Abstract
Saccharomyces cerevisiae contains an irregular telomere sequence (TG1–3)n, which differs from the regular repeat (TTAGGG)n found at the telomeres of higher organisms including humans. We have modified the entire 16-nt template region of the S. cerevisiae telomerase RNA gene (TLC1) to produce (TTAGGG)n repeats, the human telomere sequence. Haploid yeast strains with the tlc1-human allele are viable with no growth retardation and express the humanized gene at a level comparable to wild type. Southern hybridization demonstrates that (TTAGGG)n repeats are added onto the yeast chromosome ends in haploid strains with the tlc1-human allele, and sequencing of rescued yeast artificial chromosome ends has verified the addition of human telomeric repeats at the molecular level. These data suggest that the irregularity of the yeast telomere sequence is because of the template sequence of the yeast telomerase RNA. Haploid strains with the tlc1-human allele will provide an important tool for studying the function of telomerase and its regulation by telomere-binding proteins, and these strains will serve as good hosts for human artificial chromosome assembly and propagation.
The successful generation of the first human artificial chromosome (HAC) has renewed hopes of using HACs for gene delivery into human cells (1–3). However, the relatively inefficient production of HACs and the inclusion of an undefined human DNA component in this method leaves many problems to be resolved. Ideally, one would like to use a HAC of defined content that reproducibly gives rise to independent chromosomes in human cells. A HAC of defined content potentially could be constructed by the addition of human elements to yeast artificial chromosomes (YACs) containing human centromeric DNA. Such a HAC then could be propagated in yeast. One difficulty with this approach is the modification of the human telomeres with yeast telomeric DNA, presumably because of the activity of yeast telomerase during propagation (4, 5). As a consequence, such HACs may not give rise to independent chromosomes when introduced into human cells, because yeast telomeric sequences are unlikely to seed mammalian telomeres (6). Maintenance of the human telomeric sequences should provide an important advancement toward the efficient assembly and propagation of HACs in yeast cells.
Telomeres are specialized nucleoprotein complexes found at the ends of eukaryotic chromosomes that ensure the complete replication of the chromosome ends and protect the ends from degradation and fusion with other chromosome ends (7, 8). Telomere DNA sequences of all vertebrates studied share the same regular repeat (TTAGGG)n, whereas the telomere sequence in the budding yeast Saccharomyces cerevisiae is a G-rich irregular sequence (TG1–3)n. Telomeres are synthesized by a ribonucleoprotein, telomerase, first described in the ciliate Tetrahymena (9). Yeast telomerase is composed of protein subunits, including Est2p, the essential catalytic subunit, and an essential RNA that provides the template for the addition of telomeric sequence at the end of chromosomes (10, 11). The RNA component of telomerase now has been cloned from a number of organisms including Tetrahymena, yeast, and humans (12–14). The telomerase RNAs from different species all contain a domain of longer than one full telomeric repeat, which is predicted to serve as the template for telomere addition (11). Outside of this template region, there is very little sequence similarity between the RNAs of different species. A 2-nt substitution within the template domain of the S. cerevisiae telomerase RNA gene (TLC1) was incorporated into telomeric DNA, confirming that this domain serves as the template for telomere addition (13). Three-base (GUG) substitutions within and next to the template domain have been shown to be active in TLC1/tlc1 diploids and to synthesize patterns of mixed wild-type and mutant telomeric repeats into telomeric DNA, consistent with nonprocessive action of the yeast telomerase complex (15). Furthermore, disruption of the TLC1 gene led to progressive telomere shortening, which resulted in a gradual decrease in growth rate and viability (13).
To date, no complete replacement of the template region of any of the telomerase RNA genes has been described. Nor has it been demonstrated that the telomere sequence (TTAGGG)n of higher organisms, including humans, will function as telomeres in yeast, beyond the apparent ability to seed the addition of yeast telomere sequence (16). Here, we report the replacement of the entire 16-bp template domain of the TLC1 gene with a human telomere template sequence. Yeast strains with the humanized telomerase template are capable of adding human telomere sequences to the ends of yeast chromosomes. Such strains are viable, showing no signs of growth impairment. They will be useful for human artificial chromosome assembly and propagation and will provide an important tool for studying the function of both yeast and human telomerases and their regulation by telomere-binding proteins.
MATERIALS AND METHODS
Vector Construction.
