Abstract
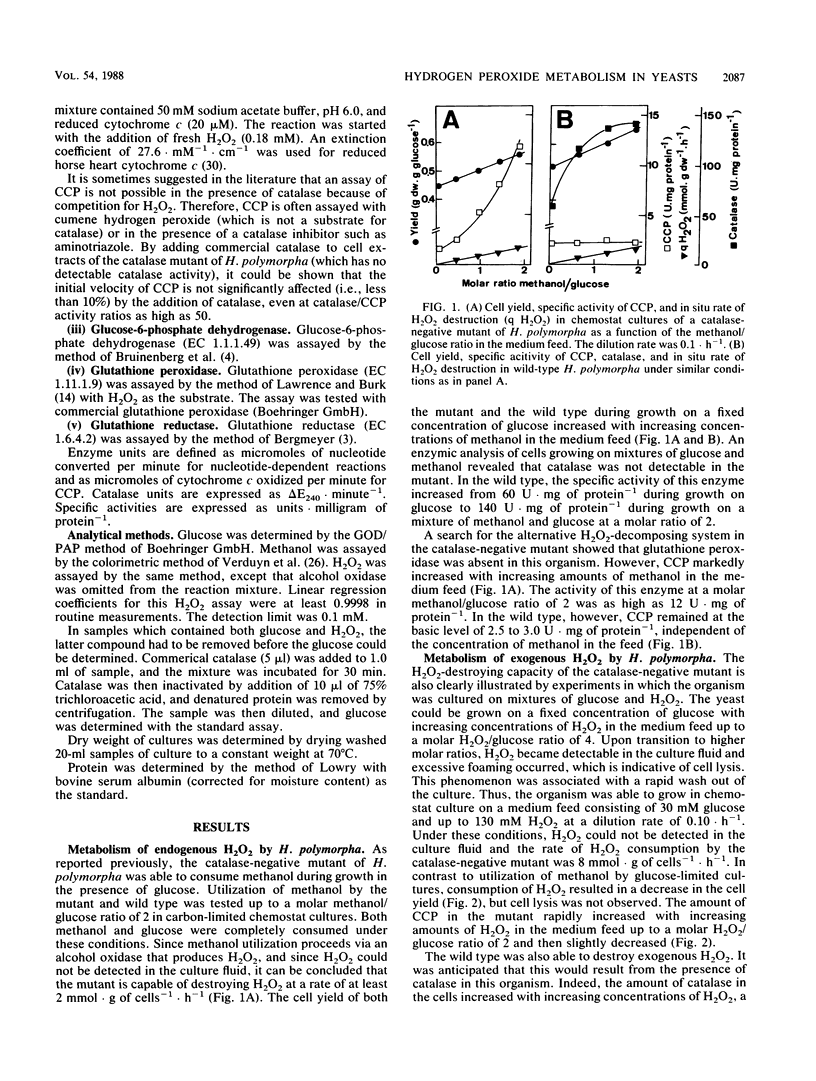
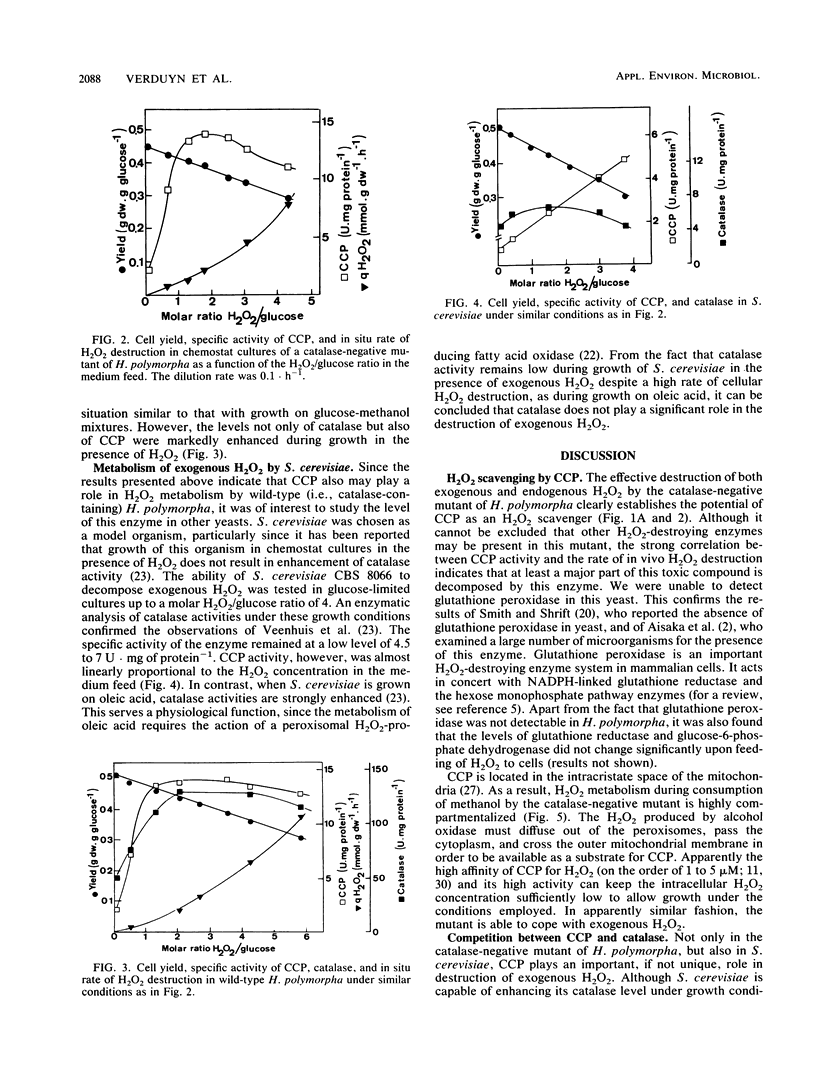
A catalase-negative mutant of the yeast Hansenula polymorpha consumed methanol in the presence of glucose when the organism was grown in carbon-limited chemostat cultures. The organism was apparently able to decompose the H2O2 generated in the oxidation of methanol by alcohol oxidase. Not only H2O2 generated intracellularly but also H2O2 added extracellularly was effectively destroyed by the catalase-negative mutant. From the rate of H2O2 consumption during growth in chemostat cultures on mixtures of glucose and H2O2, it appeared that the mutant was capable of decomposing H2O2 at a rate as high as 8 mmol · g of cells−1 · h−1. Glutathione peroxidase (EC 1.11.1.9) was absent under all growth conditions. However, cytochrome c peroxidase (CCP; EC 1.11.1.5) increased to very high levels in cells which decomposed H2O2. When wild-type H. polymorpha was grown on mixtures of glucose and methanol, the CCP level was independent of the rate of methanol utilization, whereas the level of catalase increased with increasing amounts of methanol in the substrate feed. Also, the wild type decomposed H2O2 at a high rate when cells were grown on mixtures of glucose and H2O2. In this case, an increase of both CCP and catalase was observed. When Saccharomyces cerevisiae was grown on mixtures of glucose and H2O2, the level of catalase remained low, but CCP increased with increasing rates of H2O2 utilization. From these observations and an analysis of cell yields under the various conditions, two conclusions can be drawn. (i) CCP is a key enzyme of H2O2 detoxification in yeasts. (ii) Catalase can effectively compete with mitochondrial CCP for hydrogen peroxide only if hydrogen peroxide is generated at the site where catalase is located, namely in the peroxisomes.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bruinenberg P. M., van Dijken J. P., Scheffers W. A. An enzymic analysis of NADPH production and consumption in Candida utilis. J Gen Microbiol. 1983 Apr;129(4):965–971. doi: 10.1099/00221287-129-4-965. [DOI] [PubMed] [Google Scholar]
