Abstract
Summary
Purpose:
To examine the characteristics and healthcare costs of fibromyalgia syndrome (FMS) patients in clinical practice.
Materials and methods:
Using a US health-insurance database, we identified all patients, aged ≥ 18 years, with any healthcare encounters for FMS (ICD-9-CM diagnosis code 729.1) in each year of the 3-year period, 1 July 2002 to 30 June 2005. A comparison group was then constituted, consisting of randomly selected patients without any healthcare encounters for FMS during this 3-year period. Comparison group patients were matched to FMS patients based on age and sex. Characteristics and healthcare costs of FMS patients and comparison group patients were then examined over the 1-year period, 1 July 2004 to 30 June 2005 (the most recent year for which data were available at the time of the study).
Results:
The study sample consisted of 33,176 FMS patients and an identical number in the comparison group. Mean age was 46 years, and 75% were women. FMS patients were more likely to have various comorbidities, including painful neuropathies (23% vs. 3% for comparison group), anxiety (5% vs. 1%), and depression (12% vs. 3%) (all p < 0.001); they also were more likely to have used pain-related pharmacotherapy (65% vs. 34% for comparison group; p < 0.001). Mean (SD) total healthcare costs over 12 months were about three times higher among FMS patients [$9573 ($20,135) vs. $3291 ($13,643); p < 0.001]; median costs were fivefold higher ($4247 vs. $822; p < 0.001).
Conclusions:
Patients with FMS have comparatively high levels of comorbidities and high levels of healthcare utilization and cost.
What's known
Much is known concerning the epidemiology of fibromyalgia syndrome (FMS). The efficacy of various pain-related medications in FMS has also been studied. Some information regarding utilization and cost is also available.
What's new
Our study examines levels of comorbidities and healthcare utilization and cost among patients with FMS in actual clinical practice, and seeks to place these findings in context, using an age- and sex-matched group of patients without FMS as comparators. In addition, our findings are based on data that are relatively current (1 July 2004 to 30 June 2005), as opposed to previous analyses for which data are at least a decade old.
Introduction
Fibromyalgia syndrome (FMS) is a widespread disorder of unknown aetiology that affects an estimated 2–4% of the general population (1), and over 5% of patients in general medical practice (2). Women are about nine times more likely to develop FMS than men (1). Symptoms typically appear between the ages of 20 and 55 years. The predominant symptom of FMS is widespread musculoskeletal pain. A large number of additional symptoms are also often present, including sleep disturbance, fatigue, morning stiffness, paresthesias, headaches and exercise intolerance. The symptoms of FMS can be prolonged and debilitating.
Fibromyalgia syndrome is characterized by widespread pain, tenderness and fatigue, and is typically difficult to diagnose. While various tests may be ordered to rule out other possible causes of patients’ symptoms, such as rheumatoid arthritis and lupus, none is sufficiently sensitive or specific to establish a diagnosis of FMS. In 1990, the American College of Rheumatology (ACR) published diagnostic criteria for FMS – namely, widespread pain (both sides of the body, above and below the waist, and in the cervical spine, anterior chest, thoracic spine or low back), and pain on digital palpation in at least 11 of 18 specified tender point sites (3). If a patient has typical symptoms of FMS but does not meet the ACR criteria, a diagnosis of ‘possible FMS’ is often assigned, and a therapeutic trial of standard treatment may be prescribed.
Treatment of FMS is typically geared towards reducing pain and improving quality of sleep. [It has been suggested that FMS may be due to non-restorative sleep; about three-quarters of FMS patients report non-restorative sleep (3), which often is followed by an increase, or ‘flare-up’, of symptoms.] Early controlled clinical studies demonstrated that amitriptyline, cyclobenzaprine, fluoxetine and alprazolam are effective in FMS; more recently, trials of pregabalin and duloxetine have also shown efficacy (4–13). Imipramine, steroids and non-steroidal anti-inflammatory drugs (NSAIDs) have been reported to be no better than placebo in the treatment of FMS (13). Non-pharmacological interventions, including cardiovascular fitness training, biofeedback, acupuncture and hypnotherapy, have shown limited efficacy in some patients (14–16).
Information concerning patterns of healthcare utilization and costs among FMS patients is somewhat limited. One study that examined 402 patients with chronic fatigue, chronic fatigue syndrome (CFS), FMS, and CFS and FMS reported that levels of healthcare utilization were generally high and similar across the four groups of patients (17). In a Canadian study, White et al. compared the healthcare costs of 100 FMS patients with those of 76 patients with widespread pain (but not FMS), 135 patients without widespread pain, and a random sample of 380 ‘controls’ matched on age, sex and geographical region (18). In this study, FMS patients were found to use more pain-related medications and outpatient healthcare services than patients with widespread pain; their annual healthcare costs also were CDN$493 higher compared with those of controls. In their 7-year prospective study of 538 FMS patients, Wolfe et al. found that FMS patients averaged approximately 10 outpatient visits per year, and that mean annual healthcare costs were $2274 (in 1996 dollars) (19). In their questionnaire-based study of 180 women with FMS, Penrod et al. reported that healthcare costs averaged CDN$2298 over 6 months (20). Two additional studies were based on electronic healthcare databases (US and UK respectively) and were limited to encounters with general practitioners. In the former study, based on data from a Fortune 100 manufacturer, Robinson et al. reported that mean annual healthcare costs among FMS patients (n = 4699) were twice as high as those of a 10% random sample of the overall insured population ($5945 vs. $2468; p < 0.001) (21). In the latter study, Hughes et al. reported that, among 2260 patients newly diagnosed with FMS, there were 25 office visits and 11 prescriptions, on average, in the year prior to diagnosis, and that levels of utilization were generally even higher following diagnosis (22). Another recent study by Boonen et al. examined costs of Dutch patients with FMS (n = 69), chronic low back pain (n = 110) and ankylosing spondylosis (n = 111), respectively, using cost diaries (23). The authors reported that FMS patients had the highest annual healthcare costs (€1300 vs. €1104 for chronic low back pain and €1043 for ankylosing spondylosis); mean (median) total annual costs (including healthcare, non-medical and production losses during sick leave) were estimated to be €7814 (5145) for FMS patients, €8533 (5068) for those with chronic low back pain and €3205 (1793) for those with ankylosing spondylosis.
There are substantial limitations to existing research, however. Several of the studies were conducted well over a decade ago, and many of the more recent ones have other shortcomings, including small sample size and/or choice of reference group. Moreover, many of these studies are based on questionnaire data, which may not necessarily reflect actual patterns of utilization because of problems with patient recall and/or comprehensiveness of data collection. In this study, we compare the characteristics and healthcare costs of patients with FMS with those of an age- and sex-matched comparison group, using a large US health-insurance claims database.
