Abstract
Intracerebral infection of susceptible mouse strains with Theiler’s murine encephalomyelitis virus (TMEV) results in an immune-mediated demyelinating disease similar to human multiple sclerosis. TMEV infection is widely spread via fecal-oral routes among wild mouse populations, yet these infected mice rarely develop clinical disease. Oral vaccination has often been used to protect the host against many different infectious agents, although the underlying protective mechanism of prior oral exposure is still unknown. To understand the mechanisms involved in protection from demyelinating disease following previous oral infection, immune parameters and disease progression of mice perorally infected with TMEV were compared with those of mice immunized intraperitoneally following intracerebral infection. Mice infected perorally, but not intraperitoneally, prior to CNS viral infection showed lower chronic viral persistence in the CNS and reduced TMEV-induced demyelinating disease. However, a prolonged period of post-oral infection was necessary for effective protection. Mice orally pre-exposed to the virus displayed markedly elevated levels of antibody response to TMEV in the serum, although T cell responses to TMEV in the periphery were not significantly different between perorally and intraperitoneally immunized mice. In addition, orally vaccinated mice showed higher levels of early CNS-infiltration of B cells producing anti-TMEV antibody as well as virus-specific CD4+ and CD8+ T cells compared to intraperitoneally immunized mice. Therefore, the generation of a sufficient level of protective immune responses appears to require a prolonged time period to confer protection from TMEV-induced demyelinating disease.
Keywords: Infectious immunity-Virus, Oral immunization, Vaccine, Mucosal immunity
INTRODUCTION
Once pathogens gain access to the CNS, infection often results in deadly consequences due to the unique nature of the site, as no immune cells are readily available. Thus, effective means to vaccinate against CNS pathogens such as West Nile virus and Japanese encephalomyelitis virus could be very helpful. The oral route has been used to vaccinate against a variety of viral infections (Cox, Brokstad, and Ogra, 2004; Fekadu et al., 1996; Foxwell, Cripps, and Dear, 2006; Ramakrishna et al., 1999; Vesikari, Giaquinto, and Huppertz, 2006; Zhao et al., 2006). In particular, vaccination against poliovirus is a remarkably successful example (Sabin, 1985). However, very little is known about the immunological mechanisms involved in the efficacy of oral vaccination. Despite the extensive use of oral vaccination against many viral infections, very few systemic studies have examined the time and type of immune responses required for effective protection. Although intracerebral infection of Theiler’s murine encephalomyelitis virus (TMEV) consistently induces demyelinating disease in susceptible mice (Lipton, 1975), infection via other routes seldom induces demyelinating disease (Thompson, Harrison, and Myers, 1951). The wild mouse population is widely infected with TMEV, presumably via the fecal-oral route, without apparent disease (Lipton et al., 2001). Similarly, human populations have been exposed to many different viruses without apparent clinical symptoms. Therefore, it is particularly interesting to investigate whether mice pre-exposed to the virus become resistant to developing demyelinating disease, and if they do, what are the immunological mechanisms of acquired resistance to TMEV-induced CNS disease.
TMEV belongs to the Picornavirus family. Intracerebral (ic) inoculation of susceptible mice with the BeAn 8386 or DA strains of TMEV results in a chronic progressive demyelinating disease in the CNS (Lipton, 1975). There are strong similarities between TMEV-induced demyelinating disease (TMEV-IDD) and human multiple sclerosis (MS), as evidenced by histopathological and immunological studies, the potential involvement of infectious agents, and genetic influences to disease susceptibility. Thus, this system is considered to be a relevant infectious animal model for MS (Dal Canto, 1996; Kim et al., 2000). However, the mechanism of TMEV-IDD is not well understood. Many previous studies have suggested that the CD4+ T cell response is pathogenic in susceptible SJL mice (Gerety et al., 1994; Pope et al., 1996; Yauch and Kim, 1994; Yauch et al., 1998). However, virus-specific CD4+ T cells may also be able to confer protection depending upon the timing of their presence and/or genetic background (Johnson et al., 1999; Karls, Denton, and Melvold, 2002; Mohindru, Kang, and Kim, 2006; Murray et al., 1998b; Njenga et al., 1996). It is suggested that CD8+ T cells are primarily involved in viral clearance and protection from the development of demyelinating disease (Fiette et al., 1993; Lyman et al., 2004; Pullen et al., 1993; Rodriguez et al., 1993; Rodriguez et al., 1990). However, the role of CD8+ T cells in the pathogenesis of demyelination in susceptible mice remains unclear (Begolka et al., 2001; Murray et al., 1998a; Palma and Kim, 2001). The role of antibodies to TMEV has also been previously investigated and found to be protective, despite the presence of high levels of such antibodies in clinically affected mice (Fujinami et al., 1989; Kang et al., 2005; Rodriguez et al., 1990; Rossi et al., 1991; Yahikozawa et al., 1997). The major epitopes recognized by CD4+ T cells (VP1233–250, VP274–86, VP324–37), CD8+ T cells (VP3159–166, VP3173–181, VP111–20) and several linear epitopes recognized by B cells (A1A, A1C, A2B, A3A) in the CNS and periphery of TMEV-infected susceptible SJL/J mice (Inoue, Choe, and Kim, 1994; Kang, Lyman, and Kim, 2002; Kim et al., 2000; Oleszak et al., 2004) have been identified on the capsid proteins of TMEV.
In this study, we compared the efficacy of protection from TMEV-induced demyelinating disease following oral and peritoneal administration of intact live virus in order to understand the underlying mechanisms of protection and pathogenesis. Our results indicate that susceptible SJL mice orally pre-exposed to TMEV, but not intraperitoneally, prior to intracerebral infection clear viral persistence in the CNS and become resistant to developing demyelinating disease. However, a prolonged period after oral administration is necessary for effective protection. Orally vaccinated mice displayed markedly elevated levels of antibody response to TMEV in the serum and showed increased early infiltration of B cells producing anti-TMEV antibody into the CNS. Thus, the humoral immune response appears to play an important role in oral immunization-induced protection against TMEV-induced demyelinating disease.
MATERIALS AND METHODS
Mice
Female SJL/J mice, 4–6 weeks old, were purchased from the Charles River Laboratories (Charles River, MA) via the National Cancer Institute. Mice were subsequently housed in the Northwestern University animal care facility. All mouse procedures approved by the Northwestern University Animal Care and Use Committee were used in this study.
Virus
The BeAn strain of TMEV was expanded in BHK cell monolayers in DMEM supplemented with 7% donor calf serum. Cell lysates with known PFU were used as viral stock for animal experiments. Partially purified virus was prepared following centrifugation through 30% sucrose as previously described (Yauch et al., 1998) and used for in vitro assays. Viral titer was determined by the standard plaque assay on BHK cells. Ultraviolet inactivation of virus (UV-TV) was achieved by exposure to a UV light source for 2 hr as previously described (Palma et al., 1999).
