Abstract
Numerous genes have been identified to date that contribute to the host response to systemic Salmonella Typhimurium infection in mice. We have previously identified two loci, Ity2 and Ity3, that control survival to Salmonella infection in the wild-derived inbred MOLF/Ei mouse using a (C57BL/6J × MOLF/Ei)F2cross. We validated the existence of these two loci by creating congenic mice carrying each quantitative trait locus (QTL) in isolation. Subcongenic mice generated for each locus allowed us to define the critical intervals underlying Ity2 and Ity3. Furthermore, expression profiling was carried out with the aim of identifying differentially expressed genes within the critical intervals as potential candidate genes. Genomewide expression arrays were used to interrogate expression differences in the Ity2 congenics, leading to the identification of a new candidate gene (Havcr2, hepatitis A virus cellular receptor 2). Interval-specific oligonucleotide arrays were created for Ity3, identifying one potential candidate gene (Chi3l1, chitinase 3-like 1) to be pursued further. The combination of the use of congenics in QTL confirmation and fine mapping and in the identification of candidate genes by expression profiling has been successful and represents a step toward quantitative gene(s) identification.
SALMONELLA enterica serovar Typhimurium (Salmonella Typhimurium) is a gram-negative intracellular bacterium that is responsible for a gastrointestinal illness known as salmonellosis in humans and a typhoid-like systemic disease in mice. Typhoid fever, caused by the host-specific Salmonella Typhi in humans, is a generalized systemic enteric fever, characterized by headache, nausea, abdominal pain, and diarrhea or constipation with case fatality of 16% without appropriate antibiotic treatment (Ohl and Miller 2001). Susceptibility to such infectious diseases is considered to be a complex trait involving numerous genetic and environmental factors, the interactions of which determine the ultimate outcome of infection. Only a few genes that control the host response to Salmonella infection have been identified in humans and include IFNGR1, IFNGR2, IL12B, IL12RB1, and STAT1 (Casanova and Abel 2004). However, the understanding of immunity to Salmonella infection in humans has progressed considerably through the use of mouse models of infection.
A wide range of susceptibilities to intravenous infection with Salmonella Typhimurium have been reported among various laboratory mouse strains (Roy and Malo 2002). The commonly used inbred mouse strain C57BL/6J is known to succumb to intravenous Salmonella Typhimurium infection within 5 days postinfection due to a single point mutation within Slc11a1 [previously known as Nramp1 and Ity (for immunity to Typhimurium)], a gene having a major impact in controlling the replication of Salmonella Typhimurium within the macrophage (Vidal et al. 1995). Another particular strain of interest is the wild-derived inbred mouse MOLF/Ei, which is extremely susceptible to infection despite harboring functional alleles at Slc11a1 and at another known Salmonella susceptibility locus, Toll-like receptor 4 (Tlr4). Using an F2 panel of (C57BL/6J × MOLF/Ei) mice, three quantitative trait loci (QTL) linked to the host response to Salmonella Typhimurium infection were identified: Slc11a1 (Ity), Ity2, and Ity3 with respective LOD scores of 18.8, 7.0, and 5.0. The Ity2 and Ity3 QTL were only detected in the presence of a Slc11a1 wild type (at least one MOLF/Ei allele) background. The MOLF/Ei allele has a protective effect at Ity2 on chromosome 11 under an additive mode of inheritance. MOLF/Ei alleles also contribute to susceptibility at Ity3 on distal chromosome 1 manifesting the phenotype under a recessive mode of inheritance (Sebastiani et al. 1998).
Gene identification using QTL analysis is a challenging task and is based on establishing, as precisely as possible, the chromosomal position of the QTL and identifying the genes that are located in the target region. Genes are then selected according to function, sequence, and mouse strain distribution of the QTL alleles. The creation of congenic and subcongenic strains is commonly used in the validation and in the fine mapping of QTL underlying complex traits in mice. Through repeated marker-assisted backcrossing, a congenic mouse is generated such that it carries the QTL interval from the donor strain on a homogenous background representing the recipient strain. The generation of congenics allows the assessment of the effect of a unique QTL on the disease phenotype (Rogner and Avner 2003). This congenic approach has proven fruitful in the genetic dissection of diseases such as seizure susceptibility (Ferraro et al. 2004), systemic lupus erythematosus (Haywood et al. 2004; Subramanian et al. 2005), and type 1 diabetes (Lyons et al. 2000). A combination of this classical approach to fine mapping with genomewide expression profiling techniques has been used successfully to prioritize candidate genes (Rozzo et al. 2001; Gu et al. 2002; Mcbride et al. 2003; Klein et al. 2004; Johannesson et al. 2005; De Buhr et al. 2006). In this article, we refine the map of the large Ity2 and Ity3 intervals and identify candidate genes combining two genetic dissection tools, the use of congenic strain mapping and expression profiling (genomewide and QTL interval-specific oligonucleotide arrays).
MATERIALS AND METHODS
Mice:
The parental mouse strains, MOLF/Ei and C57BL/6J, were purchased from the Jackson Laboratory at 4–6 weeks of age. The congenic mice were backcrossed for 10 generations selecting mice carrying the MOLF/Ei allele at D1Mcg4 (Ity) and between the markers D11Mit84 and Mpo (Ity2), resulting in the generation of B6.MOLF-Ity/Ity2 congenic mice. Two distinct congenic strains were created additionally as a result of recombinants selected within the Ity2 interval during the intercrossing of N10 mice to homozygosity (B6.MOLF-Ity/Ity2.RecD, B6.MOLF-Ity/Ity2.RecI) (Figure 2A). The construction of Ity3 congenic mice has been described previously (Sancho-Shimizu and Malo 2006). The Ity3 congenic and recombinant subcongenic strains (B6.MOLF-Ity/Ity3, B6.MOLF-Ity/Ity.RecA, B6.MOLF-Ity/Ity3.RecB, B6.MOLF-Ity/Ity3.RecC, B6.MOLF-Ity/Ity3.RecE) used in the present study differ from the previously published strains in that they have been backcrossed for over 10 generations. The recombinant subcongenic strains were obtained by breeding recombinant chromosomes generated during the intercrossing of the N10 B6.MOLF-Ity/Ity3 strains to homozygosity. All animal procedures were performed in accordance with regulations of the Canadian Council of Animal Care.
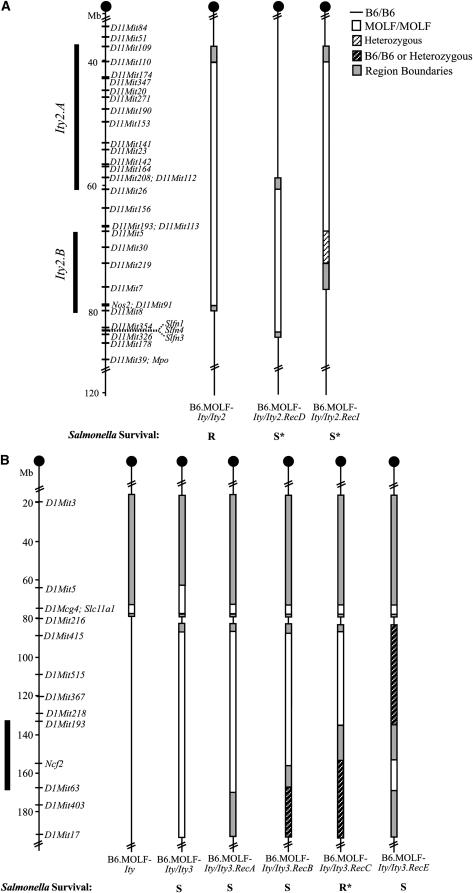
Figure 2.—
Fine mapping of Ity2 and Ity3 loci. A schematic of the Ity2 congenics on chromosome 11 (A) and the Ity3 congenics on chromosome 1 (B). The open box represents regions of MOLF/Ei homozygosity (MOLF/MOLF), the solid line regions of C57BL/6J homozygosity (B6/B6), and the hatched segment on white background, heterozygous intervals. Congenics, including individuals with intervals of C57BL/6J homozygosity and heterozygosity (B6/B6 or heterozygous), are indicated by hatched segments on black background. Unresolved intervals at the boundaries of congenic fragments, where the genotypes have not been fixed or remain undetermined, are indicated by shaded boxes. All Ity2 congenic mice also carry the MOLF/Ei congenic fragment, Ity, on chromosome 1. The bold line by the chromosomes indicates the restricted interval defined by phenotyping the congenic mice. The relative susceptibility of each congenic strain is indicated at the bottom of each chromosome as compared to the B6.MOLF-Ity referent (R, more resistant; S, more susceptible). An asterisk by the R or S indicates that the survival does not differ from the B6.MOLF-Ity referent strain.
Genotyping of congenic mice:
At every generation, Ity2 mice were genotyped using microsatellite markers D1Mcg4, D11Mit84, D11Mit109, D11Mit20, D11Mit141, D11Mit142, D11Mit112, D11Mit193, D11Mit219, D11Mit39, as well as an SSLP marker for Nos2 (due to an additional proline in MOLF/Ei) and an SSCP marker for Mpo. Nos2 primers, 5′-gtatggtgtgaggttatagagatt-3′ and 5′-gtcatgcaaaatctctccactgcc-3′ and Mpo primers, 5′-gctcactcttgcagatgtgttgac-3′ and 5′-tcgccacaggcaagacctagaccc-3′ were used for PCR amplification of genomic DNA. Animals heterozygous for the target region of chromosome 11 were further bred to obtain the next generation of mice. Additional markers—D1Mit3, D1Mit5, D1Mit318, D1Mit213, D1Mit46, D1Mit134, D1Mit216, D1Mit135, D1Mit415, D1Mit515, D1Mit367, D1Mit218, D1Mit193, D1Mit403, D1Mit17, D11Mit110, D11Mit174, D11Mit347, D11Mit208, D11Mit26, D11Mit156, D11Mit113, D11Mit5, D11Mit30, D11Mit7, D11Mit91, D11Mit8, D11Mit354, D11Mit326, D11Mit178, and D11Mit70—were used to fine map the congenic boundaries. Ity3 mice were genotyped using D1Mit3, D1Mit5, D1Mcg4, D1Mit216, D1Mit218, D1Mit193, D1Mit63, and D1Mit17 for PCR amplification at every generation. Additional restriction digests specific for Slc11a1 and Ncf2 were also carried out. Slc11a1 primers 5′-agctatttgggttcctgact-3′ and 5′-ggtcaaagccattatggtaa-3′ specific for the G169D point mutation were used to amplify genomic DNA, followed by a restriction digest using HpyCH4III. The C57BL/6J allele (D169) produces two fragments of 453 bp and 81 bp, whereas the MOLF/Ei allele (G169) is uncut with a fragment of 534 bp. Ncf2 primers 5′-atgccttacatgctcaaggtg-3′ and 5′-catgctttcttcggacaggagcagaagc-3′ specific for a point mutation (R394Q) were used to amplify genomic DNA. Digestion with BsaHI restriction enzyme was carried out resulting in two products of 278 bp and 41 bp for C57BL/6J allele (R394) and one for the MOLF/Ei allele (Q394) of 319 bp. The digested products were resolved on a 2% agarose gel.