The TLC1 gene was amplified by PCR using genomic DNA from S. cerevisiae strain YPH925 and primers TLC1-S, 5′-GATAGGTACCCTATGAAAATGTCAATGGC (23–47) and TLC1-AS, 5′-ATCGTCTAGAGGCATACCTCCGCCTAT (1292–1273), digested with KpnI and XbaI and then cloned into pRS306. The entire insert was sequenced in both directions to verify that there were no PCR errors, and the plasmid was designated pTLC1.
PCR primers TLC1.HUM, 5′-TAATTACCATGGGAAGCCTACCATCCTAACCCTAACCCTAACCAAATGTTACAG (sequence in bold type is complementary to human telomere repeats) and TLC1.HpaI, 5′-TCCAGAGTTAACGATAAGATAGAC were used to alter the template sequence to be complementary to human telomeric repeats. The PCR product was digested with NcoI and HpaI and was used to replace the NcoI-HpaI fragment of pTLC1, to give pTLC1h. The PCR portion of the insert was again sequenced to confirm the presence of the human telomere template and that there were no PCR errors.
Diploid Strain Generation.
S. cerevisiae strain AB1380 (MATa ade2–1 can1–100 lys2–1 trp1 ura3 his5-u) was crossed with strain YPH925 (MATα leu2-Δ1 trp1-Δ63 ura3–52 ade2–101 his3-Δ200 lys2–801 cyh2R kar1-Δ15) and plated onto SD8-His media, to select for diploid colonies. One colony, designated NM1, was verified to be diploid by the analysis of yeast chromosomes with pulsed-field gel electrophoresis (PFGE), as previously described (17).
Two-Step Gene Replacement.
Diploid strain NM1 was transformed with plasmid pTLC1h, linearized at the AflII site, and plated in regeneration agar onto SD8-Ura media. Colonies were picked and purified by serial restreaking, then analyzed by PCR for correct integration of the vector into the TLC1 gene. One integrant, designated C6, was verified to have plasmid pTLC1h correctly integrated into one allele of the yeast TLC1 gene by Southern hybridization. Presence of the URA3 gene was then selected against by plating strain C6 onto SD8-His + 5-fluoroorotic acid (5-FOA, Sigma) media. Accurate recombination to remove the plasmid sequences was verified by Southern hybridization. The rTth thermostable reverse transcription–PCR (RT-PCR) kit (Perkin–Elmer) and primers TLC1hum.F2, 5′-ATGGGAAGCCTACCATCCTAACCCTAACC-3′, and TLC1.R2, 5′-CAGCGCTTAGCACCGTCTGTTTGCA-3′ were used to identify clones that expressed the tlc1-human allele. Tetrad dissection and haploid spore regeneration were kindly performed by Reed B. Wickner, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health (NIH), and Thorsten Allers and Michael Lichten, National Cancer Institute, NIH.
Genotyping.
Cell lysates from each of the haploid strains were genotyped by PCR at the TLC1 locus with primers TLC1yeast.F, 5′-CCTACCATCACCACACCCAC-3′ or TLC1human.F, 5′-CCTACCATCCTAACCCTAACCC-3′, and TLC1.R, 5′-GTTCCCTGACGTTCTTTTTCC-3′. Haploid status was verified by genotyping at the MAT locus as previously described (18).
Northern Analysis.
Approximately 5 μg of total RNA per lane was hybridized in 15 ml ExpressHyb (CLONTECH) at 65°C for DNA probes, or 37°C for oligonucleotide probes and washed as per the manufacturer’s suggestions. Double-stranded DNA probes were labeled by using the Rediprime kit (Amersham) and [α-32P]dCTP. The probe for TLC1 was a 1.3-kb KpnI-SstII fragment of plasmid pTLC1h, which covers the TLC1 gene. The ACT1 probe was a 395-bp fragment of the S. cerevisiae actin gene from strain YPH925, generated by PCR using primers ACT1.S, 5′-AGACACCAAGGTATCATGGTCGGTATGGG-3′ and ACT1.AS, 5′-GAGAAACCAGCGTAAATTGGAACGACGTG-3′. Oligonucleotide probes were labeled by using polynucleotide kinase and [γ-32P]ATP. Allele-specific oligonucleotides for the yeast template and human template sequences were oligo(yT), 5′-CATTTGTGTGTGGGTGTGGTGATGGT-3′, and oligo(hT), 5′-GGTTAGGGTTAGGGTTAGGATGGT-3′, respectively.
Southern Analysis.
