Abstract
The aim of this study was to explore bactericidal activity of total and free serum simulated concentrations after the oral administration of cefditoren (400 mg, twice daily [bid]) versus the oral administration of amoxicillin-clavulanic acid extended release formulation (2,000/125 mg bid) against Haemophilus influenzae. A computerized pharmacodynamic simulation was performed, and colony counts and β-lactamase activity were determined over 48 h. Three strains were used: ampicillin-susceptible, β-lactamase-negative ampicillin-resistant (BLNAR) (also resistant to amoxicillin-clavulanic acid) and β-lactamase-positive amoxicillin-clavulanic acid-resistant (BLPACR) strains, with cefditoren MICs of ≤0.12 μg/ml and amoxicillin-clavulanic acid MICs of 2, 8, and 8 μg/ml, respectively. Against the ampicillin-susceptible and BLNAR strains, bactericidal activity (≥3 log10 reduction) was obtained from 6 h on with either total and free cefditoren or amoxicillin-clavulanic acid. Against the BLPACR strain, free cefditoren showed bactericidal activity from 8 h on. In amoxicillin-clavulanic acid simulations the increase in colony counts from 4 h on occurred in parallel with the increase in β-lactamase activity for the BLPACR strain. Since both BLNAR and BLPACR strains exhibited the same MIC, this was due to the significantly lower (P ≤ 0.012) amoxicillin concentrations from 4 h on in simulations with β-lactamase positive versus negative strains, thus decreasing the time above MIC (T>MIC). From a pharmacodynamic point of view, the theoretical amoxicillin T>MIC against strains with elevated ampicillin/amoxicillin-clavulanic acid MICs should be considered with caution since the presence of β-lactamase inactivates the antibiotic, thus rendering inaccurate theoretical calculations. The experimental bactericidal activity of cefditoren is maintained over the dosing interval regardless of the presence of a mutation in the ftsI gene or β-lactamase production.
The introduction of the Haemophilus influenzae b vaccine has decreased the incidence in invasive disease, but H. influenzae remains a key respiratory pathogen in humans. Changes in H. influenzae ecology arise together with changes in resistance phenotypes: the β-lactamase production phenotype, which has been related to antibiotic consumption (14); the β-lactamase-negative ampicillin-resistant phenotype (BLNAR), which has appeared and become more prevalent worldwide in the last decade (15, 17, 32, 35); and the β-lactamase positive amoxicillin-clavulanic acid-resistant (BLPACR) phenotype, which is increasingly important in some parts of the world such as Japan (15). The latter two phenotypes are also increasingly important in Spain (11). Resistance to amoxicillin and amoxicillin-clavulanic acid in BLNAR and BLPACR strains is associated with mutations in the ftsI gene causing alterations in the amino acid sequences of PBP3 (25), with similar β-lactamase types in BLPACR and classical ampicillin-resistant β-lactamase-positive isolates (25).
The dissemination and emergence of BLNAR and BLPACR phenotypes has led to questions about the therapeutic activity of old and new β-lactams. Clinical trials will probably not include enough number of patients with these strains, precluding correlations of these phenotypes with therapeutic activity. Experimental pharmacodynamic simulations mimicking physiological concentrations of antimicrobials can be used as alternative approaches to explore the bactericidal activity of different compounds against these types of isolates.
The aim of the present study was to explore the bactericidal activity of simulated serum concentrations of cefditoren versus amoxicillin-clavulanic acid against two H. influenzae strains exhibiting an N526K mutation in the ftsI gene (one BLNAR and one BLPACR) controlled with an ampicillin-susceptible strain. Free-drug concentrations have been considered microbiologically active in vitro (7). Since the antibiotics used in the present study exhibited different levels of protein binding, total and free serum concentrations were simulated to explore the theoretical influence of protein binding on the above-mentioned bactericidal activity, because there are some controversial data on the influence of protein binding on antibacterial activity (4, 22, 34).
MATERIALS AND METHODS
Strains.
Three H. influenzae isolates were used throughout the present study. Isolate 1 was ampicillin susceptible (no mutations in the ftsI gene and no β-lactamase TEM-1 producer), isolate 2 exhibited N526K mutation in the ftsI gene and was TEM-1 negative (BLNAR), and isolate 3 exhibited N526K mutation in the ftsI gene and was TEM-1 positive (BLPACR). Mutations in ftsI and TEM-1 genes were determined by PCR amplification and direct sequencing (8, 31).
Prior to and after the in vitro simulation process, the MICs of cefditoren and amoxicillin-clavulanic acid were determined three times by the microdilution method according to Clinical and Laboratory Standards Institute recommendations (28). Modal values were considered.
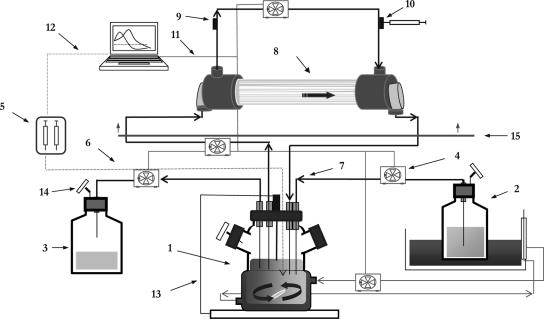
In vitro kinetic model (Fig. 1).
FIG. 1.
Diagram of the in vitro computerized device. 1, Central compartment (spinner flask, tubing, and lumen of capillary); 2, fresh broth reservoir; 3, elimination; 4, peristaltic pumps; 5, syringe pump; 6, PTFE tubing; 7, silicone tubing; 8, infection compartment (extracapillary space); 9, inoculation port; 10, sample port, 11, RS-232 connection; 12, GSIOC connection; 13, temperature probe; 14, air filter; 15, incubator.
