Abstract
Fli1 is an Ets family member that is essential for embryonic development. Increasing evidence suggests modulating Fli1 gene expression impacts lymphocyte development/function and is an important mediator in the autoimmune disease lupus. Fli1 is over-expressed in splenic lymphocytes in lupus prone mouse strains and in PBMCs of lupus patients. Presently, it is unknown how Fli1 gene expression is controlled in lymphocytes or how it becomes over-expressed in lupus. Therefore, we examined Fli1 regulation in a murine B cell line and T cell line and identified several cis-regulatory elements within a 230 bp region that contribute to Fli1 promoter activity. Ets factors Elf1, Tel and Fli1 bind in vitro to this region and increase endogenous Fli1 expression when over-expressed in a T cell line. In addition, we determined that a microsatellite located adjacent to the region containing these cis-regulatory elements is polymorphic in three lupus prone mouse strains and that the length of the microsatellite is inversely correlated with promoter activity in a T cell line. These results suggest that several Ets factors, including Fli1 itself, are involved in the transcriptional regulation of Fli1 in lymphocytes. Furthermore, the presence of a polymorphic microsatellite in the Fli1 promoter may contribute to increased Fli1 expression in T cells during lupus disease progression.
Keywords: transcription, gene regulation, transcription factors, Ets, B cells, T cells, systemic lupus erythematosus
Introduction
Fli1 is a member of the Ets family of transcription factors that share a highly conserved Ets DNA binding domain and binds to Ets binding sites (EBS) that contain the core recognition motif GGA(A/T) (Brown and McKnight, 1992; Fisher et al., 1992; Mao et al., 1994; Shore and Sharrocks, 1995; Urness and Thummel, 1990; Wasylyk et al., 1993). Murine Fli1 was first identified as a proto-oncogene as a site for retroviral integration of Friend virus-induced erythroleukemias (Ben-David et al., 1990). It is expressed in hematopoietic tissues in both embryos and adults, as well as in other tissues (Ben-David et al., 1991; Melet et al., 1996; Watson et al., 1992) and is essential for embryogenesis (Hart et al., 2000; Masuya et al., 2005; Spyropoulos et al., 2000). During lymphocyte development, Fli1 is first detected in the developing spleen and thymus of the embryo and later in mature B cells and throughout T cell development (Anderson et al., 1999; Melet et al., 1996), but is down-regulated in activated T cells (Mao et al., 1994; Zhang and Watson, 2005).
Accumulating evidence suggests strongly that Fli1 plays a key role in lymphocyte development and immune system regulation. Over-expression of Fli1 in otherwise healthy mice results in an abnormal expansion of autoreactive lymphocytes, production of autoantibodies and the development of an autoimmune kidney disease similar to that observed in murine models of lupus (Zhang et al., 1995). Subsequently, it was observed that Fli1 is over-expressed in the lymphocytes of lupus prone mouse models MRL/lpr and NZB/NZW f1 (Georgiou, 1996; Zhang et al., 2004) and in PBMCs from human lupus patients (Georgiou, 1996). Interestingly, reducing Fli1 levels by approximately 50% in the MRL/lpr mouse model decreased autoantibody production, decreased proteinuria and pathological renal scores, altered the numbers of CD8+ and naive T cells and significantly increased survival (Zhang et al., 2004). Together, these observations indicate that Fli1 expression levels play a modulatory role in the pathogenesis of lupus at least partially due to effects on lymphocyte function.
It is currently unknown how Fli1 is regulated or functions in lymphocytes. What little is known about the transcriptional regulation of Fli1 is based on studies conducted largely in non-lymphocyte cell lines (Barbeau et al., 1999; Barbeau et al., 1996; Lelievre et al., 2002; Starck et al., 1999) and may not reflect accurately how Fli1 gene expression is controlled in lymphocytes. We have identified a 230 bp region of the Fli1 promoter capable of driving Fli1 expression in a murine B cell line and a murine T cell line. Within this region, we identified four EBSs, a GATA binding site, and two STAT binding sites that contribute to promoter activity in these cells and determined that Elf1, Tel and Fli1 can bind three of the EBSs and activate Fli1 gene expression. Interestingly, we identified a polymorphic microsatellite in lupus prone strains and observed that the length of the microsatellite is inversely correlated with promoter activity in a T cell line.
Materials and Methods
Cell culture
CH1 B cells and S1A.TB.4.8.2 (S1A) T cells were obtained from American Type Culture Collection (TIB221- and TIB-27, respectively). CH1 B cells were maintained in RPMI 1640 supplemented with 10 mM HEPES, 0.05 mM betamercaptoethanol and 10% FBS. S1A T cells were maintained in RPMI 1640 supplemented with 10% FBS.
Amplification of Fli1 sequences from genomic DNA
Genomic DNA was isolated from spleens or tail biopsies using DNeasy columns (Qiagen) following manufacturer’s instructions. The -1778 to +109 Fli1 promoter and upstream regulatory sequences were amplified from genomic DNA using primers KpnEx1bU 5’GGGGTACCACCGCCACTCCAGGTCTGG and BglEx1D 5’GGAAGATCTGGGAGAGCGGCCACGTCTTC. The GA microsatellite region was amplified using primers GAUp 5’AGTGATGCGAAAAGCAG and GADn 5’GCTAATTTTGGGAAGTGACT. Comparisons of the amplified products containing the microsatellite region were subjected to electrophoresis through a 5% NuSieve agarose gel. The -1778 to -37 sequences from one BALB/c, one MRL/lpr and one NZM2410 mouse were cloned into the pGL3 Basic vector (see below) and sequenced to determine the number of GA repeats. Amplifications from these three mice were used for references in estimating the number of repeats in each animal following electrophoresis.
Promoter/Reporter plasmid construction
Fli1 sequence -1778 to +109 (relative to transcription start site +1) was amplified from genomic DNA isolated from spleens of one BALB/c, one MRL/lpr and one NZM2410 mouse as indicated above and ligated into the KpnI and BglII sites of the pGL3 Basic reporter vector (Promega). Two independent clones were established and sequenced. The region between -1778 and -37 was amplified from the -1778/+109 constructs in PCR using the pGL3 RV3 primer and Fli1 specific primer Fli1-37 5’GGAAGATCTCACATTGACCCGGTTACA. The amplified fragment was ligated into the KpnI and BglII sites of pGL3 Basic to generate Fli1 promoter/reporter (P/R) constructs containing Fli1 sequences -1778/-37. The -1778/-37 vectors were then used to generate 5’ deletion constructs -1530/-37, -986/-37, and -505/-37 by enzymatic digestion and religation. Additional deletions within the BALB/c construct (-808/-37, -690/-37, -585/-37, -411/-37, -271/-37, and -209/-37) were generated by PCR using the BALB/c -1778/-37 vector with the pGL3L 5’AGATCGATAGAGAAATGTTCTGG lower primer and the upper primer Fli1-808U 5’GAGTAGGTTTGCCTTGATCTG, Fli1-690U 5’GAGGCTGGAAAAAGCATTA, Fli1-585U 5’GAGCGTTTGGCTTTGGATT, Fli1-411U 5’GAGCGAGTGATGCGAAAAG, Fli1-271U 5’GAGAGATAGGACTTCCTCC, or Fli1-209U 5’GAGTTTCATCCGGTTAACTGTC to amplify the entire vector exclude the deleted sequences. Two clones were generated for each deletion construct and all were verified by sequencing.
All mutations within putative binding motifs were generated by site-directed mutagenesis using the BALB/c -271/-37 construct as template in PCR. Complimentary primers containing mutations in putative binding motifs were used to amplify the entire -271/-37 construct while incorporating mutations. Bases mutated are indicated in Fig. 2B. Two independent clones were generated for all constructs and mutations were verified by sequencing.
Figure 2.