Southern hybridization was carried out at 42°C in 10 ml of Rapid-hyb (Amersham) and washed as per the manufacturer’s suggestions. Oligonucleotide probes were labeled by using polynucleotide kinase and [γ-32P]ATP. The probe for yeast telomeric DNA was (TGTGGG)4, and the probe for human telomeric DNA was (TTAGGG)4.
YAC Introduction and End Rescue.
A YAC clone from the Centre d’Etudes du Polymorphisme Humain megaYAC library was kar-crossed to haploid strain 8D. Colonies were replica-plated to SD8-His, then analyzed by PFGE to verify YACductants as previously described (17). One clone, designated YACx8D.1, was chosen for further analysis. After sequentially restreaking two times, genomic DNA was isolated from 10-ml overnight cultures of the parental YAC clone and YACx8D.1 and digested with BglII, and the ends were blunted with T4 DNA polymerase. The DNA was ligated at low concentration to circularize the left YAC end, then transformed into Escherichia coli DH10B competent cells and plated on LB + 50 μg/ml ampicillin. Resultant colonies were miniprepped and digested with HindIII for analysis. Sequencing of selected clones was carried out by using the Cyclist Taq DNA sequencing kit (Stratagene) and [α-33P]dATP.
RESULTS
Humanizing the Yeast Telomerase RNA Template.
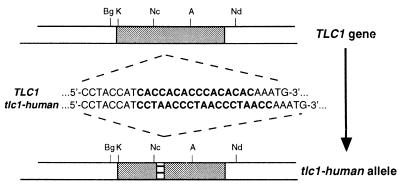
We replaced the template sequence, 5′-CACCACACCCACACAC-3′, of the S. cerevisiae telomerase RNA with a template sequence for human telomeric repeats, 5′-CCUAACCCUAACCCUAACC-3′, by two-step gene replacement in a diploid yeast strain (Fig. 1). Nine out of 10 of the resultant diploid strains analyzed were heterozygous, having both wild-type TLC1 and tlc1-human alleles, and RT-PCR analysis demonstrated that the tlc1-human allele was expressed in at least seven out of nine heterozygotes (data not shown). One heterozygote that expressed the tlc1-human allele, designated C6.1, was chosen for further analysis.
Figure 1.
Humanizing the yeast telomerase template gene, TLC1. The template region of the yeast TLC1 gene carried on pTLC1 was altered to a human template sequence (tlc1-human) and designated pTLC1h. pTLC1h then was introduced into the TLC1 gene of a diploid yeast strain by two-step gene replacement. The restriction sites are: A, AflII; Bg, BglII; K, KpnI; Nc, NcoI; Nd, NdeI.
Viability and Growth Analysis of Haploid Strains with the Humanized RNA Template.
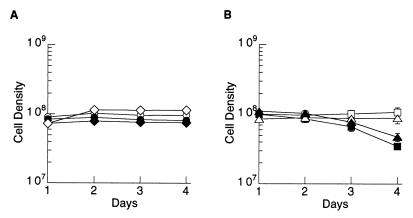
To determine whether the tlc1-human allele could support telomere maintenance, strain C6.1 was sporulated, tetrads were dissected, and haploid spores were regenerated. Twelve tetrads were dissected, with 10 of 12 tetrads having four viable spores. Genotypic analysis revealed that 8 of 12 tetrads showed 2:2 segregation of the wild-type and tlc1-human alleles, indicating that the humanization of the TLC1 gene did not cause lethality (data not shown). All 12 tetrads were analyzed for senescence by 5× serial restreaking as previously described (19). No significant difference in growth between haploid strains with the wild-type TLC1 gene and those carrying the tlc1-human allele was observed (data not shown). Two tetrads were randomly chosen for quantitative analysis of growth rate in liquid culture, as previously described for TLC1 knockouts (13). In three separate experiments, varying in length from 6 days to 2 weeks, no loss of growth rate was observed, nor was there any evidence of senescence in the tlc1-human strains (Fig. 2A). Two tetrads from strain UCC3508, which is heterozygous for disruption of the TLC1 gene, were analyzed in parallel. Haploid strains with a disruption of the TLC1 gene consistently displayed a significant drop (about 50%) in growth rate by day 4 (Fig. 2B). Shortly thereafter, however, the growth rate returned to normal (data not shown), presumably because of outgrowth of suppressor mutants, as has been previously demonstrated for tlc1 haploids derived from UCC3508 (13) (D. Gottschling, personal communication).
Figure 2.