Sequencing:
Slfn1, Slfn3, and Slfn4 genes were sequenced using spleen cDNA isolated from B6.MOLF-Ity/Ity2.RecG, B6.MOLF-Ity/Ity2.RecD, and MOLF/Ei strains to further refine the boundaries of the congenic intervals. The entire coding sequence was sequenced for Slfn1 and primers used include 5′-actgtagctcatccctcaaa-3′, 5′-caattctttgcttcaaaacc-3′, 5′-acgggggatatttgtttatt-3′, 5′-tgttacgaaaagcaagaggt-3′. The coding region of Slfn3 and Slfn4 were partially sequenced using the following primers for Slfn3, 5′-gcctatgaggagacattctg-3′ and 5′-tttcaacgagagctttttct-3′; and Slfn4, 5′-tgtctgcgtttttaaatgtg-3′ and 5′-aaatggcagaacctttgtta-3′.
Infection and survival analysis:
Mice between the ages of 6 and 16 weeks were infected intravenously with ∼1000 CFUs of Salmonella Typhimurium strain Keller through the caudal vein (Bihl et al. 2001; Sancho-Shimizu and Malo 2006). Mice were monitored daily, survival recorded, and moribund animals sacrificed by carbon dioxide asphyxiation. Kaplan-Meier survival analysis was used for statistical analysis.
Linkage analysis:
Genetic linkage was carried out using MapManagerQTX on 232 (C57BL/6J × MOLF/Ei)F2 mice using eight additional markers on chromosome 11: Il12b (5′-ttcatgtgctcgtggcctgatcca-3′ and 5′-gtacccttctaaagaaggccctgg-3′), D11Mit22, D11Mit164, D11Mit156, D11Mit30, Nos2, Mpo, and D11Mit41.
Bacterial load enumeration:
Infected mice were sacrificed on days 3 and 7 in addition to noninfected control mice. Their spleens and livers were aseptically removed and placed in tubes with 0.9% saline solution. The tissues were homogenized and plated on trypticase soy agar plates in duplicate using at least three serial dilutions. Plates were incubated at 37° overnight and the colonies enumerated the following day.
Whole-genome expression profiling:
Total RNA was isolated from the spleens of uninfected controls and infected B6.MOLF-Ity/Ity2 and B6.MOLF-Ity/Ity2.RecD mice on days 3 and 7 postinfection using Trizol reagent (Invitrogen, Carlsbad, CA). RNA from three males and three females was pooled per strain per time point for the first experiment and one male and one female pooled per strain per time point for the second experiment. The quality of the RNA was verified using BioAnalyzer. Then 10 μg of each sample was used to hybridize to the Affymetrix Mouse Chip 430v.2.0 (Affymetrix, Santa Clara, CA). Labeling of the probe, hybridization, and scanning of the microarrays were done at the McGill and Genome Quebec Innovation Center (Montreal) as previously described (Caron et al. 2006).
Construction of Ity3 interval-specific 70-mer oligonucleotide microarray:
Seventy-mer oligonucleotide probes were designed by Scienion AG (Berlin) for all transcripts possessing an accession ID and mapping to the Ity3 interval (Ensembl v19.30.1 and Celera) delineated by D1Mit135 and D1Mit63, resulting in a total of 375 transcripts. In addition to these probes, also added were probes for six other genes of interest during infection (Slc11a1, Il1b, Il6, Nos2, Mpo, Tlr2), four probes representing housekeeping genes (18srRNA, Hprt, Tbp, Gapdh), and two genes as negative controls for infection on the basis of other array experiments carried out in our laboratory (Actg2, Vcam1). The 70-mer oligos were synthesized for all transcript probes by MetaBion GmbH (Martinsried, Germany). Finally, hybridization controls, two positive (herring sperm DNA, 10 mg/ml; Cot1DNA, 1 mg/ml) and three negative controls (water, 3× SSC, 14× SSC), were added to the panel. Lyophilized oligos were resuspended in 3× SSC and spotted at a concentration of 70 μm on Sigmascreen microarray slides (Sigma, St. Louis) using the Virtek Chipwriter model SDDC2 to print oligonucleotides. The assembled Ity3 array contained a total of 2304 spots. Each transcript probe was spotted in triplicate (1152 spots), the housekeeping genes and positive hybridization controls were spotted 24 times (144 spots), and the remaining 1008 spots were represented by the negative controls. The quality of the Ity3 array was verified after each batch of prints using the SpotQC kit (Integrated DNA Technologies, Coralville, IA).
Interval-specific expression profiling:
Total RNA from spleens of uninfected and infected B6.MOLF-Ity/Ity3MOLF/B6 and B6.MOLF-Ity/Ity3 mice (at the fifth generation of backcross) was isolated as described above. Following RNA extraction, residual DNA was removed using the DNAse-I kit from Ambion and subsequent RNAeasy on-column digestion following the manufacturer's instructions (QIAGEN, Mississauga, Canada). The quality of RNA was confirmed by denaturing gel electrophoresis (formaldehyde). Microarray hybridization was performed as previously described (Charlet et al. 2005). In brief, 5–20 μg of total RNA extracted from control and infected mice was labeled with Cy3 or Cy5 dUTP by reverse transcriptase (Amersham Biosciences, Piscataway, NJ). Labeled cDNA was applied to a post-processed array, covered with a glass slip, and placed into a hybridization chamber overnight at 42°. Arrays were placed into 37° 1× SSC, 0.2% SDS to remove the cover glass, then washed in 1× SSC, 0.2% SDS for 15 min; 0.1× SSC, 0.2% SDS for 15 min; and 0.1× SSC for 15 min. Hybridized arrays were scanned with ScanArray 5000XL and hybridization results were quantified with ScanArray software (Perkin-Elmer, Freemont, CA). For each hybridization, one RNA sample from an individual B6.MOLF-Ity/Ity3 MOLF/B6 was hybridized against one RNA sample from an individual B6.MOLF-Ity/Iy3 mouse at each time point per tissue. A single reciprocal Cy-dye swap (Cy3/Cy5 and Cy5/Cy3) experiment was minimally carried out for all hybridizations, resulting in at least a single replicate for each experiment and totaling 11 arrays studied.
Microarray data analysis:
For whole-genome microarrays, the expression values were generated by probe-level analyses using the robust multi-array analysis procedure (Irizarry et al. 2003). For Ity3 arrays, analysis was performed as previously described (Charlet et al. 2005). All spots flagged as misrepresentative by ScanArray (array artifacts, etc.) were analytically ignored. Subtracting total spot intensity minus the surrounding background produced a corrected spot intensity. Negative corrected spot intensities were set to +1. Intensity ratios (Cy3/Cy5 or Cy5/Cy3) were determined using corrected spot intensities and log10 transformed. Fold change is calculated from a normalized log-ratio of that gene. Values for each gene were obtained in triplicate for each array (inherent to array design) and averaged. Only genes with fold changes of two or greater in replicate hybridization experiments are reported.
Real-time PCR:
Expression of Slfn1, Slfn4, Slfn5, Butr1, Cyfip2, Hacvr2, Itk, Sqstm1, and Tgtp in B6.MOLF-Ity/Ity2 and B6.MOLF-Ity/Ity2.RecD, as well as Chi3l1, Cdc73 Sft2d2, and Niban in B6.MOLF-Ity/Ity3 and B6.MOLF-Ity/Ity3MOLF/B6 was determined by real-time PCR using the Chromo4 Real Time PCR system (MJ Research). cDNAs were obtained from reverse transcription of infected and control spleen RNAs. The cDNAs were amplified using the following primers: Slfn1 5′-gggaacgtgctcagtaga-3′ and 5′-cctgcatttagaatcagca-3′, Slfn4 5′-aggtttaccacagaggaatg-3′ and 5′-tctggagagcatatcacctt-3′, Slfn5 5′-ggcctctcggatgatagaaa-3′ and 5′-ggtcttgctgcagggtgt-3′, Butr1 5′-cagagaaggacactggattc-3′ and 5′-tgtgactgtacatcttgacca-3′, Cyfip2 5′-gctttgacctgtttgacttc-3′ and 5′-gtcagccatcttcttcagag-3′, Hacvr2 5′-ctggtgaccctccataataa-3′ and 5′-tctgatcgtttctccagagt-3′, Itk 5′-tagcagcaagtcagatgtgt-3′ and 5′-ctgatatcttccacgacctc-3′, Sqstm1 5′-ctctaggcattgaggttgac-3′ and 5′-ttggctgagtgttactcttg-3′, Tgtp 5′-taaagacgttccctaagagg-3′ and 5′-ctctgtatggtagaagctcag-3′, Cdc73 5′-aaacgtcacttggatagacc-3′ and tcaagtgggatttatgcttt-3′, Chi3l1 5′-tgaagtacatcgactctcca-3′ and 5′-tttgacactctccttgtcct-3′, Sft2d2 5′-cgtggtatagcctttcctat-3′ and 5′-ctggcagtgtgttatgcaag-3′, Niban 5′-tgtatgagatcctcctcgat-3′ and 5′-gccatgttgtcttcaaataag-3′. Tbp was used as a housekeeping gene control and amplified using the primers 5′-cccttgtacccttcaccaat-3′ and 5′-acagccaagattcacggtag-3′ using the same cycling conditions. The cycling conditions were 95° for 30 sec, 55° for 30 sec, 72° for 30 sec, 80° for 2 sec for a total of 40 cycles. Stratagenes's Brilliant SYBR Green QPCR Master mix was used for the PCR reactions. All samples were run in duplicate along with a standard curve of four 10-fold serial dilutions of template cDNA. The expression data are expressed in relative fold-change units using uninfected B6.MOLF-Ity/Ity2 or B6.MOLF-Ity/Ity3 as the referent according to the following 2−ΔΔCt equation 2−[(Gene of interest treatment Ct − Tbp treatment Ct) − (Gene of interest reference Ct − Tbp reference Ct)] (Livak and Schmittgen 2001). The level of significance was assessed using the Student's t-test (P < 0.05).