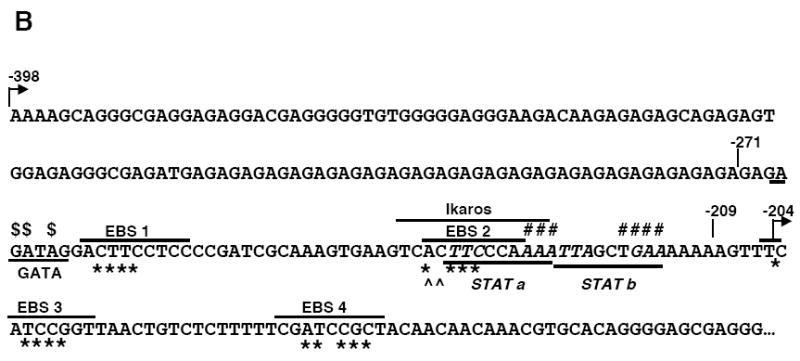
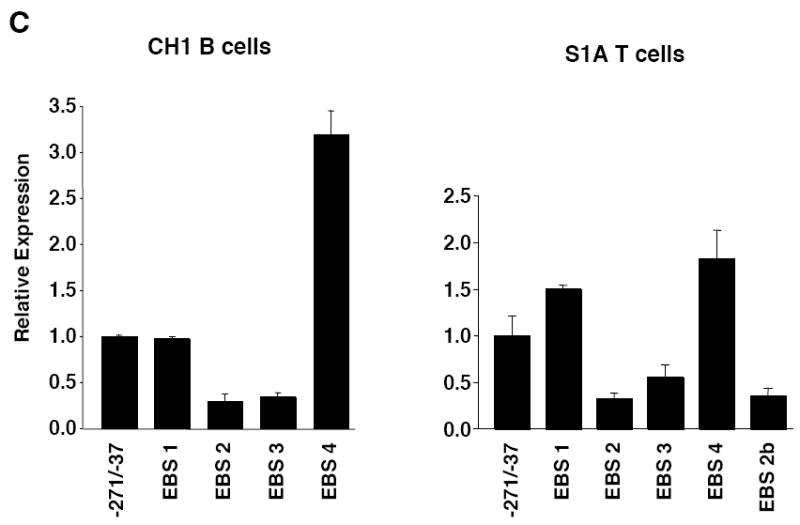
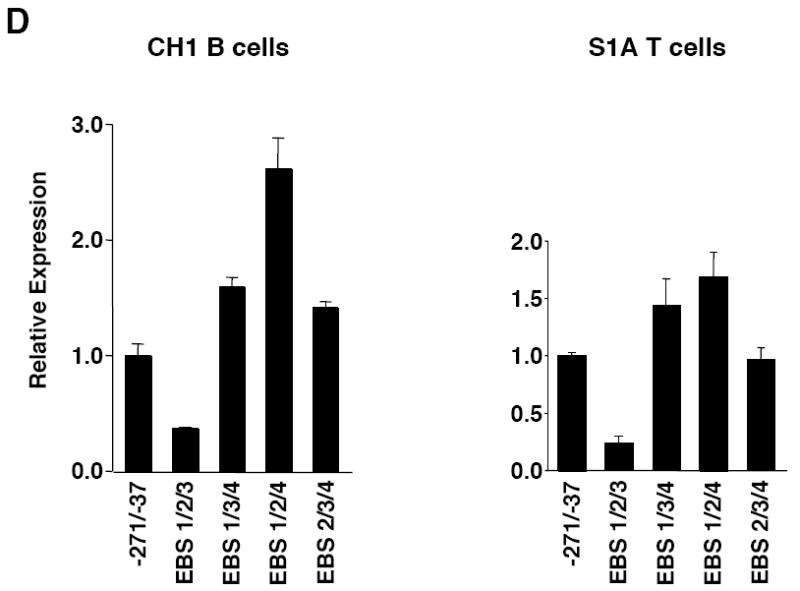
Mutational analysis of four putative Ets binding sites (EBS) in the Fli1 promoter. A) Sequence alignment showing the homology between the murine (M) and human (H) sequence in exon 1 (-398 to +18). Numbering is relative to the +1 translation start site of the murine sequence. Putative transcription factor binding sites are underlined and indicated above the sequence. The coding region of exon 1 is indicated by a shaded box. The mouse sequence was derived from direct sequencing of gDNA cloned from the BALB/c mouse. The human sequence was obtained from NCBI (Y17293.1). B) Location of putative transcription factor binding sites within the -271/-37 region of the murine sequence. Consensus sequences are underlined and mutations made within each consensus site are indicated by: * EBS1, 2, 3 or 4; ^ EBS2b; # STAT; and $ GATA. C) Transient transfection of pGL3 Fli1 -271/-37 containing mutations within each of the four EBSs into CH1 B cells and S1A T cells. D) Transient transfection of pGL3 Fli1 -271/-37 containing combinations of mutations within three of the four EBSs into CH1 B cells and S1A T cells. Results are representative of at least three independent transfections performed with two independently derived clones.
All primers were synthesized by Integrated DNA Technologies, Inc. (Iowa City, IA) and sequencing was performed by the Sequencing and Synthesis facility at Iowa State University (Ames, IA).
Expression Plasmids
The pcDNA3 Elf1 and pcDNA3 Fli1 expression vectors was obtained from Dr. Dennis Watson and the pcDNA3 Tel expression vector was obtained from Dr. Maria Trojanowska.
Transfections
The CH1 B cell line and S1A T cell line were transfected with the vectors indicated in the figure legends by nucleofection (Amaxa) following manufacturer’s instructions. To adjust for transfection efficiency, all transfections were normalized by co-transfection of a Renilla luciferase expression vector. For the transfections involving co-transfection of expression vectors, total molar amount of vector was kept constant with empty expression vector pcDNA3. Following transfection, cells were incubated for 18-24 h, harvested and analyzed for firefly and Renilla luciferase expression using the dual luciferase assay protocol (Promega) or used to isolate RNA for real-time RTPCR assays. All transfections were repeated as indicated in the figure legends with representative transfections shown. Vectors were purified using Qiagen tip columns.
Electrophoretic mobility shift assays (EMSA)
Complimentary Cy5.5 labeled oligos containing the GATA/EBS1, EBS2, EBS3 or EBS4 sites of the Fli1 promoter were synthesized by Integrated DNA Technologies, Inc. (Iowa City, IA) and annealed to generate probes. Sequences of the probes were as follows: GATA/EBS1 5’GAGAGAGAGATAGGACTTCCTCCCCGATCGC; EBS2 5’GAAGTCACTTCCCAAAATTA; EBS3 5’AAGTTTCATCCGGTTAACT; and EBS4 5’CTCTTTTTCGATCCGCTACAACA. Unlabeled oligos containing the same sequence or a consensus EBS were used as specific competitors and unlabeled oligos containing mutations within the binding motifs were used as nonspecific competitors. Mutations within each binding motif were identical to those made in the P/R constructs described above. Nuclear extracts were prepared from spleens of 10-12 week old BALB/c mice using the NE-Per nuclear extraction kit (Pierce). The GATA/EBS1, EBS3 and EBS4 probes were incubated with 10-15 μg nuclear extract and 1 μg poly dIdC in 10 mM HEPES, 10 mM Tris-HCl, 100 mM NaCl, 15 mM KCl, 0.5 mM EDTA, 1 mM DTT and 10% glycerol for 30 minutes at room temperature. The EBS2 probe was incubated with 15 μg nuclear extract and 1 μg poly dIdC in 20 mM HEPES, 0.2 mM EDTA, 100 mM KCl, 1 mM DTT, 10 μM ZnCL2 and 10% glycerol for 30 minutes at room temperature. Unlabeled specific and nonspecific competitors were added to the reaction prior to addition of the probe. For supershift assays, 1 μg of the indicated antibody was added to the reaction prior to addition of the probe. All antibodies were purchased from Santa Cruz. Reactions were run on a 4 or 5% non-denaturing 0.25X TBE 19:1 polyacrylamide gel. Gels were scanned on the Odyssey infrared imaging system (Li-Cor). All EMSAs were repeated at least three times with at least two independent preparations of nuclear extract with representative gels shown.
RNA isolation and real-time RTPCR assays
Approximately 18 hours after transfection of S1A T cells, total RNA was isolated using the RNeasy columns (Qiagen) following the manufacturer’s instructions. Real-time RTPCR was performed with each RNA sample on the MyiQ Biorad thermocycler using the iScript cDNA Synthesis and iQ SYBR Green Supermix kits (Biorad). Fli1 specific primers Fli1Ex1Up2 5’ACTTGGCCAAATGGACGGGACTAT and Fli1Ex2Dn 5’CCCGTAGTCAGGACTCCCCG were used to detect exon 1 initiated transcripts. Amplification of GAPDH transcripts using primers GAPDHFor 5’TGCACCACCAACTGCTTAGC and GAPDHRev 5’GGCATGGACTGTGGTCATGAG were used for normalization within each sample. The threshold cycle number (Ct) for each sample was measured by the Real-time iCycler iQ software (Biorad). Relative expression levels were then calculated using the Gene Expression Macro software (Biorad). Briefly, Ct values were normalized for each sample by subtracting the GAPDH Ct from the Fli1 Ct to give the ΔCt. A relative fold change was calculated using the calculation 2̂(ΔΔCt) where ΔΔCt is the difference between the ΔCt of the sample and the ΔCt of the reference sample. The empty vector (pcDNA3) transfected sample was used as the reference sample.
Statistical analysis
The non-paired student’s t-test was used to compare relative expression levels. A p value <0.05 was used to reject the null hypothesis. Error bars represent standard deviations in all figures.
