Dear Editor,
All-trans retinoic acid (ATRA) has become the mainstay in the treatment of acute promyelocytic leukemia (APL) because addition of ATRA to anthracyclines has been shown to improve disease-free survival and overall survival compared to chemotherapy alone [1]. ATRA induces the differentiation of leukemic promyelocytes into mature cells via its effect on the promyelocytic leukemia–retinoic acid receptor fusion gene product, which results from the translocation of chromosomes 15 and 17, characteristic for APL.
ATRA therapy can be complicated by the ATRA syndrome, first described by Frankel et al. [2] and consisting of respiratory distress (89%), pulmonary infiltrates (81%), fever (81%), bodyweight gain (50%), pleural effusion (47%), renal failure (39%), pericardial effusion (19%), cardiac failure (17%), and/or hypotension (12%). It occurs in about 25% of APL patients treated with ATRA monotherapy and in about 10–15% when combined with chemotherapy [2–4]. This syndrome typically starts within the first 21 days, with a median of 7–12 days [5]. In case of ATRA syndrome, retinoic acid should be discontinued and dexamethasone instituted.
We recently observed two nearly similar cases of isolated, symptomatic ATRA-induced myocarditis. Both patients had newly diagnosed t(15;17) positive APL and a negative cardiac history and were treated with ATRA in combination with idarubicin.
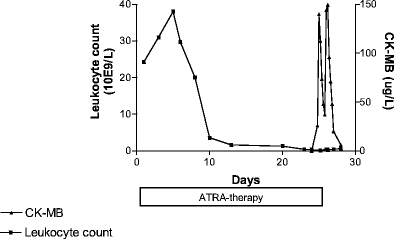
Patient 1, a 34-year-old woman, developed fever and cardiac pain (without skeletal muscle pain) on days 19 and 21, respectively. Physical examination showed no abnormalities. The electrocardiogram revealed ST elevations in most leads, and laboratory analysis showed a white blood cell count (WBC) of 1.1×109/l, a creatine kinase (CK) of 798 U/l (normal value <190 U/l), and CK-MB of 116 μg/l (normal value <6.5 μg/l) (Fig. 1). On echocardiography a pattern of perimyocarditis with minimal pericardial effusion was seen. ATRA was discontinued but dexamethasone was withheld because at that time point viral myocarditis could not be ruled out (later all serological tests for viral infections proved to be negative). The patient recovered uneventfully. ATRA was reintroduced in the following cycles of consolidation chemotherapy, which consisted of subsequent cycles with idarubicin and ATRA, mitoxantrone and ATRA, and idarubicin and ATRA without side effects. The patient achieved sustained molecular complete remission (follow-up more than 60 months) with a maintenance therapy consisting of 6-mercaptopurine, ATRA, and methotrexate once weekly during 2 years.
Fig. 1.

Case 1
Patient 2, a 46-year-old man, became febrile, hypotensive, and complained of a constant left thoracic pain (not related to respiration) on days 23 and 24 after the initiation of ATRA. There was no skeletal muscle pain. Apart from profuse sweating, physical examination was normal. The electrocardiogram showed ST elevations in most leads consistent with pericarditis. Laboratory analysis showed a WBC of 0.3×109/l, CK 1,643 U/l, and CK-MB 144 g/l (Fig. 2). Echocardiography and cardiac MRI showed a pattern of myocarditis with generalized abnormal wall movement, without pericardial effusion. ATRA was discontinued and dexamethasone was withheld because at that time point viral myocarditis could not be ruled out (also, in this patient, later all serological tests for viral infections proved to be negative). The patient recovered uneventfully and cardiac function normalized. In this patient, ATRA was reintroduced uneventfully in the following consolidation courses consisting of idarubicin, cytarabin plus ATRA, mitoxantrone plus ATRA, and idarubicin, cytarabin plus ATRA. Complete molecular remission was achieved (follow-up 6 months) and maintenance therapy consisting of 6-mercaptopurine, ATRA, and methotrexate once weekly for a period of 2 years was initiated.
Fig. 2.
Case 2
In both cases a diagnosis of isolated ATRA-induced myocarditis was made, as symptoms disappeared spontaneously when ATRA was discontinued and no other causes for myocarditis could be found.
The two cases reported here are important because the myocarditis was not only symptomatic but was also diagnosed in the absence of any symptoms or signs of the ATRA syndrome. To our knowledge, just one case of ATRA-induced generalized myositis with cardiac involvement has been reported [6]. Although in that case no myocarditis-related clinical signs or symptoms were present, electrocardiogram and echocardiography were consistent with the diagnosis of cardiac myocarditis. There have been no previous reports of ATRA-induced isolated symptomatic myocarditis.
Whether the myocarditis is part of the ATRA syndrome or toxicity-related is difficult to differentiate. The pathogenesis of the ATRA syndrome is not yet fully understood, but it is believed to be due to leukocyte infiltration and cytokine release causing endothelial damage and extravasation of APL cells upon maturation induction by ATRA treatment [3]. Necropsy of a patient with severe and fatal ATRA syndrome revealed extensive organ infiltration by the malignant cells, including myocardial involvement [7]. In that specific case, the patient had no cardiac complaints and the leukocyte count was high. In both our cases WBCs were low, making massive leukocyte infiltration a less likely cause of myocarditis. Indeed, in a series of patients with ATRA syndrome, leukocytosis was absent in one third of the patients.
In conclusion, we present two cases of ATRA-induced, isolated, symptomatic myocarditis. Both our patients recovered completely without the use of steroids and were subsequently retreated with ATRA without recurrence of the myocarditis.
Acknowledgment
S. K. Klein wrote the article. B. J. Biemond and M. H. J. van Oers provided the data of the patients and reviewed the manuscript.
References
- 1.Tallman MS, Anderson JW, Schiffer CA et al (1997) All-trans-retinoic acid in acute promyelocytic leukemia. N Engl J Med 337:1021–1028 [DOI] [PubMed]
- 2.Frankel SR, Eardley A, Lauwers G, et al (1992) The ‘retinoic acid syndrome’ in acute promyelocytic leukemia. Ann Int Med 117:292–301 [DOI] [PubMed]
- 3.Fenaux P, Boutton de S (1998) Retinoic acid syndrome, recognition, prevention and management. Drug Saf 18(4):273–279 [DOI] [PubMed]
- 4.Avvisati G, Lo Coco F, Diverio D et al (1996) AIDA (all-trans retinoic acid + idarubicin) in newly diagnosed acute promyelocytic leukemia: a gruppo Italiano Malattie Ematologiche maligne dell’ adulto (GIMEMA) pilot study. Blood 88:1390–1398 [PubMed]
- 5.De Botton S, Dombret H, Sanz M et al, European APL Group (1998) Incidence, clinical features, and outcome of all trans-retinoic acid syndrome in 413 cases of newly diagnosed acute promyelocytic leukemia. Blood 92:2712–2718 [PubMed]
- 6.Fabbiano F, Magrin S, Cangialosi C, Felice R, Mirto S, Pitrolo F (2005) All-trans retinoic acid induced cardiac and skeletal myositis in induction therapy of acute promyelocytic leukaemia. Br J Haematol 129:442–445 [DOI] [PubMed]
- 7.Kakkar N, Dhameja N, Jasmina, Das A, Radotra BD, Varma S (2002) ATRA syndrome with extensive organ infiltration. Am J Hematol 71(1):62–64 [DOI] [PubMed]