RESULTS
Ity2 and Ity3 linkage analysis:
We have previously reported the existence of two QTL, Ity2 on chromosome 11 and Ity3 on distal chromosome 1, affecting the host response of MOLF/Ei mice to infection with Salmonella Typhimurium. The MOLF/Ei allele at Ity2 confers resistance to infection in an additive fashion, accounting for 10% of the phenotypic variance. The MOLF/Ei allele at Ity3 was found to contribute to the susceptibility of MOLF/Ei mice recessively and explained 7% of the phenotypic variance (Sebastiani et al. 1998). To confirm the location of these QTL, we have added additional progeny as well as extra chromosome 11-specific markers to the existing (C57BL/6J × MOLF/Ei)F2 panel used in the initial linkage analysis.
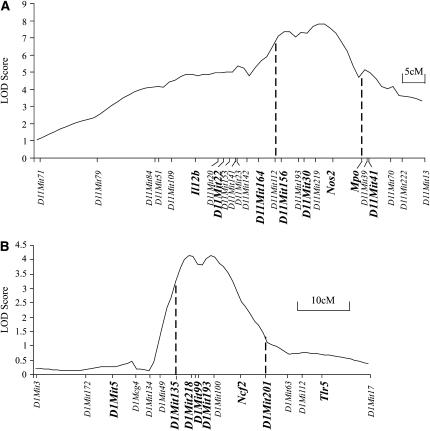
For the chromosome 11 QTL harboring Ity2, linkage was reanalyzed using eight novel markers (Il12b, D11Mit22, D11Mit164, D11Mit156, Inos, Mpo, and D11Mit41) on a total of 232 (C57BL/6J × MOLF/Ei)F2 mice, including an additional 41 mice and the original set of 191 mice. The Ity2 locus yielded a significant peak LOD score of 7.8 at Nos2 under a model of free regression, with a 2-LOD support interval (99% C.I.) spanning D11Mit112 to Mpo (Figure 1A). The addition of eight markers and additional F2 mice led to an increase in the peak LOD score from 7.0 to 7.8 and to a minor repositioning of the relatively large Ity2 interval as compared to the initial analysis, placing the peak slightly more distally.
Figure 1.—
LOD score plots of the Ity2 and Ity3 regions in (C57BL/6J × MOLF/Ei)F2 cross (A and B). The markers in bold are the additional markers used in the linkage analysis of Ity2 and Ity3 (C57BL/6J × MOLF/Ei)F2 cross as assessed by MapManagerQTX (A). The LOD score plot for Ity3 in (C57BL/6J × MOLF/Ei)F2 cross is taken from Sancho-Shimizu et al. (2006). Dashed lines indicate the boundaries of the 2-LOD support interval of the respective QTL regions.
Similarly, the Ity3 QTL on distal chromosome 1 was re-evaluated using eight additional markers, D1Mit5, D1Mit135, D1Mit218, D1Mit99, D1Mit193, D1Mit 201, Ncf2, and Tlr5, and 41 additional mice, as previously described (Sancho-Shimizu and Malo 2006). The peak LOD score of 4.1 was obtained at D1Mit218 and D1Mit100, encompassing approximately the same 2-LOD support interval from D1Mit135 to D1Mit201 as detected in the initial analysis (D1Mit135 to D1Mit63) (Figure 1B) (Sebastiani et al. 1998).
Generation of congenic mice:
Congenic mice were created for Ity2 and Ity3 by producing F1 hybrids between C57BL/6J and MOLF/Ei, followed by at least 10 successive backcross generations to the C57BL/6J parental strain. The target Ity/Ity2 and Ity/Ity3 segments were maintained using marker-assisted genotyping. Homozygous founders were established by brother-sister matings of N10 mice. Due to the impact of Slc11a1 (Ity) on the detection of Ity2 and Ity3, congenic B6.MOLF-Ity were created by transferring the wild-type allele at Slc11a1 originating from the MOLF/Ei mice onto a C57BL/6J genetic background, who naturally carry the mutant form of this well-characterized Salmonella susceptibility gene (Vidal et al. 1993). The largest Ity2 interval transferred spans from D11Mit110 to D11Mit91, a 39.0-Mb interval and the largest Ity3 interval is 62.0 Mb in size located from D1Mit218 to D1Mit17. Both regions extend well beyond the 2-LOD support interval to ensure that all genetic elements contributing to the QTL-associated phenotype would be transferred to the resulting congenic strains. Moreover, the presence of numerous immunologically relevant genes that map within the relatively large Ity2 and Ity3 intervals suggest the possibility that more than one gene may be involved in the disease phenotype underlying the QTL interval.
The recombinant congenics were generated at the same time as the B6.MOLF-Ity/Ity2 and B6.MOLF-Ity/Ity3 congenics by selecting mice sharing recombinant chromosomes at the N10 generation, which were further bred to homozygosity through brother–sister matings. This led to the establishment of two recombinant B6.MOLF-Ity/Ity2 strains (RecD and RecI) and four recombinant B6.MOLF-Ity/Ity3 strains (RecA, RecB, RecC, and RecE) shown in Figure 2, A and B.
Phenotypic characterization of Ity2 congenic strains:
All congenic strains were infected with Salmonella Typhimurium intravenously and their survival noted. As expected, the C57BL/6J mice were the most susceptible to infection and the transferal of a wild-type MOLF/Ei allele at Slc11a1 (B6.MOLF-Ity) improved resistance to infection significantly (Figure 3A) (Sancho-Shimizu and Malo 2006). Survival analysis revealed that only the B6.MOLF-Ity/Ity2 congenic improved survival time over the control B6.MOLF-Ity mice (Figure 3A and Table 1). The remaining Ity2 congenic strains, B6.MOLF-Ity/Ity2.RecD and B6.MOLF-Ity/Ity2.RecI, did not differ significantly from the B6.MOLF-Ity controls, suggesting that the portion of Ity2 that these mice carry does not contribute to improved resistance.
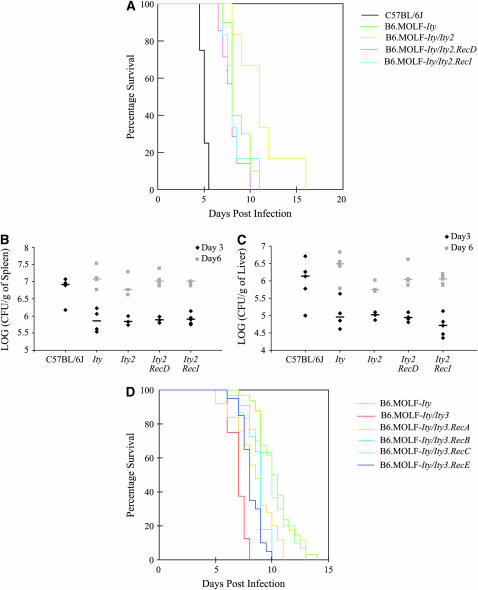
Figure 3.—
Phenotypic characterization of the Ity2 and Ity3 congenic mice. Survival curves for Ity2 congenic mice are shown in A. One representative survival experiment of seven is shown here. There were at least six mice in each group of mice tested. Bacterial load in the spleen (B) and the liver (C) of the Ity2 congenics are represented as log (CFU/g of organ). Groups of three to four mice were used for CFU enumeration for each time point; median for each group is shown as a bar. Survival of Ity3 congenic mice is shown in D. Samples from three independent experiments using at least eight mice per group were pooled and represented here.
TABLE 1.
Ity2 matrix of pairwise comparison probabilities using the logrank test (Breslow-Gehan) of Kaplan-Meier survival curves
| C57BL/6J | Ity | Ity/Ity2 | RecD | RecI | |
|---|---|---|---|---|---|
| C57BL/6J | 1.000 | ||||
| Ity | <0.001 | 1.000 | |||
| Ity/Ity2 | 0.002 | 0.036 | 1.000 | ||
| RecD | 0.001 | 0.193 | 0.012 | 1.000 | |
| RecI | 0.002 | 0.488 | 0.03 | 0.616 | 1.000 |
In an attempt to further sub-phenotype the Ity2 congenic mice, the bacterial load in the spleen and liver was determined at various time points upon infection. C57BL/6J mice had ∼10-fold higher bacterial loads in the spleen and liver at day 3 compared to the congenic mice and no C57BL/6J mice survived after day 5. This observation is attributable to the fact that these mice carry a nonfunctional mutation in Slc11a1, as previously reported (Vidal et al. 1995; Sancho-Shimizu and Malo 2006). No significant difference was observed between any of the Ity2 congenics and the B6.MOLF-Ity controls at all time points (Figure 3, B and C), strongly suggesting that the impact of Ity2 on survival to infection is not related to the level of bacterial load in these target organs.