Immunization and infection with virus
To vaccinate susceptible SJL/J mice with live TMEV, 1 × 106 − 1 × 107 PFU in 200 μl DMEM were administered either intraperitoneally or perorally (back of oral cavity) via fitted plastic tubing to avoid tissue abrasion starting from 5 weeks of age. After described time periods, live virus was intracerebrally inoculated to induce demyelinating disease. Demyelinating disease was induced by injection of 1 × 106 PFU TMEV in 30 μl into the right cerebral hemisphere of female SJL/J mice after anesthetization. Intracerebral injection with 1 × 106 PFU consistently induces chronic gait abnormalities and neurologic signs in greater than 90% of SJL/J mice. Clinical symptoms of disease were assessed weekly on the following grading scale: grade 0= no clinical signs; grade 1= mild waddling gait; grade 2= moderate waddling gait and hindlimb paresis; grade 3= severe hindlimb paralysis; grade 4= severe hindlimb paralysis and loss of righting reflex; grade 5= death.
Analysis of TMEV-specific Abs
Abs specific for viral epitopes were measured by an adaptation of the indirect ELISA previously described (Inoue, Choe, and Kim, 1994). Briefly, either 0.3 μg of UV-inactivated TMEV virus or 0.1 μg of synthetic peptides conjugated to BSA were used to coat microtiter plates and then the plates were blocked with 1% Blotto. Two-fold serial dilutions of sera in triplicates starting from 1:100 dilution were added, washed, and then allowed to react with alkaline phosphatase-conjugated goat anti-mouse secondary antibody. For assessment of IgG isotypes, polyclonal goat antibodies to mouse IgG1 and IgG2a (Southern Biotech) also recognizing IgG2ab (or IgG2c) of C57BL/6 and SJL/J strains were used (Martin, Brady, and Lew, 1998). The enzyme reaction was developed using p-nitrophenyl phosphate and measured colorimetrically by an ELISA reader at 405 nm.
Measurement of T cell proliferation
T cell proliferation was determined using splenocytes from control or virus-infected SJL/J mice. Single cell suspensions of splenocytes (5 × 105/well) in RPMI-medium supplemented with 5 × 10−5 M 2-ME and 0.5% normal syngeneic mouse serum were cultured for 72 h in triplicate in the presence of 1 or 10 μM peptides or 12.5 μg/ml UV-inactivated TMEV. Approximately 18h after the addition of 1 μCi/well of [3H]-TdR (Amersham, Arlington Heights, IL), cells were harvested and [3H]-TdR incorporation was determined in a liquid scintillation counter. Results are expressed as Δcpm (mean cpm of experimental stimulated cultures after subtraction of the background count with PBS) ± SEM from triplicate cultures.
Preparation of CNS mononuclear cells (MNC)
Sterile HBSS (30 ml) was used to perfuse the tissues through the left ventricle. Brains and spinal cords were then removed, forced through a steel screen, and the resulting cell suspensions were incubated at 37º C for 45 min in 250 μg/ml collagenase type 4 (Worthington Biochemical Corp., Lakewood, NJ). A continuous 100% percoll gradient (Pharmacia, Piscataway, NJ) in the bottom 1/3 of the gradient after centrifugation at 27,000g for 30 min was used to enrich CNS infiltrating lymphocytes. All antibodies used for flow cytometry were purchased from BD Biosciences (San Diego, CA).
ELISPOT assays
To measure IFN-γ producing cells, ELISPOT plates (Millipore, Bedford, MA) were pre-coated with 1–5 μg/ml of anti-IFN-γ antibody in 0.05 M carbonate buffer, pH 9.6. Plates were incubated with 2 × 104 CNS mononuclear cells plus 1 × 106 irradiated (3000 rad) syngeneic spleen cells or 1 × 106 splenocytes from infected mice alone in 200 μl HL-1 medium (Bio-Whittaker, Walkersville, MD) for 18 h at 37°C in the presence of 2 μM peptide. After washing, plates were incubated with biotin-conjugated anti-IFN-γ antibody (Endogen, Boston, MA) overnight. To assess anti-TMEV antibody producing cells, CNS MNC were similarly cultured in ELISPOT plates pre-coated with UV-TMEV (5 μg/ml) for 18 h followed by treatment with biotin-conjugated rabbit anti-mouse IgG. Spots were developed after incubation with streptavidin-HRP for 3 h using 3-amino-9-ethyl-carbazole (Sigma, St. Louis, MO) in 0.05M sodium acetate buffer (Targoni and Lehmann, 1998).
Flow Cytometry
Fc receptors on CNS MNC were blocked using 50 μl of 2.4G2 hybridoma (ATCC) supernatant. Cells were then stained with APC-conjugated CD8 (Clone Ly-2) and PE-conjugated anti-CD4 (clone L3T4) antibodies. To analyze B cells, cells were stained with PE-conjugated anti-CD19 (clone 1D3) antibody.
Statistical analyses
The significance (two-tailed p value) of the differences between experimental animal groups with various treatments and the control group was analyzed based on the unpaired, Student’s t-test by using the InStat Program (GraphPAD Software, San Diego, CA). Differences in disease course between experimental groups were determined by paired two-tailed t-test analysis, with the Welch correction. Values of p<0.05 were considered significant.
RESULTS
A prolonged time period is required after oral immunization to protect from TMEV-IDD
In preliminary studies, we examined whether infection of susceptible SJL/J mice via routes other than intracerebral inoculation can also lead to the development of demyelinating disease. None of the mice infected either intraperitoneally or perorally with TMEV (up to 1 × 107 PFU tested) developed clinical signs of demyelination during 150 d post-infection, whereas 100% of mice infected intracerebrally showed clinical signs at 60 d (data not shown). To assess the time required for the induction of protective immunity following oral administration of live TMEV, age-matched SJL/J mice, which were perorally pre-exposed to 1 × 107 PFU live TMEV for 30 d, 45 d or 51 d, were intracerebrally infected with 1 × 106 PFU TMEV (Fig. 1). Mice immunized perorally developed clinical symptoms of demyelinating disease indistinguishable from untreated control mice. In addition, the difference in disease frequency between these groups was not statistically significant (p=0.08), although the onset of disease appeared to be delayed and the severity reduced. The results clearly indicate that significant protection is not provided at 30 d after oral administration (p>0.05); at least 45 d appears to be required for significant protection (p<0.01) from developing demyelinating disease following intracerebral infection. Induction time of protection was not shortened by repeated oral administration or by increased viral dose (not shown). These data suggest that a prolonged time period is necessary to develop fully protective immunity following oral vaccination.