Haploid strains with the tlc1-human allele have a normal growth rate and no indication of senescence. Haploid strains were analyzed for growth rate in liquid culture. A 5.5-ml culture of each haploid strain was seeded at 3 × 105 cells/ml and grown for 24 hr at 30°C. The density of each culture was determined, then diluted back to 3 × 105, and the process was repeated. No significant difference in growth rate was observed between haploid strains carrying the wild-type allele, and those with the tlc1-human allele. This is in contrast to haploid strains with a disruption of the TLC1 gene, derived from tetrad dissection of strain UCC3508 (13), which consistently displayed a significant drop (about 50%) in growth rate by day 4. (A) Haploid strains from a tetrad of strain C6.1. Open symbols, haploid strains with the wild-type TLC1 allele; closed symbols, haploid strains with the tlc1-human allele. (B) Haploid strains from a tetrad of strain UCC35088, in which one allele of TLC1 is disrupted. Open symbols, haploid strains with the wild-type TLC1 allele; closed symbols, haploid strains with the tlc1∷LEU2 disruption. A second tetrad was analyzed for each of strains C6.1 and UCC3508, with similar results (data not shown).
Incorporation of TTAGGG Repeats into Endogenous Yeast Chromosome Ends.
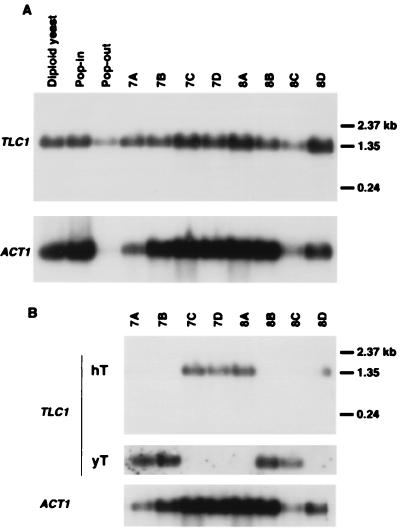
Northern analysis of the haploid strains from each of two tetrads with a full-length TLC1 probe showed that the TLC1 gene expression varied from 4- to 6-fold among haploid strains, with no correlation to TLC1 genotype (Fig. 3A). Further analysis using allele-specific oligonucleotide hybridization demonstrated that only the tlc1-human allele was expressed in strains 7C, 7D, 8A, and 8D, whereas only the wild-type TLC1 allele was expressed in strains 7A, 7B, 8B, and 8C, consistent with the genotypic analysis (Fig. 3B).
Figure 3.
Normal levels of TLC1 expression are observed in haploid strains with the tlc1-human allele. (A) TLC1 gene expression by Northern hybridization. A Northern blot of total RNA was hybridized with a full-length TLC1 probe, then stripped and reprobed with an ACT1 probe as a control for loading. Expression levels were quantitated by using a PhosphorImager. Diploid yeast, diploid strain NM1 (AB1380 × YPH925); Pop-in, clone C6 that was generated by pTLC1h plasmid insertion into NM1; Pop-out, clone C6.1, a 5-fluoroorotic acid-resistant derivative of C6 that has one copy each of the wild-type TLC1 and the tlc1-human alleles; 7A to 8D, haploid strains from two tetrad dissections of C6.1. (B) Allele-specific oligonucleotide hybridization distinguishes expression of the tlc1-human allele from the wild-type TLC1 allele. A Northern blot of total RNA was hybridized first with an oligonucleotide specific for the human template sequence (hT), then stripped and reprobed with an oligonucleotide specific for the yeast template sequence (yT), and finally stripped and reprobed with an ACT1 probe to control for loading. 7A to 8D, haploid strains from tetrads 7 and 8.
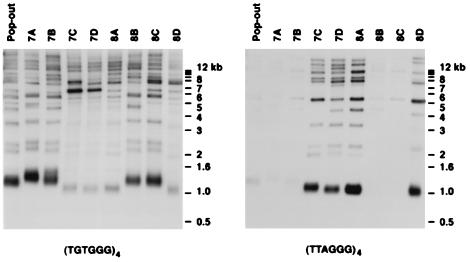
Genomic DNA was isolated from tetrads 7 and 8 after 8× serially restreaking (≈200 generations). In the strains expressing the tlc1-human allele, strong hybridization of a (TTAGGG)4 probe verified that human telomeric repeats had been added to the ends of the endogenous yeast chromosomes, and suggested that a significant portion of the wild-type telomeric DNA has been lost (Fig. 4 Right, lanes 7C, 7D, 8A, and 8D). A majority of yeast chromosomes (the Y′ class) have an XhoI site ≈1.2 kb from the end and are seen as an ≈1.2 kb heterogeneous band with the yeast telomere probe (Fig. 4 Left, lanes 7A, 7B, 8B, and 8C). In the haploid strains carrying the tlc1-human allele (7C, 7D, 8A, and 8D), the Y′-type telomere band is ≈100–200 bp shorter than in the haploid strains with the wild-type TLC1 allele, indicating that some telomere shortening has occurred in the humanized strains during the ≈200 generations in culture. A low level of hybridization of the yeast telomere probe (TGTGGG)4 also was observed in the humanized strains (Fig. 4 Left, lanes 7C, 7D, 8A, and 8D). This hybridization of the yeast telomere probe presumably is because of residual yeast telomere sequences in the humanized strains, which likely is necessary for the maintenance of chromosome stability (see below).