Consistent with previously published results, survival of B6.MOLF-Ity/Ity3 mice was significantly reduced in comparison to the B6.MOLF-Ity control (Figure 3D and Table 2). Data presented here represent B6.MOLF-Ity/Ity3 mice that have been backcrossed for over 10 generations as opposed to previously published data that tested B6.MOLF-Ity/Ity3 mice intercrossed after 5 generations (Sancho-Shimizu and Malo 2006). The survival analysis represents three independent experiments that have been pooled since no significant difference was observed in survival among the B6.MOLF-Ity mice in all experiments (mean survival time, MST = 10.7 ± 1.3; MST = 10.1 ± 0.4; and MST = 9.8 ± 0.3 days). B6.MOLF-Ity/Ity3.RecC did not differ significantly from the B6.MOLF-Ity controls, suggesting that this portion of chromosome 1 did not contribute to the susceptibility phenotype, whereas all other Ity3 recombinant congenics were more susceptible to infection. On the basis of these analyses, the B6.MOLF-Ity/Ity3 congenics were more susceptible than RecA, RecB, RecC, and RecE (Table 2). In fact, B6.MOLF-Ity/Ity3.RecA, RecB, and RecE have an intermediate phenotype as they were significantly different from B6.MOLF-Ity controls as well from B6.MOLF-Ity/Ity3 mice. However, the congenic harboring the largest MOLF interval, B6.MOLF-Ity/Ity3, was the most susceptible to Salmonella infection, suggesting the involvement of more than one gene within the Ity3 interval.
TABLE 2.
Ity3 matrix of pairwise comparison probabilities using the logrank test (Breslow-Gehan) of Kaplan-Meier survival curves
| Ity | Ity/Ity3 | RecA | RecB | RecC | RecE | |
|---|---|---|---|---|---|---|
| Ity | 1.000 | |||||
| Ity/Ity3 | <0.001 | 1.000 | ||||
| RecA | <0.001 | 0.01 | 1.000 | |||
| RecB | 0.001 | <0.001 | 0.699 | 1.000 | ||
| RecC | 0.295 | <0.001 | 0.017 | 0.054 | 1.000 | |
| RecE | <0.001 | 0.002 | 0.316 | 0.066 | <0.001 | 1.000 |
Refinement of Ity2 and Ity3 critical intervals:
The centromeric boundary of the congenic Ity2 interval in B6.MOLF-Ity/Ity2 was resolved to a 2.3-Mb region between D11Mit110 and D11Mit109. The distal end was delimited by a 790-kb region located between D11Mit91 and D11Mit8. The Ity2 RecD interval was resolved proximally by a 1.9-Mb region between D11Mit112 and D11Mit26 and distally by a 600-kb interval delimited by Slfn3 and D11Mit326. On the basis of the survival analysis of the Ity2 congenics, we were able to further restrict the large interval to two more defined regions (Figure 2A, Ity2.A and Ity2.B), which are both necessary but not sufficient in the resistance phenotype. The two intervals (Ity2.A and Ity2.B) are present in the resistant strain B6.MOLF-Ity/Ity2 and one of the two regions absent in the susceptible strains B6.MOLF-Ity/Ity2.RecD (missing Ity2.A) and B6.MOLF-Ity/Ity3.RecI (missing Ity2.B). The proximal region, Ity2.A, is flanked by D11Mit109 and D11Mit26, an interval spanning ∼23 Mb, encoding for 331 genes according to the latest update of the Ensembl genome browser (Ensembl build 43). The distal region, Ity2.B, is flanked by D11Mit5 and D11Mit8, an interval of ∼13 Mb composed of 364 genes (Ensembl build 43). Even though the survival data seem to point out these two particular regions as critical areas of interest, the region between these two intervals cannot be disregarded in any future analysis and may harbor additional genes affecting the phenotype.
The B6.MOLF-Ity/Ity3 congenics carry the Ity3 interval spanning 103 Mb between D1Mit415 and D1Mit17, with a proximal boundary between D1Mit216 and D1Mit415 (Figure 2B). The Ity interval in B6.MOLF-Ity/Ity3 mice spans D1Mcg4 to D1Mit5 with region boundaries extending proximally to D1Mit3 and distally to D1Mit216. The Ity interval in the B6.MOLF-Ity and B6.MOLF-Ity/Ity3 recombinant strains (RecA, RecB, RecC, and RecE) are defined by MOLF/Ei alleles at D1Mcg4 with region boundaries spanning D1Mcg4 to D1Mit3 and D1Mcg4 to D1Mit216. The survival analysis of the recessively inherited susceptibility locus Ity3 suggests that the region of 44.8 Mb between D1Mit415 and D1Mit193 in B6.MOLF-Ity/Ity3.RecC strain does not contribute to the susceptibility of the mice and could be eliminated as harboring genes for Ity3 susceptibility. The remaining Ity3 strains were all variably susceptible to infection, suggesting they all carry intervals necessary for the manifestation of the phenotype. The smallest interval can be deduced from the overlap between the Ity3 RecB and RecE strains, surrounding Ncf2 and delineated by D1Mit193 and D1Mit63 representing the critical interval (Figure 2B). Within this 34-Mb critical interval, there are a total of 322 genes according to Ensembl build 43.
Ity2 interval-specific expression profiling:
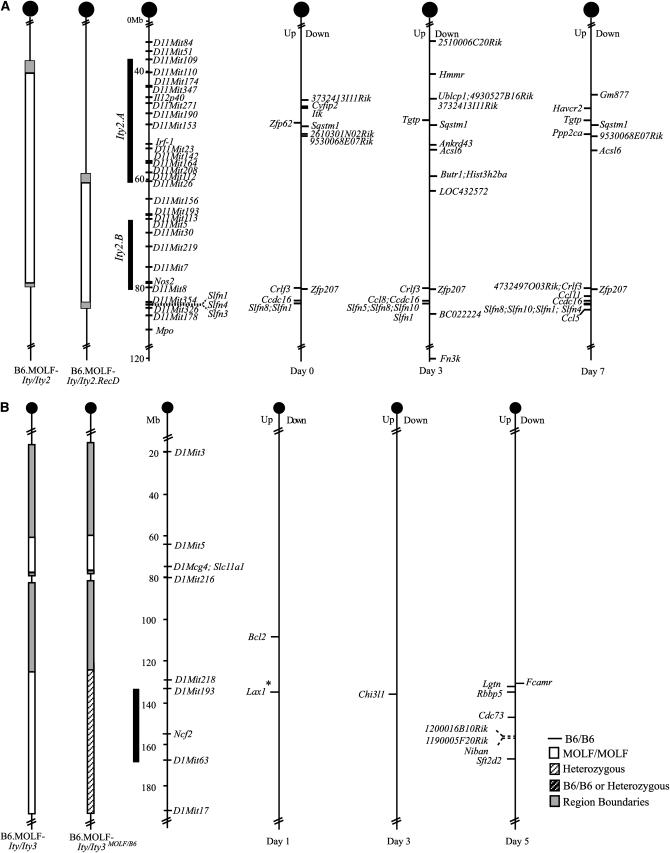
We next applied transcriptional profiling to help the identification of candidate genes underlying Ity2 and Ity3. Disease QTL have previously been shown to be linked to the heritability of variation in gene expression of positional candidate genes (Aitman et al. 1999; Hubner et al. 2005). Expression profiles of spleens from B6.MOLF-Ity/Ity2 (resistant) and B6.MOLF-Ity/Ity2.RecD (susceptible) mice were interrogated using the Affymetrix Mouse 430v2.0 chip at days 0, 3, and 7 of infection (Table 3, Figure 4A). Genes that differed by twofold and mapped to chromosome 11 were the primary focus of analysis. Differentially regulated genes were clustered within two chromosomal regions (D11Mit110–D11Mit26 and Nos2–D11Mit178) of different parental origin in the two congenic strains (Figure 4A). The proximal region delineated by D11Mit110 and D11Mit26 corresponds to the critical minimal Ity2.A interval, although the distal region (Nos2–D11Mit178) does not. No genes located within the Ity2.B interval were differentially regulated under the conditions specified.
TABLE 3.