A previously described two-compartment dynamic model exposing bacteria to changing study drug concentrations and avoiding the dilution of the bacterial inoculum together with the drug was adapted to simulate antimicrobial combinations of agents with different levels of clearance from the system (3, 33). The central compartment consists of a spinner flask, the lumina of the capillaries within the dialyzer (FX50 class; Fresenius Medical Care S.A., Barcelona, Spain), and the tubing in between. The infection site was represented by the extracapillary space of the dialyser unit combined with the intradialyser circulating tubing. The high surface area/volume ratio of the dialysis unit (>200 cm2/ml) yields a rapid equilibration of the concentration of the antimicrobial agent between the two compartments.
Before each experiment the central compartment was filled with medium Mueller-Hinton broth supplemented with 15 μg of NAD/ml, 15 μg of hemin/ml, and 5 mg/ml of yeast extract (Haemophilus test medium; Difco laboratories, Detroit, MI). The mono-exponential decay of cefditoren or clavulanic acid concentrations in the central compartment was achieved by a continuous dilution-elimination process using computerized peristaltic pumps (Masterflex; Cole-Parmer Instrument Co., Chicago, IL) at rates of 3.4 or 5.2 ml/min, respectively, to simulate their respective half-lives in human serum. Due to the sustained release of amoxicillin from the amoxicillin-clavulanic acid formulation, amoxicillin was supplemented at a rate of 3.6 ml/min to compensate for the excess of concentrations eliminated from the central compartment. Flow rates were adjusted to 4 ml/min to simulate the terminal phase of amoxicillin (from 6 h to 12 h) in the central compartment. Flow rates set in the peristaltic pumps were controlled by using Win Lin software v.2 (Masterflex).
Control drug-free simulations were performed by using a fixed rate in peristaltic pumps of 1 ml/min. Both compartments were maintained at 37°C during the simulation process.
Kinetic simulations.
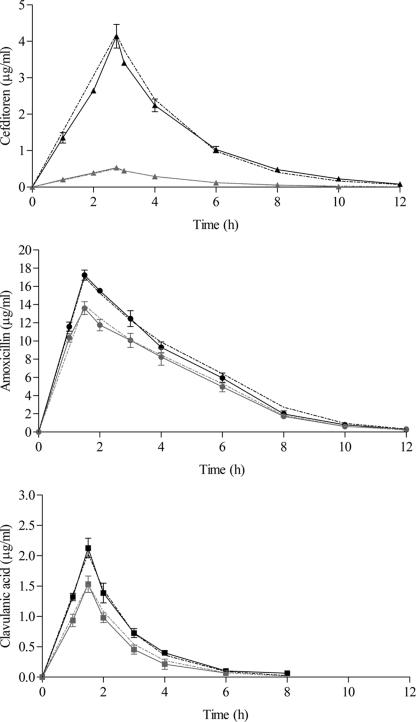
Free and total drug concentrations in serum of cefditoren and amoxicillin-clavulanic acid, after an oral twice-daily (bid) regimen of 400 mg of cefditoren-pivoxil (26, 27) and 2,000 and 125 mg of amoxicillin-clavulanic acid sustained-release formulation (19), were simulated over 48 h. Figure 2 shows target concentration-time curves. The target pharmacokinetic parameters of cefditoren were as follows: Cmax = 4.20 μg/ml (total drug), Tmax = 2.75 h, and t1/2 = 1.55 h (26, 27). Assuming a protein binding of 88% (36), the target cefditoren free Cmax was 0.5 μg/ml. The target parameters of amoxicillin-clavulanic acid were as follows: total Cmax = 17 μg/ml, free Cmax = 13.9 μg/ml (protein binding of 18%), Tmax = 1.5 h, and t1/2 = 1.3 h for amoxicillin and total Cmax = 2 μg/ml, free Cmax = 1.5 μg/ml (protein binding of 25%), Tmax = 1.5 h, and t1/2 = 1 h for clavulanic acid (19).
FIG. 2.
Target (dotted lines) and experimental (solid lines) profiles in β-lactamase-negative simulations of total (black) and free (gray) concentrations in a 400-mg cefditoren dose simulation and 2,000 mg of amoxicillin and 125 mg of clavulanic acid from the sustained release simulation.
Measurement of antibacterial effect.
Bacterial suspensions in Haemophilus test medium from an overnight culture in chocolate agar were allowed to grow to a density of 108 CFU/ml, as measured by a UV-spectrophotometer (Hitachi U-1100). A portion (60 ml) of this inoculum was introduced into the peripheral compartment. Samples (0.5 ml) from the peripheral compartment were collected at 0, 1, 2, 3, 4, 6, 8, 10, 12, 24, 25, 26, 27, 28, 30, 32, 34, 36, and 48 h and serially diluted in 0.9% of cold sodium chloride. At least four dilutions of each sample were spread onto chocolate agar, followed by incubation at 37°C, and colonies were counted after 24 h. The limit of detection was 50 CFU/ml. Each experiment was performed in triplicate.
Pharmacokinetic analysis.
For the measurement of simulated antimicrobial concentrations, additional aliquots (0.5 ml) were drawn from the peripheral compartment at 0, 1, 2, 3, 4, 6, 8, 10, 12, 24, 25, 26, 27, 28, 30, 32, 34, 36, and 48 h and at the time corresponding to the target Tmax of each antimicrobial in simulations with β-lactamase-positive and -negative strains. All samples were stored at −50°C until use. Concentrations were determined by bioassay using Morganella morganii ATCC 8076H as an indicator organism for cefditoren (lineal concentrations from 0.03 to 4 μg/ml; limit of detection = 0.015 μg/ml), Micrococcus luteus ATCC 9341 for amoxicillin, and Klebsiella pneumoniae NCTC 11228 for clavulanic acid concentrations (1). Standards and dilutions, if required, were prepared in the same broth used in the pharmacokinetic simulation and were added to wells in the plates with an even lawn of the indicator organism. Plates were incubated for 18 to 24 h at 37°C. The intraday and interday coefficients of variation were <1% for cefditoren at a concentration of 0.75 μg/ml, ≤3.2% for amoxicillin at a concentration of 0.3 μg/ml, and <2.6% for clavulanic acid at a concentration of 0.75 μg/ml.
