Abstract
Popliteal aneurysms are rare and tend to occur in older men with significant co-morbidity. Historically, management of popliteal aneurysms can be considered in three broad groups: (i) the technique of Antyllus; (ii) techniques relying upon a collateral circulation; and (iii) techniques involving maintenance or restoration of circulation. Bypass and exclusion is currently been challenged by endovascuiar techniques which show promise in selected cases.
Current controversies in popliteal aneurysms management are: when to repair asymptomatic aneurysms, what operation to do and how to manage acute thrombosis. These have been addressed by studying, prospectively, 73 patients presenting with 116 popliteal aneurysms. Diameter greater than 2 cm is often stated as being an indication for elective operation in asymptomatic popliteal aneurysms. However, distortion of the aneurysm appears to be at least as important as size in determining whether symptoms are likely to develop. Of 17 popliteal aneurysms followed for a median of 34 months with a diameter 2–3 cm and distortion less than 45°, none thrombosed. This is no worse than patency following elective bypass (P= 0.064). Popliteal aneurysms greater than 3 cm in diameter in patients who are unfit or who declined an operation were significantly more likely to develop thrombosis or any other symptom (P = 0.01 and P = 0.004, respectively). Popliteal aneurysms less than 3 cm in diameter with distortion less than 45° can safely be managed by ultrasound surveillance. Popliteal aneurysms with greater diameter or distortion are best operated upon.
Bypass, combined with proximal and distal ligation of the aneurysm, resulted in 5-year graft patency of 78% and 65% for popliteal aneurysms originally patent or thrombosed, respectively, with good long-term exclusion of the aneurysm.
In addition to the general complications of intra-arterial thrombolysis, acute deterioration of the limb during lysis appears to be a particular problem when dealing with thrombosed popliteal aneurysms. It occurs in about 13% of cases which compares with 2% when dealing with thrombosed grafts or native arteries. Intra-arterial thrombolysis for thrombosed popliteal aneurysms is associated with unacceptabiy high numbers of complications and thrombolysis should be reserved for intra-operative use only.
Keywords: Popliteal aneurysm, Distortion and size, Thrombosis, Treatment, Thrombolysis
Popliteal aneurysms are defined as localised dilatations of the popliteal artery greater than 2 cm in diameter or an increase of 1.5 times the normal arterial calibre.1,2 Popliteal aneurysms are the commonest peripheral artery aneurysm and have a prevalence of about 1% in men aged 65–80 years.3 They are uncommon in women. True aneurysms are mainly atherosclerotic in origin. Rare causes include mycotic aneurysm, Bechet's and Marfan's syndrome. The aneurysms may be single or multiple at any point along the popliteal artery. They are often associated with other aneurysms, e.g. abdominal aortic aneurysm (AAA) or contralateral popliteal aneurysms. They tend to occur in older men with significant co-morbidity. This needs to be borne in mind when planning treatment.
Historical overview4–6
Until the 20th century, the main indications for operation for popliteal aneurysms were pain, swelling of the leg and rupture of the aneurysm. Surgical treatment can be considered in three main groups.
Antyllus' technique
Antyllus, a Greek surgeon (3rd century AD) advocated treatment of aneurysms by proximal and distal ligation followed by opening the sac and evacuating its contents. For a popliteal aneurysm, the technique was to open and evacuate the aneurysm under tourniquet. The tourniquet was then released so that the popliteal artery entering and leaving the sac could be identified and the vessels ligated immediately above and below the aneurysm. This operation, deep behind the knee, without the benefits of modern anaesthesia and antiseptics would have been extremely dangerous. Wilmer, a London surgeon, stated in 1779 that ‘there is not, that I know, a single case upon record where that operation has succeeded’. Indeed, Percivall Pott (1714–1788), advocated amputation as being the best treatment for popliteal aneurysms that had become symptomatic.
Techniques relying on a collateral circulation
Kirkland, Murray and Desault all advocated proximal ligation of the aneurysm at its neck. Desault also described distal rather than proximal ligation. John Hunter's (1728–1793) contribution was to carry out proximal ligation remote from the aneurysm. Although a good story, it is doubtful whether Hunter carried out his famous experiment on the Richmond Park deer to examine collateral circulation before performing his first case. There is no record of the experiments having being done other than for studies on bone growth in any of John Hunter's case notes. Furthermore, there is no mention of these experiments in any contemporary lecture. They were not described until 1879 in a lecture by Richard Owen defending vivisection. Finally, development of a collateral circulation following arterial occlusion had been known for many years. The rationale for proximal ligation proposed by Hunter may well have been provoked by wanting to deal with normal, rather than diseased, vessels close to the aneurysm. Hunter described a case where he carried out a classical proximal ligation at the neck of a false symptomatic popliteal aneurysm. The operation was difficult and on the fifth day the artery ruptured where the ligature had been tied. Hunter stated: ‘I then took up the artery a little higher, where I was sure it was sound’. First carried out in December 1785, Hunter performed four more procedures. Three of the five operations were successful, with one patient surviving for 50 years.