Ity2 whole genome array list of differentially regulated genes on chromosome 11
| Up/downa | Affymetrix ID | Gene name | Position (Mb)b | Fold change | Function |
|---|---|---|---|---|---|
| Day 0 | |||||
| Up | 1451655_at | Slfn8 | 82.8 | 10.8 | T cell development |
| 1438235_at | Crlf3 | 79.9 | 8.0 | Cytokine receptor | |
| 1418612_at | Slfn1 | 83.1 | 5.6 | T cell development | |
| 1450942_at | Ccdc16 | 82.6 | 3.9 | Cell cycle | |
| 1451730_at | Zfp62 | 49.1 | 2.2 | Skeletal muscle | |
| Down | 1423546_at | Zfp207 | 80.2 | −3.3 | Metal binding nuclear protein |
| 1440076_at | Sqstm1 | 50.0 | −3.3 | Regulates NF-κB signaling | |
| 1443435_at | 3732413I11Rik | 44.4 | −3.3 | Ubiquitin conjugation | |
| 1456836_at | Itk | 46.2 | −2.5 | T cell development | |
| 1430177_at | 2610301N02Rik | 51.8 | −2.5 | Unknown | |
| 1427108_at | 9530068E07Rik | 52.2 | −2.0 | Unknown | |
| 1449273_at | Cyfip2 | 46.0 | −2.0 | Fragile X/T cell adhesion | |
| Day 3 | |||||
| Up | 1451655_at | Slfn8 | 82.8 | 15.6 | T cell development |
| 1438235_at | Crlf3 | 79.9 | 7.1 | Cytokine receptor | |
| 1450942_at | Ccdc16 | 82.6 | 5.5 | Cell cycle | |
| 1418612_at | Slfn1 | 82.9 | 5.1 | T cell development | |
| 1425728_at | Tgtp | 48.7 | 3.4 | T cell GTPase | |
| 1419684_at | Ccl8 | 81.9 | 2.3 | Chemotaxis | |
| 1458458_at, 1456288_at | Slfn5 | 82.8 | 2.2, 2.0 | T cell development | |
| 1444350_at | Slfn10 | 82.8 | 2.0 | T cell development | |
| Down | 1451257_at | Acsl6 | 54.1 | −3.3 | Fatty acid metabolism |
| 1445845_at | Ublcp1 | 44.3 | −3.3 | RNA polymerase II CTD | |
| 1423546_at | Zfp207 | 80.2 | −3.3 | Metal binding nuclear protein | |
| 1429871_at, 1427541_x_at | Hmmr | 40.5 | −3.3, −2.5 | Cell motility/ERK kinase | |
| 1429606_at | 4930527B16Rik | 44.3 | −2.5 | Unknown | |
| 1436998_at | Ankrd43 | 53.3 | −2.5 | DNA binding | |
| 1418311_at | Fn3k | 121.3 | −2.5 | Fructosamine kinase | |
| 1419074_at | 2510006C20Rik | 30.9 | −2.5 | Unknown | |
| 1421264_at | Butr1 | 58.7 | −2.5 | Immunoglobulin-like | |
| 1449482_at | Hist3h2ba | 58.8 | −2.5 | Histone/nucleosome | |
| 1440076_at | Sqstm1 | 50.0 | −2.5 | Regulates NF-κB signaling | |
| 1458440_at | LOC432572 | 62.0 | −2.5 | Unknown | |
| 1443435_at | 3732413I11Rik | 44.4 | −2.0 | Ubiquitin conjugation | |
| 1425704_at | BC022224 | 84.6 | −2.0 | Short chain dehydrogenase/reductase | |
| Day 7 | |||||
| Up | 1451655_at | Slfn8 | 82.8 | 10.2 | T cell development |
| 1418612_at | Slfn1 | 82.9 | 4.5 | T cell development | |
| 1425728_at | Tgtp | 48.7 | 3.8 | T cell GTPase | |
| 1438235_at | Crlf3 | 79.9 | 3.8 | Cytokine receptor | |
| 1418126_at | Ccl5 | 83.3 | 3.0 | T cell/macrophage chemokine | |
| 1450942_at | Ccdc16 | 82.6 | 3.0 | Cell cycle | |
| 1417789_at | Ccl11 | 81.9 | 2.4 | Eosinophil chemokine | |
| 1451584_at | Havcr2 | 46.3 | 2.3 | Macrophage/TH1 response | |
| 1444875_at | Ppp2ca | 51.9 | 2.1 | Kinase | |
| 1424501_at | 4732497O03Rik | 79.8 | 2.0 | RNA processing | |
| 1444350_at | Slfn10 | 82.8 | 2.0 | T cell development | |
| 1427102_at | Slfn4 | 83.0 | 2.0 | T cell development | |
| Down | 1440076_at | Sqstm1 | 50.0 | −3.3 | Regulates NF-κB signaling |
| 1436789_at | Ccnjl | 43.4 | −2.0 | Cyclin/cell cycle | |
| 1423546_at | Zfp207 | 80.2 | −2.0 | Metal binding nuclear protein | |
| 1427108_at | 9530068E07Rik | 52.2 | −2.0 | Unknown | |
| 1451257_at | Acsl6 | 54.1 | −2.0 | Fatty acid metabolism | |
Up- or downregulated in B6.MOLF-Ity/Ity2 compared to B6.MOLF-Ity/Ity2.RecD.
Physical map positions based on Ensembl build 39.
Figure 4.—
Location of differentially expressed genes on chromosome 11, Ity2 (A), and on chromosome 1, Ity3 (B) during infection. Genes found to be upregulated in the resistant B6.MOLF-Ity/Ity2 in A or B6.MOLF-Ity/Ity3 strain in B are on the left of the chromosome and downregulated genes on the right. The asterisk indicates that the position of Lax1 is approximated on the basis of the fact that it is syntenic to human 1q32.
Among those differentially regulated, there were a total of five genes that were differentially expressed at all time points (Table 3, Figure 4A). Three genes, Slfn8, Crlf3, and Ccdc16, were consistently expressed at higher levels in the resistant B6.MOLF-Ity/Ity2 strain and two genes, Zfp207 and Sqstm1, had lower expression at all time points. Of these consistently differentially regulated genes, only Sqstm1 (sequestosome 1) was mapped to the critical minimal Ity2.A interval. Sqstm1 is involved in the polyubiquitination of TRAF6 (TNF receptor-associated factor 6) and in the regulation of NF-κB signaling (Babu et al. 2005). Many of the pathways that lead to the upregulation of pro-inflammatory cytokine expression are dependent on proper NF-κB signaling, suggesting a role for Sqstm1 in the immune response.
In control mice (day 0), there were a total of 12 chromosome 11-specific genes that were differentially regulated (Table 3). Of the 12 genes, 7 were located within the minimal critical Ity2.A interval and included in addition to Sqstm1, Zfp62 (zinc-finger protein 62), Itk (IL-2 inducible T-cell kinase), Cyfip2 (cytoplasmic FMR1 interacting protein 2), and three ESTs. The gene Itk is clearly involved in T-cell activation (Au-Yeung et al. 2006) and Cyfip2 may also play a role in T-cell regulation, on the basis of recent work showing that high expression of CYFIP2 in multiple sclerosis patients is associated with increased T-cell adhesion (Mayne et al. 2004).
At day 3, we detected an increase in the number of differentially regulated genes on chromosome 11 for a total of 22 genes, of which 14 had lower expression and 8 genes had higher expression levels in B6.MOLF-Ity/Ity2 mice. Ten genes were located within the minimal Ity2.A interval, including several genes involved in the regulation of transcription (Ublcp1, Ankrd43, Hist3h2ba) and genes with potential relevance to the immune response, Hmmr (hyaluronan mediated motility receptor), Tgtp (T-cell specific GTPase), and Butr1 (butyrophilin related 1). Hmmr is a receptor involved in cell motility and in various kinase signaling cascades, including the ERK1 kinase (Turley et al. 2002), Tgtp is implicated in anti-viral responses (Carlow et al. 1998), and Butr1 according to UniProtKB/Swiss-Prot (http://ca.expasy.org/sprot/) is predicted to be a member of the immunoglobulin gene family.
At day 7, there were 5 transcripts with reduced expression and 12 transcripts with increased expression in B6.MOLF-Ity/Ity2 congenics, of which 7 were located within the minimal Ity2.A interval. We observed increased expression of two additional transcripts involved in T-cell activation, Tgtp and Havcr2, a gene involved in dampening TH1 immune responses and immunological tolerance (Carlow et al. 1998; Sanchez-Fueyo et al. 2003).
For all time points, the genes with lower expression levels were almost exclusively found in the proximal portion of the minimal Ity2.A interval and high transcriptional activity was observed at the distal limit of the Ity2 QTL interval, outside of the critical interval. This distal region contains members of the Schlafen family of genes (Slfn1, Slfn4, Slfn5, Slfn8, and Slfn10) that were expressed at higher levels at different time points during infection in the resistant B6.MOLF-Ity/Ity2 mice. Slfn1, and more recently Slfn8, has been demonstrated to be involved in the negative regulation of peripheral T-cell growth (Schwarz et al. 1998; Geserick et al. 2004). The differential regulation observed here may represent either epigenetic effects on gene expression due to rearrangement in the chromatin structure at the limit of the congenic interval or downstream cis effects due to the Ity2 QTL gene(s).
Validation of Ity2 microarray data:
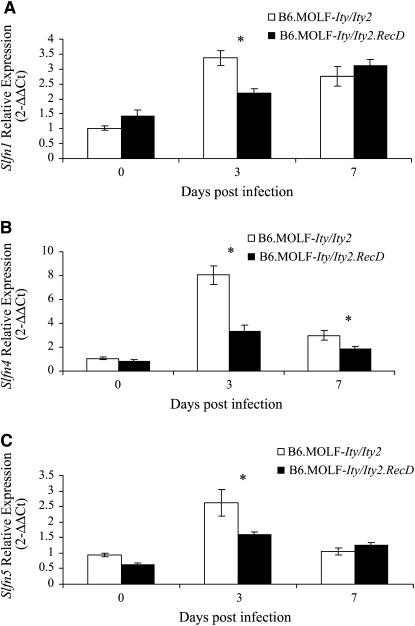
In general, alleles of MOLF/Ei origin present lower levels of expression for several genes located within the Ity2 QTL interval. We have measured mRNA levels of specific transcripts by quantitative PCR (QPCR) in the congenic strains to validate the observed differential expression in congenic mice and to determine if the interstrain differences in gene expression may be caused by the high genetic diversity known to be present between MOLF/Ei and C57BL/6J mice. We initially focused our validation on the Slfn family of genes that were consistently expressed at higher levels in the resistant B6.MOLF-Ity/Ity2 mice. We first sequenced the target region for Slfn1 and found numerous sequence variants between MOLF/Ei and C57BL/6J alleles that could have interfered with probe binding, accounting for the apparent differential expression observed using the microarray. Primers were then carefully designed to ensure that MOLF/Ei and C57BL/6J alleles were appropriately amplified by QPCR for all genes tested. We confirmed an expression difference for Slfn1, Slfn4, and Slfn5 (Figure 5). The expression of all genes was significantly increased in B6.MOLF-Ity/Ity2 during infection at day 3. Consistent with microarray results, Slf5 expression was higher in B6.MOLF-Ity/Ity2 mice on day 3, as was Slfn4 on day 7 but also on day 3. Slfn1 results did not correlate with microarray data due to the numerous sequence variants within the Affymetrix probe.
Figure 5.—
Real-time PCR validation of the Slfn cluster of genes during infection. Expression was determined using uninfected B6.MOLF-Ity/Ity2 mice as the referent. All values are expressed as 2−ΔΔCt units, where the referent is set to 1 and the housekeeping gene used was Tbp. Statistical significance was assessed using the Student's t-test and is indicated by an asterisk (P < 0.05). Slfn1 (A), Slfn4 (B), and Slfn5 (C) are shown here.