Antimicrobial concentrations were analyzed by a noncompartmental approach (model 200) using WinNonlin professional program version 5.2 (Pharsight, Mountain View, CA). Cmax and Tmax values were obtained directly from the observed data. The area under the concentration-time curve from 0 to 24 h (AUC0-24) was calculated by the linear-log trapezoidal rule.
Measurement of β-lactamase activity.
β-Lactamase activity was measured at each sampling time for the BLPACR strain and at time zero and 24 h for the β-lactamase-nonproducing strains by using a modification of a previously described method (24, 30). In brief, 0.025 ml of a 500-μg/ml solution of nitrocefin was added to 225-μl portions of samples collected at sampling times for colony counting, followed by incubation for 30 min at 37°C. Afterward, 0.5 ml of 0.05 M phosphate buffer was added, and the absorbance at 486 nm (A486) was spectrophotometrically read, using broth without inoculum as the baseline absorbance.
Statistical analysis.
Differences in log10 colony counts at each sampling time with respect to initial inocula were calculated. Differences in log10 reductions between antibiotics at dosing times (12, 24, 36, and 48 h) and at 6 h for each strain were determined by the Student t test. Since experiments were performed in triplicate, P ≤ 0.012 was considered statistically significant after alpha-adjustment.
RESULTS
The pre- and postsimulation MICs (μg/ml) were identical for cefditoren and amoxicillin-clavulanic acid: 0.015 and 2 for the ampicillin-susceptible strain, 0.12 and 8 for the BLNAR strain, and 0.06 and 8 for the BLPACR strain, respectively.
Figure 2 shows experimental concentration-time curves determined in simulations with β-lactamase-negative strains. Similar experimental versus target concentrations were obtained. Table 1 shows the pharmacokinetic parameters experimentally obtained in β-lactamase-negative simulations and time above MIC (T>MIC) values. The experimental pharmacokinetic parameter values were similar to the target values described above in Materials and Methods.
TABLE 1.
Experimentally obtained pharmacokinetic parameters in β-lactamase-negative simulations and T>MICs
| Pharmacokinetic parameter | Cefditoren
|
Amoxicillin
|
Clavulanic acid
|
|||
|---|---|---|---|---|---|---|
| Total | Free | Total | Free | Total | Free | |
| Mean Cmax (μg/ml) ± SD | 4.1 ± 0.3 | 0.5 ± 0.0 | 17.2 ± 0.6 | 13.6 ± 0.7 | 2.1 ± 0.2 | 1.5 ± 0.1 |
| Mean Tmax (h) ± SD | 2.8 ± 0.0 | 2.8 ± 0.0 | 1.5 ± 0.0 | 1.5 ± 0.0 | 1.5 ± 0.0 | 1.5 ± 0.0 |
| Mean t1/2 (h) ± SD | 1.6 ± 0.05 | 1.5 ± 0.1 | 1.3 ± 0.1 | 1.4 ± 0.1 | 1.0 ± 0.1 | 1.0 ± 0.1 |
| Mean AUC0-24 (μg/ml·h) ± SD | 14.8 ± 0.3 | 1.9 ± 0.3 | 73.1 ± 1.2 | 60.7 ± 3.3 | 4.7 ± 0.2 | 2.9 ± 0.1 |
| T>MIC indexa | ||||||
| Ampicillin susceptible | 100 | 100 | 65.4 | 63.3 | ||
| BLNAR | 99.3 | 44.1 | 34.6 | 28.3 | ||
| BLPACR | 100 | 62.5 | 34.6 | 28.3 | ||
Expressed as the percent dosing interval. The value was determined on an amoxicillin basis in the case of the amoxicillin-clavulanic MIC.
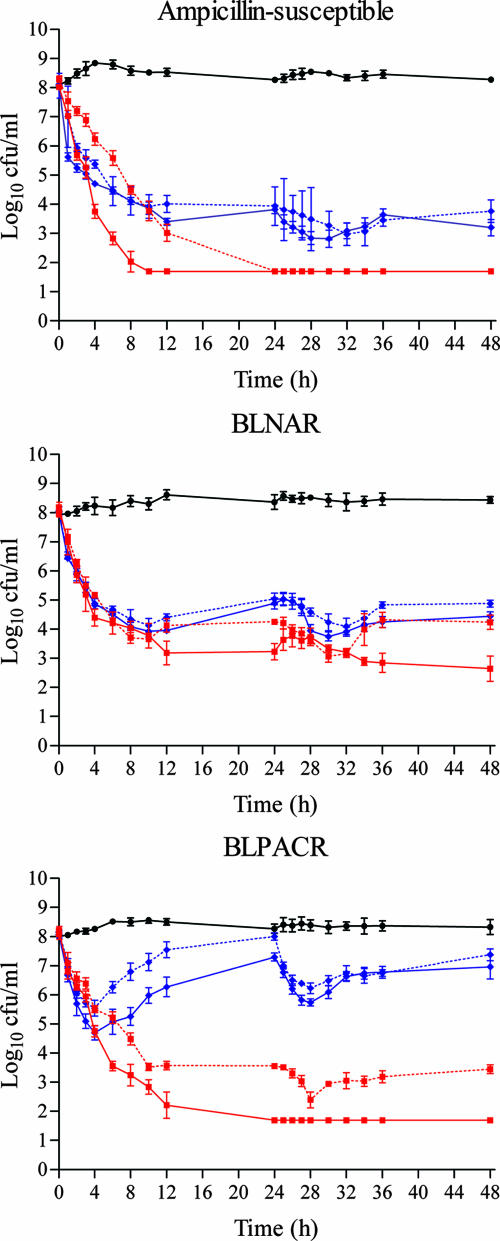
Figure 3 shows the colony counts over time in antibiotic-free simulations and those carried out with total and free concentrations of both study drugs.
FIG. 3.
Colony counts over 48 h in antibiotic-free simulations (black), simulations with total cefditoren (red solid line), free cefditoren (red dotted line), total amoxicillin-clavulanic acid (blue solid line), and free amoxicillin-clavulanic acid (blue dotted line). The regimens simulated were cefditoren (400 mg bid) and amoxicillin-clavulanic acid (2,000/125 mg bid) sustained release.