Methods
Data were obtained from the PharMetrics Patient-Centric Database. The database is composed of facility, professional service and retail (i.e. outpatient) pharmacy claims from over 85 US health plans. The plans provide healthcare coverage to approximately 11 million persons annually throughout the US (Midwest, 35%; Northeast, 21%; South, 31%; West, 13%). All patient identifiers in the database have been fully encrypted, and the database is fully compliant with the Health Insurance Portability and Accountability Act of 1996.
Information available for each facility and professional service claim includes date and place of service, diagnoses (in ICD-9-CM format), procedures [in ICD-9-CM (selected plans only) and HCPCS formats], provider specialty, and charged and paid amounts. Data available for each retail pharmacy claim include the drug dispensed (in NDC format), the dispensing date, and the quantity dispensed and number of days of therapy supplied (selected plans only). All claims include a charged amount; the database also provides paid (i.e. total reimbursed, including patient deductible, copayment, and/or coinsurance) amounts.
Selected demographic and eligibility information is also available, including age, gender, geographical region, coverage type and the dates of insurance coverage. All patient-level data can be arrayed in chronological order to provide a detailed, longitudinal profile of all medical and pharmacy services used by each insured person.
Using the PharMetrics Patient-Centric Database, we identified all patients, aged ≥ 18 years, who had one or more healthcare encounters with a diagnosis of FMS (ICD-9-CM diagnosis code 729.1) in each year of the 3-year period, 1 July 2002 to 30 June 2005 (i.e. all patients were required to have encounters with a diagnosis of FMS in three concurrent years). As FMS is often difficult to diagnosis correctly, we used this selection algorithm to maximize the likelihood that subjects in our study actually had FMS. Patients enrolled in Medicaid programmes and those aged ≥ 65 years who were enrolled in Medicare supplemental or capitated plans were also excluded, as their claims histories may be incomplete.
A comparison group was constituted, consisting of a cohort of randomly selected patients without any encounters with a diagnosis of FMS over the 3-year period, 1 July 2002 to 30 June 2005. The comparison group was matched to FMS patients on the basis of age and sex. All other inclusion/exclusion criteria used to constitute the cohort of FMS patients were used to constitute the comparison group.
All pharmacy, professional service and facility claims were then compiled for all study subjects (FMS and comparator group) between 1 July 2004 and 30 June 2005 (or the patient's period of continuous eligibility, if less than 365 days).
The prevalence of selected (medically attended) comorbidities was examined for both FMS patients and patients in the comparison group (Table 1); patients were deemed to have any of the conditions of interest if they had either one or more hospitalizations or two or more outpatient claims on different days with a relevant diagnosis code during the year of interest.
Table 1.
Definitions of selected comorbidities*
| Comorbidity | ICD-9-CM diagnosis codes |
|---|---|
| Neoplasms | 140.X–239.X |
| Diabetes | 250–250.XX |
| Mental and mood disorders | |
| Anxiety | 300.01, 300.3, 309.81, 300.23, 300.21, 300.22, 300.2,300.20, 300.29, 300.02, 293.84, 309.21, 300.0, 300.00, 300.09,300.1X |
| Depression | 300.4, 309.0, 309.1, 296.5, 296.2, 296.3, 290.21, 292.84, 296.20 - 296.XX, 298.0 |
| Tension headache | 307.81 |
| Migraine | 346–346.XX |
| Diseases of the circulatory system | 390–459.XX |
| Diseases of the respiratory system | 460–519.X |
| Diseases of the digestive system | |
| Irritable bowel syndrome | 564.1 |
| GERD | 530.11, 530.81 |
| Gastritis | 535.0–535.5 |
| Other | 520.5–537.X, 540–543.X, 550.0–553.X, 555.0–558.X,560–560.X, 562.00–562.XX, 564–579.X |
| Diseases of the musculoskeletal system and connective tissue | |
| Back pain | 722.92–722.93, 724–724.X |
| Cervical pain | 722.4–722.5, 722.81, 722.91, 723.1, 723.5–723.6 |
| Arthritis | 711.00–716.XX |
| Other body/joint pain | 710.0, 717–720.X, 725–729.X |
| Other | 710–710.X, 721–723.X, 730–739.X |
| Symptoms, signs and ill-defined conditions | |
| Fatigue | 780.71, 780.79 |
| Headache | 784 |
| Chest pain | 786.5–786.5X |
| Abdominal pain | 789.0–789.0X |
| Anxiety-related symptoms | 780.4, 785.0–785.1, 786.01, 786.05, 786.09 |
| Gastric-related symptoms | 787.0, 787.01–787.03, 787.1–787.3, 787.9, 787.91, 787.99 |
| Other | 780.02–796.X, 799–799.X |
| Painful neuropathic disorders | |
| Diabetic neuropathy | 250.6X, 357.2 |
| Post-herpetic neuralgia | 53.1X |
| Back pain withneuropathic involvement | 721.41, 721.42, 721.91, 722.1, 722.10, 722.11, 722.2, 722.70,722.72, 722.73, 724.0X, 724.3, 724.4 |
| Neck pain with neuropathicinvolvement | 721.1, 722.0, 722.71, 723.0, 723.4 |
| Cancer with neuropathic pain | Malignant neoplasms (140.XX–172.XX, 174.XX–208.XX) in conjunction with neuropathy337.2X, 353.2, 353.3, 353.4, 354.4, 355.7X, 355.9, 729.2, 353.0, 353.1, 353.8, 353.9,354.0, 354.1, 354.2, 354.3, 354.5, 354.8, 354.9, 355.0, 355.1, 355.2, 355.3, 355.4,355.5, 355.6, 355.8, 357.3, 357.8, 357.9 |
| Causalgia | 337.2X, 353.2, 353.3, 353.4, 354.4, 355.7X, 355.9, 729.2 |
| Phantom limb pain | 353.6 |
| Trigeminal neuralgia | 350.1 |
| Atypical facial pain | 350.2, 352.1 |
| Other painful neuropathies | 353.0, 353.1, 353.8, 353.9, 354.0, 354.1, 354.2, 354.3, 354.5, 354.8, 354.9, 355.0, 355.1,355.2, 355.3, 355.4, 355.5, 355.6, 355.8 |
| Sleep disorders | 780.51, 780.52, 307.41, 307.42, 307.49, 780.53, 780.57, 786.03, 347.0X, 347.1X, V69.4,780.5, 780.50, 780.54, 780.55, 780.56, 780.58, 780.59 |
*ICD-9-CM diagnosis codes may be three, four or five digits; ‘X’ for the fourth or fifth digit indicates a wildcard. Codes with fewer than five digits and no Xs in the fourth or fifth digit were selected only if recorded without subsequent digits.
