Abstract
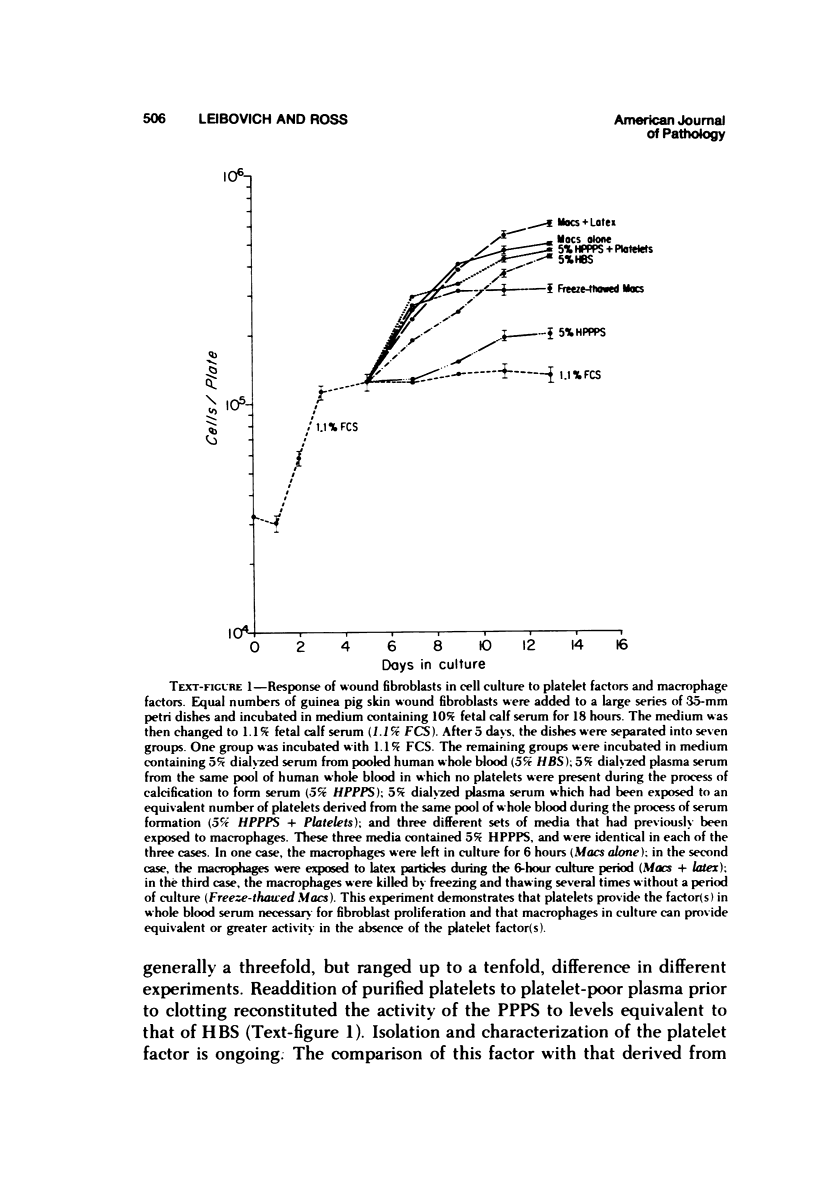
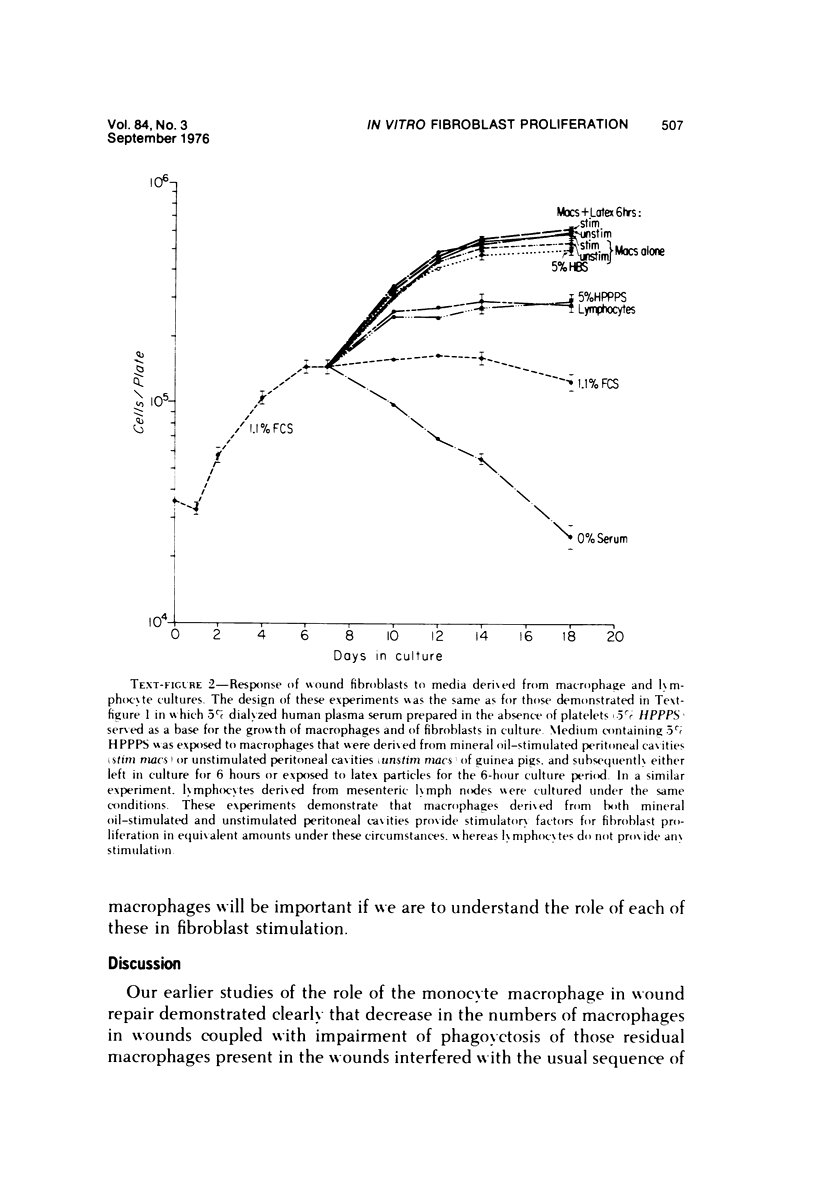
Whole blood serum (HBS) stimulates the proliferation of fibroblasts in vitro, while platelet-poor plasma serum (PPPS) does not. Fibroblasts grown in the presence of PPPS are truly quiescent in that they are not deprived of nutrients in the culture medium and less than 3% of the cells synthesize DNA and divide. In vivo experiments have suggested that macrophages are necessary for stimulation of fibroplasia during wound repair. We have utilized the difference in growth-promoting activity between HBS and PPPS to study the ability of macrophages to produce growth-promoting activity in cell culture. Guinea pig peritoneal macrophages cultured in vitro in medium containing PPPS release into the medium, either directly or indirectly, a factor (or factors) that stimulates the proliferation of guinea pig wound fibroblasts. This macrophage-dependent, fibroblast-stimulating activity (MFSA) is nondialyzable, heat stable (56 C for 30 minutes), and requires culture in vitro for demonstration of activity. The relationship between MFSA and other growth factor(s) has not yet been determined. In contrast to the macrophage, lymphocytes prepared from mesenteric lymph nodes produced no figroblast-stimulating activity.
Full text
PDF











Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armelin H. A. Pituitary extracts and steroid hormones in the control of 3T3 cell growth. Proc Natl Acad Sci U S A. 1973 Sep;70(9):2702–2706. doi: 10.1073/pnas.70.9.2702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosmann H. B. Elevated glycosidases and proteolytic enzymes in cells transformed by RNA tumor virus. Biochim Biophys Acta. 1972 Apr 21;264(2):339–343. doi: 10.1016/0304-4165(72)90298-x. [DOI] [PubMed] [Google Scholar]
- Burger M. M. Proteolytic enzymes initiating cell division and escape from contact inhibition of growth. Nature. 1970 Jul 11;227(5254):170–171. doi: 10.1038/227170a0. [DOI] [PubMed] [Google Scholar]
- Calderon J., Williams R. T., Unanue E. R. An inhibitor of cell proliferation released by cultures of macrophages. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4273–4277. doi: 10.1073/pnas.71.11.4273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L. B., Buchanan J. M. Mitogenic activity of blood components. I. Thrombin and prothrombin. Proc Natl Acad Sci U S A. 1975 Jan;72(1):131–135. doi: 10.1073/pnas.72.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corvol M. T., Malemud C. J., Sokoloff L. A pituitary growth-promoting factor for articular chondrocytes in monolayer culture. Endocrinology. 1972 Jan;90(1):262–271. doi: 10.1210/endo-90-1-262. [DOI] [PubMed] [Google Scholar]
