Abstract
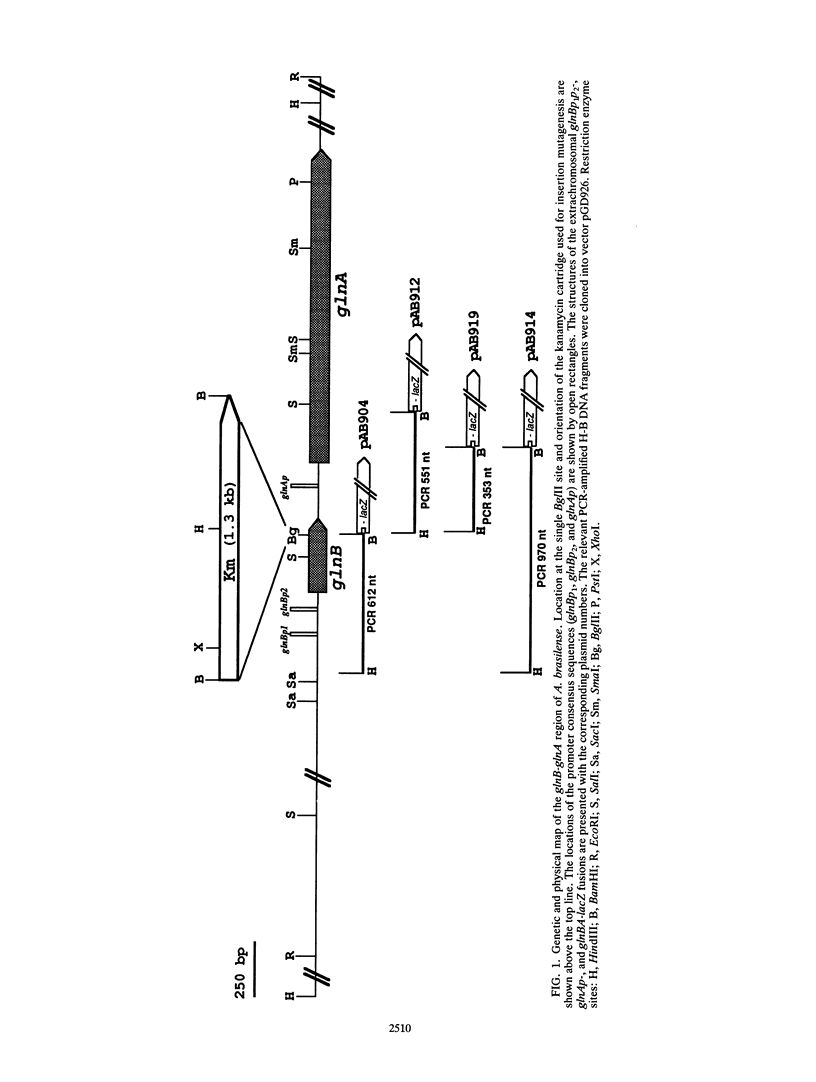
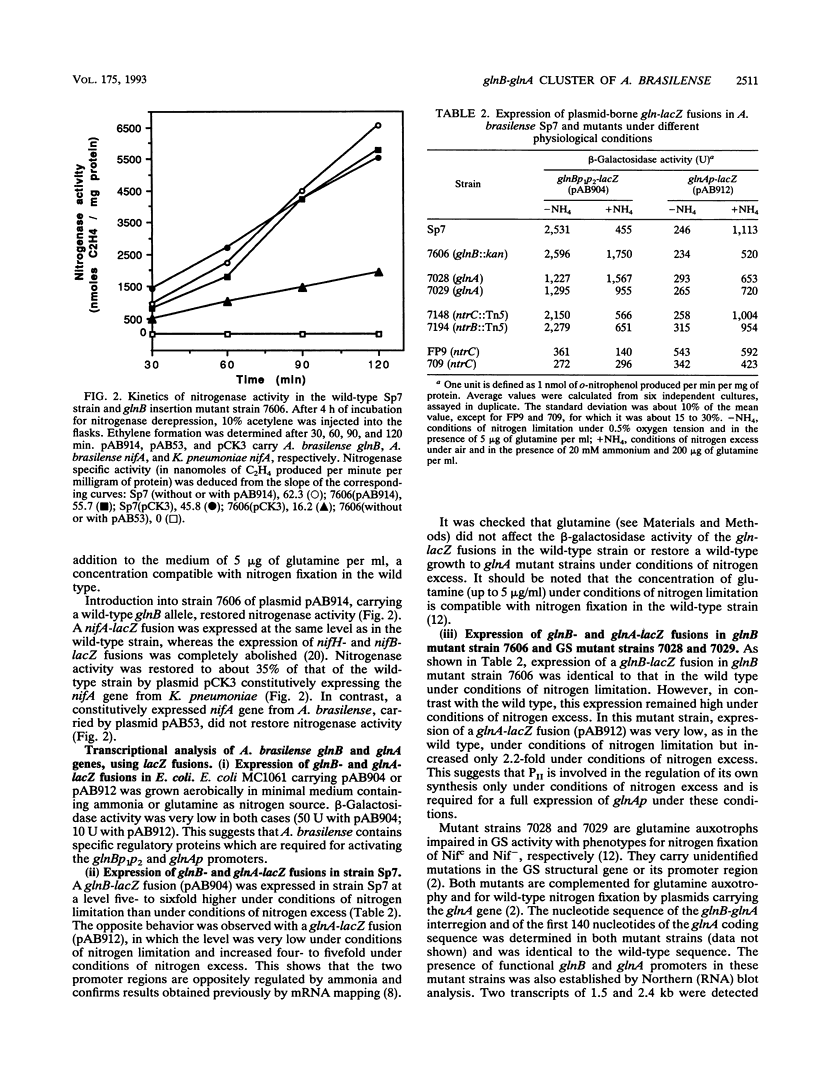
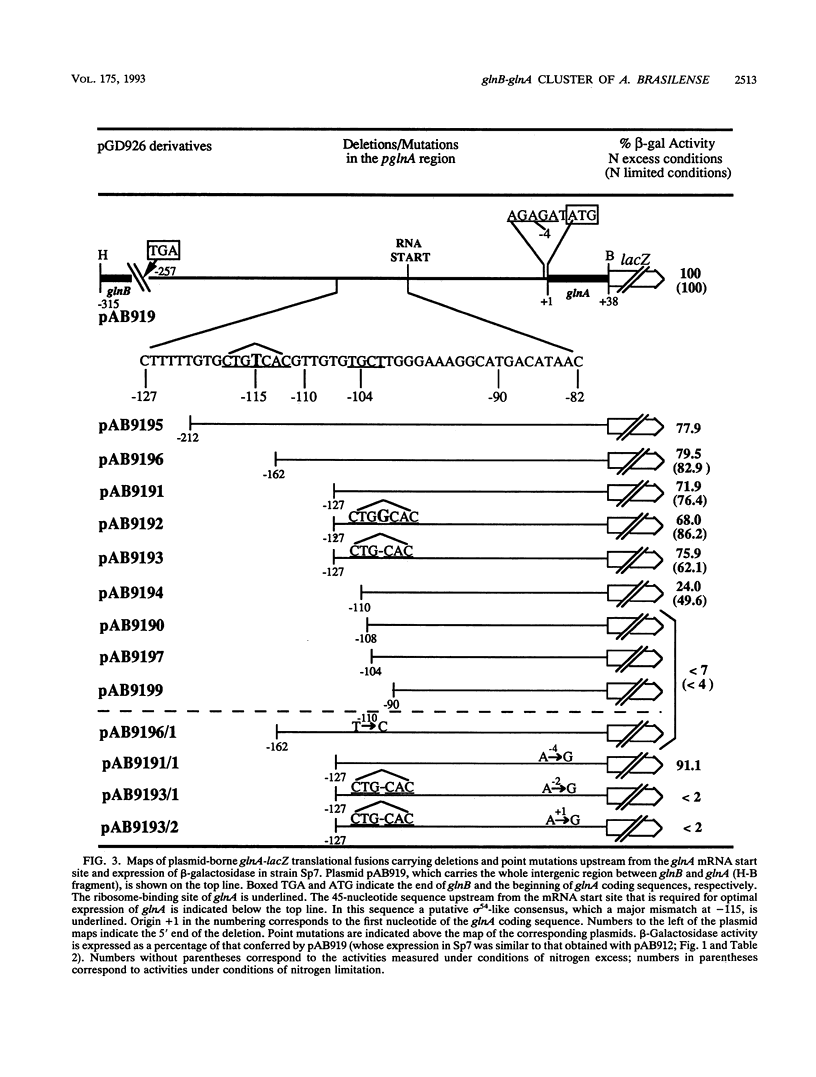
The functional organization of the glnB-A cluster of Azospirillum brasilense, which codes for the PII protein and glutamine synthetase, respectively, was studied with the aid of lacZ fusions, deletion mapping, site-directed mutagenesis, and complementation. It was shown previously by mRNA mapping that the cluster contains two tandemly organized promoters, glnBp1 and glnBp2, of the sigma 70 and sigma 54 types, respectively, upstream of glnB and a third unidentified promoter upstream of glnA. Data obtained with lacZ fusions in the wild-type strain confirmed that cotranscription of glnBA and transcription of glnA alone were oppositely regulated by the cell N status. Quantification of promoter activities showed a high level of transcription from glnBp1p2 and a low level from glnAp under conditions of nitrogen limitation. The opposite situation prevails under conditions of nitrogen excess. As a consequence, PII polypeptide synthesis is increased under conditions of nitrogen fixation, which strongly suggests that PII plays an important role under these conditions. Null mutant strains of glnB, ntrB-ntrC, nifA, and point mutant strains in glnA were analyzed. NtrB and NtrC are not involved in the regulation of glnBA expression, in contrast to PII and glutamine synthetase. Glutamine synthetase probably acts by modulating the intracellular N status, and PII acts by modifying the properties of an unidentified regulator which might be a functional homolog of NtrC. In addition, a Nif- null mutant strain of glnB was characterized further. A Nif+ phenotype was restored to the strain by nifA from Klebsiella pneumoniae but not by nifA from A. brasilense. This mutant strain is not impaired in NifA synthesis, which is relatively independent of the growth conditions in A. brasilense. It is therefore most likely that PII is required for NifA activation under conditions of nitrogen fixation. Deletion mapping and site-directed mutagenesis showed glnAp was located within a 45-bp DNA fragment upstream of the mRNA start site, dissimiar to previously described consensus sites for sigma factors.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bozouklian H., Elmerich C. Nucleotide sequence of the Azospirillum brasilense Sp7 glutamine synthetase structural gene. Biochimie. 1986 Oct-Nov;68(10-11):1181–1187. doi: 10.1016/s0300-9084(86)80062-1. [DOI] [PubMed] [Google Scholar]