Table 2 shows initial inoculum reduction (log10 colony counts at time zero − log10 colony counts at each sampling time) in simulations with total and free cefditoren and amoxicillin-clavulanic acid concentrations against the three study strains.
TABLE 2.
Log10 reductions in simulations with total and free cefditoren and amoxicillin-clavulanic acid concentrations
| Phenotype and antibiotic(s)a | Mean log10 reductions (log10 colony counts at time zero − log10 colony counts) ± SD at each sampling time
|
|||||||
|---|---|---|---|---|---|---|---|---|
| 6 h | 8 h | 12 h | 24 h | 28 h | 30 h | 36 h | 48 h | |
| Ampicillin susceptible | ||||||||
| Total CDN | 5.2 ± 0.3 | 6.0 ± 0.4 | >6.3 | >6.3 | >6.3 | >6.3 | >6.3 | >6.3 |
| Total AMC | 3.6 ± 0.1 | 4.0 ± 0.0 | 4.7 ± 0.2 | 4.3 ± 0.1 | 5.3 ± 0.2 | 5.3 ± 0.2 | 4.5 ± 0.1 | 4.9 ± 0.2 |
| Free CDN | 2.7 ± 0.2 | 3.8 ± 0.1 | 5.3 ± 0.3 | >6.3 | >6.3 | >6.3 | >6.3 | >6.3 |
| Free AMC | 3.6 ± 0.4 | 4.0 ± 0.3 | 4.1 ± 0.1 | 4.1 ± 0.2 | 4.6 ± 0.3 | 4.8 ± 0.0 | 4.6 ± 0.2 | 4.3 ± 0.2 |
| BLNAR | ||||||||
| Total CDN | 4.0 ± 0.5 | 4.2 ± 0.4 | 5.0 ± 0.5 | 5.0 ± 0.4 | 4.5 ± 0.5 | 4.9 ± 0.1 | 5.3 ± 0.5 | 5.5 ± 0.6 |
| Total AMC | 3.5 ± 0.2 | 3.9 ± 0.1 | 4.0 ± 0.1 | 3.1 ± 0.3 | 4.1 ± 0.3 | 4.3 ± 0.2 | 3.7 ± 0.2 | 3.5 ± 0.1 |
| Free CDN | 3.7 ± 0.1 | 4.3 ± 0.3 | 3.8 ± 0.2 | 3.7 ± 0.0 | 4.3 ± 0.2 | 4.9 ± 0.3 | 3.6 ± 0.3 | 3.7 ± 0.3 |
| Free AMC | 3.4 ± 0.3 | 3.7 ± 0.5 | 3.7 ± 0.3 | 3.0 ± 0.3 | 3.5 ± 0.3 | 3.8 ± 0.4 | 3.2 ± 0.3 | 3.2 ± 0.3 |
| BLPACR | ||||||||
| Total CDN | 4.7 ± 0.2 | 5.0 ± 0.3 | 6.0 ± 0.5 | >6.3 | >6.3 | >6.3 | >6.3 | >6.3 |
| Total AMC | 2.9 ± 0.5 | 2.8 ± 0.4 | 1.8 ± 0.5 | 0.8 ± 0.1 | 2.3 ± 0.2 | 2.0 ± 0.2 | 1.3 ± 0.3 | 1.1 ± 0.5 |
| Free CDN | 2.9 ± 0.3 | 3.6 ± 0.0 | 4.5 ± 0.3 | 4.5 ± 0.2 | 5.7 ± 0.4 | 5.1 ± 0.2 | 5.0 ± 0.4 | 4.6 ± 0.1 |
| Free AMC | 1.9 ± 0.3 | 1.4 ± 0.1 | 0.6 ± 0.2 | 0.1 ± 0.2 | 1.9 ± 0.1 | 1.7 ± 0.0 | 1.4 ± 0.2 | 0.8 ± 0.4 |
CDN, cefditoren; AMC, amoxicillin-clavulanic acid.
Against the ampicillin-susceptible strain, a bactericidal activity (≥3 log10 reduction) was obtained from 6 h onward with either total or free cefditoren or amoxicillin-clavulanic acid. However, significantly higher initial inocula reductions were obtained with total and free cefditoren (>6.3 log10) at 12, 24, 36, and 48 h (P < 0.001).
Against the BLNAR strain, bactericidal activity (≥3 log10 reduction) was obtained from 6 h on with both study drugs in total and free simulations. Significantly (P < 0.001) higher bactericidal activity was observed with cefditoren total drug—but not with free drug—at 24, 36, and 48 h.
Against the BLPACR strain, free cefditoren showed bactericidal activity from 8 h on. This activity was significantly (P < 0.001) better than free amoxicillin-clavulanic acid, which achieved only small reductions of <2 log10.
When exploring activity at 6 h (early bactericidal activity; 50% of the first dosing interval), no significant differences were found between free cefditoren and amoxicillin-clavulanic acid against the ampicillin-susceptible strain (P = 0.02) and the BLNAR isolate (P = 0.14), but differences were significant against the BLPACR strain (P = 0.011), favoring cefditoren.
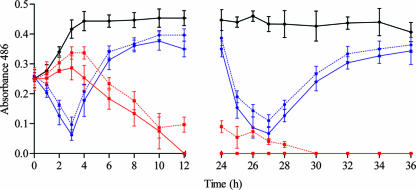
No β-lactamase activity was observed for the ampicillin-susceptible and the BLNAR strains both at 0 h and at 24 h. Figure 4 shows the β-lactamase activity (absorbance units) of the BLPACR strain in antibiotic-free simulations and in simulations with total and free concentrations of study drugs. The β-lactamase activity in antibiotic-free simulations was >0.4 from 3 h on. The β-lactamase activity in cefditoren simulations decreased to nondetectable values from 12 h on in total drug simulations and from 27 h on in free-drug simulations. The β-lactamase activity in total and free amoxicillin-clavulanic acid simulations decreased within the first 4 h after dosing but increased to values of 0.4 absorbance units at 12, 24, 36, and 48 h, values similar to those measured in antibiotic-free simulations. Colony counts for the BLPACR strain (Fig. 3) in amoxicillin-clavulanic acid simulations increased in parallel with the increase in β-lactamase activity from 4 h on (Fig. 4).