- Davies P., Page R. C., Allison A. C. Changes in cellular enzyme levels and extracellular release of lysosomal acid hydrolases in macrophages exposed to group A streptococcal cell wall substance. J Exp Med. 1974 May 1;139(5):1262–1282. doi: 10.1084/jem.139.5.1262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dulak N. C., Temin H. M. Multiplication-stimulating activity for chicken embryo fibroblasts from rat liver cell conditioned medium: a family of small polypeptides. J Cell Physiol. 1973 Apr;81(2):161–170. doi: 10.1002/jcp.1040810205. [DOI] [PubMed] [Google Scholar]
- Gerschenson L. E., Okigaki T., Andersson M., Molson J., Davidson M. B. Fine structural and growth characteristics of cultured rat liver cells. Insulin effects. Exp Cell Res. 1972 Mar;71(1):49–58. doi: 10.1016/0014-4827(72)90262-5. [DOI] [PubMed] [Google Scholar]
- Gery I., Gershon R. K., Waksman B. H. Potentiation of the T-lymphocyte response to mitogens. I. The responding cell. J Exp Med. 1972 Jul 1;136(1):128–142. doi: 10.1084/jem.136.1.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gery I., Waksman B. H. Potentiation of the T-lymphocyte response to mitogens. II. The cellular source of potentiating mediator(s). J Exp Med. 1972 Jul 1;136(1):143–155. doi: 10.1084/jem.136.1.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gospodarowicz D. Localisation of a fibroblast growth factor and its effect alone and with hydrocortisone on 3T3 cell growth. Nature. 1974 May 10;249(453):123–127. doi: 10.1038/249123a0. [DOI] [PubMed] [Google Scholar]
- Henney C. S. Studies on the mechanism of lymphocyte-mediated cytolysis. II. The use of various target cell markers to study cytolytic events. J Immunol. 1973 Jan;110(1):73–84. [PubMed] [Google Scholar]
- Hoffmann M., Dutton R. W. Immune response restoration with macrophage culture supernatants. Science. 1971 Jun 4;172(3987):1047–1048. doi: 10.1126/science.172.3987.1047. [DOI] [PubMed] [Google Scholar]
- Holley R. W., Kiernan J. A. "Contact inhibition" of cell division in 3T3 cells. Proc Natl Acad Sci U S A. 1968 May;60(1):300–304. doi: 10.1073/pnas.60.1.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohler N., Lipton A. Platelets as a source of fibroblast growth-promoting activity. Exp Cell Res. 1974 Aug;87(2):297–301. doi: 10.1016/0014-4827(74)90484-4. [DOI] [PubMed] [Google Scholar]
- Leibovich S. J., Ross R. The role of the macrophage in wound repair. A study with hydrocortisone and antimacrophage serum. Am J Pathol. 1975 Jan;78(1):71–100. [PMC free article] [PubMed] [Google Scholar]