Figure 4.
Human telomeric DNA is present at the ends of chromosomes in haploid strains expressing the tlc1-human allele. A Southern blot of XhoI-digested yeast genomic DNA was hybridized first with an oligonucleotide probe specific for yeast telomeric DNA, (TGTGGG)4, stripped, and then reprobed with an oligonucleotide probe specific for human telomeric DNA, (TTAGGG)4. DNA loading varied less than 3-fold between lanes, as determined by rehybridization with a probe for actin and quantitation by using a PhosphorImager. Pop-out, diploid strain C6.1, which has one copy each of the wild-type TLC1 and the tlc1-human alleles; 7A to 8D, haploid strains from the dissection of two tetrads of C6.1.
Addition of Human Telomeres to a YAC.
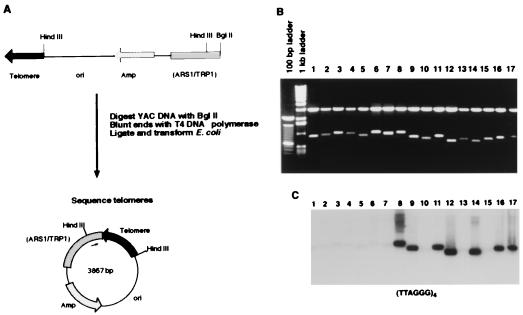
To analyze newly synthesized telomeres in a tlc1-human strain at the molecular level, the addition of human telomeric repeats to a YAC was assessed. A YAC from the Centre d’Etudes du Polymorphisme Humain megaYAC library was introduced into the tlc1-human strain by kar-crossing (17). After 2× serial restreaking (≈50 generations), the left YAC end was rescued as a plasmid to E. coli (Fig. 5A). Digestion of rescued YAC ends from strain AB1380 and from the tlc1-human strain with HindIII released a variable-sized band that includes the YAC telomere sequence (Fig. 5B). Variation in the size of the telomere band would be expected if telomerase is active at the YAC ends. On average, the YAC telomeres rescued from the tlc1-human strain were about 175 bp shorter than those rescued from strain AB1380, consistent with the previous hybridization analysis of yeast chromosome telomeres in the tlc1-human strain (Fig. 4). Hybridization of the rescued YAC telomeres with an oligonucleotide probe for human telomeric sequences (TTAGGG)4 revealed that 3 of 10 YAC telomeres rescued from the tlc1-human strain had no human telomere sequences, whereas the remaining 7 of 10 contained human telomeric repeats (Fig. 5C, lanes 8–17). The shortened YAC ends with TTAGGG repeats and the absence of TTAGGG sequence at some YAC ends rescued from the tlc1-human strain may have resulted from the removal of some TTAGGG repeats during the telomere-cloning procedure (during removal of the single-stranded 3′ tails of the YAC telomeres), or perhaps the regular TTAGGG repeats are unstable in E. coli. However, it is also possible that some YAC ends have not yet been acted on by telomerase, consistent with models of telomere length regulating telomerase activity (11).
Figure 5.