Results
Three Ets binding sites contribute to positive and negative regulation in B and T cells
The Fli1 promoter region from -1778 to -37 was amplified from genomic DNA (gDNA) isolated from the spleen of a BALB/c mouse and ligated into the pGL3 Basic luciferase reporter vector. This region includes the distal and proximal promoters (Barat et al., 2000; Barbeau et al., 1996; Dhulipala et al., 1998). To define the sequences necessary to drive expression in lymphocytes, successive deletions from the 5’ end of the amplified Fli1 sequence were made, generating a series of Fli1 promoter/reporter (P/R) constructs (Fig. 1A). These constructs were transiently transfected into the CH1 B cell line and the S1A T cell line and luciferase expression analyzed. In order to analyze Fli1 regulation in cells similar to primary cells, we employed cell lines derived from tumors not induced by chemical or viral carcinogens and bearing properties similar to normal cells (Horibata and Harris, 1970; Lynes et al., 1978). In both the CH1 and S1A cell lines, sequences from -1778 to -505 were not required for promoter activity while deletion of the region between -271 and -209 resulted in little to no promoter activity in both cell lines (Fig. 1B and 1C). These results suggest that the region from -271 to -209 contains regulatory elements necessary for promoter activity in these cell lines. The region between -1778 and -505 may contain negative regulatory elements as promoter activity increased when this region was removed. The -271/-37 construct contains only one transcription start site at -204, which was identified as the major transcription start site in spleen (Starck et al., 1999). Sequence comparison of the mouse and human Fli1 sequences show high homology throughout this region (Fig. 2A). Therefore, further analyses focused on the -271/-37 region.
Figure 1.

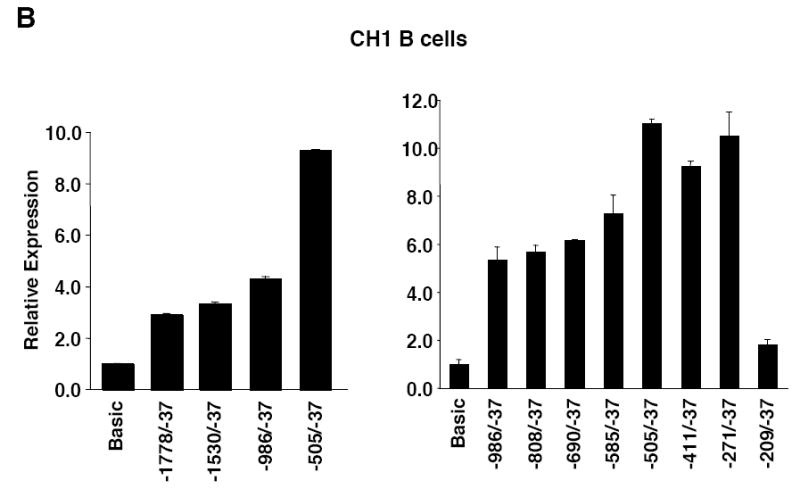
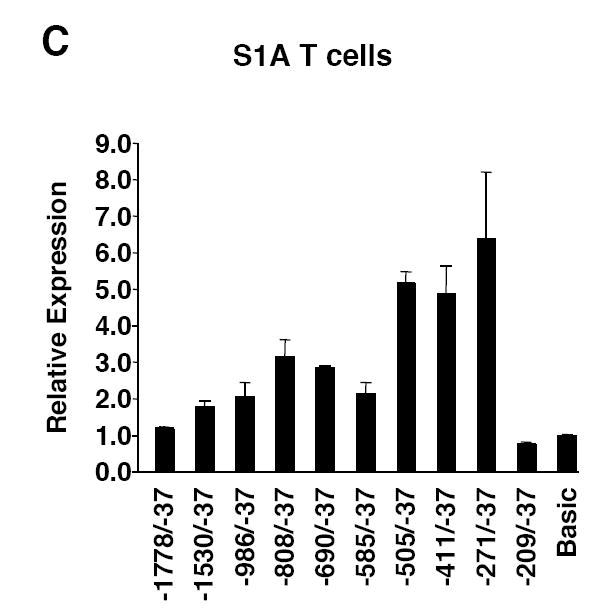
5’ deletion analysis of Fli1 promoter activity in B and T cells. A) Schematic of the Fli1 -1778 to -37 region. The Fli1 upstream regulatory region -1778 to -37 was amplified from genomic DNA isolated from a BALB/c spleen and ligated into the pGL3 Basic vector to generate the pGL3 Fli1 -1778/-37 promoter/reporter construct. Deletions were made from the 5’ end of the pGL3 Fli1 -1778/-37 construct to generate a series of promoter/reporter constructs. B) Transient transfection of the constructs shown in (A) into the CH1 B cell line. C) Transient transfection of the constructs shown in (A) into the S1A T cell line. Expression is presented relative to the pGL3 Basic empty vector, which was set to 1. Results are representative of at least three independent transfections performed with two independently derived clones.
Putative cis-regulatory elements were identified by scanning the -271 to -37 sequence for known transcription factor binding sites using the MatInspector program (Genomatix) and from studies conducted previously in other cell types (Barbeau et al., 1999; Barbeau et al., 1996; Hodge et al., 2002; Lelievre et al., 2002). Putative binding sites analyzed included four Ets binding sites (EBS1-4), two STAT binding sites (STATa and STATb) and a GATA binding site (Fig. 2B). To determine whether the putative cis-regulatory elements are important for promoter activity in lymphocytes, site-directed mutagenesis of the -271/-37 P/R construct was used to mutate the putative cis-elements within bases critical for binding (Fig. 2B). These constructs were transiently transfected into the CH1 B cell and S1A T cell lines. Mutations within either EBS2 or EBS3 resulted in a 40-50% decrease, while mutating EBS4 resulted in a 2-3-fold increase in promoter activity. Mutation of EBS1 had no effect on expression in the CH1 B cells and resulted in an approximate 1.5-fold increase in expression in the S1A T cells (Fig. 2C).
EBS2 overlaps with putative STAT and Ikaros binding sites. The base pair changes made in generating the EBS2 mutation potentially disrupts binding to all three sites. Therefore, to determine whether the Ets binding site specifically is necessary for promoter activity, the EBS2b mutation was generated. This mutation was engineered to impact binding to the Ets binding site only while leaving the STAT and Ikaros binding sites intact. The EBS2b mutation resulted in a decrease in expression in the B cell line (data not shown) and the T cell line (Fig. 2C) similar to that observed with the EBS2 mutation. Mutating two of the four EBSs (data not shown) or three of the four EBSs (Fig. 2D) in all possible combinations confirmed the data with the single EBS mutations in both cell lines. Together these results suggest that both EBS2 and 3 are positive regulatory elements and EBS4 is a negative regulatory element.
A GATA/EBS dual element contributes to positive regulation in B and T cells
Next we examined the possible function of the GATA site. The GATA site is immediately adjacent to the EBS1 site and together the sites comprise a potential GATA/EBS dual element, an element which has been shown to function cooperatively in the transactivation of other genes (Galson et al., 1993 ; Henkel and Brown, 1994). Similar to the results above with EBS1 site, mutation of the GATA site alone had little effect on promoter activity in either cell line. However, when both the EBS1 and GATA sites were mutated a 50% decrease in expression was observed in both cell lines (Fig. 3A). These results suggest that the GATA/EBS1 dual element may function in a cooperative manner in lymphocytes.
Figure 3.