- Daum G., Böhni P. C., Schatz G. Import of proteins into mitochondria. Cytochrome b2 and cytochrome c peroxidase are located in the intermembrane space of yeast mitochondria. J Biol Chem. 1982 Nov 10;257(21):13028–13033. [PubMed] [Google Scholar]
- De Duve C., Baudhuin P. Peroxisomes (microbodies and related particles). Physiol Rev. 1966 Apr;46(2):323–357. doi: 10.1152/physrev.1966.46.2.323. [DOI] [PubMed] [Google Scholar]
- Eggeling L., Sahm H. Regulation of alcohol oxidase synthesis in Hansenula polymorpha: oversynthesis during growth on mixed substrates and induction by methanol. Arch Microbiol. 1980 Sep;127(2):119–124. doi: 10.1007/BF00428015. [DOI] [PubMed] [Google Scholar]
- Erecínska M., Oshino N., Loh P., Brocklehurst E. In vitro studies on yeast cytochrome c peroxidase and its possible function in the electron transfer and energy coupling reactions. Biochim Biophys Acta. 1973 Jan 18;292(1):1–12. doi: 10.1016/0005-2728(73)90245-4. [DOI] [PubMed] [Google Scholar]
- Kang C. H., Ferguson-Miller S., Margoliash E. Steady state kinetics and binding of eukaryotic cytochromes c with yeast cytochrome c peroxidase. J Biol Chem. 1977 Feb 10;252(3):919–926. [PubMed] [Google Scholar]
- Kaput J., Goltz S., Blobel G. Nucleotide sequence of the yeast nuclear gene for cytochrome c peroxidase precursor. Functional implications of the pre sequence for protein transport into mitochondria. J Biol Chem. 1982 Dec 25;257(24):15054–15058. [PubMed] [Google Scholar]
- Lawrence R. A., Burk R. F. Glutathione peroxidase activity in selenium-deficient rat liver. Biochem Biophys Res Commun. 1976 Aug 23;71(4):952–958. doi: 10.1016/0006-291x(76)90747-6. [DOI] [PubMed] [Google Scholar]