Analysis of YAC ends rescued from a haploid yeast strain expressing the tlc1-human allele. (A) Diagrammatic representation of the rescue of the acentric arm of pYAC4. Genomic DNA was isolated from yeast carrying the YAC and digested with BglII to release an approximately 3.9-kb left-end fragment from the YAC. The fragment was blunted with T4 DNA polymerase and recircularized before introduction into E. coli, where it was propagated as a plasmid. The telomeres were sequenced by using a primer located in the TRP/ARS region of the plasmid, indicated by the arrow within the vector circle. (B) Rescued YAC telomeres have a variable amount of telomeric DNA. Ethidium bromide-stained gel of miniprep DNA from the rescued YAC ends, digested with HindIII, which releases a 2.9-kb constant vector band, and a variable-sized band, which includes the telomere. Lanes 1–7, ends rescued from the original YAC clone in strain AB1380; lanes 8–17, ends rescued from YACx8D.1, which contains a YAC kar-crossed to tlc1-human haploid strain 8D. (C) YAC ends rescued from the haploid strains carrying the tlc1-human allele have different amounts of human telomeric DNA. Southern hybridization of the gel seen in B with an oligonucleotide for human telomeric repeats, (TTAGGG)4.
Sequence analysis revealed that from 0 to 234 bp of human telomeric DNA was present on the YAC ends rescued from the tlc1-human strain (Table 1). Yeast typically contain ≈300 bp of telomeric DNA (20). Therefore, after only ≈50 generations in the humanized yeast strain, a significant portion of the telomeric DNA present on a YAC end was replaced with human sequences. These data conclusively show that the yeast telomerase is using the humanized template to synthesize new telomere repeats.
Table 1.
Sequence of rescued YAC ends
| Sample | Telomere sequence |
|---|---|
| 8 | …GTGTGTGGGTGTGGG(TTAGGG)45TTA |
| 9 | …GGGTGTGTGGGTGGGG(TTAGGG)7TTA |
| 10 | …TGTGGTGTGTGGGTGTGTGGGTGTGGTG |
| 11 | …GGGTGTGGTGTGTGTGGGG(TTAGGG)9 |
| 12 | …TGTGGGTGTGGTGTGTGGG(TTAGGG)23 |
| 13 | …TGGGTGTGGGTGTGGGTGTGGTGTGTGG |
| 14 | …TGTGGGTGTGGTGTGTGGG(TTAGGG)23 |
| 15 | …TGTGGTGTGTGGGTGTGTGGGTGTGGTG |
| 16 | …TGGGTGTGTGGGTGGG(TTAGGG)7TTA |
| 17 | …GGGTGTGGTGTGTGTGGGG(TTAGGG)9 |
Shown is the sequence ending at the distal end of the rescued YAC telomere. All 10 samples contained sequences conforming to the S. cerevisiae telomere consensus sequence, TG1–3, followed by a variable number of human telomeric (TTAGGG) repeats. Sample numbers correspond to the lanes of Fig. 5B.
All rescued YAC ends retained substantial amounts of yeast telomeric sequences (more than 100 bp in some cases; data not shown), regardless of the amount of human telomeric DNA present. It is likely that this yeast telomeric DNA is required for stable maintenance of the YAC in yeast. It therefore will be important to determine how far from the distal ends of the YAC these sequences can occur and still be functional, when designing HAC vectors.
Several rescued YAC ends contain four Gs before TTAGGG repeats (samples 9, 11, and 17 in Table 1). Most likely this resulted from the action of the humanized telomerase template. A G4 stretch can be generated if the template RNA annealed to the last two Gs of a TG3 with the last two Cs of the template “… CTAACCC… ”
DISCUSSION
In most species, telomere repeats are added in precise tandemly repeated units, consistent with a telomerase activity that faithfully copies its RNA template (21). In contrast, S. cerevisiae telomeric repeats are irregular (TG)1–6TG2–3, often abbreviated as TG1–3. This irregularity has been explained both by the irregular nature of the 5′-CACCACACCCACACAC-3′ template sequence found in the S. cerevisiae telomerase RNA, and as an intrinsic mechanistic property of the S. cerevisiae telomerase (13, 15). The replacement of the yeast template sequence with the humanized template resulted in the incorporation of regular, TTAGGG repeats into telomeric DNA. This strongly suggests that the irregularity of S. cerevisiae telomere sequences is due solely to the template region of the TLC1 gene.
It has been suggested that the activity of telomerase may be regulated by the number of telomere-binding proteins bound at the telomere ends (22). Rap1p, which interacts with telomeres in vivo, appears to be the major telomere-binding protein in yeast, and the Rap1p consensus-binding site, RC/AAYCCRYNCAYY, can be found as frequently as once per 18 bp of yeast telomeric DNA (23, 24). Mutation or overexpression of RAP1 affects telomere length (20), and reduced Rap1p binding of telomeric DNA has been suggested as the cause of telomere lengthening in Kluyveromyces lactis strains with telomerase RNA template mutations (25). In contrast, strains with three base pair mutations in the yeast TLC1 gene demonstrate telomere shortening, likely because of reduced enzymatic activity of telomerase (15). In the present study, the replacement of the TLC1 template with the human template sequence is expected to reduce the number of Rap1p-binding sites in telomeric DNA, yet resulted in shortened telomeres, suggesting that telomerase activity may be affected in the tlc1-human strains.