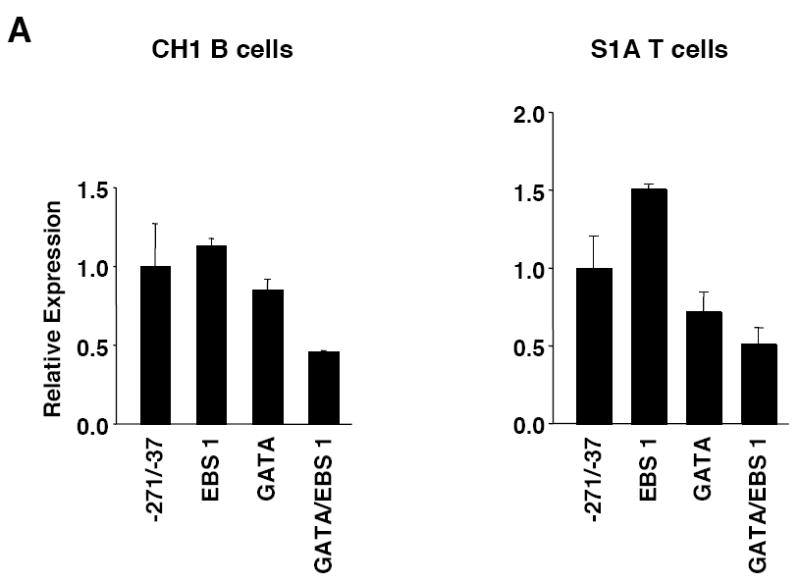
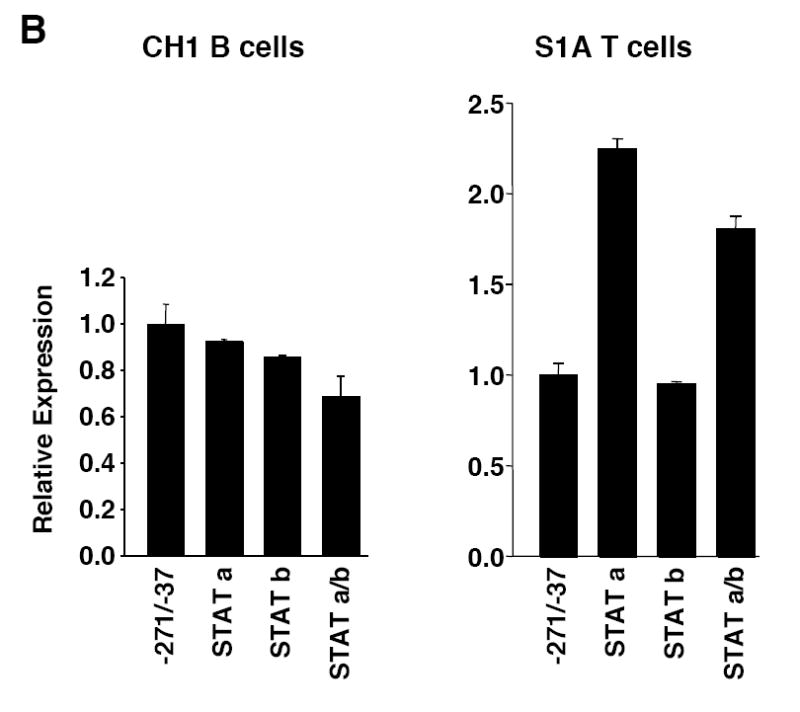
Mutational analysis of the putative GATA site and STAT sites. A) Transient transfection of pGL3 Fli1 -271/-37 containing mutations within the GATA site alone or in combination with the EBS1 site of the GATA/EBS dual element into CH1 B cells or S1A T cells. B) Transient transfection of pGL3 Fli1 -271/-37 containing mutations within each STAT site alone or in combination into CH1 B cells or S1A T cells. Results are representative of at least three independent transfections performed with two independently derived clones.
Two STAT binding sites function differently to regulate expression in B and T cells
The EBSs and GATA site functioned similarly in both the B and T cell lines; however, the STAT sites do not. In the B cell line, mutation of either the STATa or STATb site alone had little effect on promoter activity while mutation of both STAT sites (STATa/b) decreased promoter activity 20-25% (Fig. 3B). Conversely, in the T cell line, mutation of the STATa site alone resulted in a 2-fold increase, mutation of the STATb site had little effect, and mutation of both sites resulted in a nearly 2-fold increase in promoter activity (Fig. 3B). These results suggest that the STATa site may function as a negative regulatory element in T cells while the two STAT sites may serve as redundant positive regulatory elements in B cells.
Elf1, Tel and Fli1 bind to EBS1, EBS2 and EBS3, respectively, in vitro and stimulate Fli1 expression in vivo
Fli1 is co-expressed with several other Ets factors in lymphocytes. To identify which factors in lymphocytes bind to each of the EBSs in the -271/-37 region, nuclear extracts were isolated from BALB/c spleen and used in electrophoretic mobility shift assays (EMSA) without or with antibodies to Ets factors that are co-expressed with Fli1 in lymphocytes. Specific binding to probes containing the GATA/EBS1 dual element (Fig. 4A), EBS2 (Fig. 4B) and EBS3 (Fig. 4C) was detected to the EBSs of all three probes through the use of unlabeled specific and nonspecific competitors as indicated in the figures. No specific binding was observed using a probe containing EBS4 (data not shown). Addition of Elf1 antibodies to the binding reactions with the GATA/EBS1 probe resulted in the disappearance of the Ets specific shift (Fig. 4A), indicating Elf1 is able to bind the Ets site in the GATA/EBS1 element. Although the GATA/EBS1 probe was similar to that used in a previous study in which GATA1 binding was observed in extracts from another cell type (Barbeau et al., 1999), specific binding to the GATA site was not observed under the binding conditions that resulted in specific binding to EBS1 (Fig. 4A) nor under the binding conditions used in the previous study (data not shown). Ets specific binding to the EBS2 probe was supershifted by addition of Tel antibodies (Fig. 4B) and Ets specific binding to the EBS3 probe was supershifted by addition of Fli1 antibodies (Fig. 4C). These results indicate that three different Ets factors, including Fli1 itself, may be regulating Fli1 transcription in lymphocytes. To address whether these factors are able to activate the Fli1 promoter in vivo, Elf1, Tel and Fli1 expression plasmids were transfected into the S1A T cell line and endogenous Fli1 message levels were measured by real-time RTPCR. Our results indicate that over-expression of Fli1 and Tel significantly increased expression of the endogenous Fli1 gene (Fig. 5). Over-expression of Elf1 also resulted in a slight increase in Fli1 message levels, but the increase was not statistically significant. Together these results suggest that Fli1 and Tel can directly bind and regulate Fli1 gene expression in lymphocytes.
Figure 4.
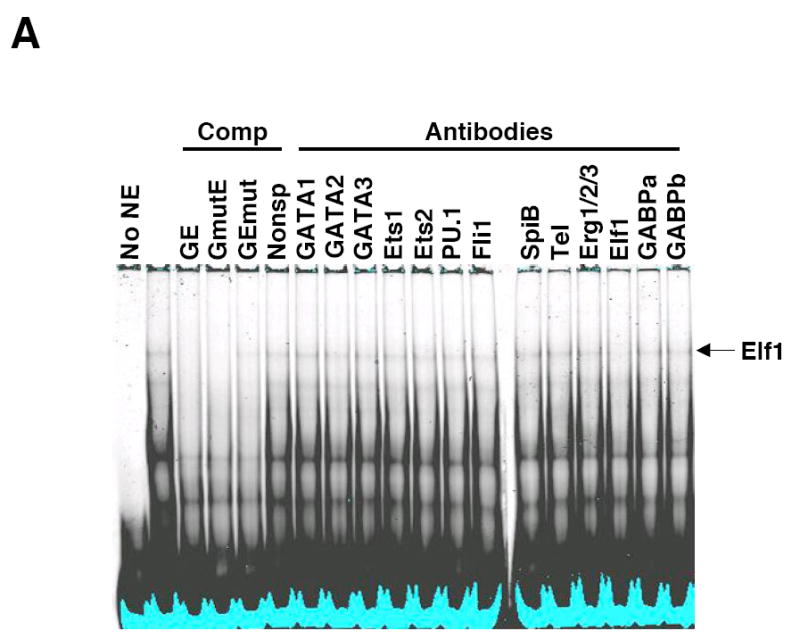
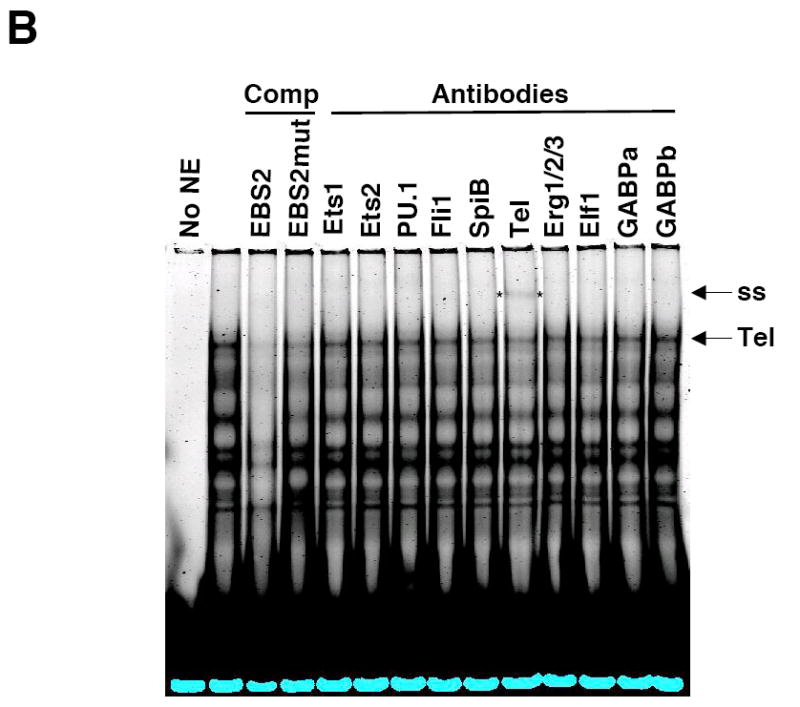
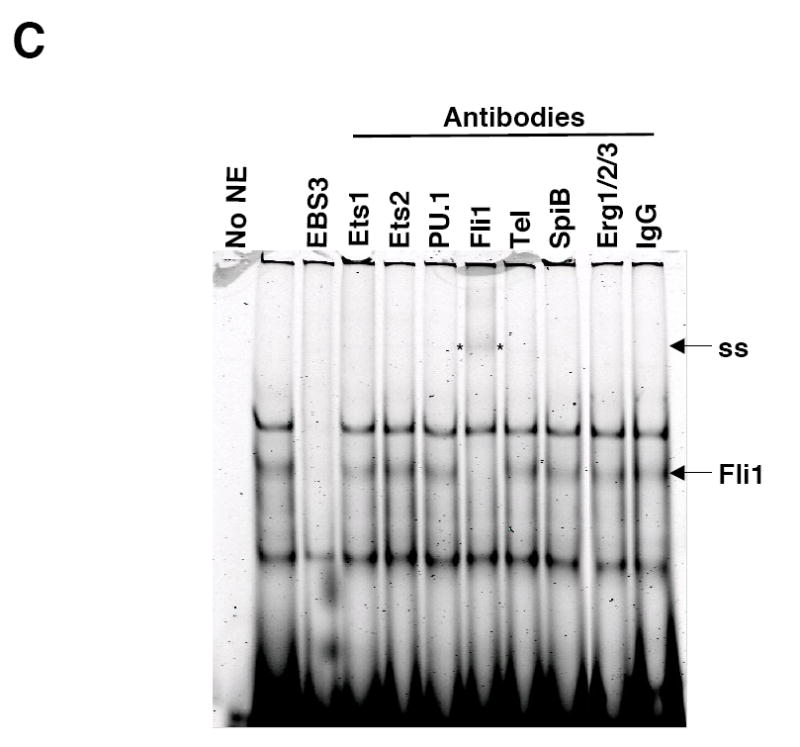