The number of patients receiving ≥ 1 prescriptions for various ‘pain-related’ medications during the year of interest was examined, including: (i) antiepileptics (AEDs); (ii) benzodiazepines; (iii) corticosteroids; (iv) cyclo-oxygenase (COX)-2 inhibitors and other prescription NSAIDs; (v) muscle relaxants; (vi) sedatives/hypnotics; (vii) opioids (both short- and long-acting agents); (viii) antidepressants [including tricyclic antidepressants (TCAs), monoamine oxidase (MAO) inhibitors, and selective serotonin reuptake inhibitors (SSRIs)]; (ix) antimigraine agents (triptans, all other); and (x) miscellaneous agents [injectable analgesics (e.g. bupivacaine), topical analgesics (e.g. lidocaine), clonidine]. Medications were designated as being ‘pain-related’ based on their designation as analgesics or adjuvant medications in the World Health Organization's (WHO) ‘analgesic ladder’, which was developed initially for the treatment of cancer pain (24). Published literature has supported the use of adjuvant medications for the treatment of neuropathic pain in patients with and without cancer (25–36). Patients were considered to have received combinations of pain-related medications if, during the year of interest, they received at least one prescription for ≥ 2 classes of pain-related medications (e.g. antidepressants and opioids). All other medications were defined to be non-pain-related.
Utilization of healthcare services [i.e. outpatient pharmacy dispenses, doctor office visits, other outpatient visits, emergency department (ED) visits and hospitalizations] was examined over the 1-year period, 1 July 2004 to 30 June 2005, based on paid medical claims. No attempt was made to attribute care specifically to the treatment of FMS. Costs were tallied by category of care, as follows: (i) pain-related medications; (ii) non-pain-related medications; (iii) doctor office visits; (iv) ED visits; (v) hospital outpatient visits; and (vi) inpatient care. Reimbursed amounts (including patient co-pays) were used in all analyses of healthcare costs. Where applicable, measures of counts and costs were annualized for patients not continuously enrolled in the database over the entire year of interest.
The statistical significance of differences between FMS patients and the comparison group was calculated using paired t-tests for normally distributed continuous measures; otherwise, a Wilcoxon signed-rank test was used. McNemar's and Bowker's tests were used to determine the statistical significance of differences in categorical measures, as appropriate. All analyses were conducted, using PC-SAS® v.9.1 (37).
Results
The study sample consisted of 33,176 FMS patients and an identical number in the comparison group. Mean age was 46 years, and 75% were women. FMS patients were more likely than those in the comparison group to have various comorbidities, including diseases of the circulatory system [OR (95% CI) = 2.1 (2.0–2.1)], diabetes [1.5 (1.4–1.6)], anxiety [4.3 (3.8–4.7)], depression [4.9 (4.5–5.2)], irritable bowel syndrome [6.2 (4.9–7.9)], gastro-oesophageal reflux disease [3.8 (3.4–4.2)] and sleep disorders [6.1 (5.4–6.9)] (all p < 0.001) (Table 2). They also were more likely to have pain-related comorbidities, including painful neuropathies [10.3 (9.6–11.0)], back pain [14.2 (13.3–15.2)], cervical pain [16.3 (14.9–17.9)], arthritis [6.3 (5.8–6.8)] and migraine headache [6.9 (6.0–8.0)] (all p < 0.001).
Table 2.
Demographic and clinical characteristics of study subjects*
| Characteristic | FMS patients (n = 33,176) | Comparison group (n = 33,176) | OR (95% CI) | p-Value |
|---|---|---|---|---|
| Age, mean (SD), years | 45.5 (10.2) | 45.5 (10.2) | – | N/A |
| Sex | ||||
| Male | 8471 (25.5) | 8471 (25.5) | – | N/A |
| Female | 24,705 (74.5) | 24,705 (74.5) | ||
| Comorbidities | ||||
| Neoplasms | 2569 (7.7) | 1702 (5.1) | 1.6 (1.5–1.7) | < 0.001 |
| Diabetes | 1945 (5.9) | 1296 (3.9) | 1.5 (1.4–1.6) | < 0.001 |
| Mental and mood disorders | ||||
| Anxiety | 1800 (5.4) | 441 (1.3) | 4.3 (3.8–4.7) | < 0.001 |
| Depression | 4073 (12.3) | 926 (2.8) | 4.9 (4.5–5.2) | < 0.001 |
| Tension headache | 385 (1.2) | 23 (0.1) | 16.9 (11.1–25.8) | < 0.001 |
| Migraine | 1478 (4.5) | 222 (0.7) | 6.9 (6.0–8.0) | < 0.001 |
| Diseases of the circulatory system | 7284 (22.0) | 4000 (12.1) | 2.1 (2.0–2.1) | < 0.001 |
| Diseases of the respiratory system | 8696 (26.2) | 3288 (9.9) | 3.2 (3.1–3.4) | < 0.001 |
| Diseases of the digestive system | ||||
| Irritable bowel syndrome | 484 (1.5) | 79 (0.2) | 6.2 (4.9–7.9) | < 0.001 |
| GERD | 1785 (5.4) | 495 (1.5) | 3.8 (3.4–4.2) | < 0.001 |
| Gastritis | 201 (0.6) | 61 (0.2) | 3.3 (2.5–4.4) | < 0.001 |
| Other | 4111 (12.4) | 1424 (4.3) | 3.2 (3.0–3.4) | < 0.001 |
| Any of above | 5371 (16.2) | 1806 (5.4) | 3.4 (3.2–3.5) | < 0.001 |
| Diseases of the musculoskeletal system and connective tissue | ||||
| Back pain | 10,518 (31.7) | 1051 (3.2) | 14.2 (13.3–15.2) | < 0.001 |
| Cervical pain | 6692 (20.2) | 506 (1.5) | 16.3 (14.9–17.9) | < 0.001 |
| Arthritis | 3743 (11.3) | 659 (2.0) | 6.3 (5.8–6.8) | < 0.001 |