Subtelomeric DNA may also play a role in telomere length regulation. Conserved structural features have been identified in yeast and human subtelomeric regions (26). Every yeast chromosome has an array of degenerate TTAGGG repeats, and a comparison of human and yeast subtelomeric regions suggested that degenerate TTAGGG repeats identify an evolutionarily conserved boundary between subtelomeric domains (26). The yeast (TTAGGG)n-binding factor 1 (Tbf1 or Tbfα) was identified and cloned through its ability to interact with the vertebrate telomere sequence, (TTAGGG)n, but not the irregular yeast telomeric repeats in vitro (27). Tbf1 has been shown to share epitopes with the human telomere repeat factor (TRF1), and it has been suggested that Tbf1 may serve as an anchor protein to target other telomere factors, such as telomerase, to the conserved junction sequence distant from the terminus and thereby allow heterologous telomere sequences such as human telomere repeats to function in yeast (28). However, mutation of the TBF1, an essential gene in yeast, yields no detectable telomere phenotype (29). Further studies will be necessary to determine whether the ability of Tbf1 to bind to the (TTAGGG)n repeats is allowing the tlc1-human strains to survive the progressive loss of Rap1p-binding sites at the chromosome termini. Disruption of the TBF1 gene in the tlc1-human strains may help to answer this question.
Yeast strains with the humanized telomerase template sequence now can be used to determine the requirements for human telomeres to be propagated in yeast, yet to retain function in mammalian cells. One area of research in which this information will be valuable is the development of HACs by modification of YACs containing human centromeric sequences. Stable YACs containing alpha-satellite DNA are a reasonable starting point for HAC construction, because of their large capacity for DNA, the ease with which they can be manipulated and modified in yeast, and the fact that they can be introduced intact into mammalian cells. The tlc1-human yeast strains should be more suitable hosts for the assembly and maintenance of such HAC constructs than wild-type yeast strains, because they can add and maintain human telomere sequences at the ends of the HAC constructs. Moreover, the tlc1-human yeast strains could be used to address the question of what amount or location of human telomeric DNA is necessary for HAC telomeres to be functional when transferred to human cells.
Acknowledgments
We thank Daniel Gottschling (Fred Hutchinson Cancer Research Center) for the gift of strain UCC3508, Reed B. Wickner [National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health (NIH)], Thorsten Allers and Michael Lichten (National Cancer Institute, NIH) for help with tetrad dissection, and Francis Collins and William Pavan (National Human Genome Research Institute, NIH) for critical reading of the manuscript. K.H. is supported in part by a postdoctoral fellowship from the Cystic Fibrosis Foundation.
Footnotes
This paper was submitted directly (Track II) to the Proceedings Office.
Abbreviations: YAC, yeast artificial chromosome; HAC, human artificial chromosome.
References
- 1.Harrington J J, Van Bokkelen G, Mays R W, Gustashaw K, Willard H F. Nat Genet. 1997;15:345–355. doi: 10.1038/ng0497-345. [DOI] [PubMed] [Google Scholar]
- 2.Rosenfeld M A. Nat Genet. 1997;15:333–335. doi: 10.1038/ng0497-333. [DOI] [PubMed] [Google Scholar]
- 3.Warburton P E, Kipling D. Nature (London) 1997;386:553–555. doi: 10.1038/386553a0. [DOI] [PubMed] [Google Scholar]