- Ossowski L., Unkeless J. C., Tobia A., Quigley J. P., Rifkin D. B., Reich E. An enzymatic function associated with transformation of fibroblasts by oncogenic viruses. II. Mammalian fibroblast cultures transformed by DNA and RNA tumor viruses. J Exp Med. 1973 Jan 1;137(1):112–126. doi: 10.1084/jem.137.1.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul D., Lipton A., Klinger I. Serum factor requirements of normal and simian virus 40-transformed 3T3 mouse fibroplasts. Proc Natl Acad Sci U S A. 1971 Mar;68(3):645–652. doi: 10.1073/pnas.68.3.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierson R. W., Jr, Temin H. M. The partial purification from calf serum of a fraction with multiplication-stimulating activity for chicken fibroblasts in cell culture and with non-suppressible insulin-like activity. J Cell Physiol. 1972 Jun;79(3):319–330. doi: 10.1002/jcp.1040790302. [DOI] [PubMed] [Google Scholar]
- ROSS R., BENDITT E. P. Wound healing and collagen formation. I. Sequential changes in components of guinea pig skin wounds observed in the electron microscope. J Biophys Biochem Cytol. 1961 Dec;11:677–700. doi: 10.1083/jcb.11.3.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross R., Glomset J. A. Atherosclerosis and the arterial smooth muscle cell: Proliferation of smooth muscle is a key event in the genesis of the lesions of atherosclerosis. Science. 1973 Jun 29;180(4093):1332–1339. doi: 10.1126/science.180.4093.1332. [DOI] [PubMed] [Google Scholar]
- Ross R., Glomset J., Kariya B., Harker L. A platelet-dependent serum factor that stimulates the proliferation of arterial smooth muscle cells in vitro. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1207–1210. doi: 10.1073/pnas.71.4.1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross R. The fibroblast and wound repair. Biol Rev Camb Philos Soc. 1968 Feb;43(1):51–96. doi: 10.1111/j.1469-185x.1968.tb01109.x. [DOI] [PubMed] [Google Scholar]
- Ross R. The smooth muscle cell. II. Growth of smooth muscle in culture and formation of elastic fibers. J Cell Biol. 1971 Jul;50(1):172–186. doi: 10.1083/jcb.50.1.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rutherford R. B., Ross R. Platelet factors stimulate fibroblasts and smooth muscle cells quiescent in plasma serum to proliferate. J Cell Biol. 1976 Apr;69(1):196–203. doi: 10.1083/jcb.69.1.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmon W. D., Jr, Hosse B. R. Stimulation of HeLa cell growth by a serum fraction with sulfation factor activity. Proc Soc Exp Biol Med. 1971 Mar;136(3):805–808. doi: 10.3181/00379727-136-35370. [DOI] [PubMed] [Google Scholar]
- Tangen O., Berman H. J., Marfey P. Gel filtration. A new technique for separation of blood platelets from plasma. Thromb Diath Haemorrh. 1971 Jun 30;25(2):268–278. [PubMed] [Google Scholar]
- Unanue E. R. The regulatory role of macrophages in antigenic stimulation. Adv Immunol. 1972;15:95–165. doi: 10.1016/s0065-2776(08)60684-7. [DOI] [PubMed] [Google Scholar]