| Other body/joint pain | 12,560 (37.9) | 1962 (5.9) | 9.7 (9.2–10.2) | < 0.001 |
| Other | 16,602 (50.0) | 1596 (4.8) | 19.8 (18.8–20.9) | < 0.001 |
| Any of above | 25,718 (77.5) | 4086 (12.3) | 24.6 (23.5–25.6) | < 0.001 |
| Symptoms, signs and ill-defined conditions | ||||
| Fatigue | 2375 (7.2) | 419 (1.3) | 6.0 (5.4–6.7) | < 0.001 |
| Headache | 3254 (9.8) | 365 (1.1) | 9.8 (8.8–10.9) | < 0.001 |
| Chest pain | 2506 (7.6) | 870 (2.6) | 3.0 (2.8–3.3) | < 0.001 |
| Abdominal pain | 3121 (9.4) | 949 (2.9) | 3.5 (3.3–3.8) | < 0.001 |
| Anxiety-related symptoms | 2567 (7.7) | 773 (2.3) | 3.5 (3.2–3.8) | < 0.001 |
| Gastric-related symptoms | 1866 (5.6) | 442 (1.3) | 4.4 (4.0–4.9) | < 0.001 |
| Other | 10,933 (33.0) | 3809 (11.5) | 3.8 (3.6–3.9) | < 0.001 |
| Any of above | 15,656 (47.2) | 5701 (17.2) | 4.3 (4.2–4.5) | < 0.001 |
| Painful neuropathic disorders | ||||
| Diabetic neuropathy | 162 (0.5) | 43 (0.1) | 3.8 (2.7–5.3) | < 0.001 |
| Post-herpetic neuralgia | 51 (0.2) | 9 (0.0) | 5.7 (2.8–11.5) | < 0.001 |
| Back pain with neuropathic involvement | 4421 (13.3) | 512 (1.5) | 9.8 (8.9–10.8) | < 0.001 |
| Neck pain with neuropathic involvement | 2229 (6.7) | 219 (0.7) | 10.8 (9.4–12.5) | < 0.001 |
| Cancer with neuropathic pain | 34 (0.1) | 4 (0.0) | 8.5 (3.0–24.0) | < 0.001 |
| Causalgia | 1087 (3.3) | 33 (0.1) | 34.0 (24.1–48.1) | < 0.001 |
| Phantom limb pain | 2 (0.0) | 0 (0.0) | – | < 0.001 |
| Trigeminal neuralgia | 30 (0.1) | 6 (0.0) | 5.0 (2.1–12.0) | < 0.001 |
| Atypical facial pain | 34 (0.1) | 2 (0.0) | 17.0 (4.1–70.8) | < 0.001 |
| Other painful neuropathies | 1259 (3.8) | 186 (0.6) | 7.0 (6.0–8.2) | < 0.001 |
| Any of above | 7565 (22.8) | 925 (2.8) | 10.3 (9.6–11.0) | < 0.001 |
| Sleep disorders | 1906 (5.7) | 328 (1.0) | 6.1 (5.4–6.9) | < 0.001 |
| Any of above | 31,519 (95.0) | 16,473 (49.7) | 19.3 (18.3–20.4) | < 0.001 |
| Mean (SD) duration of follow-up (days) | 334.6 (70.5) | 305.6 (92.3) | – | < 0.001 |
*Unless otherwise indicated, all values are n (%). FMS, fibromyalgia syndrome; OR, odds ratio; CI, confidence interval; GERD, gastro-oesophageal reflux disease; SD, standard deviation.
Fibromyalgia syndrome patients were nearly four times as likely as patients in the comparison group to have received pain-related medications, including AEDs, benzodiazepines, opioids, antidepressants and muscle relaxants (Table 3). The most commonly received medications among FMS patients were antidepressants [4.7 (4.5, 4.9)], opioids [4.3 (4.1–4.5)] and NSAIDs (including COX-2 inhibitors) [3.3 (3.2–3.5)] (all p < 0.001). Thirty-four per cent of FMS patients received some combination of pain-related medications vs. only 7% in the comparison group (p < 0.001) (Table 4). The most commonly received combinations were antidepressants and opioids [8.9 (8.3–9.5)], AEDs and antidepressants [12.4 (11.0–14.1)], and sedatives/hypnotics and opioids [8.2 (7.3–9.1)] (all p < 0.001).
Table 3.
Number of study subjects receiving pain-related medications*
| Pain-related medication | FMS patients (n = 33,176) | Comparison group (n = 33,176) | OR (95% CI) | p-Value |
|---|---|---|---|---|
| Antiepileptics | 3925 (11.8) | 534 (1.6) | 8.2 (7.5–9.0) | < 0.001 |
| Benzodiazepines | 6669 (20.1) | 1912 (5.8) | 4.1 (3.9–4.3) | < 0.001 |
| Corticosteroids | 4819 (14.5) | 1655 (5.0) | 3.2 (3.1–3.4) | < 0.001 |
| COX-2 inhibitors and other prescription NSAIDs | ||||
| COX-2 inhibitors | 3863 (11.6) | 914 (2.8) | 4.7 (4.3–5.0) | < 0.001 |
| Other NSAIDs | 7479 (22.5) | 3042 (9.2) | 2.9 (2.8–3.0) | < 0.001 |
| Any of above | 9719 (29.3) | 3662 (11.0) | 3.3 (3.2–3.5) | < 0.001 |
| Muscle relaxants | 7422 (22.4) | 1290 (3.9) | 7.1 (6.7–7.6) | < 0.001 |
| Sedatives and hypnotics | 4120 (12.4) | 1069 (3.2) | 4.3 (4.0–4.6) | < 0.001 |
| Opioids | ||||
| Short-acting opioids | 12,300 (37.1) | 4106 (12.4) | 4.2 (4.0–4.3) | < 0.001 |
| Long-acting opioids | 2258 (6.8) | 92 (0.3) | 4.3 (4.1–4.5) | < 0.001 |
| Any of above | 12,541 (37.8) | 4123 (12.4) | 4.3 (4.1–4.5) | < 0.001 |
| Antidepressants | ||||
| TCAs | 3711 (11.2) | 468 (1.4) | 8.8 (8.0–9.7) | < 0.001 |
| MAOs | 7 (0.0) | 4 (0.0) | 1.8 (0.5–6.0) | 0.366 |
| SSRIs | 7450 (22.5) | 2,597 (7.8) | 3.4 (3.3–3.6) | < 0.001 |
| Other antidepressants | 6516 (19.6) | 1,503 (4.5) | 4.7 (4.5–4.9) | < 0.001 |
| Any of above | 12,842 (38.7) | 3935 (11.9) | 4.7 (4.5–4.9) | < 0.001 |
| Antimigraines | ||||
| Triptans | 1881 (5.7) | 438 (1.3) | 4.5 (4.0–5.0) | < 0.001 |
| Other antimigraines | 493 (1.5) | 116 (0.3) | 4.3 (3.5–5.3) | < 0.001 |
| Any of above | 2248 (6.8) | 539 (1.6) | 4.4 (4.0–4.8) | < 0.001 |
| Miscellaneous | 3623 (10.9) | 1513 (4.6) | 2.6 (2.4–2.7) | < 0.001 |
| Any of above | 21,402 (64.5) | 11,243 (33.9) | 3.5 (3.4–3.7) | < 0.001 |
*Unless otherwise indicated, all values are n (%). FMS, fibromyalgia syndrome; OR, odds ratio; CI, confidence interval; COX, cyclo-oxygenase; NSAID, non-steroidal anti-inflammatory drug; TCA, tricyclic antidepressant; MAO, monoamine oxidase; SSRI, selective serotonin reuptake inhibitor.
Table 4.