Figure 1. Requirement of greater than 45 days after oral immunization for effective protection from TMEV-IDD.

Female SJL/J mice were orally vaccinated with 1×107 PFU TMEV BeAn at 30 (n=10), 45 (n=10) or 51 (n=6) days prior to intracerebral infection with 1×106 PFU TMEV. All mouse groups were intracerebrally infected with TMEV on the same day at 15 wk of age. Peroral (PO) immunization resulted in significantly lower incidence of TMEV-IDD compared to those non-immunized. Mice immunized at 45 days or earlier (51 days) prior to intracerebral (ic) infection were effectively protected, but mice immunized at 30 days prior to infection were not. Differences in disease incidences between the non-immunized group and the orally immunized group are as follows, based on a paired, two-tailed Student’s t test with Welch correction between 28 and 56 d post infection: at −30 d, p>0.05 (not significant); −45 d, p<0.01 (very significant); and −51 d, p<0.01 (very significant).
Effective protection is induced following oral, but not peritoneal, infection
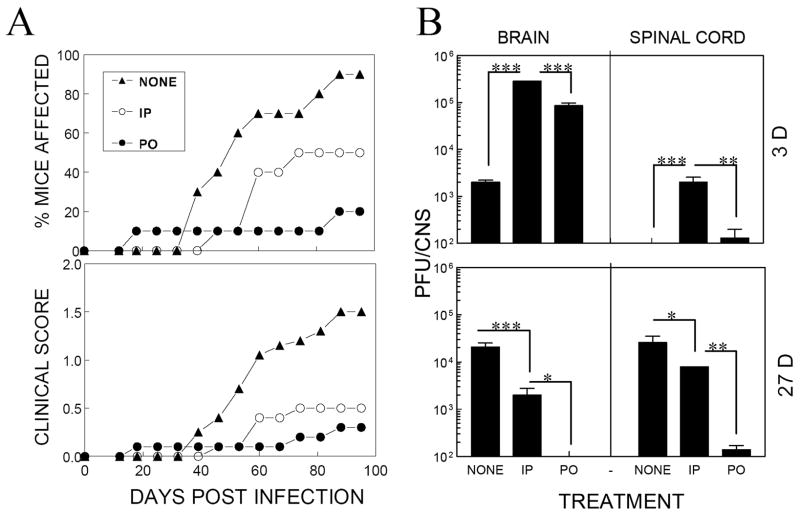
To compare the relative efficacy for protection by the same virus given via different routes, 1 × 107 PFU live virus was administered either perorally or intraperitoneally at 45 d prior to intracerebral infection with 1 × 106 PFU TMEV (Fig. 2). The results clearly indicate that intraperitoneal exposure to the virus prior to intracerebral infection confers significant protection (p<0.0001) against the development of clinical demyelinating disease (Fig. 2A). However, the same dose of virus given perorally induced much more effective protection (p=0.01) compared to that administered intraperitoneally. In contrast, exacerbation was seen after intracerebral administration prior to CNS viral infection instead of protection (data not shown). These data clearly demonstrate that oral administration of intact TMEV induces superior protective immunity against demyelinating disease subsequent to intracerebral infection with the virus, compared to intraperitoneal delivery.
Figure 2. Protection from TMEV-IDD by oral, but not intraperitoneal, immunization.
Female SJL/J mice (n=10 each group) were perorally (po) or intraperitoneally (ip) administered with 1×107 PFU intact BeAn at 45 days prior to intracerebral infection. Untreated mice were intracerebrally infected as a control group. A. Oral immunization resulted in a significantly lower incidence of TMEV-IDD compared to intraperitoneally immunized mice or the untreated control group. Differences among the experimental groups are significant based on ANOVA Tukey-Kramer multiple comparison test between days 39 and 95. **p<0.01, untreated (NONE) vs intraperitoneally (IP) immunized groups; ***p<0.001, NONE vs perorally (PO) immunized groups; *p<0.05, IP vs PO. B. Viral persistence levels in the CNS (brains and spinal cords) of these groups (n=3–4) were assessed by plaque assay at 3 and 27 d post CNS viral infection. TMEV levels at both 3 and 27 days post-infection were significantly lower in PO immunized mice compared to those of IP immunized or unimmunized mice. *, p<0.05; **, p<0.01; and ***; p<0.001.
Viral persistence is drastically reduced in the CNS of orally vaccinated mice
In order to correlate the protection induced by prior exposure to virus with viral persistence following intracerebral infection, levels of replicating TMEV in the CNS of mice untreated and pre-exposed to the virus either orally or peritoneally were compared (Fig. 2B). It is interesting to note that higher virus levels were observed in the CNS (brains and spinal cords) of intraperitoneally vaccinated mice at 3 d post intracerebral infection compared to untreated mice. Similarly, viral levels of orally vaccinated mice were significantly higher (p<0.001) compared to untreated control mice. However, viral levels in the CNS of orally vaccinated mice were significantly lower (p<0.03) than those of peritoneally vaccinated mice. Any plaque-forming viral titers were detectable in the CNS of either ip-infected or po-infected mice at 45 dpi prior to ic-infection (not shown). It is not clear at this time why administration of live virus orally or peritoneally prior to intracerebral infection elevates the initial viral load in the CNS. Perhaps free TMEV or cells harboring persistent TMEV in the periphery of these mice may have rapidly migrated to the CNS. Alternatively, preexisting immune responses in the periphery may have activated glial cells to enhance initial viral replication in the CNS. Nevertheless, initial levels of virus in the CNS (3 d post infection) were not correlated to subsequent disease susceptibility, as control mice without pre-exposure to the virus showed the lowest levels of virus at this time point, yet developed the highest incidence of disease (Fig. 2B). In contrast to viral levels seen immediately (3 d) after intracerebral infection, viral levels in peritoneally treated mice were significantly (p=0.02) reduced at 27 d post infection compared to those of untreated control mice. Orally administered mice showed further reductions of viral persistence both in the brain (p=0.04) and spinal cord (p<0.0001) compared to those seen in peritoneally exposed mice. Therefore, prolonged viral replication in the CNS of intracerebrally infected mice that were previously exposed orally, but not peritoneally, is severely restricted. The viral persistence level of mice orally preexposed to TMEV is consistent with the most effective protection by the treatment against the development of TMEV-IDD (Fig. 2A).