- Ohnishi T., Kawaguchi K., Hagihara B. Preparation and some properties of yeast mitochondria. J Biol Chem. 1966 Apr 25;241(8):1797–1806. [PubMed] [Google Scholar]
- Reid G. A., Yonetani T., Schatz G. Import of proteins into mitochondria. Import and maturation of the mitochondrial intermembrane space enzymes cytochrome b2 and cytochrome c peroxidase in intact yeast cells. J Biol Chem. 1982 Nov 10;257(21):13068–13074. [PubMed] [Google Scholar]
- Seah T. C., Kaplan J. G. Purification and properties of the catalase of bakers' yeast. J Biol Chem. 1973 Apr 25;248(8):2889–2893. [PubMed] [Google Scholar]
- Veenhuis M., Mateblowski M., Kunau W. H., Harder W. Proliferation of microbodies in Saccharomyces cerevisiae. Yeast. 1987 Jun;3(2):77–84. doi: 10.1002/yea.320030204. [DOI] [PubMed] [Google Scholar]
- Veenhuis M., Van Dijken J. P., Harder W. The significance of peroxisomes in the metabolism of one-carbon compounds in yeasts. Adv Microb Physiol. 1983;24:1–82. doi: 10.1016/s0065-2911(08)60384-7. [DOI] [PubMed] [Google Scholar]
- Williams P. G., Stewart P. R. The intramitochondrial location of cytochrome c peroxidase in wild-type and petite Saccharomyces cerevisiae. Arch Microbiol. 1976 Feb;107(1):63–70. doi: 10.1007/BF00427868. [DOI] [PubMed] [Google Scholar]
- Yonetani T. Cytochrome c peroxidase. Adv Enzymol Relat Areas Mol Biol. 1970;33:309–335. doi: 10.1002/9780470122785.ch6. [DOI] [PubMed] [Google Scholar]
- Yonetani T., Ray G. S. Studies on cytochrome c peroxidase. 3. Kinetics of the peroxidatic oxidation of ferrocytochrome c catalyzed by cytochrome c peroxidase. J Biol Chem. 1966 Feb 10;241(3):700–706. [PubMed] [Google Scholar]
- van Dijken J. P., Veenhuis M., Vermeulen C. A., Harder W. Cytochemical localization of catalase activity in methanol-grown Hansenula polymorpha. Arch Microbiol. 1975 Nov 7;105(3):261–267. doi: 10.1007/BF00447145. [DOI] [PubMed] [Google Scholar]