Number of study subjects receiving selected combinations of pain-related medications*
| Measure | FMS patients (n = 33,176) | Comparison group (n = 33,176) | OR (95% CI) | p-Value |
|---|---|---|---|---|
| Number receiving | ||||
| No combinations | 22,000 (66.3) | 30,932 (93.2) | 0.1 (0.1–0.1) | < 0.001 |
| One combination | 4168 (12.6) | 1474 (4.4) | 3.1 (2.9–3.3) | |
| Two combinations | 2008 (6.1) | 385 (1.2) | 5.5 (4.9–6.1) | |
| Three combinations | 1551 (4.7) | 184 (0.6) | 8.8 (7.5–10.3) | |
| Four combinations | 1106 (3.3) | 86 (0.3) | 13.3 (10.6–16.5) | |
| Five combinations | 705 (2.1) | 58 (0.2) | 12.4 (9.5–16.2) | |
| ≥ 6 combinations | 1638 (4.9) | 57 (0.2) | 30.2 (23.2–39.3) | |
| Number receiving | ||||
| AEDs and antidepressants | 3146 (9.5) | 277 (0.8) | 12.4 (11.0–14.1) | < 0.001 |
| TCAs and sedatives/hypnotics | 880 (2.7) | 68 (0.2) | 13.3 (10.4–17.0) | < 0.001 |
| TCAs and SSRIs | 1406 (4.2) | 106 (0.3) | 13.8 (11.3–16.8) | < 0.001 |
| TCAs and NSAIDs | 1580 (4.8) | 137 (0.4) | 12.1 (10.1–14.4) | < 0.001 |
| Benzodiazepines and sedatives/hypnotics | 2010 (6.1) | 297 (0.9) | 7.1 (6.3–8.1) | < 0.001 |
| AEDs and sedatives/hypnotics | 1216 (3.7) | 75 (0.2) | 16.8 (13.3–21.2) | < 0.001 |
| Antidepressants and opioids | 7937 (23.9) | 1132 (3.4) | 8.9 (8.3–9.5) | < 0.001 |
| Muscle relaxants and sedatives/hypnotics | 1914 (5.8) | 143 (0.4) | 14.1 (11.9–16.8) | < 0.001 |
| Benzodiazepines and NSAIDs | 2507 (7.6) | 408 (1.2) | 6.6 (5.9–7.3) | < 0.001 |
| AEDs and antidepressants and opioids | 2589 (7.8) | 133 (0.4) | 21.0 (17.7–25.0) | < 0.001 |
| Sedatives/hypnotics and opioids | 2971 (9.0) | 395 (1.2) | 8.2 (7.3–9.1) | < 0.001 |
| Corticosteroids and AEDs | 1093 (3.3) | 82 (0.2) | 13.7 (11.0–17.2) | < 0.001 |
| Miscellaneous and AEDs | 879 (2.6) | 68 (0.2) | 13.3 (10.3–17.0) | < 0.001 |
| Miscellaneous and TCAs | 672 (2.0) | 56 (0.2) | 12.2 (9.3–16.1) | < 0.001 |
| Miscellaneous and opioids | 2148 (6.5) | 395 (1.2) | 5.7 (5.2–6.4) | < 0.001 |
| Antimigraine and AEDs | 174 (0.5) | 11 (0.0) | 15.9 (8.6–29.2) | < 0.001 |
| Antimigraine and TCAs | 137 (0.4) | 17 (0.1) | 8.1 (4.9–13.4) | < 0.001 |
| Antimigraine and opioids | 361 (1.1) | 42 (0.1) | 8.7 (6.3–12.0) | < 0.001 |
| Sedatives/hypnotics and AEDs and TCAs | 329 (1.0) | 17 (0.1) | 19.5 (12.0–31.8) | < 0.001 |
*Unless otherwise indicated, all values are n (%). FMS, fibromyalgia syndrome; OR, odds ratio; CI, confidence interval; AEDs, antiepileptic drugs; NSAID, non-steroidal anti-inflammatory drugs; SSRI, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants.
Fibromyalgia syndrome patients also were significantly more likely to have received various non-pain-related medications, including antibiotics [2.6 (2.5–2.6)], ulcer medications [3.5 (3.3–3.6)], prescription cough/cold/allergy medications [2.3 (2.2–2.4)] and dermatologicals [2.3 (2.2–2.4)] (all p < 0.001) (Table 5). Overall use of non-pain-related pharmacotherapy among FMS patients was substantially higher than that among patients in the comparison group. For example, 37% of FMS patients received four or more different types of these medications compared with only 15% among comparison patients.
Table 5.
Number of study subjects receiving non-pain-related medications*
| Medication type | FMS patients (n = 33,176) | Comparison group (n = 33,176) | OR (95% CI) | p-Value |
|---|---|---|---|---|
| Antibiotics | 15,236 (45.9) | 8331 (25.1) | 2.6 (2.5–2.6) | < 0.001 |
| Cough/cold/allergy | 6870 (20.7) | 3431 (10.3) | 2.3 (2.2–2.4) | < 0.001 |
| Ulcer drugs | 7465 (22.5) | 2575 (7.8) | 3.5 (3.3–3.6) | < 0.001 |
| Antihistamines | 4841 (14.6) | 2000 (6.0) | 2.7 (2.5–2.8) | < 0.001 |
| Dermatologicals | 6687 (20.2) | 2191 (6.6) | 2.3 (2.2–2.4) | < 0.001 |
| Miscellaneous anti-infective agents | 3848 (11.6) | 1703 (5.1) | 2.4 (2.3–2.6) | < 0.001 |
| Nasal agents | 4449 (13.4) | 1790 (5.4) | 2.7 (2.6–2.9) | < 0.001 |
| Antiasthmatic and bronchodilators | 4243 (12.8) | 1825 (5.5) | 2.5 (2.4–2.7) | < 0.001 |
| Oestrogens | 4260 (12.8) | 1759 (5.3) | 2.6 (2.5–2.8) | < 0.001 |
| Antihyperlipidemics | 5042 (15.2) | 3252 (9.8) | 1.6 (1.6–1.7) | < 0.001 |
| Ophthalmic agents | 2718 (8.2) | 1566 (4.7) | 1.8 (1.7–1.9) | < 0.001 |
| Antihypertensives | 4839 (14.6) | 3556 (10.7) | 1.4 (1.4–1.5) | < 0.001 |
| Diuretics | 4061 (12.2) | 2173 (6.5) | 2.0 (1.9–2.1) | < 0.001 |
| Antifungals | 2767 (8.3) | 971 (2.9) | 3.0 (2.8–3.3) | < 0.001 |
| Tetracyclines | 2039 (6.1) | 888 (2.7) | 1.9 (1.8–2.1) | < 0.001 |
| Beta blockers | 3430 (10.3) | 1866 (5.6) | 2.1 (2.0–2.3) | < 0.001 |
| Thyroid agents | 3697 (11.1) | 1838 (5.5) | 1.3 (1.2–1.4) | < 0.001 |
| Contraceptives | 2196 (6.6) | 1727 (5.2) | 2.1 (1.9–2.2) | < 0.001 |
| Vaginal products | 1658 (5.0) | 825 (2.5) | 2.8 (2.5–3.1) | < 0.001 |
| Laxatives | 1532 (4.6) | 562 (1.7) | 2.1 (2.0–2.3) | < 0.001 |
| Antivirals | 1711 (5.2) | 826 (2.5) | 3.2 (2.8–3.5) | < 0.001 |
| Mouth/throat/dental agents | 1294 (3.9) | 422 (1.3) | 3.8 (3.4–4.3) | < 0.001 |
| Miscellaneous gastrointestinal agents | 1464 (4.4) | 394 (1.2) | 1.7 (1.5–1.8) | < 0.001 |
| Calcium channel blockers | 1858 (5.6) | 1146 (3.5) | 2.5 (2.3–2.8) | < 0.001 |
| Urinary anti-infectives | 1128 (3.4) | 454 (1.4) | 2.5 (2.2–2.8) | < 0.001 |
| Any of above | 24,714 (74.5) | 17,141 (51.7) | 2.7 (2.6–2.8) | < 0.001 |
| Number receiving | ||||
| None of the above | 8462 (25.5) | 16,035 (48.3) | 0.4 (0.4–0.4) | < 0.001 |
| One of the above | 4097 (12.3) | 5156 (15.5) | 0.8 (0.7–0.8) | |
| Two of the above | 4372 (13.2) | 4096 (12.3) | 1.1 (1.0–1.1) | |
| Three of the above | 3995 (12.0) | 2971 (9.0) | 1.4 (1.3–1.5) | |
| ≥ 4 of the above | 12,250 (36.9) | 4918 (14.8) | 3.4 (3.2–3.5) | |
*Unless otherwise indicated, all values are n (%). FMS, fibromyalgia syndrome; OR, odds ratio; CI, confidence interval.