Levels of anti-viral T cell responses in the periphery do not correlate with protection
To examine whether differential levels of T cell responses to viral determinants are induced between orally vaccinated and intraperitoneally vaccinated mice, levels of splenic T cell proliferation responses to viral antigens were assessed at various time points after the administration of live TMEV (Fig. 3). Two weeks (15 d) after the administration of live virus, orally vaccinated mice exhibited slightly higher levels of T cell proliferative responses. However, by the time of intracerebral infection (45 d post immunization), response levels in this group were significantly lower compared to those in intraperitoneally received mice (Fig. 3A). Some of our results indicated that there were detectable levels of antibodies in intraperitoneally immunized mice at 47 d post immunization and small but increased levels of antibody titer following intracerebral infection. However, these levels were consistently lower than those in orally immunized mice. Only marginal differences were detected thereafter between mice that received the virus intraperitoneally or perorally. In order to compare levels of proinflammatory Th1 cell responses, IFNγ levels produced by CD4+ T cells were assessed (Fig. 3B). Levels of IFNγ produced by CD4+ T cells during the early stage of CNS viral infection (45–52 d) were also marginally reduced in mice pre-exposed to the virus orally compared to those in mice pre-exposed intraperitoneally. Similarly, virus-specific IFNγ-producing CD8+ T cell levels in the periphery were increased in both orally and peritoneally pre-exposed mice compared to untreated mice (Fig. 3C). However, no such significant difference was detected between orally and peritoneally immunized mice. These results suggest that the level of peripheral T cell responses to viral determinants induced by immunization with live virus does not correlate with the level of protection from demyelinating disease.
Figure 3. Peripheral T cell responses to TMEV antigens in mice vaccinated via different routes.

A. Proliferative responses of splenic T cells to TMEV antigens in mice orally or intraperitoneally vaccinated with live TMEV (n=3). Splenocytes from mice at days 15/& −30d, 45/0d, 52/+22d or 115/+70d (post immunization/intracerebral infection, respectively) were cultured for 4 d in the presence of UV-inactivated TMEV or synthetic peptides bearing the predominant CD4+ T cell epitopes (VP1 233–250, VP2 74–86, and VP3 24–37). B. IFNγ-producing splenic CD4+ T cells in response to TMEV capsid epitopes determined by ELISPOT assays. C. IFNγ-producing CD8+ T cells in response to TMEV epitopes (VP111–20, VP3159–166 and VP3173–181) determined by ELISPOT assays. NS, not significant; *, p<0.05; **, p<0.01; and ***, p<0.001.
Levels of anti-viral T cell responses in the CNS are higher in orally pre-exposed mice
To further correlate immune responses to TMEV with the efficacy of protection induced by live virus immunizations, levels of T cell responses to viral antigens in the CNS, which is the site of viral persistence and demyelination, were determined by flow cytometry (Fig. 4A). The infiltration levels of overall CD4+ and CD8+ T cells to the CNS of vaccinated mice were significantly increased at 3 d post intracerebral infection. However, these levels became similar to those of untreated mice after 6 d post infection. Further ELISPOT results determining the levels of IFNγ-producing T cells upon stimulation with individual epitopes were consistent with the above flow cytometric analysis (Fig. 4B). Levels of IFNγ-producing CD4+ and CD8+ T cells specific for viral determinants in the CNS of orally vaccinated mice were consistently higher than those of intraperitoneally vaccinated mice throughout the course of viral infection. These results suggest that oral administration of live virus is capable of effectively enhancing the level of virus-specific T cells, in particular the level of virus-specific CD8+ T cells, in the CNS following intracerebral infection of TMEV (Fig. 4), in contrast to those in the periphery (Fig. 3).
Figure 4. Levels of T cells specific to TMEV antigens in the CNS of mice vaccinated via different routes.

A. Flow cytometric analyses of T cells among the CNS mononuclear cells from vaccinated or untreated mice at 3, 6, and 27 d post intracerebral TMEV infection. The proportion of IFNγ-producing cells in response to the mixture of CD4+ T cell epitope peptides and CD8+ T cell epitope peptides was determined by flow cytometry following intracellular cytokine staining. One representative of three separate experiments with similar results is shown here. B. The assessment of IFNγ-producing T cells infiltrating the CNS of vaccinated mice in response to individual TMEV epitope peptides by ELISPOT assay at 6 and 14 d post CNS TMEV infection. The statistic values represent the significance between vaccinated and non-vaccinated mice and the values inside the parentheses represent the difference between orally vaccinated and peritoneally vaccinated mice. *, p<0.05; **, p<0.01; and ***, p<0.001. When the values of three CD8+ T cell epitopes (VP111–20, VP3159–166 and VP3173–181) were pooled, ***, NONE vs IP; ***, NONE vs PO; **, IP vs PO at 6 d post infection and *, NONE vs IP; ***, NONE vs PO; *, IP vs PO at 14 d post infection.
Antibody levels in serum are higher in mice immunized perorally than peritoneally
To examine whether higher levels of antibodies produced following oral immunization are responsible for effective prevention from the development of demyelinating disease, TMEV-specific antibody levels in mice vaccinated via different routes were assessed during the course of viral infection (Fig. 5). It is interesting to note that only orally vaccinated mice showed a significant level of anti-TMEV antibodies in the serum at the time of intracerebral TMEV infection (47 d post immunization) and during early (7 d) CNS viral infection (Fig. 5A). However, the level of antibodies in intraperitoneally immunized mice gradually increased to the level of orally vaccinated mice at 22 d post CNS infection and thereafter; such an increase also followed in control mice that were not vaccinated. In vitro virus-neutralizing levels of sera correlated with anti-TMEV antibody levels, but did not correlate with disease levels (not shown), as previously shown (Inoue et al., 1994). Both IgG1 and IgG2a isotypes of anti-TMEV antibodies were produced, suggesting the lack of Th type preference for antibody production (Fig. 5B). No difference in the levels of anti-TMEV antibodies (IgA and IgG) was observed in the saliva of these mice at the time of intracerebral infection, although oral administration initially induced a higher level of these antibodies (Fig. 5C). These results suggest that the presence of a high level of serum anti-TMEV antibodies at the early stage of intracerebral infection correlates with effective protection from demyelinating disease.
Figure 5. Antibody levels to TMEV in mice vaccinated orally and peritoneally.

A. Mice were bled at 17/−30d, 47/0d, 54/+7d, 69/+22d, 117/+70d or 159/+112d post-immunization followed by intracerebral infection, respectively. Antibody Levels to TMEV in pooled serum (n=5) were analyzed by ELISA. Sera from orally vaccinated mice show markedly higher levels of antibodies to TMEV compared to those from peritoneally vaccinated mice (p<0.001) in the early stages of viral infection (47/0 and 54/+7 d post immunization/infection). B. The isotype determination of anti-TMEV antibody in pooled sera from mice at 47/0 d post immunization/infection. Serum IgG subclasses (IgG1 and IgG2a) were determined by ELISA. C. Antibody titers detected in saliva reactive to UV-inactivated TMEV. Saliva washes were collected from mice at 17/−30d and 54/7d post immunization/infection. Samples were pooled and analyzed by ELISA. Total IgA levels were not different between the groups (data not shown).
