invest. 1983;71:221–230. doi: 10.1172/JCI110762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hakonarson H, Herrick D J, Gonzalez-Serrano P, Grunstein M M. J Clin Invest. 1997;99:117–124. doi: 10.1172/JCI119122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hakonarson H, Herrick D, Grunstein M M. Am J Physiol. 1995;269:L645–L652. doi: 10.1152/ajplung.1995.269.5.L645. [DOI] [PubMed] [Google Scholar]
- 20.Kessler S W. J Immunol. 1975;115:1617–1624. [PubMed] [Google Scholar]
- 21.Hakonarson H, Herrick D J, Gonzalez Serrano P, Grunstein M M. J Clin Invest. 1996;97:2593–2600. doi: 10.1172/JCI118708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Noveral J P, Grunstein M M. Am J Physiol. 1992;263:L555–L561. doi: 10.1152/ajplung.1992.263.5.L555. [DOI] [PubMed] [Google Scholar]
- 23.Tanaka D T, Grunstein M M. J Clin Invest. 1990;85:345–350. doi: 10.1172/JCI114444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chomczynski P, Sacchi N. Anal Biochem. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- 25.Capel P J, van de Winkel J G, van den Herik-Oudijk I E, Verbeek J S. Immunomethods. 1994;4:25–34. doi: 10.1006/immu.1994.1004. [DOI] [PubMed] [Google Scholar]
- 26.Kikutani H, Inui S, Sato R, Barsumian E L, Owaki H, et al. Cell. 1986;47:657–665. doi: 10.1016/0092-8674(86)90508-8. [DOI] [PubMed] [Google Scholar]
- 27.Putney S D, Herlihy W C, Schimmel P. Nature (London) 1983;302:718–721. doi: 10.1038/302718a0. [DOI] [PubMed] [Google Scholar]
- 28.Seshadri T, Uzman J A, Oshima J, Campisi J. J Biol Chem. 1993;268:18474–18480. [PubMed] [Google Scholar]
- 29.Wardlaw A J, Dunnette S, Gleich G J, Collins J V, Kay A B. Am Rev Respir Dis. 1988;140:1745–1753. doi: 10.1164/ajrccm/137.1.62. [DOI] [PubMed] [Google Scholar]
- 30.Sears M R, Burrows B, Flannery E M, Herbison G P, Hewitt C J, Holdaway M D. N Engl J Med. 1991;325:1067–1071. doi: 10.1056/NEJM199110103251504. [DOI] [PubMed] [Google Scholar]
- 31.Burrows B, Fernando D, Martinez M D, Barbee R A, Clive M G. N Engl J Med. 1989;320:272–277. doi: 10.1056/NEJM198902023200502. [DOI] [PubMed] [Google Scholar]
- 32.Burrows B, Martinez F D, Cline M A, Lebowitz M D. Am J Respir Crit Care Med. 1995;152:1497–1500. doi: 10.1164/ajrccm.152.5.7582283. [DOI] [PubMed] [Google Scholar]
- 33.Burrows B, Sears M R, Flannery E M, Herbison G P, Holdaway M D. J Allergy Clin Immunol. 1995;95:548–556. doi: 10.1016/s0091-6749(95)70317-9. [DOI] [PubMed] [Google Scholar]
- 34.Spiegelberg H L, Simon R A. J Clin Invest. 1981;68:845–852. doi: 10.1172/JCI110339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Conroy M C, Adkinson N F, Jr, Lichtenstein L M. J Immunol. 1977;118:1317–1321. [PubMed] [Google Scholar]
- 36.Malveaux F J, Conroy M C, Adkinson N F, Jr, Lichtenstein L M. J Clin Invest. 1978;62:176–181. doi: 10.1172/JCI109103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lantz C S, Yamaguchi M, Oettgen H C, Katona I M, Miyajima I, Kinet J P, Galli S J. J Immunol. 1997;158:2517–2521. [PubMed] [Google Scholar]
- 38.Yamaguchi, M., Lantz, C. S., Oettgen, H. C., Katona, I. M., Fleming, T., Miyajima, I., Kinet, J.-P. & Galli, S. J. (1997) J. Exp. Med. 663–672. [DOI] [PMC free article] [PubMed]
- 39.Lee W T, Rao M, Conrad D H. J Immunol. 1987;139:1191–1198. [PubMed] [Google Scholar]
- 40.Daeron M, Ishizaka K. J Immunol. 1986;136:1612–1619. [PubMed] [Google Scholar]
- 41.Lee W T, Conrad D H. J Immunol. 1986;136:4573–4580. [PubMed] [Google Scholar]
- 42.Sutton B J, Gould H J. Nature (London) 1993;366:421–428. doi: 10.1038/366421a0. [DOI] [PubMed] [Google Scholar]
- 43.Aubry J-P, Pochon S, Graber P, Jansen K U, Bonnefoy J-Y. Nature (London) 1992;358:505–507. doi: 10.1038/358505a0. [DOI] [PubMed] [Google Scholar]
- 44.Delespesse G, Sarfati M, Wu C Y, Fournier S, Letellier M. Immunol Rev. 1992;125:77–97. doi: 10.1111/j.1600-065x.1992.tb00626.x. [DOI] [PubMed] [Google Scholar]
- 45.Matsumoto A K, Kopicky-Burd J, Carter R H, Tuveson D A, Tedder T F, Fearon D T. J Exp Med. 1991;173:55–64. doi: 10.1084/jem.173.1.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Coyle A J, Wagner K, Bertrand C, Tsuyuki S, Bews J, Heusser C. J Exp Med. 1996;183:1303–1310. doi: 10.1084/jem.183.4.1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Peterson L H, Conrad D H. J Immunol. 1985;135:2654–2660. [PubMed] [Google Scholar]