FIG. 4.
β-Lactamase activity (absorbance units) of the BLPACR in simulations with total cefditoren (red line), free cefditoren (dotted red line), total amoxicillin-clavulanic acid (blue line), and free amoxicillin-clavulanic acid (dotted blue line) versus antibiotic-free simulations (black line).
The increase in colony counts for the BLPACR strain but not with the BLNAR strain, both exhibiting the same experimental T>MIC (when calculated with concentrations determined in simulations with β-lactamase-negative strains; Table 1), was due to the significantly lower (P ≤ 0.012) amoxicillin concentrations from 4 h on in total drug or 2 h on in free drug simulations when we compared simulations with the β-lactamase-positive versus β-lactamase-negative strains (Table 3).
TABLE 3.
Amoxicillin concentrations, pharmacokinetic parameters, and T>MICs in β-lactamase-positive versus β-lactamase-negative simulationsa
| Pharmacokinetic parameter | Amoxicillin
|
Clavulanic acid
|
||||||
|---|---|---|---|---|---|---|---|---|
| Total
|
Free
|
Total
|
Free
|
|||||
| β pos | β neg | β pos | β neg | β pos | β neg | β pos | β neg | |
| Mean Concn (μg/ml) ± SD at time (h) | ||||||||
| 1 | 11.6 ± 0.6 | 11.6 ± 0.5 | 9.9 ± 2.2 | 10.4 ± 0.5 | 1.2 ± 0.0 | 1.3 ± 0.1 | 0.9 ± 0.1 | 0.9 ± 0.1 |
| 1.5 | 17.4 ± 0.8 | 17.2 ± 0.6 | 13.3 ± 0.9 | 13.6 ± 0.7 | 2.1 ± 0.1 | 2.1 ± 0.2 | 1.4 ± 0.2 | 1.5 ± 0.1 |
| 2 | 14.1 ± 0.5 | 15.5 ± 0.2 | 7.8 ± 0.9* | 11.8 ± 0.6 | 1.5 ± 0.2 | 1.4 ± 0.2 | 1.0 ± 0.1 | 1.0 ± 0.1 |
| 3 | 10.5 ± 0.1 | 12.5 ± 0.9 | 2.9 ± 0.0* | 10.1 ± 0.8 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.4 ± 0.0 | 0.5 ± 0.1 |
| 4 | 5.5 ± 0.7* | 9.3 ± 0.6 | 0.8 ± 0.2* | 8.2 ± 0.9 | 0.4 ± 0.0 | 0.4 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.1 |
| 6 | 2.1 ± 0.2* | 5.9 ± 0.5 | <0.03 | 5.0 ± 0.5 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 8 | <0.03 | 2.0 ± 0.3 | <0.03 | 1.7 ± 0.3 | 0.1 ± 0.0 | 0.1 ± 0.0 | <0.03 | <0.03 |
| 10 | <0.03 | 0.8 ± 0.0 | <0.03 | 0.6 ± 0.2 | <0.03 | <0.03 | <0.03 | <0.03 |
| 12 | <0.03 | 0.3 ± 0.1 | <0.03 | 0.3 ± 0.1 | <0.03 | <0.03 | <0.03 | <0.03 |
| Mean Cmax (μg/ml) ± SD | 17.4 ± 0.6 | 17.2 ± 0.6 | 13.3 ± 0.9 | 13.6 ± 0.7 | 2.1 ± 0.0 | 2.1 ± 0.2 | 1.4 ± 0.1 | 1.5 ± 0.1 |
| Mean AUC0-24 (μg/ml·h) ± SD | 48.7 ± 0.2* | 73.1 ± 1.2 | 22.5 ± 2.4* | 60.7 ± 3.3 | 4.5 ± 0.0 | 4.7 ± 0.0 | 2.8 ± 0.2 | 2.9 ± 0.1 |
| Mean T>MIC (% dosing interval)b | 23.3 | 34.6 | 10.4 | 28.3 | ||||
β pos, β-lactamase-positive simulations; β neg, β-lactamase-negative simulations. *, P ≤ 0.012 versus β-negative simulations.
For the BLPACR strain.
The presence of β-lactamase activity led to significantly lower amoxicillin AUC0-24 in simulations with β-lactamase-positive versus β-lactamase-negative strains (Table 3) and a pronounced decrease in T>MIC (from 34.6 to 23.3% in total simulations and from 28.3 to 10.4% in free simulations) for the BLPACR strain. No differences were found in cefditoren concentrations over time measured in simulations with β-lactamase-positive versus β-lactamase-negative strains.
DISCUSSION
In Spain, while β-lactamase production in H. influenzae was found at a constant frequency of ca. 20 to 25% in successive nationwide surveillance studies (SAUCE Program) carried out in 1996 to 1997 (13), 1998 and 1999 (23), and 2001 and 2002 (29), the BLNAR phenotype prevalences were 9 and 4.5% in the 1998-1999 and 2001-2002 surveillance studies, respectively, and the BLPACR phenotype was not detected in the first surveillance study and was anecdotal (0.1%) in the other two. Similar BLNAR rates have been reported in United States (16) and Europe (18), with this last report suggesting the highest rates in Spain (up to ca. 30%). In Japan isolation rates as high as ca. 40% have been reported (32).
The increase and/or appearance of these new BLNAR and BLPACR phenotypes raises the question of the clinical activity of old and new β-lactams. The resistance of BLNAR and BLPACR phenotypes to previous antibiotics such as amoxicillin with or without clavulanic acid is due to changes in PBP3 (25). In the BLPACR phenotype, in addition to this resistance mechanism, β-lactamase production is present, and the β-lactamase type is primary TEM-1 (25). These phenotypes should also be considered resistant to previous cephalosporins such as cefaclor, cefamandole, cefetamet, cefuroxime, cefonicid, cefprozil, cefpodoxime, or cefdinir (5).