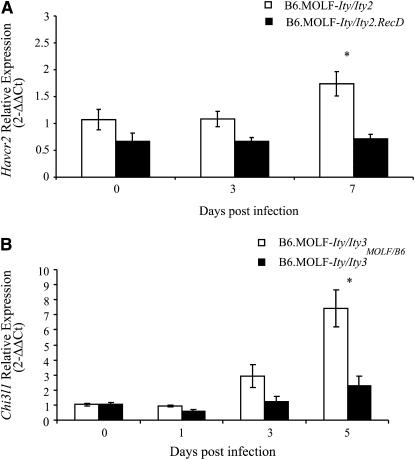
Six positional candidate genes (Sqstm1, Itk, Cyfip2, Butr1, Tgtp, and Havcr2) have been selected on the basis of their differential levels of expression and on their potential role in the host immune response for QPCR analyses. QPCR of Cypfip2, Itk, Butr1, Tgtp, and Sqstm1 expression revealed results that were inconsistent with the microarray data (data not shown). Interestingly, four of these genes, with the exception of Tgtp, were all found to be expressed at lower levels in the MOLF-derived allele represented in the B6.MOLF-Ity/Ity2 congenic, suggesting that the probes for these genes may contain sequence variants that affect hybridization as observed in the Slfn genes. All genes tested were either induced or downregulated upon infection. QPCR validation was confirmed for Havcr2 demonstrating a difference in the regulation of this gene upon infection (Figure 6A). In B6.MOLF-Ity/Ity2 mice there was an upregulation during the course of infection whereas this gene was not induced upon infection in B6.MOLF-Ity/Ity2.RecD mice.
Ity3 interval-specific expression profiling:
To investigate expression differences in the Ity3 interval, we employed a slightly different approach by creating a custom oligonucleotide array including probes representing genes that map within the Ity3 interval. The assembled array included 375 genes mapping between the markers D1Mit135 and D1Mit63 (based on Ensembl build v19.30.1), which corresponds to the region approximating the 2-LOD support interval. The quality of the array hybridization experiments was confirmed by positive hybridization signals emitted by the positive controls (18srRNA, Hprt, Tbp, Gapdh, herring sperm DNA, cot1 DNA). Spleen RNA from B6.MOLF-Ity/Ity3 (N5) and B6.MOLF-Ity/Ity3MOLF/B6 (N5) uninfected (day 0), day 1, day 3, and day 5 Salmonella Typhimurium-infected mice was hybridized to the arrays for analysis. The recessively inherited susceptibility phenotype that the Ity3 locus confers has previously been established using B6.MOLF-Ity/Ity3 and B6.MOLF-Ity/Ity3MOLF/B6 mice such that B6.MOLF-Ity/Ity3MOLF/B6 were more resistant to infection, as were the B6.MOLF-Ity mice shown in Figure 3D (Sancho-Shimizu and Malo 2006). Probes that were differentially regulated by twofold in dye swap experiments were considered for further analysis.
No genes were found to be differentially regulated at day 0; however, two genes, Bcl2 (B-cell leukemia/lymphoma 2) and Lax1 (lymphocyte transmembrane adaptor 1) (Table 4 and Figure 4B), had higher expression levels in the susceptible B6.MOLF-Ity/Ity3 mice 1 day after infection with Salmonella Typhimurium. Bcl2 is involved in suppressing apoptosis and it has been implicated in apoptosis associated with infections; however, this gene was found outside the critical candidate region for Ity3 (Kroemer 1997; Rios-Barrera et al. 2006). Lax1 has yet to be mapped definitively on the Ensembl mouse genome browser build 43; however, it is located on human 1p32.1, placing it on mouse chromosome 1 around 135.5 Mb within the Ity3 minimal interval, making it an excellent candidate gene on the basis of its role in lymphocyte signaling (Zhu et al. 2002).
TABLE 4.
Ity3 interval specific oligonucleotide array list of differentially regulated genes on chromosome 1 in spleen
| Up/downa | Accession ID | Gene name | Position (Mb)b | Fold change (average ± SEM) | Function |
|---|---|---|---|---|---|
| Day 1 | |||||
| Up | NM_172842 | Lax1 | c | 2.7 ± 0.7 | Regulates TCR/BCR signaling |
| NM_009741 | Bcl2 | 108.4 | 2.3 ± 0.2 | Suppresses apoptosis | |
| Day 3 | |||||
| Up | NM_007695 | Chi3l1 | 136.0 | 2.4 ± 0.3 | Bacterial adhesion/invasion |
| Day 5 | |||||
| Up | NM_145512 | Sft2d2 | 167.0 | 2.4 ± 0.2 | Retrograde vesicle transport |
| NM_022018 | Niban | 153.3 | 2.3 ± 0.2 | Unknown | |
| NM_02876 | 1190005F20Rik | 153.2 | 2.3 ± 0.3 | N(2),N(2)-dimethylguanosine tRNA methyltransferase | |
| NM_025819 | 1200016B10Rik | 153.1 | 2.2 ± 0.1 | Unknown | |
| NM_145991 | Cdc73 | 145.4 | 2.2 ± 0.4 | Tumor suppressor; cell cycle regulation | |
| NM_010709 | Lgtn | 133.0 | 2.1 ± 0.2 | Trafficking receptor for phosphoglycoproteins | |
| NM_172517 | Rbbp5 | 134.3 | 2.1 ± 0.4 | Binds retinoblastoma protein | |
| Down | NM_144960 | Fcamr | 132.6 | −3.7 ± 1.1 | Fc receptor (IgA, IgM) |
Up- or downregulated in B6.MOLF-Ity/Ity3 compared to B6.MOLF-Ity/Ity3MOLF/B6.
Physical map positions based on Ensembl build 39.
Physical map position is unknown on the basis of Ensembl build 39; however, it is syntenic to human 1q32.1, corresponding to a region around 135 Mb on mouse chromosome 1.
Only one gene, chitinase 3-like 1 (Chi3l1), had higher expression levels in the B6.MOLF-Ity/Ity3 mice on day 3. Chi3l1 is located within the critical Ity3 interval and has been shown recently to mediate bacterial adhesion and invasion in intestinal epithelial cells and to influence the outcome of oral Salmonella Typhimurium infections in mice, making it an interesting candidate gene to pursue (Mizoguchi 2006).
Seven genes had increased expression and one gene had lower expression on day 5 of infection in B6.MOLF-Ity/Ity3 mice as detected by array experiments. The only gene to have lower expression in B6.MOLF-Ity/Ity3 mice was Fcamr (FcαμR or Fc receptor, IgA, IgM, high affinity), the Fc receptor responsible for binding to IgM and IgA; however, it was found to be outside the critical interval (Shibuya et al. 2000). Of the seven genes with higher expression, six mapped within the critical Ity3 interval identified through fine mapping and include Rbbp5 (retinoblastoma-binding protein 5), Cdc73 (also known as Hprt2), Sft2d2 (SFT2 domain containing 2), Niban, 1200016B10Rik, and 1190005F20Rik (Table 2 and Figure 4B). Among these genes, only Sft2d2 presented with a relevant putative function in Salmonella infection, as it is predicted to be involved in the retrograde vesicle transport by similarity to the yeast Sft2p protein (Conchon et al. 1999).
Validation of Ity3 microarray data:
On the basis of position and function, four differentially regulated genes, Chi3l1, Cdc73, Sft2d2, and Niban, were further interrogated by QPCR. Cdc73 and Niban genes were constitutively expressed and not affected by infection whereas Sft2d2 was downregulated and Chi3l1 induced upon infection (data not shown and Figure 6B). Of those tested, only Chi3l1 was confirmed by QPCR, showing elevated expression levels in B6.MOLF-Ity/Ity3 spleens throughout infection but only significantly differing from B6.MOLF-Ity/Ity3MOLF/B6 mRNA levels on day 5, unlike the array data which found a difference at day 3 (Figure 6B). The discrepancy in these results may be due to the difference in the sensitivities of the two expression assays used, arrays vs. QPCR.
Figure 6.—
Real-time PCR validation of genes confirmed as differentially regulated in Ity2 (A) and Ity3 (B). Expression was determined using uninfected B6.MOLF-Ity/Ity2 or B6.MOLF-Ity/Ity3 mice as the referent. All values are expressed as 2−ΔΔCt units, where the referent is set to 1 and the housekeeping gene used was Tbp. Statistical significance was assessed using the Student's t-test and is indicated by an asterisk (P < 0.05). Ity2 candidate Havcr2 (A) and Ity3 candidate Chi3l1 (B) are shown here.
DISCUSSION
Ity2 and Ity3 are two QTL implicated in survival to systemic Salmonella Typhimurium infection that have been identified in the wild-derived inbred mouse MOLF/Ei (Sebastiani et al. 1998).We have used congenic mouse strains in combination with expression profiling to prioritize candidate genes for each QTL, an approach that has been used successfully in finding disease-causing genes in QTL mapping studies by other groups (Rozzo et al. 2001; Gu et al. 2002; Mcbride et al. 2003; Klein et al. 2004; Johannesson et al. 2005; De Buhr et al. 2006). A weakness of the expression profiling approach to keep in mind, however, is that the causative effect underlying quantitative traits, such as survival to Salmonella Typhimurium infection, may not be due to differential gene expression and this approach may be associated with the potential risk of not identifying the disease gene(s). In fact, several important Salmonella-susceptibility loci including Slc11a1 and Tlr4 are not regulated at the transcript levels (Vidal et al. 1993; Qureshi et al. 1999). On the other hand, differential expression of a candidate gene does not necessarily imply causation. An excellent example of this situation is provided by the gene Tlr5 in Salmonella susceptibility of MOLF/Ei mice. Tlr5 was clearly shown to be downregulated in the liver of MOLF/Ei mice; however, in vitro and in vivo functional analyses clearly showed that Tlr5 was not the gene underlying the Ity3 locus (Sebastiani et al. 2000; Angers et al. 2006). Despite these limitations, the use of congenic mice in microarray analyses remains extremely valuable in the prioritization of candidate genes for future analysis.
We have confirmed the genetic effect of Ity2 on resistance to infection and Ity3 on susceptibility to infection using congenic and subcongenic mouse strains. These mouse strains were also very useful to map Ity2 and Ity3 more precisely and in defining the minimal critical region to search for candidate genes. Congenics carrying the chromosome 11 Ity2 interval as well as Ity3 on distal chromosome 1 were created in the context of a protective allele at Slc11a1 within Ity (Sancho-Shimizu and Malo 2006). B6.MOLF-Ity/Ity2 congenics showed improved survival as compared to the B6.MOLF-Ity controls, consistent with the prediction from linkage analyses, in which the MOLF/Ei allele at Ity2 conferred protection and was inherited additively (Sebastiani et al. 1998). The two subcongenics, B6.MOLF-Ity/Ity2.RecD and B6.MOLF-Ity/Ity2RecI, enabled us to identify the minimal intervals necessary for resistance, Ity2.A and Ity2.B, suggesting the involvement of at least two loci. Ity2.A covers a 22-Mb proximal portion spanning D11Mit109 to D11Mit26 containing 331 genes, and Ity2.B consists of a 13-Mb distal interval from D11Mit5 to D11Mit8 including 364 genes.