Elf1, Tel and Fli1 in splenic NE bind to EBS1, EBS2 and EBS3, respectively. Labeled oligos containing the GATA/EBS1 (A), EBS2 (B) or EBS3 (C) cis-regulatory elements from the murine Fli1 promoter were incubated with NE isolated from BALB/c spleens and unlabeled specific or nonspecific oligos as competitors (Comp) or the antibodies indicated. Elf1, Elf1 binding; Tel, Tel binding; Fli1, Fli1 binding; and ss, supershift are indicated to the right of each gel. Specific competitors: GE (GATA/EBS1), EBS2 and EBS3 are unlabeled wild type oligos. Nonspecific competitors: GmutE contains a wild type EBS1 site and mutated GATA site; GEmut contains a wild type GATA site and a mutated EBS1 site; EBS2mut, contains a mutation disrupting binding to the overlapping EBS2, STATa and Ikaros sites. EMSAs were performed at least three times with multiple NE preparations. EBS3 binding reactions with antibodies to Elf1, GABPα, and GABPβ and unlabeled nonspecific competitor containing a mutated EBS3 site showed no change in binding (data not shown).
Figure 5.
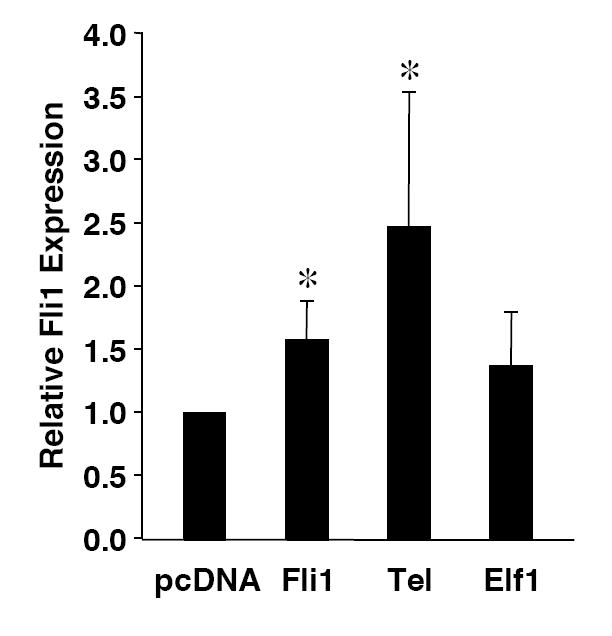
Over-expression of Elf1, Tel and Fli1 stimulate endogenous Fli1 expression. Real-time RTPCR for endogenous Fli1 transcripts was performed, as described in Materials and Methods, on RNA isolated from S1A T cells transfected with expression vectors for Elf1, Tel or Fli1 or the empty expression vector pcDNA3 as indicated. Endogenous Fli1 expression levels are presented relative to the expression level in the cells transfected with pcDNA3, which was set to 1. Two independent preparations of cDNA were prepared on RNA isolated following each transfection and real-time PCR was performed on each preparation of cDNA. Results are presented as an average of the four real-time PCR data. * p< 0.05.
Identification of a polymorphism in lupus prone mice that affects expression in a T cell line
To determine whether any polymorphisms are present in the promoters and upstream regulatory region of Fli1 in lupus prone mice that might impact the regulation of Fli1 transcription, the -1778 to -37 region was cloned from an MRL/lpr mouse and an NZM2410 mouse into pGL3 Basic. Upon comparison of the MRL/lpr and NZM2410 sequences to the BALB/c sequence cloned above, we identified polymorphisms in two microsatellites. One is located around -876 and consists of CTTT and CTTC repeats. The sequence cloned from the MRL/lpr mouse was one CTTT repeat longer and one CTTC shorter while the sequence cloned from the NZM2410 mouse was three CTTT repeats longer compared to the BALB/c sequence (data not shown). The other polymorphism is located at the 5’ end of exon 1 and consists of GA dinucleotide repeats. The GAn microsatellite was three repeats shorter in the MRL/lpr sequence (GA23) and eight repeats shorter in the NZM2410 sequence (GA18) compared to the BALB/c sequence (GA26) (Fig. 6A). The CTTT/CTTC microsatellite is not present in the human sequence while the GAn microsatellite is present in the human sequence and is located in a region exhibiting high homology with the mouse sequence (Fig. 2A). Therefore, further analyses focused on the GAn microsatellite.
Figure 6.
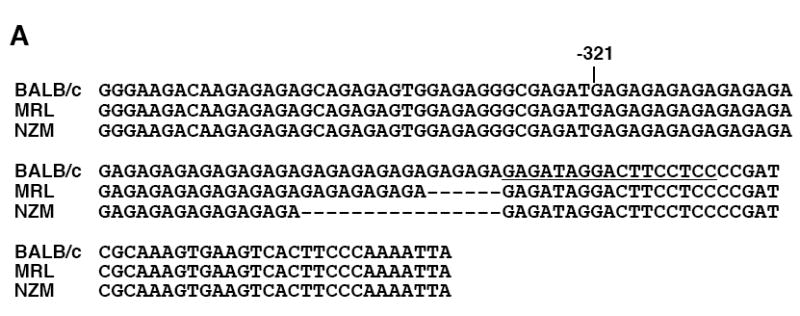
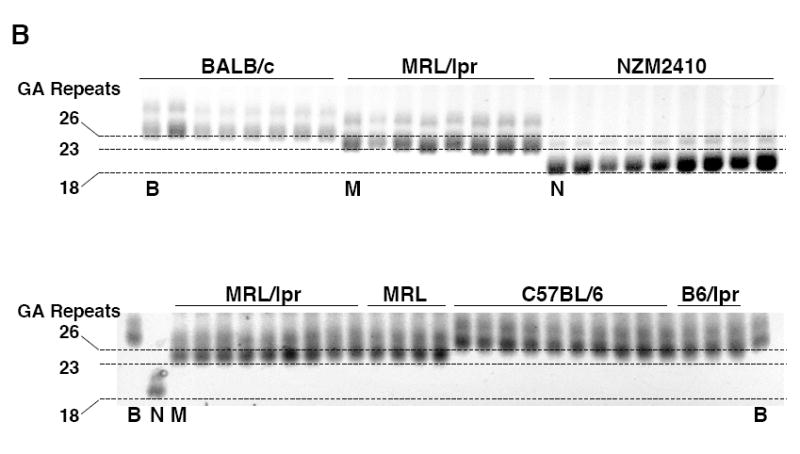
A GAn microsatellite in the Fli1 promoter is polymorphic in lupus prone mice. A) Sequence comparison of the Fli1 promoter region containing the GAn microsatellite from three mouse strains. The GAn microsatellite is located in the 5’ end of exon 1 and begins at -321. The location of the GATA/EBS1 dual element is located at the 3’ end of the microsatellite and is underlined. B) The GAn microsatellite region was amplified from genomic DNA isolated from two non-autoimmune prone strains BALB/c and C57BL/6 and three lupus prone strains MRL/lpr, MRL/mpj (MRL) and NZM2410 and run on a 5% agarose gel. Each lane is an individual animal from the indicated strain. The BALB/c (B), MRL/lpr (M) and NZM2410 (N) animals that were originally cloned and sequenced in (A) and contain 26, 23 and 18 repeats, respectively, are indicated underneath the lane. These samples were run on both gels for comparison and to estimate the number of GA repeats in the other samples.