The evaluation of old and new β-lactams should be based on the study of their pharmacodynamic activity. The presence of mutations in the ftsI gene in the BLNAR phenotype and the additional TEM-1 β-lactamase production in the BLPACR phenotype may influence the values of the pharmacokinetic/pharmacodynamic index predicting bactericidal activity, by increasing the MIC and by reducing target site concentrations due to enzymatic degradation by β-lactamases. The T>MIC has been considered the pharmacokinetic/pharmacodynamic index predicting efficacy for β-lactams. Values of ca. 40% of the dosage interval for penicillins and of ca. 50% for cephalosporins have been related to bacterial eradication and subsequent therapeutic efficacy in animal models and human infections (2, 6, 9). In the present study we used a computerized device to explore the bactericidal activity over the dosing interval of simulated serum concentrations obtained after the oral administration of amoxicillin-clavulanic acid (2,000/125 mg) or cefditoren (400 mg) against an ampicillin-susceptible, a BLNAR, and a BLPACR strain. Free drug was tested because the unbound form of an antimicrobial is the active form in vitro and presumably in vivo (7), but since there are some controversial data on the influence of protein binding on the antibacterial activity both in vivo (22) and in vitro (4, 34), experiments were also carried out with total drug to explore the potential maximal and minimal activities (i.e., the range containing the actual activity).
Against the ampicillin-susceptible strain, free cefditoren showed significantly higher bactericidal activity after the second dose than did amoxicillin-clavulanic acid. The T>MIC was >50% for both drugs. This high bactericidal activity of cefditoren is maintained against the two strains harboring the N526K mutation in the ftsI gene (free cefditoren T>MIC of >40%). Surprisingly, in the case of amoxicillin-clavulanic acid the activity of total and free drug against the BLNAR strain was high, with bactericidal activity (>3 log10 reduction) from 6 h onward despite its high MIC (8 μg/ml), a value considered resistant. Total and free amoxicillin T>MIC values were 34.6 and 28.3% of the dosing interval, respectively; values lower than those classically considered for bactericidal activity with penicillins. However, the production of TEM-1 β-lactamase as an additional resistance mechanism in the BLPACR strain rendered amoxicillin-clavulanic acid nonbactericidal regardless of the same MIC (8 μg/ml) for this strain and the BLNAR strain. The reason for this lack of bactericidal activity against the BLPACR strain was that clavulanic acid concentrations were <0.5 μg/ml from 4 h on, with nondetectable values from 8 h on. From 4 h on, the β-lactamase activity of the BLPACR strain began to increase, reaching values similar to those in antibiotic-free simulations from 8 to 12 h. β-Lactamase activity inactivated amoxicillin, producing lower amoxicillin concentrations in simulations with the BLPACR strain. The experimental free/total ratios were very similar over time (range, 0.8 to 0.9) in β-lactamase negative simulations (Table 3). These free/total ratios do not have the same values in β-lactamase-positive simulations (ranging from 0.9 at 1 h to 0.01 at 6 h) due to the influence of the β-lactamase inactivation of amoxicillin. Although the value is equal at 1 h, differences increase over time, resulting in a high difference at 6 h (0.8 versus 0.01). This is due to the β-lactamase action, which logically affects low concentrations (free profile) more than high concentrations (total profile). Due to this, the experimentally measured T>MIC decreased from 34.6% (in β-lactamase negative simulations) to 23% in BLPACR simulations of total concentrations and from 28.3 to 10.4% for free concentrations.
Since both BLNAR and BLPACR strains had the same MIC, calculation of T>MIC using concentrations from phase I trials results in information far from the actual situation because it does not consider the influence of β-lactamase activity, when present, on the T>MIC. This suggests that bid regimens of clavulanic acid may be inadequate against H. influenzae strains exhibiting elevated amoxicillin-clavulanic acid MICs. In a previous study (21) using similar methodology, brief exposure to clavulanic acid seemed not to have affected the bactericidal activity of amoxicillin concentrations (corresponding also to those obtained after the 2,000/125-mg sustained release formulation) against β-lactamase-positive strains. Differences between both studies are due to the lower MICs values of amoxicillin-clavulanic acid for the strains used (0.5 to 0.75 μg/ml) in the previous study; thus, isolates probably did not present mutation in the ftsI gene (12) and were not BLPACR.
It may be argued that, in vivo, the decrease in average amoxicillin concentrations might be less pronounced than the decrease in in vitro systems, although previous in vivo experiments have shown lower ampicillin concentrations when the infection was caused by β-lactamase-positive versus β-lactamase-negative strains (20).
The results of this experimental study point out that, from the pharmacodynamic point of view, the theoretical amoxicillin T>MIC (calculated using pharmacokinetic data from phase I studies) against β-lactamase-positive strains with elevated ampicillin-amoxicillin-clavulanic acid MICs should be viewed with caution since the presence of TEM-1 β-lactamase inactivates the antibiotic, thus rendering inaccurate theoretical calculations. The experimental bactericidal activity of cefditoren is maintained over the dosing interval regardless of the presence of mutation in the ftsI gene or β-lactamase production. Although cefditoren exhibits high protein binding, free concentrations were enough to be active against H. influenzae that, as in the present study, exhibit low cefditoren MICs regardless of the resistance phenotype (10, 12).
Acknowledgments
This study was supported by an unrestricted grant from Tedec-Meiji Farma SA, Madrid, Spain.
We thank M. Gimeno for critical review of the manuscript and J. García-de-Lomas (Instituto Valenciano de Microbiología) for providing the ftsI gene sequenced strains.
Footnotes
Published ahead of print on 30 July 2007.
REFERENCES
- 1.Andrews, J. M. 1999. Microbiological assays, p. 35-44. In D. S. Reeves, R. Wise, J. M. Andrews, and L. O. White (ed.), Clinical antimicrobial assays. Oxford University Press, Oxford, United Kingdom.