The mean (SD) number of doctor office visits was fourfold higher among FMS patients over 12 months vs. that in the comparison group – 17.8 (15.0) and 4.3 (7.0) respectively (p < 0.001). They also had twice as many other outpatient visits [1.8 (4.6) vs. 0.9 (3.1)] and four times as many emergency room visits [0.4 (1.5) vs. 0.1 (0.5)] (both p < 0.001) (Table 6).
Table 6.
Use of healthcare services
| FMS patients (n = 33,176) | Comparison group (n = 33,176) | p-Value | |
|---|---|---|---|
| Number of office visits | |||
| 0 | 251 (0.8) | 10,543 (31.8) | < 0.001 |
| 1 | 680 (2.0) | 4907 (14.8) | |
| 2 | 1105 (3.3) | 3934 (11.9) | |
| 3 | 1300 (3.9) | 3122 (9.4) | |
| ≥ 4 | 29,840 (89.9) | 10,670 (32.2) | |
| Mean (SD) | 17.8 (15.0) | 4.3 (7.0) | < 0.001 |
| Median (IQR) | 14 (8–23) | 2 (0–5) | |
| Number of other outpatient visits | |||
| 0 | 20,477 (61.7) | 24,765 (74.6) | < 0.001 |
| 1 | 4149 (12.5) | 3961 (11.9) | |
| 2 | 2468 (7.4) | 1837 (5.5) | |
| 3 | 1594 (4.8) | 957 (2.9) | |
| ≥ 4 | 4488 (13.5) | 1656 (5.0) | |
| Mean (SD) | 1.8 (4.6) | 0.9 (3.1) | < 0.001 |
| Median (IQR) | 0 (0–2) | 0 (0–1) | |
| Number of ED visits | |||
| 0 | 26,656 (80.3) | 30,235 (91.1) | < 0.001 |
| 1 | 4294 (12.9) | 2304 (6.9) | |
| 2 | 1211 (3.7) | 476 (1.4) | |
| 3 | 451 (1.4) | 110 (0.3) | |
| ≥ 4 | 564 (1.7) | 51 (0.2) | |
| Mean (SD) | 0.4 (1.5) | 0.1 (0.5) | < 0.001 |
| Median (IQR) | 0 (0–0) | 0 (0–0) | |
| Number of hospitalizations | |||
| 0 | 30,146 (90.9) | 31,934 (96.3) | < 0.001 |
| 1 | 2431 (7.3) | 1094 (3.3) | |
| 2 | 403 (1.2) | 115 (0.3) | |
| 3 | 104 (0.3) | 21 (0.1) | |
| ≥ 4 | 92 (0.3) | 12 (0.0) | |
| Mean (SD) | 0.1 (0.6) | 0.1 (0.4) | < 0.001 |
| Median (IQR) | 0 (0–0) | 0 (0–0) | |
| Inpatient days | |||
| Mean (SD) | 0.7 (4.7) | 0.2 (2.0) | < 0.001 |
| Median (IQR) | 0 (0–0) | 0 (0–0) | |
*Unless otherwise indicated, all values are n (%). Mean values and medians annualized to account for differential follow-up. FMS, fibromyalgia syndrome; SD, Standard deviation; IQR, interquartile range.
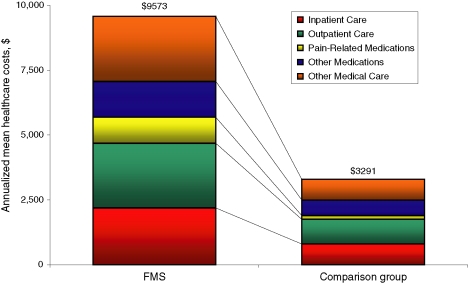
Mean (SD) total healthcare costs over 12 months also were about three times higher among FMS patients vs. patients in the comparison group [$9573 ($20,135) vs. $3291 ($13,643) respectively; p < 0.001] (Figure 1); median healthcare costs were fivefold higher among FMS patients ($4247 vs. 822; p < 0.001). Inpatient care represented about one-quarter of total healthcare costs in both groups; pain-related medications represented 11% and 4.3% of total healthcare costs among FMS patients and comparison group patients respectively.
Figure 1.
Mean annualized healthcare costs among study subjects. All differences (e.g. total healthcare costs, inpatient care, pain-related medications) were statistically significant (p < 0.001 for all)
Discussion
Patients with FMS often were seen for other medical problems, including back pain, cervical pain, painful neuropathic disorders, respiratory infections, hypertension and sleep disorders; they were also commonly seen for mood disorders, including depression and anxiety. Almost one-half of FMS patients had encounters with diagnoses of ‘symptoms, signs and ill-defined conditions’, including headache, abdominal pain, chest pain, fatigue and gastric-related symptoms. Without exception, the prevalence of these medical and psychiatric comorbidities was significantly higher among FMS patients than among age- and sex-matched patients without diagnoses of FMS.