To confirm this observation in lupus prone strains, we compared the length of the microsatellite in multiple animals from three lupus prone mouse strains and three non-autoimmune prone mouse strains. A 200 bp region containing the GAn microsatellite was amplified from gDNA isolated from BALB/c, C57BL/6 and C57BL/6/lpr non-autoimmune prone strains and MRL/lpr, MRL/mpj and NZM2410 lupus prone strains. The MRL/mpj and MRL/lpr strains are genetically similar except for the presence of the Fas defect in the MRL/lpr strain. Amplified products were analyzed by high percentage gel electrophoresis. The BALB/c, MRL/lpr and NZM2410 animals that were originally cloned and sequenced were used as references to approximate the number of repeats present in each animal. Our results indicate that the BALB/c animals had the longest GAn microsatellites consisting of approximately 25-27 repeats, the NZM2410 animals had the shortest with approximately 18-19 repeats, while the MRL/lpr animals had approximately 23-24 repeats (Fig. 6B). Our estimates of microsatellite length from the gels were confirmed by direct sequencing of the amplified products from four animals from each of the BALB/c, C57BL/6, MRL/lpr and NZM2410 strains.
To determine whether the length of the GAn microsatellite effects promoter activity, P/R constructs containing the regions -1530/-37, -986/-37 and -505/-37 were generated for the MRL/lpr and NZM2410 strains in the same manner as those for the BALB/c strain (Fig. 1A). These constructs, as well as the -1778/-37 constructs, were transiently transfected into the CH1 B and S1A T cell lines and their expression compared to the corresponding BALB/c constructs. The -1778/-37, -1530/-37 and -986/-37 constructs contain both the CTTT/CTTC and GAn polymorphic microsatellites while the -505/-37 constructs contain only the polymorphic GAn microsatellite. Direct sequencing of the constructs confirmed that the -505/-37 BALB/c, MRL/lpr and NZM2410 sequences are identical except for the number of GA repeats. Therefore, any differences in promoter activity of the -505/-37 constructs can be attributed to the difference in length of the GAn microsatellite. The BALB/c, MRL/lpr and NZM2410 constructs contain 26, 23 and 18 GA repeats, respectively. Expression of each of the constructs across strains was similar in the CH1 B cell line (Fig. 7A). However, in the S1A T cell line, we observed a 1.2-fold to 2-fold increase in promoter activity of the -1778/-37, -1530/-37 and -986/-37 constructs from the MRL/lpr and NZM2410 strains compared to the respective BALB/c constructs (Fig. 7B). These increases in promoter activity may be due to either or both of the polymorphic microsatellites. Interestingly, the -505/-37 MRL/lpr and NZM2410 constructs expressed approximately 1.5-fold and 2-fold higher, respectively than the -505/-37 BALB/c construct in the S1A T cells (Fig. 7B). As the length of the microsatellite is the only sequence difference in the -505/-37 constructs between the three strains, these results demonstrate an inverse relationship between the length of the GAn microsatellite and promoter activity in these cells.
Figure 7.
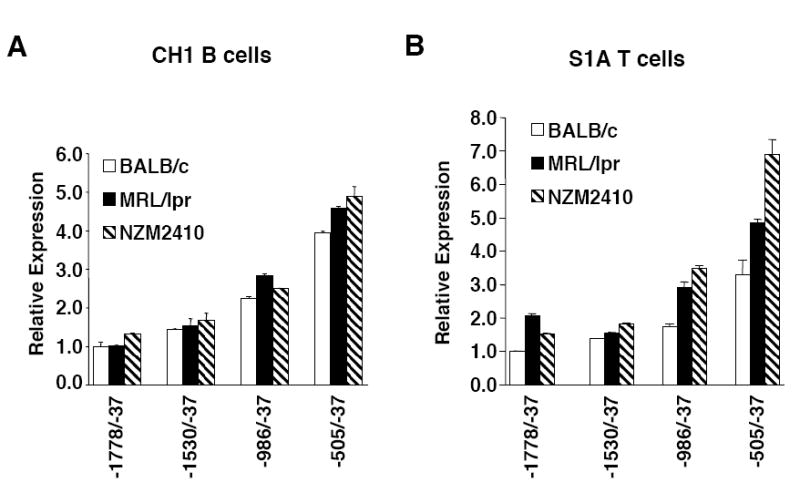
Analysis of GAn microsatellite length on Fli1 promoter activity. P/R constructs containing the BALB/c (GA26), MRL/lpr (GA23) or NZM2410 (GA18) sequences indicated were transiently transfected into the CH1 B cells (A) and S1A T cells (B). The -505/-37 P/R constructs are identical except for the length of the GAn microsatellite, whereas the other constructs contain another microsatellite that varies between the three strains. Results are representative of at least three independent transfections performed with two independently derived clones.
Discussion
The Fli1 gene does not map within or near any of the identified mouse or human lupus susceptibility loci (Lee and Nath, 2005; Morel et al., 1994). However, the induction of lupus by increasing Fli1 levels in non-autoimmune prone mice (Zhang et al., 1995) and reduction of disease by reducing Fli1 levels in lupus prone mice (Zhang et al., 2004) indicate strongly that the immune system and lupus disease progression are sensitive to the levels of Fli1. Gaining a better understanding of how Fli1 gene expression is controlled normally will be critical for determining how it becomes dysregulated in lupus. As little is known about how Fli1 is transcriptionally regulated, specifically in lymphocytes, we analyzed the promoters and upstream regulatory regions of Fli1 for cis-regulatory elements necessary for expression in lymphocytes.
Deletion analysis of the Fli1 promoter region in P/R constructs identified a region between -271 and -37 capable of driving expression in a B cell line and a T cell line. Site-directed mutagenesis of putative cis-regulatory elements within the -271/-37 construct identified several Ets binding sites, a GATA binding site and two STAT binding sites that contribute to promoter activity. Two of the Ets binding sites, EBS2 and EBS3 act as positive regulatory elements, while a third EBS, EBS4, acts as a negative regulatory element. A fourth EBS, EBS1, acts as a positive regulatory element in conjunction with the adjacent GATA site. These results were observed in both the B and T cell lines.
During lymphocyte development, Fli1 is co-expressed with several Ets factors and their expression is regulated in a lineage and stage-specific manner (Anderson et al., 1999). Fli1 expression levels are highest in pre-T and resting mature T cell stages (Anderson et al., 1999), with expression becoming down-regulated as T cells are activated (Zhang and Watson, 2005). Ets factors co-expressed with Fli1 in resting mature T cells include Ets1, Elf1, Ets2 and Tel1. In B cells, Fli1 is expressed at very low levels in the pro- and pre-B cells stages and highly at the mature B cell stage along with PU.1, Ets1, Ets2, SpiB, Erg, Elf1 and Tel1 (Anderson et al., 1999). Addition of antibodies to these Ets factors in our in vitro binding studies identified specific binding of Ets factors Elf1, Tel and Fli1 in splenic nuclear extracts to the Ets binding sites in EBS1, EBS2 and EBS3, respectively. However, some Ets factors do not readily bind in vitro (Hagman and Grosschedl, 1992). Therefore, it is possible that other Ets factors that were not observed to bind in vitro may preferentially bind these EBSs in vivo.
To determine whether Elf1, Tel and Fli1 are able to stimulate Fli1 expression in vivo, we over-expressed these factors in the S1A T cell line and measured the message levels of the endogenous Fli1 gene. We observed that over-expression of Tel and Fli1 significantly increased Fli1 expression. Elf1 also increased expression, but the increase was not statistically significant. We speculate that Elf1 may require the presence of a GATA factor for significant stimulation through the GATA/EBS dual element (see below). These results suggest that Fli1 may autoregulate its expression in lymphocytes. Indeed, Fli1 has been shown to bind and stimulate its own promoter in other cell lines (Lelievre et al., 2002; Starck et al., 1999) and to up-regulate endogenous Fli1 expression in Fli1 transgenic mice (Zhang et al., 1995).
EBS1 contains an Ets binding site that lies adjacent to a GATA binding site and composes a potential GATA/EBS dual element. GATA and Ets factors have been shown to bind to GATA/EBS dual elements and to function cooperatively to regulate expression of other genes (Galson et al., 1993; Henkel and Brown, 1994). Although the GATA/EBS dual element in the Fli1 promoter has been shown to act as an enhancer in an erythroid cell line and to bind GATA1 and Spi1/PU.1 in vitro, cooperative binding was not observed (Barbeau et al., 1999; Starck et al., 1999). In the CH1 B cells and S1A T cells, mutating either the Ets site or the GATA site alone had little effect on promoter activity. However, when both sites were mutated promoter activity decreased by 50%. These results suggest that the factors binding the dual element may be binding cooperatively and require only either the GATA site or the EBS for binding and transactivation. Alternatively, either a GATA factor or an Ets factor alone is sufficient for transactivation. Although we observed specific binding to EBS1 by Elf1 in splenic nuclear extracts, we did not observe specific binding to the GATA site under all conditions tested including conditions used to observe GATA1 binding to this element in another cell type (Barbeau et al., 1999).