B6.MOLF-Ity/Ity3 and subcongenics were used to further delineate the critical Ity3 interval to 34 Mb composed of 320 genes. The region boundaries marked by D1Mit415 and D1Mit216, as well as region boundaries surrounding the Ity interval, will be resolved further in future experiments, as their contribution to the susceptibility of B6.MOLF-Ity/Ity3 mice could not be determined. In spite of this, a clear and consistent phenotype was observed in the Ity3 congenic strains tested (B6.MOLF-Ity/Ity3, Rec.A, Rec.B, Rec.E), suggesting that the homozygous MOLF/Ei interval that they carry is responsible for the observed susceptibility.
Faced with a large number of candidate genes within the Ity2 and Ity3 intervals and the laborious and time-consuming creation of high resolution congenic mapping, we have used genomewide expression (Ity2) and QTL-specific arrays (Ity3) to obtain a list of candidate genes for each QTL. The advantage of microarrays lies partly in its unbiased approach to identify differentially expressed genes, at times leading to the discovery of genes that may not have been previously considered as candidates. For Ity2, the primary focus was to identify genes dysregulated in the QTL interval with the aim of identifying pathways that may be implicated in the phenotype. We noted first a bias of differentially regulated transcripts that appear as clusters on chromosomes 1 and 11 (supplemental Tables 1–3 at http://www.genetics.org/supplemental/). These happen to correspond to the chromosomes that carry the congenic intervals. This is probably due to the introduction of a MOLF/Ei genomic interval and may represent epigenetic effects on transcription. The mere introduction of a portion of the chromosome may affect the chromatin structure, e.g., affecting the transcription of a number of genes. Resolving expression differences due to the different congenic fragments in the context of Salmonella infection can be complicated by these extraneous influences.
Within the chromosome 11 interval, we have identified two clusters of differentially regulated genes, a cluster of genes around Ity2.A and another just outside the distal interval Ity2.B. We observed an overall trend that the differentially regulated genes in the proximal interval were almost always expressed at higher levels in the spleens of the resistant B6.MOLF-Ity/Ity2 mice, whereas those that were at the distal region had lower expression levels. An example of differentially regulated genes in the distal region is the Slfn family of genes. Upon sequencing of Slfn1, numerous sequence variants were identified (21 SNPs/1-kb coding sequence), suggesting that difference in intensity signal is probably due to poor probe hybridization in the B6.MOLF-Ity/Ity2.RecD mice rather than low transcript levels. Although some expression differences were confirmed upon infection (Figure 5), it is unlikely that these genes are the primary genes underlying Ity2 as they map outside the target congenic interval. The differential regulation may be due to the allelic differences in the genes, since the susceptible B6.MOLF-Ity/Ity2.RecD mice carry the MOLF/Ei allele in this interval, or may be due to epigenetic effects as discussed above.
The candidates of the Ity2 locus would be those that are differentially regulated in the proximal interval coincident with Ity2.A since the B6.MOLF-Ity/Ity2 mice carry the resistant MOLF/Ei allele in this interval as compared to the susceptible B6.MOLF-Ity/Ity2.RecD mice that harbor the C57BL/6J allele. We have identified six such genes in the spleen. Among these genes, the differential expression was confirmed for Havcr2 (Figure 6A). On day 7, Havcr2 (also known as Tim3) was found to be upregulated in the resistant congenic B6.MOLF-Ity/Ity2. Havcr2 is specifically expressed on TH1 cells and thought to negatively regulate TH1 responses and has been linked to asthma susceptibility (Meyers et al. 2005). This may indicate that the resistant B6.MOLF-Ity/Ity2 mice are more efficient at controlling the inflammatory response than their susceptible counterparts, suggesting a crucial role in the response to infection and leading to their prolonged survival. Hence, Havcr2 distinguishes itself as promising candidate for Ity2.
Many of the genes that were differentially expressed were involved in T cell activities, which is most probably due to tissue bias (Table 3). The apparent lack of differentially expressed genes in the Ity2.B interval may suggest that it may contain gene(s) with a protein defect that may in turn affect the expression of the genes within the Ity2.A interval. This may be a proposed mechanism by which these two critical intervals interact and are hence both necessary for the resistance phenotype observed in the B6.MOLF-Ity/Ity2 mice.
Custom oligonucleotide arrays were constructed for the Ity3 interval and led to the identification of four new candidates, Chi3l1, Cdc73, Sft2d2, and Niban, on the basis of position, expression, and potential function in the host response to Salmonella infection. These four genes had higher expression levels in the susceptible B6.MOLF-Ity/Ity3 mice; however, only one, Chi3l1, was validated by QPCR. Chi3l1 appears to be a very promising candidate on the basis of its function to enhance bacterial invasion and adherence in intestinal epithelial cells. In fact, the administration of Chi3l1-neutralizing antibodies prior to oral Salmonella Typhimurium infection was protective in mice (Mizoguchi 2006). Moreover, overexpression of this gene in vitro resulted in the presence of greater numbers of intracellular Salmonella Typhimurium (Mizoguchi 2006). Therefore, the upregulation of Chi3l1 may be consistent with increased susceptibility to infection as observed in B6.MOLF-Ity/Ity3 mice.
In this study, we have restricted the Ity2 and Ity3 intervals and proposed a list of candidate genes to be investigated further. One differentially regulated gene, Havcr2 in Ity2, and another such gene, Chi3l1 in Ity3, have been identified as potential candidates for these respective loci. The wild-derived inbred mouse MOLF/Ei has been separated by over 1 MY of evolution from the classical strains such as C57BL/6J, within which time they have accumulated numerous sequence variants, on the order of 1 SNP/100 bp (Abe et al. 2004; Ideraabdullah et al. 2004). The evolutionary divergence between the two strains is well illustrated in this study as reflected by the high sequence variation between C57BL/6J and MOLF/Ei alleles at specific genes, such as the Slfn's, which has the potential of affecting interpretation of array studies. Other than sequence variation, the possibility to affect transcriptional regulation on the basis of chromatin structure or due to genetic background effects is also demonstrated through the cluster of differentially regulated genes found distal to the Ity2.B interval. Ity3 was found to account for only 7% of the phenotypic variance, suggesting strongly that Salmonella susceptibility in MOLF/Ei mice results from polygenic inheritance in which the accumulation of weak effects at several different loci and interaction between loci explained the disease phenotype. The use of larger F2 populations and the creation of novel informative crosses will be necessary to reveal other loci that determine susceptibility to Salmonella infection in this wild-derived mouse. Nevertheless, the new genetic tools developed in these studies will contribute to further resolve the intricacies of Salmonella pathogenesis in the MOLF/Ei strain, contributing to our understanding of their extreme susceptibility to infection.
Acknowledgments
We thank Béatrice Kenol, Kyoko Yuki, and Catherine Paré for their technical assistance, Makeda Semret for helpful discussions in the creation of the Ity3 custom oligonucleotide arrays, and Fiona MacIntosh for her help in the printing of the arrays as well as in several hybridization experiments. This project was supported by research grants to D.M. from the Howard Hughes Medical Institute and the Canadian Institute for Health Research. D.M. is a McGill William Dawson Scholar. V.S-S. was supported by studentships from the McGill University Health Center Research Institute and by the McGill University Faculty of Medicine.