More TMEV-specific B cells are initially present in the CNS of orally immunized mice
To further correlate the level of antibodies elevated in vaccinated mice at the early stage of CNS viral infection with the efficacy of protection against TMEV-IDD, levels of CD19+ B cells specific for TMEV in the CNS and the periphery were determined (Fig. 6). Levels of B cells infiltrating the CNS were relatively higher in orally vaccinated mice compared to those in peritoneally vaccinated or untreated mice during the early stage (3–6 d) of CNS viral infection (Fig. 6A). Levels of anti-TMEV antibody-producing B cells in the CNS and periphery were further determined by ELISPOT assay using UV-TMEV coated plates (Fig. 6B). The results clearly indicate that significantly higher levels of anti-TMEV antibody-producing B cells are present in the CNS and periphery during the early phase (3–14 d) of viral infection in orally vaccinated mice compared to intraperitoneally vaccinated mice. Therefore, both the level of serum antibodies and the number of antibody-producing B cells in the CNS correlate well with the efficacy of protection, consistent again with the possibility that the primary mechanism of protection from TMEV-IDD following oral immunization is via the production of virus-specific antibodies.
Figure 6. TMEV-specific B cells in the CNS and periphery during the course of viral infection.

A. Flow cytometric analysis of B cells infiltrating the CNS of mice (47 d prior to viral infection) at 50/3, 53/6 and 74/27 d post immunization/infection, respectively. The percentages of CD19+ cells in the CNS MNC from non-vaccinated, orally and peritoneally vaccinated mice were determined at 3, 6, and 27 d post infection. B. Enumeration of anti-TMEV antibody-producing cells in the CNS and periphery during the course of viral infection. *, p<0.05; **, p<0.01; and ***, p<0.001. A representative of three separate experiments is shown here.
Antibodies to a predominant VP1 epitope in orally vaccinated mice are elevated
To further analyze the nature of antibodies at the beginning of viral infection in mice vaccinated via different routes, the reactivity of antibodies to previously characterized predominant, linear antibody epitopes (Inoue, Choe, and Kim, 1994) was assessed (Fig. 7). Susceptible SJL mice intracerebrally infected with TMEV were shown to strongly recognize the A1C epitope (VP1262–276) as compared to resistant BALB/c or C57BL/6 mice (Inoue, Choe, and Kim, 1994). The antibody population reactive to this viral epitope was predominant in orally vaccinated mice, but not in peritoneally vaccinated mice, during the early stage (within 7 d) of viral infection (Fig. 7A). This pattern is consistent with our previous results demonstrating that preimmunization with either VP1 protein or A1C-epitope peptide can protect mice against subsequent intracerebral TMEV infection (Yahikozawa et al., 1997). However, antibody levels to this epitope become predominant during late infection regardless of the route of immunization, as previously shown in mice with clinical symptoms of demyelination (Yahikozawa et al., 1997). It is unclear why mice displaying elevated levels of such antibodies during late infection are not protected against TMEV-IDD. In order to determine the potential role of antibodies to this epitope during the late stage of viral infection, pooled serum from mice immunized with A1C-KLH was transfused to naïve SJL/J mice before or after intracerebral TMEV infection. The disease courses of these groups were then compared to each other and to an untreated mouse group (Fig. 7B). The results clearly indicate that the presence of antibodies during the beginning of viral infection to the CNS, but not during late infection, is protective against TMEV-IDD. In fact, the presence of such antibodies after the establishment of viral persistence appears to exacerbate the disease. These results are consistent with previous reports demonstrating that pre-existing antibodies to TMEV are protective, although clinically affected mice with demyelinating disease display high levels of such antibodies, which are capable of neutralizing viral infection/replication in vitro (Kang et al., 2005; Mohindru, Kang, and Kim, 2006; Yahikozawa et al., 1997; Inoue, Choe, and Kim, 1994).
Figure 7. Epitope reactivity and the potential role of antibodies in virus-infected mice.

A. Reactivity of anti-TMEV antibodies to linear epitopes in sera from mice (n=4) at 47/0 d and 103/+56 d post immunization/infection. Antibodies to A1C (VP1262–276) and A3A (VP324–37) were relatively higher in orally vaccinated mice at 47/0 d compared to peritoneally vaccinated mice; the level of antibodies to A1C selectively increased after viral infection regardless of immunization. B. Effect of transfusion of antibodies specific to the A1C epitope on the development of demyelinating disease. Naïve SJL/J mice were repeatedly immunized with A1C-KLH and the serum was transfused into SJL/J mice (n=10/group) before (−5, 0 and 5 d) or after (12, 19, 25 d) intracerebral viral infection. Differences between groups that received serum antibodies before and after viral infection (p<0.002) and between the group that received antibodies after viral infection and the group that did not receive any antibodies (P=0.01) were very significant based on a paired, two-tailed Student’s t test between 33 and 61 d post infection. Also, the difference between the group that received antibodies prior to viral infection and the group that did not receive antibodies was significant (P<0.03) based on a paired, two-tailed Student’s t test between 47–75 d post infection. Similar results were observed in a separate additional experiment.
DISCUSSION
Despite the fact that oral vaccination is widely used to protect against many viral infections (Cox, Brokstad, and Ogra, 2004; Fekadu et al., 1996; Foxwell, Cripps, and Dear, 2006; Ramakrishna et al., 1999; Sabin, 1985), very few systematic studies have been conducted to examine the time required to obtain maximal protection and the type of immune responses involved in this protection. In this study, we investigated the potential protective effects of pre-exposure to live virus through non-pathogenic routes, i.e. intraperitoneally and perorally, against TMEV-induced demyelinating disease. This virus is widely spread via fecal-oral routes among the wild type mouse population without apparent disease (Lipton et al., 2001; Thompson, Harrison, and Myers, 1951; and not shown). Therefore, such studies may provide important information regarding the relationship between the pathogenesis of inflammatory demyelination upon access to the CNS and previous exposure to the virus via oral infection. Our results indicate that susceptible SJL/J mice pre-exposed to live TMEV perorally, but not intraperitoneally, are resistant against TMEV-induced demyelinating disease (Figs. 1 & 2). It is interesting to note that a prolonged time period (>45 d) is necessary to acquire effective protection following oral immunization. The mechanisms involved in such delayed protection are unclear. However, a similar delayed period is also required to produce significant levels of antibodies in the serum (Fig. 5), which apparently provide important protective immunity (Kang et al., 2005; Rodriguez et al., 1990). In addition, it was previously shown that intraperitoneal immunization with attenuated TMEV significantly reduce viral load in the CNS through the production of neutralizing antibodies (Kurtz, Sun, and Fujinami, 1995). Thus, it is most likely that the presence of a high level of anti-viral antibodies prior to CNS viral infection is responsible for the protection provided by oral immunization (Fig. 7). Since the majority of picornaviruses, including TMEV, are enteric viruses that spread through the fecal-oral route, a prolonged stimulation of immune responses by viruses residing in the mucosal system may provide an effective generation of such protective immunity. However, TMEV introduced intraperitoneally may be cleared more rapidly and/or may be ineffective at inducing vigorous antibody responses.