The GATA and EBSs in the -271/-37 P/R construct functioned similarly in both the B and T cells. Alternatively, the function of the STAT sites varied between the B and T cells. The STATa site was not required for promoter activity in the B cell line but acted as a negative regulatory element in the T cell line. The STATa site overlaps with the Ets binding site in the EBS2 element. Because the Ets binding site acts as a positive regulatory element in both cell lines, these results suggest a possible competition for binding to the EBS2 element between an Ets factor for activation and a STAT factor for repression in T cells possibly during T cell activation. The STATb site located a few base pairs downstream of the STATa site also appeared to not be required for promoter activity in the B cell line. However, if both the STATa and STATb sites were mutated, promoter activity decreased. These results indicate that the two sites may serve a redundant function as a positive element in B cells. A previous study demonstrated that the endogenous human Fli1 gene, which contains an additional STAT site just upstream of the two STAT sites in the murine gene, is up-regulated in response to IL-6 in a human erythroleukemia cell line and that STAT3 activates human Fli1 P/R constructs in response to IL-6 (Hodge et al., 2002). However, stimulation of the CH1 B cells and S1A T cells with various concentrations of IL-6 did not result in any significant changes in expression of the endogenous Fli1 gene in either of the cell lines nor of wild type or STAT mutant P/R constructs in the CH1 B cells (data not shown). Together, our results suggest that the STAT sites may have different functions in B cells and T cells and that the IL-6/STAT 3 pathway may not regulate murine promoter activity in lymphocytes. However, activation of STAT factors through other pathways requires further investigation.
The EBS4 element contains an Ets binding site and functions as a negative element in both the B and T cell lines. No specific binding of factors from splenic nuclear extracts was observed under various binding conditions. It remains to be determined what binds this element to contribute to the repression of promoter activity. Future studies aimed at identifying the factor binding this element may be beneficial in determining how Fli1 is down-regulated during T cell activation and early development of B cells.
In addition to the cis-regulatory elements, we identified two polymorphic microsatellites in the Fli1 promoter region. Polymorphic dinucleotide microsatellites in or near promoters have been identified and found to regulate many other genes (Borrmann et al., 2003; Gebhardt et al., 1999; Shimajiri et al., 1999; Wang et al., 2005). One of the microsatellites that consists of GA repeats is highly conserved from mouse to human and is therefore likely to have human relevance. The GAn microsatellite was shorter in the MRL/lpr, MRL/lpj and NZM2410 lupus prone strains compared to the non-autoimmune prone BALB/c and C57BL/6 strains. Deleting the GAn microsatellite did not appear to have an impact on promoter activity in either a B cell line or a T cell line, indicating the microsatellite is not necessary for expression in these cells. However, the presence of the GAn microsatellite appears to modulate Fli1 promoter activity in the T cell line such that promoter activity is inversely correlated with the length of the microsatellite. This result is similar to those observed with a dinucleotide microsatellite in the promoter of the Cyr61 gene (Wang et al., 2005).
Together, these results suggest that the GAn polymorphism may be associated with over-expression of Fli1 in lupus mice and that the length of the microsatellite may have functional consequences in modulating Fli1 promoter activity in T cells. How the microsatellite functions to modulate promoter activity remains to be determined. Of particular significance, the GAn microsatellite appears to be polymorphic in the human population following a small study of 40 individuals (data not shown). Because this polymorphism is likely to be a subtle modifier of promoter activity and not sufficient alone to cause over-expression, a larger study of lupus patients is underway to determine whether there is any correlation between the length of the GAn microsatellite and aspects of disease.
It is unknown whether the over-expression of Fli1 in the lupus prone mice is due to dysregulation at the transcriptional, translational or post-translational level. Fli1 over-expression has been observed at both the mRNA and protein levels (Van Parijs et al., 1999; Zhang et al., 2004), indicating that over-expression is likely due to dysregulation of a transcriptional event. It is possible that more than one mechanism is involved in the over-expression of Fli1 in the different mouse strains. The GAn microsatellite is located in the 5’ end of exon 1 and could be involved in a post-transcriptional event. Further studies will be required to determine the exact role of this polymorphism in Fli1 gene expression.
In conclusion, we determined that in lymphocytes, Fli1 promoter activity is controlled by at least six cis-regulatory elements, including four putative Ets binding sites. Our results in lymphocytic cell lines are similar to those conducted in other cell types (Barbeau et al., 1999; Lelievre et al., 2002; Starck et al., 1999). However, in addition to Fli1 itself, we demonstrate the novel finding that Efl1 and Tel are able to bind to the Fli1 promoter and up-regulate its expression. Together, these results indicate that Elf1, Tel and Fli1 may be involved in regulating Fli1 gene expression in lymphocytes. Additionally, we identified a GAn microsatellite in the Fli1 promoter that is polymorphic in lupus prone mice and its length is inversely proportional to promoter activity in a T cell line. Because the immune system and lupus disease expression are sensitive to the levels of Fli1 (Georgiou, 1996; Zhang et al., 1995; Zhang et al., 2004), further studies to decipher the precise control of Fli1 gene expression in lymphocytes will be important for understanding the pathogenesis of lupus and also may provide insight into the role of Fli1 in hematopoietic malignancies.
Acknowledgments
The authors thank Dr. Dennis Watson and Dr. Angie Rizzino for critical reading of the manuscript and valuable comments. This work was supported in part by NIH grant DK072306, the Kirkland Scholar Program, the Medical University of South Carolina University Research Committee, Medical Research Service Ralph H. Johnson VAMC, and NIH grant AR47451.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Anderson MK, Hernandez-Hoyos G, Diamond RA, Rothenberg EV. Precise developmental regulation of Ets family transcription factors during specification and commitment to the T cell lineage. Development. 1999;126:3131–48. doi: 10.1242/dev.126.14.3131. [DOI] [PubMed] [Google Scholar]
- Barat C, Barbeau B, Delattre O, Rassart E. Presence of new alternative exons in human and mouse Fli-1 genes. Biochim Biophys Acta. 2000;1517:164–70. doi: 10.1016/s0167-4781(00)00239-6. [DOI] [PubMed] [Google Scholar]
- Barbeau B, Barat C, Bergeron D, Rassart E. The GATA-1 and Spi-1 transcriptional factors bind to a GATA/EBS dual element in the Fli-1 exon 1. Oncogene. 1999;18:5535–45. doi: 10.1038/sj.onc.1202913. [DOI] [PubMed] [Google Scholar]
- Barbeau B, Bergeron D, Beaulieu M, Nadjem Z, Rassart E. Characterization of the human and mouse Fli-1 promoter regions. Biochim Biophys Acta. 1996;1307:220–32. doi: 10.1016/0167-4781(96)00060-7. [DOI] [PubMed] [Google Scholar]
- Ben-David Y, Giddens EB, Bernstein A. Identification and mapping of a common proviral integration site Fli-1 in erythroleukemia cells induced by Friend murine leukemia virus. Proc Natl Acad Sci U S A. 1990;87:1332–6. doi: 10.1073/pnas.87.4.1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-David Y, Giddens EB, Letwin K, Bernstein A. Erythroleukemia induction by Friend murine leukemia virus: insertional activation of a new member of the ets gene family, Fli-1, closely linked to c-ets-1. Genes Dev. 1991;5:908–18. doi: 10.1101/gad.5.6.908. [DOI] [PubMed] [Google Scholar]