Fibromyalgia syndrome is difficult to diagnose. In fact, some clinicians believe that FMS does not truly exist, and that resolution of FMS symptoms will result from proper treatment of all other, better-established conditions. Unfortunately, it is beyond the scope of our study to ascertain whether or not patients actually had FMS (the database does not contain patients’ medical records), or whether FMS is in fact an actual disease. However, we note that in addition to the higher frequency with which other conditions were noted among FMS patients, all such patients in our study also had at least one encounter for the treatment of FMS in each consecutive year of a 3-year period. This finding is perhaps suggestive that the clinicians of these patients were not satisfied that these conditions fully explained their symptoms.
It is important to note that these differences in the prevalence of various medical and psychiatric comorbidities are not necessarily suggestive of an aetiological link between these other conditions and FMS. Various possible explanations exist for this finding. For one, as FMS is characterized by widespread pain, tenderness and fatigue, patients with this condition may present more frequently to their medical providers than would otherwise be the case. More frequent visits may lead to opportunistic case finding and hence a higher prevalence of diagnosed medical and psychiatric comorbidities. Another possible explanation is the difficulty in making a diagnosis of FMS; patients might receive a variety of other diagnoses as various diseases and conditions are considered as possible explanations for the overlapping symptoms with which patients present.
During the year of study, FMS patients were nearly twice as likely as those in the comparison group to have received pain-related medications, and they were approximately fivefold more likely to have received multiple pain-related medications (primarily, antidepressants and opioids); use of non-pain-related medications was also higher. Given their higher levels of comorbidities and use of medications, it is not surprising that levels of utilization of healthcare services were much greater among FMS patients than patients in the comparison group; compared with the latter, the former averaged four times as many doctor office visits, twice as many other outpatient visits and four times more emergency room visits during the 12-month period of study. Mean healthcare costs were nearly three times higher among FMS patients vs. comparison patients. Our findings therefore indicate that patients with FMS are generally in poorer health and have greater levels of healthcare utilization and cost than patients of similar age and sex without this condition.
In epidemiological studies, which generally require patients to meet ACR criteria for FMS, women have constituted between 86% and 95% of study samples (1,17–19,38). In our study, the number was only 75%. However, the proportion of women in studies such as ours, which used FMS diagnoses on health insurance claims, was 61–81%– consistent with the 75% reported in our study (21,22).
Our study has several limitations. First and most important is our case-selection algorithm. Given the difficulty in making a correct diagnosis of FMS, it is likely that some patients who in fact do not have FMS receive a diagnosis of the disease on at least one occasion. To increase the specificity, we required that all patients in our sample have at least one encounter with a diagnosis of FMS in each of three consecutive years. One consequence of this decision is that we may have selected a cohort of FMS patients with relatively high levels of utilization. In the database, a total of 253,556 patients (6.8% of all patients in the database) had at least one claim with a diagnosis of FMS in any one year of the 3-year period that was used to identify FMS patients; 71,549 (1.9%) had claims with diagnoses of FMS in 2 years of the 3-year period. While it is unlikely that all 253,556 of these patients had FMS, it is entirely probably that our identified sample of 33, 176 patients is an underestimate of the total number of FMS patients in the database during this period of time. Accordingly, the generalizability of our findings to the population of FMS patients as a whole is unknown.
Second, information on medication use in healthcare claims databases is limited to prescription drugs, and specifically to prescriptions that are filled at outpatient (i.e. retail) pharmacies. Thus, to the extent that patients with FMS self-medicate with over-the-counter medications, we would have underestimated their total use of medications. We also do not know whether FMS patients in our study received pain-related medications for the treatment of FMS or for pain associated with other conditions (e.g. arthritis). In addition, as some pain-related medications are also used to treat conditions that are not commonly considered painful (e.g. AEDs in seizure disorders, antidepressants in depression), it would be incorrect to infer that all use of such agents was necessarily for the treatment of FMS pain. Because pharmacy records do not contain information on diagnosis, the extent to which pain-related medications were actually prescribed for the treatment of FMS is unknown. We note further that the database records only whether or not a prescription for a particular medication was filled; not how much, if any, the patient actually took.
Finally, we were limited to examining healthcare costs only. FMS is also associated with disability and may negatively impact productivity. One previous estimate, based on 4699 persons with at least one claim with a diagnosis of FMS employed by a US Fortune 100 manufacturer, estimated that FMS was associated with approximately $1552 (1998 dollars) in indirect costs (21). Another estimate, based on 180 women identified by rheumatologists as having FMS using ACR criteria, estimated the 6-month impact of FMS on indirect costs [including market and non-market (i.e. household) work] to be $4335 (2001 dollars) (20). Accordingly, our estimate likely understates the total economic burden of FMS.
In conclusion, our findings suggest that patients with FMS have strikingly high levels of comorbidities, and high levels of healthcare utilization and costs. Effective new treatments for FMS may lead to reductions in utilization and costs to the extent that these high levels are attributable to the disease.
Funding
Funding for this research was provided by Pfizer Inc., New York, NY.