- 2.Ball, P., F. Baquero, O. Cars, T. File, J. Garau, K. Klugman, D. E. Low, E. Rubinstein, R. Wise, et al. 2002. Antibiotic therapy of community respiratory tract infections: strategies for optimal outcomes and minimized resistance emergence. J. Antimicrob. Chemother. 49:31-40. [DOI] [PubMed] [Google Scholar]
- 3.Blaser, J. 1985. In-vitro model for simultaneous simulation of the serum kinetics of two drugs with different half-lives. J. Antimicrob. Chemother. 15(Suppl. A):125-130. [DOI] [PubMed] [Google Scholar]
- 4.Cafini, F., L. Aguilar, N. Gonzalez, M. J. Gimenez, M. Torrico, L. Alou, D. Sevillano, P. Vallejo, and J. Prieto. 2007. In vitro effect of the presence of human albumin or human serum on the bactericidal activity of daptomycin against strains with the main resistance phenotypes in gram-positives. J. Antimicrob. Chemother. 59:1185-1189. [DOI] [PubMed] [Google Scholar]
- 5.Clinical and Laboratory Standards Institute. 2005. Performance standards for antimicrobial susceptibility testing; fifteenth informational supplement. CLSI/NCCLS document M100-S15. Clinical and Laboratory Standards Institute, Wayne, PA.
- 6.Craig, W. A. 1998. Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin. Infect. Dis. 26:1-10. [DOI] [PubMed] [Google Scholar]
- 7.Craig, W. A., and C. M. Kunin. 1976. Significance of serum protein and tissue binding of antimicrobial agents. Annu. Rev. Med. 27:287-300. [DOI] [PubMed] [Google Scholar]
- 8.Dabernat, H., C. Delmas, M. Seguy, R. Pelissier, G. Faucon, S. Bennamani, and C. Pasquier. 2002. Diversity of beta-lactam resistance-conferring amino acid substitutions in penicillin-binding protein 3 of Haemophilus influenzae. Antimicrob. Agents Chemother. 46:2208-2218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dagan, R., K. P. Klugman, W. A. Craig, and F. Baquero. 2001. Evidence to support the rationale that bacterial eradication in respiratory tract infection is an important aim of antimicrobial therapy. J. Antimicrob. Chemother. 47:129-140. [DOI] [PubMed] [Google Scholar]
- 10.Fenoll, A., L. Aguilar, O. Robledo, M. J. Gimenez, D. Tarrago, J. J. Granizo, M. Gimeno, and P. Coronel. 2007. Influence of the β-lactam resistance phenotype on the cefuroxime versus cefditoren susceptibility of Streptococcus pneumoniae and Haemophilus influenzae recovered from children with acute otitis media. J. Antimicrob. Chemother. 60:323-327. [DOI] [PubMed] [Google Scholar]
- 11.García-Cobos, S., J. Campos, E. Lázaro, F. Román, E. Cercenado, C. García-Rey, M. Pérez-Vázquez, J. Oteo, and F. de Abajo. 2007. Ampicillin-resistant non-β-lactamase producing Haemophilus influenzae in Spain: recent emergence of clonal isolates with increased resistance to cefotaxime and cefixime. Antimicrob. Agents Chemother. 51:2564-2573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garcia-de-Lomas, J., M. Lerma, L. Cebrian, J. L. Juan-Banon, P. Coronel, M. J. Gimenez, and L. Aguilar. 2007. Influence of Haemophilus influenzae beta-lactamase production and/or ftsI gene mutations on in vitro activity of and susceptibility rates to aminopenicillins and second- and third-generation cephalosporins. Int. J. Antimicrob. Agents 30:190-192. [DOI] [PubMed] [Google Scholar]
- 13.Garcia-Rodriguez, J. A., F. Baquero, J. Garcia de Lomas, L. Aguilar, et al. 1999. Antimicrobial susceptibility of 1,422 Haemophilus influenzae isolates from respiratory tract infections in Spain: the results of a 1-year (1996-97) multicenter surveillance study. Infection 27:265-267. [DOI] [PubMed] [Google Scholar]
- 14.Gómez, J., J. Ruiz-Gómez, J. L. Hernández-Cardona, M. L. Nuñez, M. Canteras, and M. Valdés. 1994. Antibiotic resistance patterns of Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis: a prospective study in Murcia, Spain, 1983-1992. Chemotherapy 40:299-303. [DOI] [PubMed] [Google Scholar]
- 15.Hasegawa, K., N. Chiba, R. Kobayashi, S. Y. Murayama, S. Iwata, K. Sunakawa, and K. Ubukata. 2004. Rapidly increasing prevalence of beta-lactamase-nonproducing, ampicillin-resistant Haemophilus influenzae type b in patients with meningitis. Antimicrob. Agents Chemother. 48:1509-1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hasegawa, K., K. Yamamoto, N. Chiba, R. Kobayashi, K. Nagai, M. R. Jacobs, P. C. Appelbaum, K. Sunakawa, and K. Ubukata. 2003. Diversity of ampicillin-resistance genes in Haemophilus influenzae in Japan and the United States. Microb. Drug Resist. 9:39-46. [DOI] [PubMed] [Google Scholar]
- 17.Jacobs, M. R., S. Bajaksouzian, A. Zilles, G. Lin, G. A. Pankuch, and P. C. Appelbaum. 1999. Susceptibilities of Streptococcus pneumoniae and Haemophilus influenzae to 10 oral antimicrobial agents based on pharmacodynamic parameters: 1997 U.S. surveillance study. Antimicrob. Agents Chemother. 43:1901-1908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jansen, W. T., A. Verel, M. Beitsma, J. Verhoef, and D. Milatovic. 2006. Longitudinal European surveillance study of antibiotic resistance of Haemophilus influenzae. J. Antimicrob. Chemother. 58:873-877. [DOI] [PubMed] [Google Scholar]
- 19.Kaye, C. M., A. Allen, S. Perry, M. McDonagh, M. Davy, K. Storm, N. Bird, and O. Dewit. 2001. The clinical pharmacokinetics of a new pharmacokinetically enhanced formulation of amoxicillin/clavulanate. Clin. Ther. 23:578-584. [DOI] [PubMed] [Google Scholar]
- 20.Lavoie, G. Y., and M. G. Bergeron. 1985. Influence of four modes of administration on penetration of aztreonam, cefuroxime, and ampicillin into interstitial fluid and fibrin clots and on in vivo efficacy against Haemophilus influenzae. Antimicrob. Agents Chemother;. 28:404-412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lowdin, E., O. Cars, and I. Odenholt. 2002. Pharmacodynamics of amoxicillin/clavulanic acid against Haemophilus influenzae in an in vitro kinetic model: a comparison of different dosage regimens including a pharmacokinetically enhanced formulation. Clin. Microbiol. Infect;. 8:646-653. [DOI] [PubMed] [Google Scholar]
- 22.Moellering, R. C., and G. M. Eliopoulos. 2005. Principles of anti-infective therapy, p. 242-253. In G. L. Mandell, J. E. Bennett, and R. Dolin (ed.), Mandell, Douglas, and Bennett principles and practice of infectious diseases, 6th ed. Elsevier Churchill Livingstone, Philadelphia, PA.
