Abstract
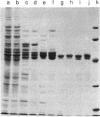
The nucleotide sequence of the celG gene of Clostridium thermocellum, encoding endoglucanase CelG, was determined. The open reading frame extended over 1,698 bp and encoded a 566-amino-acid polypeptide (molecular weight of 63,128) similar to the C. thermocellum endoglucanase CelB (51.5% identical residues). The N terminus displayed a typical signal peptide, followed by a catalytic domain. The C terminus, which was separated from the catalytic domain by a 25-amino-acid segment rich in Pro, Thr, and Ser, contained two conserved stretches of 22 amino acids closely similar to those previously described in other cellulases from the same organism. Expression of the gene in Escherichia coli was increased by fusing the fragment coding for the catalytic domain in frame with the start of the lacZ' gene present in the vector. A low- and a high-M(r) form of the protein were purified. The two forms displayed identical enzymatic properties. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis analysis showed that both forms consist of a major polypeptide of M(r) 50,000 and two minor polypeptides of M(r)s 49,000 and 48,000, resulting from heterogeneous proteolytic cleavage at the C terminus. An antiserum raised against the forms purified from E. coli reacted with an immunoreactive polypeptide of M(r) 66,000, which was associated with the extracellular cellulolytic complex of C. thermocellum known as the cellulosome.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beguin P., Cornet P., Millet J. Identification of the endoglucanase encoded by the celB gene of Clostridium thermocellum. Biochimie. 1983 Aug-Sep;65(8-9):495–500. doi: 10.1016/s0300-9084(83)80131-x. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Béguin P., Millet J., Aubert J. P. Cellulose degradation by Clostridium thermocellum: from manure to molecular biology. FEMS Microbiol Lett. 1992 Dec 15;100(1-3):523–528. doi: 10.1111/j.1574-6968.1992.tb14087.x. [DOI] [PubMed] [Google Scholar]
- Chauvaux S., Beguin P., Aubert J. P., Bhat K. M., Gow L. A., Wood T. M., Bairoch A. Calcium-binding affinity and calcium-enhanced activity of Clostridium thermocellum endoglucanase D. Biochem J. 1990 Jan 1;265(1):261–265. doi: 10.1042/bj2650261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durrant A. J., Hall J., Hazlewood G. P., Gilbert H. J. The non-catalytic C-terminal region of endoglucanase E from Clostridium thermocellum contains a cellulose-binding domain. Biochem J. 1991 Jan 15;273(Pt 2):289–293. doi: 10.1042/bj2730289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujino T., Béguin P., Aubert J. P. Cloning of a Clostridium thermocellum DNA fragment encoding polypeptides that bind the catalytic components of the cellulosome. FEMS Microbiol Lett. 1992 Jul 1;73(1-2):165–170. doi: 10.1016/0378-1097(92)90602-k. [DOI] [PubMed] [Google Scholar]
- Grépinet O., Béguin P. Sequence of the cellulase gene of Clostridium thermocellum coding for endoglucanase B. Nucleic Acids Res. 1986 Feb 25;14(4):1791–1799. doi: 10.1093/nar/14.4.1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grépinet O., Chebrou M. C., Béguin P. Nucleotide sequence and deletion analysis of the xylanase gene (xynZ) of Clostridium thermocellum. J Bacteriol. 1988 Oct;170(10):4582–4588. doi: 10.1128/jb.170.10.4582-4588.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grépinet O., Chebrou M. C., Béguin P. Purification of Clostridium thermocellum xylanase Z expressed in Escherichia coli and identification of the corresponding product in the culture medium of C. thermocellum. J Bacteriol. 1988 Oct;170(10):4576–4581. doi: 10.1128/jb.170.10.4576-4581.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall J., Hazlewood G. P., Barker P. J., Gilbert H. J. Conserved reiterated domains in Clostridium thermocellum endoglucanases are not essential for catalytic activity. Gene. 1988 Sep 15;69(1):29–38. doi: 10.1016/0378-1119(88)90375-7. [DOI] [PubMed] [Google Scholar]
- Hazlewood G. P., Davidson K., Clarke J. H., Durrant A. J., Hall J., Gilbert H. J. Endoglucanase E, produced at high level in Escherichia coli as a lacZ' fusion protein, is part of the Clostridium thermocellum cellulosome. Enzyme Microb Technol. 1990 Sep;12(9):656–662. doi: 10.1016/0141-0229(90)90004-a. [DOI] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Henrissat B., Claeyssens M., Tomme P., Lemesle L., Mornon J. P. Cellulase families revealed by hydrophobic cluster analysis. Gene. 1989 Sep 1;81(1):83–95. doi: 10.1016/0378-1119(89)90339-9. [DOI] [PubMed] [Google Scholar]
- Johnson E. A., Sakajoh M., Halliwell G., Madia A., Demain A. L. Saccharification of Complex Cellulosic Substrates by the Cellulase System from Clostridium thermocellum. Appl Environ Microbiol. 1982 May;43(5):1125–1132. doi: 10.1128/aem.43.5.1125-1132.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lamed R., Setter E., Bayer E. A. Characterization of a cellulose-binding, cellulase-containing complex in Clostridium thermocellum. J Bacteriol. 1983 Nov;156(2):828–836. doi: 10.1128/jb.156.2.828-836.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mead D. A., Szczesna-Skorupa E., Kemper B. Single-stranded DNA 'blue' T7 promoter plasmids: a versatile tandem promoter system for cloning and protein engineering. Protein Eng. 1986 Oct-Nov;1(1):67–74. doi: 10.1093/protein/1.1.67. [DOI] [PubMed] [Google Scholar]
- Navarro A., Chebrou M. C., Béguin P., Aubert J. P. Nucleotide sequence of the cellulase gene celF of Clostridium thermocellum. Res Microbiol. 1991 Nov-Dec;142(9):927–936. doi: 10.1016/0923-2508(91)90002-r. [DOI] [PubMed] [Google Scholar]
- Tokatlidis K., Salamitou S., Béguin P., Dhurjati P., Aubert J. P. Interaction of the duplicated segment carried by Clostridium thermocellum cellulases with cellulosome components. FEBS Lett. 1991 Oct 21;291(2):185–188. doi: 10.1016/0014-5793(91)81279-h. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson M. E. Compilation of published signal sequences. Nucleic Acids Res. 1984 Jul 11;12(13):5145–5164. doi: 10.1093/nar/12.13.5145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagüe E., Béguin P., Aubert J. P. Nucleotide sequence and deletion analysis of the cellulase-encoding gene celH of Clostridium thermocellum. Gene. 1990 Apr 30;89(1):61–67. doi: 10.1016/0378-1119(90)90206-7. [DOI] [PubMed] [Google Scholar]
- Zabarovsky E. R., Allikmets R. L. An improved technique for the efficient construction of gene libraries by partial filling-in of cohesive ends. Gene. 1986;42(1):119–123. doi: 10.1016/0378-1119(86)90158-7. [DOI] [PubMed] [Google Scholar]
- von Heijne G. Patterns of amino acids near signal-sequence cleavage sites. Eur J Biochem. 1983 Jun 1;133(1):17–21. doi: 10.1111/j.1432-1033.1983.tb07424.x. [DOI] [PubMed] [Google Scholar]