- Bozouklian H., Fogher C., Elmerich C. Cloning and characterization of the glnA gene of Azospirillum brasilense Sp7. Ann Inst Pasteur Microbiol. 1986 Jul-Aug;137B(1):3–18. doi: 10.1016/s0769-2609(86)80089-8. [DOI] [PubMed] [Google Scholar]
- Bueno R., Pahel G., Magasanik B. Role of glnB and glnD gene products in regulation of the glnALG operon of Escherichia coli. J Bacteriol. 1985 Nov;164(2):816–822. doi: 10.1128/jb.164.2.816-822.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casadaban M. J., Martinez-Arias A., Shapira S. K., Chou J. Beta-galactosidase gene fusions for analyzing gene expression in escherichia coli and yeast. Methods Enzymol. 1983;100:293–308. doi: 10.1016/0076-6879(83)00063-4. [DOI] [PubMed] [Google Scholar]
- Chiurazzi M., Iaccarino M. Transcriptional analysis of the glnB-glnA region of Rhizobium leguminosarum biovar viciae. Mol Microbiol. 1990 Oct;4(10):1727–1735. doi: 10.1111/j.1365-2958.1990.tb00550.x. [DOI] [PubMed] [Google Scholar]
- Colonna-Romano S., Riccio A., Guida M., Defez R., Lamberti A., Iaccarino M., Arnold W., Priefer U., Pühler A. Tight linkage of glnA and a putative regulatory gene in Rhizobium leguminosarum. Nucleic Acids Res. 1987 Mar 11;15(5):1951–1964. doi: 10.1093/nar/15.5.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ditta G., Schmidhauser T., Yakobson E., Lu P., Liang X. W., Finlay D. R., Guiney D., Helinski D. R. Plasmids related to the broad host range vector, pRK290, useful for gene cloning and for monitoring gene expression. Plasmid. 1985 Mar;13(2):149–153. doi: 10.1016/0147-619x(85)90068-x. [DOI] [PubMed] [Google Scholar]
- Galimand M., Perroud B., Delorme F., Paquelin A., Vieille C., Bozouklian H., Elmerich C. Identification of DNA regions homologous to nitrogen fixation genes nifE, nifUS and fixABC in Azospirillum brasilense Sp7. J Gen Microbiol. 1989 May;135(5):1047–1059. doi: 10.1099/00221287-135-5-1047. [DOI] [PubMed] [Google Scholar]