References
- 1.Wolfe F. The epidemiology of fibromyalgia. J Musculoskelet Pain. 1993;1:137–48. [Google Scholar]
- 2.Campbell SM, et al. Clinical characteristics of fibrositis. I. A ‘blinded’, controlled study of symptoms and tender points. Arthritis Rheum. 1983;26:817–24. doi: 10.1002/art.1780260701. [DOI] [PubMed] [Google Scholar]
- 3.Wolfe F, et al. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia: report of the multicenter criteria committee. Arthritis Rheum. 1990;33:160–72. doi: 10.1002/art.1780330203. [DOI] [PubMed] [Google Scholar]
- 4.Crofford LJ, et al. Pregabalin for the treatment of fibromyalgia syndrome: results of a randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2005;52:1264–73. doi: 10.1002/art.20983. [DOI] [PubMed] [Google Scholar]
- 5.Arnold LM, et al. A randomized, double-blind, placebo-controlled trial of duloxetine in the treatment of women with fibromyalgia with or without major depressive disorder. Pain. 2005;119:5–15. doi: 10.1016/j.pain.2005.06.031. [DOI] [PubMed] [Google Scholar]
- 6.Arnold LM, et al. A double-blind, multicenter trial comparing duloxetine with placebo in the treatment of fibromyalgia patients with or without major depressive disorder. Arthritis Rheum. 2004;50:2974–84. doi: 10.1002/art.20485. [DOI] [PubMed] [Google Scholar]
- 7.Tofferi JK. Treatment of fibromyalgia with cyclobenzaprine: a meta-analysis. Arthritis Rheum. 2004;51:9–13. doi: 10.1002/art.20076. [DOI] [PubMed] [Google Scholar]
- 8.Arnold LM, et al. A randomized, placebo-controlled, double-blind, flexible-dose study of fluoxetine in the treatment of women with fibromyalgia. Am J Med. 2002;112:191–7. doi: 10.1016/s0002-9343(01)01089-0. [DOI] [PubMed] [Google Scholar]
- 9.Goldenberg D, et al. A randomized, double-blind crossover trial of fluoxetine and amitriptyline in the treatment of fibromyalgia. Arthritis Rheum. 1996;39:1852–9. doi: 10.1002/art.1780391111. [DOI] [PubMed] [Google Scholar]
- 10.Carette S, et al. Comparison of amitriptyline, cyclobenzaprine, and placebo in the treatment of fibromyalgia. Arthritis Rheum. 1994;37:32–40. doi: 10.1002/art.1780370106. [DOI] [PubMed] [Google Scholar]
- 11.Santandrea S, et al. A double-blind crossover study of two cyclobenzaprine regimens in primary fibromyalgia syndrome. J Int Med Res. 1993;21:74–80. doi: 10.1177/030006059302100202. [DOI] [PubMed] [Google Scholar]
- 12.Russell IJ, et al. Treatment of primary fibrositis/fibromyalgia syndrome with ibuprofen and alprazolam: a double-blind, placebo-controlled study. Arthritis Rheum. 1991;34:552–60. doi: 10.1002/art.1780340507. [DOI] [PubMed] [Google Scholar]
- 13.Goldenberg DL. Fibromyalgia: treatment problems. J Musculoskelet Pain. 1993;1:71–81. [Google Scholar]
- 14.Dadabhoy D. Therapy insight: Fibromyalgia—a different type of pain needing a different type of treatment. Nat Clin Pract Rheum. 2006;2:364–72. doi: 10.1038/ncprheum0221. [DOI] [PubMed] [Google Scholar]
- 15.Dadabhoy D. Fibromyalgia: progress in diagnosis and treatment. Curr Pain Headache Rep. 2005;9:399–404. doi: 10.1007/s11916-005-0019-2. [DOI] [PubMed] [Google Scholar]
- 16.Rao SG. The management of fibromyalgia. Drugs Today. 2004;40:539–54. doi: 10.1358/dot.2004.40.6.850485. [DOI] [PubMed] [Google Scholar]
- 17.Bombardier CH. Chronic fatigue, chronic fatigue syndrome, and fibromyalgia: disability and health-care use. Med Care. 1996;34:924–30. doi: 10.1097/00005650-199609000-00005. [DOI] [PubMed] [Google Scholar]
- 18.White KP. The London Fibromyalgia Epidemiology Study: comparing the demographic and clinical characteristics in 100 random community cases of fibromyalgia versus controls. J Rheumatol. 1999;26:1577–85. [PubMed] [Google Scholar]
- 19.Wolfe F, et al. A prospective, longitudinal, multicenter study of service utilization and costs in fibromyalgia. Arthritis Rheum. 1997;40:1560–70. doi: 10.1002/art.1780400904. [DOI] [PubMed] [Google Scholar]
- 20.Penrod JR, et al. Health services costs and their determinants in women with fibromyalgia. J Rheumatol. 2004;31:1391–8. [PubMed] [Google Scholar]
- 21.Robinson RL, et al. Economic cost and epidemiological characteristics of patients with fibromyalgia claims. J Rheumatol. 2003;30:1318–25. [PubMed] [Google Scholar]
- 22.Hughes G, et al. The impact of a diagnosis of fibromyalgia on health care resource use by primary care patients in the UK: An observational study based on clinical practice. Arthritis Rheum. 2006;54:177–83. doi: 10.1002/art.21545. [DOI] [PubMed] [Google Scholar]
- 23.Boonen A, et al. Large differences in cost of illness and wellbeing between patients with fibromyalgia, chronic low back pain, or ankylosing spondylosis. Ann Rheum Dis. 2005;64:396–402. doi: 10.1136/ard.2003.019711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cancer Pain Relief. Geneva, Switzerland: World Health Organization; 1996. World Health Organization. [Google Scholar]
- 25.Brant JM. Cancer-related neuropathic pain. Nurse Pract Forum. 1998;9:154–62. [PubMed] [Google Scholar]
- 26.Kanner R. Diagnosis and management of neuropathic pain in patients with cancer. Cancer Invest. 2001;19:324–33. doi: 10.1081/cnv-100102559. [DOI] [PubMed] [Google Scholar]
- 27.George RM. The management of neuropathic pain in cancer: Clinical guidelines for the use of adjuvant analgesics. Indian J Cancer. 2000;37:4–9. [PubMed] [Google Scholar]
- 28.Allen RR. Neuropathic pain in the cancer patient. Neuropathic Pain Syndr. 1998;16:869–87. doi: 10.1016/s0733-8619(05)70102-x. [DOI] [PubMed] [Google Scholar]
- 29.Katz N. Neuropathic pain in cancer and AIDS. Clin J Pain. 2000;16:S41–8. doi: 10.1097/00002508-200006001-00008. [DOI] [PubMed] [Google Scholar]
- 30.Arner S. Lack of analgesic effect of opioids on neuropathic and idiopathic forms of pain. Pain. 1988;33:11–23. doi: 10.1016/0304-3959(88)90198-4. [DOI] [PubMed] [Google Scholar]
- 31.Cohen KL. Efficacy and safety of nonsteroidal anti-inflammatory drugs in the therapy of diabetic neuropathy. Arch Intern Med. 1987;147:1442–4. [PubMed] [Google Scholar]
- 32.Ripamonti C. Strategies for the treatment of cancer pain in the new millennium. Drugs. 2001;61:955–77. doi: 10.2165/00003495-200161070-00005. [DOI] [PubMed] [Google Scholar]
- 33.Farrar JT. Neuropathic cancer pain: the role of adjuvant analgesics. Oncology. 2001;15:1435–45. [PubMed] [Google Scholar]
- 34.Oster G, et al. Use of potentially inappropriate pain-related medications in older adults with painful neuropathic disorders. Am J Geriatr Pharmacother. 2004;2:163–70. doi: 10.1016/j.amjopharm.2004.09.005. [DOI] [PubMed] [Google Scholar]
- 35.Berger A. Clinical characteristics and economic costs of patients with painful neuropathic disorders. J Pain. 2004;5:143–9. doi: 10.1016/j.jpain.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 36.Berger A. Use of antiepileptics and tricyclic antidepressants in cancer patients with neuropathic pain. Eur J Cancer Care. 2006;15:138–45. doi: 10.1111/j.1365-2354.2005.00624.x. [DOI] [PubMed] [Google Scholar]
- 37.Release 9.1. Cary, NC: SAS Institute Inc.; 2004. SAS® Proprietary Software. [Google Scholar]
- 38.Oliver K. Effects of social support and education on health care costs for patients with fibromyalgia. J Rheumatol. 2001;28:2711–9. [PubMed] [Google Scholar]