The roles of antibodies in controlling viral persistence and protecting mice against TMEV-induced demyelinating disease are not yet completely understood. It is generally accepted that the antibody response to various viruses is protective by either neutralizing viral infection or inhibiting viral replication (Burton, 2002; Griffin et al., 1997). In addition, several neurotropic viruses, including mouse hepatitis virus, require the continuous presence of antibodies in order to effectively control viral persistence, as a lack of antibodies results in viral recrudesces (Ramakrishna et al., 2003). However, antibodies specific to viral determinants do not play a significant role in either the protection or pathogenesis of CNS diseases in some viral systems (Burton, 2002; Schneider-Schaulies, Meulen, and Schneider-Schaulies, 2003). Therefore, it is not yet clear whether antibodies play protective and/or pathogenic roles in certain virus-induced diseases.
The potential role of antibodies specific for TMEV in viral clearance and protection from TMEV-IDD has also been previously investigated. B cells producing anti-TMEV antibodies have been identified in the demyelinating lesion (Cash et al., 1989). Some antibodies to TMEV appear to cross-react with autoantigens on the myelin and oligodendrocytes, suggesting a pathogenic role of such antibodies (Yamada, Zurbriggen, and Fujinami, 1990). Furthermore, elevated antibody responses are seen in the CNS as well as in the periphery of susceptible mice undergoing demyelinating disease compared to healthy TMEV-infected resistant mice (Inoue, Choe, and Kim, 1994). These results suggest a potential pathogenic role of antibodies to TMEV. However, the removal of B cells by treatment with anti-IgM antibody exacerbates disease (Kurtz, Sun, and Fujinami, 1995; Rodriguez et al., 1990). A marked decrease in the number of virus-infected cells in the CNS of susceptible mice was also observed after passive transfer of sera from virus-infected resistant donors (Borrow, Welsh, and Nash, 1993; Rossi et al., 1991). Furthermore, susceptible SJL mice preimmunized with isolated capsid proteins or linear antibody epitopes conjugated to carrier molecules are resistant to TMEV-induced demyelination (Yahikozawa et al., 1997). Recently, we demonstrated that antibody response is critical in protecting mice against TMEV-induced disease, especially when the level of CD8+ T cell response is low (Kang et al., 2005). These findings are consistent with our present study indicating that the presence of antibodies specific for TMEV prior to and/or during the initial stage of viral infection following oral immunization (Fig. 5) or serum transfusion (Fig. 7) provides powerful protection against virally induced demyelinating disease. However, such antibodies during chronic viral infection may not be protective and/or may even be pathogenic (Fig. 7). Taken together, these results indicate that the time of availability of a sufficient level of anti-viral antibodies is critical for effective protection.
The correlation between the level of peripheral T cell responses and the efficacy of protection from TMEV-induced demyelinating disease is less clear. It was previously shown that CD4+ T cells raise antibody levels, which are critical for protection during the early stage of viral infection (Borrow, Welsh, and Nash, 1993). Peripheral T cell responses in both CD4+ and CD8+ T cell populations in either peritoneally or orally vaccinated mice showed significant increases compared to untreated mice. However, differences in peripheral T cell responses between the vaccinated groups were not consistent with their levels of protection (Fig. 3). If anything, these levels were often somewhat lower in orally vaccinated mice compared to those in peritoneally vaccinated mice. It is unclear at this time why there is a discrepancy between the level of peripheral T cell responses and the efficacy of protection, which correlates with the level of anti-TMEV antibody in the blood. It is conceivable that virus-specific T cells may have been sequestered at the mucosal site in orally vaccinated mice, leading to a lower T cell level in the periphery compared to peritoneally vaccinated mice. This possibility is consistent with the higher levels of virus-specific antibodies found in the circulation and the number of T cells present in the CNS during the beginning of intracerebral infection (Figs. 4 & 5). Therefore, it is most likely that both virus-specific T cells and antibodies play protective roles against TMEV-induced demyelinating disease during the early stage of viral infection, as was recently demonstrated for TMEV-specific CD8+ T cells (Lyman et al., 2004), CD4+ T cells (Mohindru, Kang, and Kim, 2006) and antibodies (Kang et al., 2005).
The level of CNS-infiltrating virus-specific immune cells showed a clearer correlation to the efficacy of vaccine-induced protection. Levels of both anti-viral CD4+ and CD8+ T cell responses in the CNS are the highest in orally vaccinated mice, followed by peritoneally vaccinated and then untreated mice during the early stage of viral infection (Fig. 4). In particular, levels of CD8+ T cells in the CNS of orally vaccinated mice are significantly higher. Similarly, levels of CNS-infiltrating B cells specific for TMEV are also highest in orally vaccinated mice among the groups during the early stage of CNS viral infection (Fig. 6). These results strongly suggest that the levels of virus-specific immune cells present in the CNS during early infection are critically important in controlling viral load and its consequent viral persistence in the CNS. The significant elevation of immune responses in the CNS during early viral infection appears to be most effectively achieved following oral immunization, resulting in superior protection against TMEV-induced demyelinating disease. Therefore, the widely spread infection of TMEV in wild type mice may function as a powerful means to provide strong protection against this virus, which causes devastating disease when it accesses the CNS.
Acknowledgments
* This work was supported by United States Public Health Service Grants, RO1 NS28752, RO1 NS33008, and PO1 NS23349.