- 4.Brown W R A, MacKinnon P J, Villasanté A, Spurr N, Buckle V J, Dobson M J. Cell. 1990;63:119–132. doi: 10.1016/0092-8674(90)90293-n. [DOI] [PubMed] [Google Scholar]
- 5.Taylor S S, Larin Z, Tyler Smith C. Hum Mol Genet. 1994;3:1383–1386. doi: 10.1093/hmg/3.8.1383. [DOI] [PubMed] [Google Scholar]
- 6.Hanish J P, Yanowitz J L, De Lange T. Proc Natl Acad Sci USA. 1994;91:8861–8865. doi: 10.1073/pnas.91.19.8861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Blackburn E H. Cell. 1994;77:621–623. doi: 10.1016/0092-8674(94)90046-9. [DOI] [PubMed] [Google Scholar]
- 8.Zakian V A. Science. 1995;270:1601–1607. doi: 10.1126/science.270.5242.1601. [DOI] [PubMed] [Google Scholar]
- 9.Greider C W, Blackburn E H. Cell. 1985;43:405–413. doi: 10.1016/0092-8674(85)90170-9. [DOI] [PubMed] [Google Scholar]
- 10.Counter C M, Meyerson M, Eaton E N, Weinberg R A. Proc Natl Acad Sci USA. 1997;94:9202–9207. doi: 10.1073/pnas.94.17.9202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Greider C W. Annu Rev Biochem. 1996;65:337–365. doi: 10.1146/annurev.bi.65.070196.002005. [DOI] [PubMed] [Google Scholar]
- 12.Greider C W, Blackburn E H. Nature (London) 1989;337:331–337. doi: 10.1038/337331a0. [DOI] [PubMed] [Google Scholar]
- 13.Singer M S, Gottschling D E. Science. 1994;266:404–409. doi: 10.1126/science.7545955. [DOI] [PubMed] [Google Scholar]
- 14.Feng J, Funk W D, Wang S-S, Weinrich S L, Avilion A A, Chiu C-P, Adams R R, Chang E, Allsopp R C, Yu J, et al. Science. 1995;269:1236–1241. doi: 10.1126/science.7544491. [DOI] [PubMed] [Google Scholar]
- 15.Prescott J, Blackburn E. Genes Dev. 1997;11:528–540. doi: 10.1101/gad.11.4.528. [DOI] [PubMed] [Google Scholar]
- 16.Cross S H, Allshire R C, McKay S J, McGill N I, Cooke H J. Nature (London) 1989;338:771–774. doi: 10.1038/338771a0. [DOI] [PubMed] [Google Scholar]
- 17.Spencer F, Ketner G, Connelly C, Hieter P. Methods. 1993;5:161–175. [Google Scholar]
- 18.Silverman G A. In: YAC Protocols. Markie D, editor. Totowa, NJ: Humana; 1996. pp. 199–216. [Google Scholar]
- 19.Lendvay T S, Morris D K, Sah J, Balasubramanian B, Lundblad V. Genetics. 1996;144:1399–1412. doi: 10.1093/genetics/144.4.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zakian V A. Annu Rev Genet. 1996;30:141–172. doi: 10.1146/annurev.genet.30.1.141. [DOI] [PubMed] [Google Scholar]
- 21.Greider C W, Collins K, Autexier C. DNA Replication in Eukaryotic Cells. Cold Spring Harbor, NY: Cold Spring Harbor Lab. Press; 1996. pp. 619–638. [Google Scholar]
- 22.Marcand S, Gilson E, Shore D. Science. 1997;275:986–990. doi: 10.1126/science.275.5302.986. [DOI] [PubMed] [Google Scholar]
- 23.Conrad M N, Wright J H, Wolf A J, Zakian V A. Cell. 1990;63:739–750. doi: 10.1016/0092-8674(90)90140-a. [DOI] [PubMed] [Google Scholar]
- 24.Gilson E, Roberge M, Giraldo R, Rhodes D, Gasser S M. J Mol Biol. 1993;231:293–310. doi: 10.1006/jmbi.1993.1283. [DOI] [PubMed] [Google Scholar]
- 25.Krauskopf A, Blackburn E H. Nature (London) 1996;383:354–357. doi: 10.1038/383354a0. [DOI] [PubMed] [Google Scholar]
- 26.Flint J, Bates G P, Clark K, Dorman A, Willingham D, Roe B A, Micklem G, Higgs D R, Louis E J. Hum Mol Genet. 1997;6:1305–1314. doi: 10.1093/hmg/6.8.1305. [DOI] [PubMed] [Google Scholar]
- 27.Liu Z, Tye B-K. Genes Dev. 1991;5:49–59. doi: 10.1101/gad.5.1.49. [DOI] [PubMed] [Google Scholar]
- 28.Fang G, Cech T R. In: Telomeres. Blackburn E H, Greider C W, editors. Cold Spring Harbor, NY: Cold Spring Harbor Lab. Press; 1995. pp. 69–105. [Google Scholar]
- 29.Brigati C, Kurtz S, Balderes D, Vidali G, Shore D. Mol Cell Biol. 1993;13:1306–1314. doi: 10.1128/mcb.13.2.1306. [DOI] [PMC free article] [PubMed] [Google Scholar]