References
- Abe, K., H. Noguchi, K. Tagawa, M. Yuzuriha, A. Toyoda et al., 2004. Contribution of Asian mouse subspecies Mus musculus molossinus to genomic constitution of strain C57BL/6J, as defined by BAC-end sequence-SNP analysis. Genome Res. 14(12): 2439–2447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aitman, T. J., A. M. Glazier, C. A. Wallace, L. D. Cooper, P. J. Norsworthy et al., 1999. Identification of Cd36 (Fat) as an insulin-resistance gene causing defective fatty acid and glucose metabolism in hypertensive rats. Nat. Genet. 21(1): 76–83. [DOI] [PubMed] [Google Scholar]
- Angers, I., V. Sancho-Shimizu, A. Descoteaux, A. T. Gewirtz and D. Malo, 2006. Tlr5 is not primarily associated with susceptibility to Salmonella Typhimurium infection in MOLF/Ei mice. Mamm. Genome 17(5): 385–397. [DOI] [PubMed] [Google Scholar]
- Au-Yeung, B. B., S. D. Katzman and D. J. Fowell, 2006. Cutting edge: Itk-dependent signals required for CD4+ T cells to exert, but not gain, Th2 effector function. J. Immunol. 176(7): 3895–3899. [DOI] [PubMed] [Google Scholar]
- Babu, J. R., T. Geetha and M. W. Wooten, 2005. Sequestosome 1/p62 shuttles polyubiquitinated tau for proteasomal degradation. J. Neurochem. 94(1): 192–203. [DOI] [PubMed] [Google Scholar]
- Bihl, F., L. Lariviere, S. Qureshi, L. Flaherty and D. Malo, 2001. LPS-hyporesponsiveness of mnd mice is associated with a mutation in Toll-like receptor 4. Genes Immun. 2: 56–59. [DOI] [PubMed] [Google Scholar]
- Carlow, D. A., S. J. Teh and H. S. Teh, 1998. Specific antiviral activity demonstrated by TGTP, a member of a new family of interferon-induced GTPases. J. Immunol. 161(5): 2348–2355. [PubMed] [Google Scholar]
- Caron, J., L. Lariviere, M. Nacache, M. Tam, M. M. Stevenson et al., 2006. Influence of Slc11a1 on the outcome of Salmonella enterica serovar Enteritidis infection in mice is associated with Th polarization. Infect. Immun. 74(5): 2787–2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casanova, J. L., and L. Abel, 2004. The human model: a genetic dissection of immunity to infection in natural conditions. Nat. Rev. Immunol. 4(1): 55–66. [DOI] [PubMed] [Google Scholar]
- Charlet, D., S. Mostowy, D. Alexander, L. Sit, H. G. Wiker et al., 2005. Reduced expression of antigenic proteins MPB70 and MPB83 in Mycobacterium bovis BCG strains due to a start codon mutation in sigK. Mol. Microbiol. 56(5): 1302–1313. [DOI] [PubMed] [Google Scholar]
- Conchon, S., X. Cao, C. Barlowe and H. R. Pelham, 1999. Got1p and Sft2p: membrane proteins involved in traffic to the Golgi complex. EMBO J. 18(14): 3934–3946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Buhr, M. F., M. Mahler, R. Geffers, W. Hansen, A. M. Westendorf et al., 2006. Cd14, Gbp1, and Pla2g2a: three major candidate genes for experimental IBD identified by combining QTL and microarray analyses. Physiol. Genomics 25(3): 426–434. [DOI] [PubMed] [Google Scholar]
- Ferraro, T. N., G. T. Golden, G. G. Smith, J. F. Martin, F. W. Lohoff et al., 2004. Fine mapping of a seizure susceptibility locus on mouse Chromosome 1: nomination of Kcnj10 as a causative gene. Mamm. Genome 15(4): 239–251. [DOI] [PubMed] [Google Scholar]
- Geserick, P., F. Kaiser, U. Klemm, S. H. Kaufmann and J. Zerrahn, 2004. Modulation of T cell development and activation by novel members of the Schlafen (slfn) gene family harbouring an RNA helicase-like motif. Int. Immunol. 16(10): 1535–1548. [DOI] [PubMed] [Google Scholar]
- Gu, W., X. Li, K. H. Lau, B. Edderkaoui, L. R. Donahae et al., 2002. Gene expression between a congenic strain that contains a quantitative trait locus of high bone density from CAST/EiJ and its wild-type strain C57BL/6J. Funct. Integr. Genomics 1(6): 375–386. [DOI] [PubMed] [Google Scholar]
- Haywood, M. E., N. J. Rogers, S. J. Rose, J. Boyle, A. McDermott et al., 2004. Dissection of BXSB lupus phenotype using mice congenic for chromosome 1 demonstrates that separate intervals direct different aspects of disease. J. Immunol. 173(7): 4277–4285. [DOI] [PubMed] [Google Scholar]
- Hubner, N., C. A. Wallace, H. Zimdahl, E. Petretto, H. Schulz et al., 2005. Integrated transcriptional profiling and linkage analysis for identification of genes underlying disease. Nat. Genet. 37(3): 243–253. [DOI] [PubMed] [Google Scholar]
- Ideraabdullah, F. Y., E. de la Casa-Esperon, T. A. Bell, D. A. Detwiler, T. Magnuson et al., 2004. Genetic and haplotype diversity among wild-derived mouse inbred strains. Genome Res. 14(10A): 1880–1887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irizarry, R. A., B. Hobbs, F. Collin, Y. D. Beazer-Barclay, K. J. Antonellis et al., 2003. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4(2): 249–264. [DOI] [PubMed] [Google Scholar]
- Johannesson, M., L. M. Olsson, A. K. Lindqvist, S. Moller, D. Koczan et al., 2005. Gene expression profiling of arthritis using a QTL chip reveals a complex gene regulation of the Cia5 region in mice. Genes Immun. 6(7): 575–583. [DOI] [PubMed] [Google Scholar]
- Klein, R. F., J. Allard, Z. Avnur, T. Nikolcheva, D. Rotstein et al., 2004. Regulation of bone mass in mice by the lipoxygenase gene Alox15. Science 303(5655): 229–232. [DOI] [PubMed] [Google Scholar]
- Kroemer, G., 1997. The proto-oncogene Bcl-2 and its role in regulating apoptosis. Nat. Med. 3(6): 614–620. [DOI] [PubMed] [Google Scholar]
- Livak, K. J., and T. D. Schmittgen, 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4): 402–408. [DOI] [PubMed] [Google Scholar]
- Lyons, P. A., N. Armitage, F. Argentina, P. Denny, N. J. Hill et al., 2000. Congenic mapping of the type 1 diabetes locus, Idd3, to a 780-kb region of mouse chromosome 3: identification of a candidate segment of ancestral DNA by haplotype mapping. Genome Res. 10(4): 446–453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayne, M., T. Moffatt, H. Kong, P. J. McLaren, K. R. Fowke et al., 2004. CYFIP2 is highly abundant in CD4+ cells from multiple sclerosis patients and is involved in T cell adhesion. Eur. J. Immunol. 34(4): 1217–1227. [DOI] [PubMed] [Google Scholar]
- McBride, M. W., F. J. Carr, D. Graham, N. H. Anderson, J. S. Clark et al., 2003. Microarray analysis of rat chromosome 2 congenic strains. Hypertension 41(3): 847–853. [DOI] [PubMed] [Google Scholar]
- Meyers, J. H., C. A. Sabatos, S. Chakravarti and V. K. Kuchroo, 2005. The TIM gene family regulates autoimmune and allergic diseases. Trends Mol. Med. 11(8): 362–369. [DOI] [PubMed] [Google Scholar]
- Mizoguchi, E., 2006. Chitinase 3-like-1 exacerbates intestinal inflammation by enhancing bacterial adhesion and invasion in colonic epithelial cells. Gastroenterology 130(2): 398–411. [DOI] [PubMed] [Google Scholar]
- Ohl, M. E., and S. I. Miller, 2001. Salmonella: a model for bacterial pathogenesis. Annu. Rev. Med. 52: 259–274. [DOI] [PubMed] [Google Scholar]
- Qureshi, S., L. Lariviere, G. Leveque, S. Clermont, K. Moore et al., 1999. Endotoxin-tolerant mice have mutations in Toll-like reseptor 4 (Tlr4). J. Exp. Med. 189: 615–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rios-Barrera, V. A., V. Campos-Pena, D. Aguilar-Leon, L. R. Lascurain, M. A. Meraz-Rios et al., 2006. Macrophage and T lymphocyte apoptosis during experimental pulmonary tuberculosis: their relationship to mycobacterial virulence. Eur. J. Immunol. 36(2): 345–353. [DOI] [PubMed] [Google Scholar]
- Rogner, U. C., and P. Avner, 2003. Congenic mice: cutting tools for complex immune disorders. Nat. Rev. Immunol. 3(3): 243–252. [DOI] [PubMed] [Google Scholar]
- Roy, M. F., and D. Malo, 2002. Genetic regulation of host responses to Salmonella infection in mice. Genes Immun. 3(7): 381–393. [DOI] [PubMed] [Google Scholar]
- Rozzo, S. J., J. D. Allard, D. Choubey, T. J. Vyse, S. Izui et al., 2001. Evidence for an interferon-inducible gene, Ifi202, in the susceptibility to systemic lupus. Immunity 15(3): 435–443. [DOI] [PubMed] [Google Scholar]
- Sanchez-Fueyo, A., J. Tian, D. Picarella, C. Domenig, X. X. Zheng et al., 2003. Tim-3 inhibits T helper type 1-mediated auto- and alloimmune responses and promotes immunological tolerance. Nat. Immunol. 4(11): 1093–1101. [DOI] [PubMed] [Google Scholar]
- Sancho-Shimizu, V., and D. Malo, 2006. Sequencing, expression, and functional analyses support the candidacy of Ncf2 in susceptibility to Salmonella typhimurium infection in wild-derived mice. J. Immunol. 176(11): 6954–6961. [DOI] [PubMed] [Google Scholar]
- Schwarz, D. A., C. D. Katayama and S. M. Hedrick, 1998. Schlafen, a new family of growth regulatory genes that affect thymocyte development. Immunity 9(5): 657–668. [DOI] [PubMed] [Google Scholar]
- Sebastiani, G., L. Olien, S. Gauthier, E. Skamene, K. Morgan et al., 1998. Mapping of genetic modulators of natural resistance to infection with Salmonella typhimurium in wild-derived mice. Genomics 47(2): 180–186. [DOI] [PubMed] [Google Scholar]
- Sebastiani, G., G. Leveque, L. Lariviere, L. Laroche, E. Skamene et al., 2000. Cloning and characterization of the murine Toll-like Receptor 5 (Tlr5) gene: sequence and mRNA expression studies in Salmonella-susceptible MOLF/Ei mice. Genomics 64(3): 230–240. [DOI] [PubMed] [Google Scholar]
- Shibuya, A., N. Sakamoto, Y. Shimizu, K. Shibuya, M. Osawa et al., 2000. Fc alpha/mu receptor mediates endocytosis of IgM-coated microbes. Nat. Immunol. 1(5): 441–446. [DOI] [PubMed] [Google Scholar]
- Subramanian, S., Y. S. Yim, K. Liu, K. Tus, X. J. Zhou et al., 2005. Epistatic suppression of systemic lupus erythematosus: fine mapping of Sles1 to less than 1 mb. J. Immunol. 175(2): 1062–1072. [DOI] [PubMed] [Google Scholar]
- Turley, E. A., P. W. Noble and L. Y. Bourguignon, 2002. Signaling properties of hyaluronan receptors. J. Biol. Chem. 277(7): 4589–4592. [DOI] [PubMed] [Google Scholar]
- Vidal, S., D. Malo, K. Vogan, E. Skamene and P. Gros, 1993. Natural resistance to infection with intracellular parasites: isolation of a candidate for Bcg. Cell 73: 469–485. [DOI] [PubMed] [Google Scholar]
- Vidal, S., M. Tremblay, G. Govoni, S. Gauthier, G. Sebastiani et al., 1995. The Ity/Lsh/Bcg locus: natural resistance to infection with intracellular parasites is abrogated by disruption of the Nramp1 gene. J. Exp. Med. 182(3): 655–666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, M., E. Janssen, K. Leung and W. Zhang, 2002. Molecular cloning of a novel gene encoding a membrane-associated adaptor protein (LAX) in lymphocyte signaling. J. Biol. Chem. 277(48): 46151–46158. [DOI] [PubMed] [Google Scholar]