- Borrmann L, Seebeck B, Rogalla P, Bullerdiek J. Human HMGA2 promoter is coregulated by a polymorphic dinucleotide (TC)-repeat. Oncogene. 2003;22:756–60. doi: 10.1038/sj.onc.1206073. [DOI] [PubMed] [Google Scholar]
- Brown TA, McKnight SL. Specificities of protein-protein and protein-DNA interaction of GABP alpha and two newly defined ets-related proteins. Genes Dev. 1992;6:2502–12. doi: 10.1101/gad.6.12b.2502. [DOI] [PubMed] [Google Scholar]
- Dhulipala PD, Lee L, Rao VN, Reddy ES. Fli-1b is generated by usage of differential splicing and alternative promoter. Oncogene. 1998;17:1149–57. doi: 10.1038/sj.onc.1202030. [DOI] [PubMed] [Google Scholar]
- Fisher RJ, Koizumi S, Kondoh A, Mariano JM, Mavrothalassitis G, Bhat NK, Papas TS. Human ETS1 oncoprotein. Purification, isoforms, -SH modification, and DNA sequence-specific binding. J Biol Chem. 1992;267:17957–65. [PubMed] [Google Scholar]
- Galson DL, Hensold JO, Bishop TR, Schalling M, D’Andrea AD, Jones C, Auron PE, Housman DE. Mouse beta-globin DNA-binding protein B1 is identical to a proto-oncogene, the transcription factor Spi-1/PU.1, and is restricted in expression to hematopoietic cells and the testis. Mol Cell Biol. 1993;13:2929–41. doi: 10.1128/mcb.13.5.2929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gebhardt F, Zanker KS, Brandt B. Modulation of epidermal growth factor receptor gene transcription by a polymorphic dinucleotide repeat in intron 1. J Biol Chem. 1999;274:13176–80. doi: 10.1074/jbc.274.19.13176. [DOI] [PubMed] [Google Scholar]
- Georgiou P, Markoulakou IG, Green JE, Dantis P, Romano-Spica V, Kottardid S, Lautenberger JA, Watson DK, Papas TS, Fischinger PJ, Bhat NK. Expression of ets family of genes in systemic lupus erythematosus and Sjogren’s syndrome. International Journal of Oncology. 1996;9:9–18. [PubMed] [Google Scholar]
- Hagman J, Grosschedl R. An inhibitory carboxyl-terminal domain in Ets-1 and Ets-2 mediates differential binding of ETS family factors to promoter sequences of the mb-1 gene. Proc Natl Acad Sci U S A. 1992;89:8889–93. doi: 10.1073/pnas.89.19.8889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hart A, Melet F, Grossfeld P, Chien K, Jones C, Tunnacliffe A, Favier R, Bernstein A. Fli-1 is required for murine vascular and megakaryocytic development and is hemizygously deleted in patients with thrombocytopenia. Immunity. 2000;13:167–77. doi: 10.1016/s1074-7613(00)00017-0. [DOI] [PubMed] [Google Scholar]
- Henkel G, Brown MA. PU.1 and GATA: components of a mast cell-specific interleukin 4 intronic enhancer. Proc Natl Acad Sci U S A. 1994;91:7737–41. doi: 10.1073/pnas.91.16.7737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodge DR, Li D, Qi SM, Farrar WL. IL-6 induces expression of the Fli-1 proto-oncogene via STAT3. Biochem Biophys Res Commun. 2002;292:287–91. doi: 10.1006/bbrc.2002.6652. [DOI] [PubMed] [Google Scholar]
- Horibata K, Harris AW. Mouse myelomas and lymphomas in culture. Exp Cell Res. 1970;60:61–77. doi: 10.1016/0014-4827(70)90489-1. [DOI] [PubMed] [Google Scholar]
- Lee YH, Nath SK. Systemic lupus erythematosus susceptibility loci defined by genome scan meta-analysis. Hum Genet. 2005;118:434–43. doi: 10.1007/s00439-005-0073-1. [DOI] [PubMed] [Google Scholar]
- Lelievre E, Lionneton F, Mattot V, Spruyt N, Soncin F. Ets-1 regulates fli-1 expression in endothelial cells. Identification of ETS binding sites in the fli-1 gene promoter. J Biol Chem. 2002;277:25143–51. doi: 10.1074/jbc.M201628200. [DOI] [PubMed] [Google Scholar]
- Lynes MA, Lanier LL, Babcock GF, Wettstein PJ, Haughton G. Antigen-induced murine B cell lymphomas. I. Induction and characterization of CH1 and CH2. J Immunol. 1978;121:2352–7. [PubMed] [Google Scholar]
- Mao X, Miesfeldt S, Yang H, Leiden JM, Thompson CB. The FLI-1 and chimeric EWS-FLI-1 oncoproteins display similar DNA binding specificities. J Biol Chem. 1994;269:18216–22. [PubMed] [Google Scholar]
- Masuya M, Moussa O, Abe T, Deguchi T, Higuchi T, Ebihara Y, Spyropoulos DD, Watson DK, Ogawa M. Dysregulation of granulocyte, erythrocyte, and NK cell lineages in Fli-1 gene-targeted mice. Blood. 2005;105:95–102. doi: 10.1182/blood-2003-12-4345. [DOI] [PubMed] [Google Scholar]
- Melet F, Motro B, Rossi DJ, Zhang L, Bernstein A. Generation of a novel Fli-1 protein by gene targeting leads to a defect in thymus development and a delay in Friend virus-induced erythroleukemia. Mol Cell Biol. 1996;16:2708–18. doi: 10.1128/mcb.16.6.2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morel L, Rudofsky UH, Longmate JA, Schiffenbauer J, Wakeland EK. Polygenic control of susceptibility to murine systemic lupus erythematosus. Immunity. 1994;1:219–29. doi: 10.1016/1074-7613(94)90100-7. [DOI] [PubMed] [Google Scholar]
- Shimajiri S, Arima N, Tanimoto A, Murata Y, Hamada T, Wang KY, Sasaguri Y. Shortened microsatellite d(CA)21 sequence down-regulates promoter activity of matrix metalloproteinase 9 gene. FEBS Lett. 1999;455:70–4. doi: 10.1016/s0014-5793(99)00863-7. [DOI] [PubMed] [Google Scholar]
- Shore P, Sharrocks AD. The ETS-domain transcription factors Elk-1 and SAP-1 exhibit differential DNA binding specificities. Nucleic Acids Res. 1995;23:4698–706. doi: 10.1093/nar/23.22.4698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spyropoulos DD, Pharr PN, Lavenburg KR, Jackers P, Papas TS, Ogawa M, Watson DK. Hemorrhage, impaired hematopoiesis, and lethality in mouse embryos carrying a targeted disruption of the Fli1 transcription factor. Mol Cell Biol. 2000;20:5643–52. doi: 10.1128/mcb.20.15.5643-5652.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Starck J, Doubeikovski A, Sarrazin S, Gonnet C, Rao G, Skoultchi A, Godet J, Dusanter-Fourt I, Morle F. Spi-1/PU.1 is a positive regulator of the Fli-1 gene involved in inhibition of erythroid differentiation in friend erythroleukemic cell lines. Mol Cell Biol. 1999;19:121–35. doi: 10.1128/mcb.19.1.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urness LD, Thummel CS. Molecular interactions within the ecdysone regulatory hierarchy: DNA binding properties of the Drosophila ecdysone-inducible E74A protein. Cell. 1990;63:47–61. doi: 10.1016/0092-8674(90)90287-o. [DOI] [PubMed] [Google Scholar]
- Van Parijs L, Refaeli Y, Lord JD, Nelson BH, Abbas AK, Baltimore D. Uncoupling IL-2 signals that regulate T cell proliferation, survival, and Fas-mediated activation-induced cell death. Immunity. 1999;11:281–8. doi: 10.1016/s1074-7613(00)80103-x. [DOI] [PubMed] [Google Scholar]
- Wang B, Ren J, Ooi LL, Chong SS, Lee CG. Dinucleotide repeats negatively modulate the promoter activity of Cyr61 and is unstable in hepatocellular carcinoma patients. Oncogene. 2005;24:3999–4008. doi: 10.1038/sj.onc.1208550. [DOI] [PubMed] [Google Scholar]
- Wasylyk B, Hahn SL, Giovane A. The Ets family of transcription factors. Eur J Biochem. 1993;211:7–18. doi: 10.1007/978-3-642-78757-7_2. [DOI] [PubMed] [Google Scholar]
- Watson DK, Smyth FE, Thompson DM, Cheng JQ, Testa JR, Papas TS, Seth A. The ERGB/Fli-1 gene: isolation and characterization of a new member of the family of human ETS transcription factors. Cell Growth Differ. 1992;3:705–13. [PubMed] [Google Scholar]
- Zhang L, Eddy A, Teng YT, Fritzler M, Kluppel M, Melet F, Bernstein A. An immunological renal disease in transgenic mice that overexpress Fli-1, a member of the ets family of transcription factor genes. Mol Cell Biol. 1995;15:6961–70. doi: 10.1128/mcb.15.12.6961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang XK, Gallant S, Molano I, Moussa OM, Ruiz P, Spyropoulos DD, Watson DK, Gilkeson G. Decreased expression of the Ets family transcription factor Fli-1 markedly prolongs survival and significantly reduces renal disease in MRL/lpr mice. J Immunol. 2004;173:6481–9. doi: 10.4049/jimmunol.173.10.6481. [DOI] [PubMed] [Google Scholar]
- Zhang XK, Watson DK. The FLI-1 transcription factor is a short-lived phosphoprotein in T cells. J Biochem (Tokyo) 2005;137:297–302. doi: 10.1093/jb/mvi032. [DOI] [PubMed] [Google Scholar]