Abbreviations
- MS
Multiple sclerosis
- TMEV
Theiler’s murine encephalomyelitis virus
- TMEV-IDD
TMEV-induced demyelinating disease
- CNS
central nervous system
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Begolka WS, Haynes LM, Olson JK, Padilla J, Neville KL, Dal Canto M, Palma J, Kim BS, Miller SD. CD8-deficient SJL mice display enhanced susceptibility to Theiler’s virus infection and increased demyelinating pathology. J Neurovirol. 2001;7:409–20. doi: 10.1080/135502801753170264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borrow P, Welsh CJ, Nash AA. Study of the mechanisms by which CD4+ T cells contribute to protection in Theiler’s murine encephalomyelitis. Immunology. 1993;80:502–6. [PMC free article] [PubMed] [Google Scholar]
- Burton D. Antibodies, viruses and vaccines. Nat Rev Immunol. 2002;2:706–13. doi: 10.1038/nri891. [DOI] [PubMed] [Google Scholar]
- Cash E, Bandeira A, Chirinian S, Brahic M. Characterization of B lymphocytes present in the demyelinating lesions induced by Theiler’s virus [published erratum appears in J Immunol 1989 Sep 15;143(6):2081] J Immunol. 1989;143:984–8. [PubMed] [Google Scholar]
- Cox RJ, Brokstad KA, Ogra P. Influenza virus: immunity and vaccination strategies. Comparison of the immune response to inactivated and live, attenuated influenza vaccines. Scand J Immunol. 2004;59:1–15. doi: 10.1111/j.0300-9475.2004.01382.x. [DOI] [PubMed] [Google Scholar]
- Dal Canto MC, Kim BS, Miller SD, Melvold RW. Theiler’s murine encephalomyelitis virus (TMEV)-induced demyelination: a model for human multiple clerosis. Methods. 1996;10:453–461. doi: 10.1006/meth.1996.0123. [DOI] [PubMed] [Google Scholar]
- Fekadu M, Nesby SL, Shaddock JH, Schumacher CL, Linhart SB, Sanderlin DW. Immunogenicity, efficacy and safety of an oral rabies vaccine (SAG-2) in dogs. Vaccine. 1996;14:465–8. doi: 10.1016/0264-410x(95)00244-u. [DOI] [PubMed] [Google Scholar]
- Fiette L, Aubert C, Brahic M, Rossi CP. Theiler’s virus infection of beta 2-microglobulin-deficient mice. J Virol. 1993;67:589–92. doi: 10.1128/jvi.67.1.589-592.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foxwell AR, Cripps AW, Dear KB. Haemophilus influenzae oral whole cell vaccination for preventing acute exacerbations of chronic bronchitis. Cochrane Database Syst Rev. 2006;(4):CD001958. doi: 10.1002/14651858.CD001958.pub2. [DOI] [PubMed] [Google Scholar]
- Fujinami RS, Rosenthal A, Lampert PW, Zurbriggen A, Yamada M. Survival of athymic (nu/nu) mice after Theiler’s murine encephalomyelitis virus infection by passive administration of neutralizing monoclonal antibody. J Virol. 1989;63:2081–2087. doi: 10.1128/jvi.63.5.2081-2087.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerety SJ, Rundell MK, Dal Canto MC, Miller SD. Class II-restricted T cell responses in Theiler’s murine encephalomyelitis virus-induced demyelinating disease. VI. Potentiation of demyelination with and characterization of an immunopathologic CD4+ T cell line specific for an immunodominant VP2 epitope. J Immunol. 1994;152:919–29. [PubMed] [Google Scholar]
- Griffin D, Levine B, Tyor W, Ubol S, Despres P. The role of antibody in recovery from alphavirus encephalitis. Immunol Rev. 1997;159:155–61. doi: 10.1111/j.1600-065x.1997.tb01013.x. [DOI] [PubMed] [Google Scholar]
- Inoue A, Choe YK, Kim BS. Analysis of antibody responses to predominant linear epitopes of Theiler’s murine encephalomyelitis virus. J Virol. 1994;68:3324–33. doi: 10.1128/jvi.68.5.3324-3333.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson AJ, Njenga MK, Hansen MJ, Kuhns ST, Chen L, Rodriguez M, Pease LR. Prevalent class I-restricted T-cell response to the Theiler’s virus epitope Db:VP2121-130 in the absence of endogenous CD4 help, tumor necrosis factor alpha, gamma interferon, perforin, or costimulation through CD28. J Virol. 1999;73:3702–8. doi: 10.1128/jvi.73.5.3702-3708.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang BS, Lyman MA, Kim BS. The majority of infiltrating CD8+ T cells in the central nervous system of susceptible SJL/J mice infected with Theiler’s virus are virus specific and fully functional. J Virol. 2002;76:6577–85. doi: 10.1128/JVI.76.13.6577-6585.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang BS, Palma JP, Lyman MA, Dal Canto M, Kim BS. Antibody response is required for protection from Theiler’s virus-induced encephalitis in C57BL/6 mice in the absence of CD8+T cells. Virology. 2005;340:84–94. doi: 10.1016/j.virol.2005.06.028. [DOI] [PubMed] [Google Scholar]
- Karls KA, Denton PW, Melvold RW. Susceptibility to Theiler’s murine encephalomyelitis virus-induced demyelinating disease in BALB/cAnNCr mice is related to absence of a CD4+ T-cell subset. Mult Scler. 2002;8:469–74. doi: 10.1191/1352458502ms850oa. [DOI] [PubMed] [Google Scholar]
- Kim BS, Palma JP, Inoue A, Koh CS. Pathogenic immunity in Theiler’s virus-induced demyelinating disease: a viral model for multiple sclerosis. Arch Immunol Ther Exp. 2000;48:373–9. [PubMed] [Google Scholar]
- Kurtz CI, Sun XM, Fujinami RS. B-lymphocyte requirement for vaccine-mediated protection from Theiler’s murine encephalomyelitis virus-induced central nervous system disease. J Virol. 1995;69(8):5152–5. doi: 10.1128/jvi.69.8.5152-5155.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipton HL. Theiler’s virus infection in mice: an unusual biphasic disease process leading to demyelination. Infect Immun. 1975;11:1147–1155. doi: 10.1128/iai.11.5.1147-1155.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipton HL, Kim BS, Yahikozawa H, Nadler CF. Serological evidence that Mus musculus is the natural host of Theiler’s murine encephalomyelitis virus. Virus Res. 2001;76:79–86. doi: 10.1016/s0168-1702(01)00256-8. [DOI] [PubMed] [Google Scholar]
- Lyman MA, Myoung J, Mohindru M, Kim BS. Quantitative, not qualitative, differences in CD8+ T cell responses to Theiler’s murine encephalomyelitis virus between resistant C57BL/6 and susceptible SJL/J mice. Eur J Immunol. 2004;34:2730–9. doi: 10.1002/eji.200324811. [DOI] [PubMed] [Google Scholar]
- Martin RM, Brady JL, Lew AM. The need for IgG2c specific antiserum when isotyping antibodies from C57BL/6 and NOD mice. J Immunol Methods. 1998;212:187–92. doi: 10.1016/s0022-1759(98)00015-5. [DOI] [PubMed] [Google Scholar]