- Holtel A., Merrick M. J. The Klebsiella pneumoniae PII protein (glnB gene product) is not absolutely required for nitrogen regulation and is not involved in NifL-mediated nif gene regulation. Mol Gen Genet. 1989 Jun;217(2-3):474–480. doi: 10.1007/BF02464920. [DOI] [PubMed] [Google Scholar]
- Holtel A., Merrick M. Identification of the Klebsiella pneumoniae glnB gene: nucleotide sequence of wild-type and mutant alleles. Mol Gen Genet. 1988 Dec;215(1):134–138. doi: 10.1007/BF00331314. [DOI] [PubMed] [Google Scholar]
- Kaminski P. A., Elmerich C. Involvement of fixLJ in the regulation of nitrogen fixation in Azorhizobium caulinodans. Mol Microbiol. 1991 Mar;5(3):665–673. doi: 10.1111/j.1365-2958.1991.tb00738.x. [DOI] [PubMed] [Google Scholar]
- Kranz R. G., Pace V. M., Caldicott I. M. Inactivation, sequence, and lacZ fusion analysis of a regulatory locus required for repression of nitrogen fixation genes in Rhodobacter capsulatus. J Bacteriol. 1990 Jan;172(1):53–62. doi: 10.1128/jb.172.1.53-62.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang Y. Y., Kaminski P. A., Elmerich C. Identification of a nifA-like regulatory gene of Azospirillum brasilense Sp7 expressed under conditions of nitrogen fixation and in the presence of air and ammonia. Mol Microbiol. 1991 Nov;5(11):2735–2744. doi: 10.1111/j.1365-2958.1991.tb01982.x. [DOI] [PubMed] [Google Scholar]
- Liang Y. Y., de Zamaroczy M., Arsène F., Paquelin A., Elmerich C. Regulation of nitrogen fixation in Azospirillum brasilense Sp7: involvement of nifA, glnA and glnB gene products. FEMS Microbiol Lett. 1992 Dec 15;100(1-3):113–119. doi: 10.1111/j.1574-6968.1992.tb14028.x. [DOI] [PubMed] [Google Scholar]
- Magasanik B. Reversible phosphorylation of an enhancer binding protein regulates the transcription of bacterial nitrogen utilization genes. Trends Biochem Sci. 1988 Dec;13(12):475–479. doi: 10.1016/0968-0004(88)90234-4. [DOI] [PubMed] [Google Scholar]
- Martin G. B., Thomashow M. F., Chelm B. K. Bradyrhizobium japonicum glnB, a putative nitrogen-regulatory gene, is regulated by NtrC at tandem promoters. J Bacteriol. 1989 Oct;171(10):5638–5645. doi: 10.1128/jb.171.10.5638-5645.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Ninfa A. J., Magasanik B. Covalent modification of the glnG product, NRI, by the glnL product, NRII, regulates the transcription of the glnALG operon in Escherichia coli. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5909–5913. doi: 10.1073/pnas.83.16.5909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okon Y., Albrecht S. L., Burris R. H. Carbon and ammonia metabolism of Spirillum lipoferum. J Bacteriol. 1976 Nov;128(2):592–597. doi: 10.1128/jb.128.2.592-597.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Son H. S., Rhee S. G. Cascade control of Escherichia coli glutamine synthetase. Purification and properties of PII protein and nucleotide sequence of its structural gene. J Biol Chem. 1987 Jun 25;262(18):8690–8695. [PubMed] [Google Scholar]
- Stock J. B., Ninfa A. J., Stock A. M. Protein phosphorylation and regulation of adaptive responses in bacteria. Microbiol Rev. 1989 Dec;53(4):450–490. doi: 10.1128/mr.53.4.450-490.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarrand J. J., Krieg N. R., Döbereiner J. A taxonomic study of the Spirillum lipoferum group, with descriptions of a new genus, Azospirillum gen. nov. and two species, Azospirillum lipoferum (Beijerinck) comb. nov. and Azospirillum brasilense sp. nov. Can J Microbiol. 1978 Aug;24(8):967–980. doi: 10.1139/m78-160. [DOI] [PubMed] [Google Scholar]
- Wain-Hobson S., Sonigo P., Danos O., Cole S., Alizon M. Nucleotide sequence of the AIDS virus, LAV. Cell. 1985 Jan;40(1):9–17. doi: 10.1016/0092-8674(85)90303-4. [DOI] [PubMed] [Google Scholar]
- Westby C. A., Enderlin C. S., Steinberg N. A., Joseph C. M., Meeks J. C. Assimilation of 13NH4+ by Azospirillum brasilense grown under nitrogen limitation and excess. J Bacteriol. 1987 Sep;169(9):4211–4214. doi: 10.1128/jb.169.9.4211-4214.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Zamaroczy M., Delorme F., Elmerich C. Characterization of three different nitrogen-regulated promoter regions for the expression of glnB and glnA in Azospirillum brasilense. Mol Gen Genet. 1990 Dec;224(3):421–430. doi: 10.1007/BF00262437. [DOI] [PubMed] [Google Scholar]
- de Zamaroczy M., Delorme F., Elmerich C. Regulation of transcription and promoter mapping of the structural genes for nitrogenase (nifHDK) of Azospirillum brasilense Sp7. Mol Gen Genet. 1989 Dec;220(1):88–94. doi: 10.1007/BF00260861. [DOI] [PubMed] [Google Scholar]