- 23.Marco, F., J. Garcia-de-Lomas, C. Garcia-Rey, E. Bouza, L. Aguilar, C. Fernandez-Mazarrasa, et al. 2001. Antimicrobial susceptibilities of 1,730 Haemophilus influenzae respiratory tract isolates in Spain in 1998-1999. Antimicrob. Agents Chemother. 45:3226-3228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Martin, M., L. Aguilar, I. P. Balcabao, M. L. Gomez-Lus, R. Dal-Re, and J. Prieto. 1997. In-vitro pharmacodynamic simulation of clavulanic acid concentrations: effect on Staphylococcus aureus and Haemophilus influenzae beta-lactamase activity. J. Antimicrob. Chemother. 39:290-292. [DOI] [PubMed] [Google Scholar]
- 25.Matic, V., B. Bozdogan, M. R. Jacobs, K. Ubukata, and P. C. Appelbaum. 2003. Contribution of beta-lactamase and PBP amino acid substitutions to amoxicillin/clavulanic acid resistance in beta-lactamase-positive, amoxicillin/clavulanic acid-resistant Haemophilus influenzae. J. Antimicrob. Chemother. 52:1018-1021. [DOI] [PubMed] [Google Scholar]
- 26.Mulford, D., M. Mayer, and G. Witt. 2000. Effect of age and gender on the pharmacokinetics of cefditoren. Abstr. 40th Intersci. Conf. Antimicrob. Agents Chemother. 2000, abstr. 310. American Society for Microbiology, Washington, DC.
- 27.Mulford, D., M. Mayer, and G. Witt. 2000. Effect of renal impairment on the pharmacokinetic of cefditoren. Abstr. 40th Intersci. Conf. Antimicrob. Agents Chemother. 2000, abstr. 311. American Society for Microbiology, Washington, DC.
- 28.National Committee for Clinical Laboratory Standards. 2003. Methods for dilution antimicrobial susceptibility tests for bacteria that growth aerobically; approved standard, 6th ed. NCCLS M7-A6. NCCLS, Wayne, PA.
- 29.Perez-Trallero, E., C. Garcia-de-la-Fuente, C. Garcia-Rey, F. Baquero, L. Aguilar, R. Dal-Re, J. Garcia-de-Lomas, et al. 2005. Geographical and ecological analysis of resistance, coresistance, and coupled resistance to antimicrobials in respiratory pathogenic bacteria in Spain. Antimicrob. Agents Chemother. 49:1965-1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Prieto, J., L. Aguilar, M. J. Gimenez, D. Toro, M. L. Gomez-Lus, R. Dal-Re, and I. P. Balcabao. 1998. In vitro activities of co-amoxiclav at concentrations achieved in human serum against the resistant subpopulation of heteroresistant Staphylococcus aureus: a controlled study with vancomycin. Antimicrob. Agents Chemother. 42:1574-1577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Scriver, S. R., S. L. Walmsley, C. L. Kau, D. J. Hoban, J. Brunton, A. McGeer, T. C. Moore, and E. Witwicki. 1994. Determination of susceptibilities of Canadian isolates of Haemophilus influenzae and characterization of their β-lactamases. Antimicrob. Agents Chemother. 38:1678-1680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Seki, H., Y. Kasahara, K. Ohta, Y. Saikawa, R. Sumita, A. Yachie, S. Fujita, and S. Koizumi. 1999. Increasing prevalence of ampicillin-resistant, non-beta-lactamase-producing strains of Haemophilus influenzae in children in Japan. Chemotherapy 45:15-21. [DOI] [PubMed] [Google Scholar]
- 33.Sevillano, D., A. Calvo, M. J. Gimenez, L. Alou, L. Aguilar, E. Valero, A. Carcas, and J. Prieto. 2004. Bactericidal activity of amoxicillin against non-susceptible Streptococcus pneumoniae in an in vitro pharmacodynamic model simulating the concentrations obtained with the 2000/125 mg sustained-release co-amoxiclav formulation. J. Antimicrob. Chemother. 54:1148-1151. [DOI] [PubMed] [Google Scholar]
- 34.Sevillano, D., M. J. Giménez, L. Alou, L. Aguilar, F. Cafini, M. Torrico, N. González, O. Echeverría, P. Coronel, and J. Prieto. Effects of human albumin and serum on the in vitro bactericidal activity of cefditoren against penicillin resistant Streptococcus pneumoniae. J. Antimicrob. Chemother., in press. [DOI] [PubMed]
- 35.Ubukata, K., Y. Shibasaki, K. Yamamoto, N. Chiba, K. Hasegawa, Y. Takeuchi, K. Sunakawa, M. Inoue, and M. Konno. 2001. Association of amino acid substitutions in penicillin-binding protein 3 with beta-lactam resistance in beta-lactamase-negative ampicillin-resistant Haemophilus influenzae. Antimicrob. Agents Chemother. 45:1693-1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wellington, K., and M. P. Curran. 2004. Cefditoren pivoxil: a review of its use in the treatment of bacterial infections. Drugs 64:2597-2618. [DOI] [PubMed] [Google Scholar]