- Mohindru M, Kang B, Kim BS. Initial capsid-specific CD4+ T cell responses protect against Theiler’s murine encephalomyelitisvirus-induced demyelinating disease. Eur J Immunol. 2006;36:2106–2115. doi: 10.1002/eji.200535785. [DOI] [PubMed] [Google Scholar]
- Murray PD, McGavern DB, Lin X, Njenga MK, Leibowitz J, Pease LR, Rodriguez M. Perforin-dependent neurologic injury in a viral model of multiple sclerosis. J Neurosci. 1998a;18:7306–14. doi: 10.1523/JNEUROSCI.18-18-07306.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray PD, Pavelko KD, Leibowitz J, Lin X, Rodriguez M. CD4+ and CD8(+) T cells make discrete contributions to demyelination and neurologic disease in a viral model of multiple sclerosis. J Virol. 1998b;72:7320–9. doi: 10.1128/jvi.72.9.7320-7329.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Njenga MK, Pavelko KD, Baisch J, Lin X, David C, Leibowitz J, Rodriguez M. Theiler’s virus persistence and demyelination in major histocompatibility complex class II-deficient mice. J Virol. 1996;70:1729–37. doi: 10.1128/jvi.70.3.1729-1737.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleszak EL, Chang JR, Friedman H, Katsetos CD, Platsoucas CD. Theiler’s virus infection: a model for multiple sclerosis. Clin Microbiol Rev. 2004;17:174–207. doi: 10.1128/CMR.17.1.174-207.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palma JP, Kim BS. Induction of selected chemokines in glial cells infected with Theiler’s virus. J Neuroimmunol. 2001;117:166–70. doi: 10.1016/s0165-5728(01)00326-5. [DOI] [PubMed] [Google Scholar]
- Palma JP, Yauch RL, Lang S, Kim BS. Potential role of CD4+ T cell-mediated apoptosis of activated astrocytes in Theiler’s virus-induced demyelination. J Immunol. 1999;162:6543–51. [PubMed] [Google Scholar]
- Pope JG, Karpus WJ, VanderLugt C, Miller SD. Flow cytometric and functional analyses of central nervous system- infiltrating cells in SJL/J mice with Theiler’s virus-induced demyelinating disease. Evidence for a CD4+ T cell-mediated pathology. J Immunol. 1996;156:4050–8. [PubMed] [Google Scholar]
- Pullen LC, Miller SD, Dal Canto MC, Kim BS. Class I-deficient resistant mice intracerebrally inoculated with Theiler’s virus show an increased T cell response to viral antigens and susceptibility to demyelination. Eur J Immunol. 1993;23:2287–93. doi: 10.1002/eji.1830230935. [DOI] [PubMed] [Google Scholar]
- Ramakrishna C, Bergmann CC, Atkinson R, Stohlman SA. Control of central nervous system viral persistence by neutralizing antibody. J Virol. 2003;77:4670–8. doi: 10.1128/JVI.77.8.4670-4678.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramakrishna C, Desai A, Shankar SK, Chandramuki A, Ravi V. Oral immunisation of mice with live Japanese encephalitis virus induces a protective immune response. Vaccine. 1999;17:3102–8. doi: 10.1016/s0264-410x(99)00137-1. [DOI] [PubMed] [Google Scholar]
- Rodriguez M, Dunkel AJ, Thiemann RL, Leibowitz J, Zijlstra M, Jaenisch R. Abrogation of resistance to Theiler’s virus-induced demyelination in H-2b mice deficient in beta 2-microglobulin. J Immunol. 1993;151:266–76. [PubMed] [Google Scholar]
- Rodriguez M, Kenny JJ, Thiemann RL, Woloschak GE. Theiler’s virus-induced demyelination in mice immunosuppressed with anti-IgM and in mice expressing the xid gene. Microb Pathog. 1990;8:23–35. doi: 10.1016/0882-4010(90)90005-b. [DOI] [PubMed] [Google Scholar]
- Rossi CP, Cash E, Aubert C, Coutinho A. Role of the humoral immune response in resistance to Theiler’s virus infection. J Virol. 1991;65:3895–9. doi: 10.1128/jvi.65.7.3895-3899.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabin AB. Oral poliovirus vaccine: history of its development and use and current challenge to eliminate poliomyelitis from the world. J Infect Dis. 1985;151:420–36. doi: 10.1093/infdis/151.3.420. [DOI] [PubMed] [Google Scholar]
- Schneider-Schaulies J, Meulen V, Schneider-Schaulies S. Measles infection of the central nervous system. J Neurovirol. 2003;9:247–52. doi: 10.1080/13550280390193993. [DOI] [PubMed] [Google Scholar]
- Targoni OS, Lehmann PV. Endogenous myelin basic protein inactivates the high avidity T cell repertoire. J Exp Med. 1998;187:2055–63. doi: 10.1084/jem.187.12.2055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson R, Harrison VM, Myers FP. A spontaneous epizootic of mouse encephalomyelitis. Proc Soc Exp Biol Med. 1951;77:262–6. doi: 10.3181/00379727-77-18744. [DOI] [PubMed] [Google Scholar]
- Vesikari T, Giaquinto C, Huppertz HI. Clinical trials of rotavirus vaccines in Europe. Pediatr Infect Dis J. 2006;25:S42–7. doi: 10.1097/01.inf.0000197565.45345.4e. [DOI] [PubMed] [Google Scholar]
- Yahikozawa H, Inoue A, Koh CS, Choe YK, Kim BS. Major linear antibody epitopes and capsid proteins differentially induce protective immunity against Theiler’s virus-induced demyelinating disease. J Virol. 1997;71:3105–13. doi: 10.1128/jvi.71.4.3105-3113.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada M, Zurbriggen A, Fujinami RS. Monoclonal antibody to Theiler’s murine encephalomyelitis virus defines a determinant on myelin and oligodendrocytes, and augments demyelination in experimental allergic encephalomyelitis. J Exp Med. 1990;171:1893–1907. doi: 10.1084/jem.171.6.1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yauch RL, Kim BS. A predominant viral epitope recognized by T cells from the periphery and demyelinating lesions of SJL/J mice infected with Theiler’s virus is located within VP1(233–244) J Immunol. 1994;153:4508–19. [PubMed] [Google Scholar]
- Yauch RL, Palma JP, Yahikozawa H, Koh CS, Kim BS. Role of individual T-cell epitopes of Theiler’s virus in the pathogenesis of demyelination correlates with the ability to induce a Th1 response. J Virol. 1998;72:6169–6174. doi: 10.1128/jvi.72.7.6169-6174.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X, Zhang M, Li Z, Frankel FR. Vaginal protection and immunity after oral immunization of mice with a novel vaccine strain of Listeria monocytogenes expressing human immunodeficiency virus type 1 gag. J Virol. 2006;80:8880–90. doi: 10.1128/JVI.00894-06. [DOI] [PMC free article] [PubMed] [Google Scholar]