Though widely acclaimed by several contemporary surgeons, results were often unpredictable and the procedure itself difficult. It is not surprising that occlusion of the aneurysm by non-operative means was attempted through most of the 19th century. These comprised three main groups of treatment – flexion, compression bandaging, and direct compression of the superficial femoral artery. The principle of flexion was to hold the limb in a position such that the knee and hip were tightly flexed. This could be used alone or after ligation of the popliteal aneurysms. It was ‘by no means painless’! Successful occlusion of the popliteal aneurysms and limb salvage were achieved in about half of the cases treated. Similar results were claimed by tight Esmarch bandaging of the limb. Compression of the superficial femoral artery in the thigh, so as to cause thrombosis in the aneurysm, could be achieved by digital pressure, application of weights, use of a compression device or by a combination of these means. By applying two compressors, one could be released and the other tightened to avoid skin damage. Of 148 popliteal aneurysms so treated and described in the British, Continental and American literature during 1870–1879, successful thrombosis was achieved in 68. The majority of the remainder went on to have ligation of the popliteal aneurysms. The average duration of treatment using a compression device was 12 days (range, 24 h to 7 weeks). This was slightly shorter with digital compression, but combining the two techniques took even longer to achieve thrombolysis. The longest recorded duration of treatment was 6 months! During a similar time frame, data from 67 popliteal aneurysms which underwent ligation were obtained from six London hospitals. The mortality was 15% but only two patients required amputation.
Rudolph Matas (1860–1957) advocated exposing and opening the aneurysm under tourniquet and suturing from within the sac all vessels entering and leaving it. The sac was then obliterated by successive layers of sutures; this he called obliterative endoaneurismorrhaphy.
Maintenance of circulation
Restorative endoaneurismorrhaphy was applied to saccular aneurysms. Once the sac had been opened and the contents evacuated, the connection between the main vessel and the aneurysm was sutured so as to restore flow. Reconstructive endoaneurismorrhaphy was used for fusiform aneurysms where continuity of the main artery was restored by making a new channel by suturing the walls of the sac together over a catheter. In 1906, Matas described the results of 19 popliteal aneurysms treated by endoaneurismorrhaphy.6 One patient died. There were no cases of secondary haemorrhage and none of gangrene. Two patients ultimately required late amputation.
The next type of operation described was excision of the aneurysm with restoration of circulation using popliteal vein (Goyanes, 1905), end-to-end anastomosis (Enderlen, 1907), reversed saphenous vein (Prlngle, 1913) and synthetic grafts (Crawford, 1957). The most commonly performed operation today, exclusion and vein bypass, was introduced by Edwards in 1969. Endovascular repair using an intravascular stent was described by Marln in 1994.
Clinical details
Between January 1988 and December 2004, all patients presenting with popliteal aneurysms were prospectively studied.8–10 Statistical analysis was carried out using a Mann Whitney U-test, χ2 test, Fisher's exact test and linear regression analysis, as appropriate.
Some 73 patients presented with 116 popliteal aneurysms, bilateral popliteal aneurysms being present in 43 patients (59%). There were 71 men and two women. The median age was 69 years (range, 46–89 years). Risk factors included hypertension (32), ischaemic heart disease (19), cerebrovascular accident (12), diabetes (5) and other significant co-morbidity in 31. At the time of presentation, 33 patients still smoked. From the time of diagnosis, 5-, 8- and 10-year survival was 80%, 48% and 42%, respectively. Median survival was 96 months.
Some 36 patients (49%) had co-existing abdominal aortic aneurysms (AAA), five had iliac and six femoral aneurysms. Some 45 patients presented with thrombosed popliteal aneurysms. In 31 of these cases, aneurysms were bilateral. Only 12 of 28 patients who did not present with a thrombosed popliteal aneurysms had bilateral aneurysms (χ2 4.83; df 1; P < 0.03).
At the time of initial diagnosis, 44 (38%) popliteal aneurysms were asymptomatic, 39 (34%) produced acute ischaemia (thrombosis 31, embolus 8), 29 (25%) intermittent claudication (thrombosed 16, patent 13) and four (3.4%) were producing pressure symptoms in the popliteal fossa. No patient presented with a ruptured popliteal aneurysm, though one aneurysm did rupture when a patient was being treated with heparin for a thrombosed popliteal aneurysm on the other side.
Whenever possible, patients underwent angiography (digital subtraction or magnetic resonance) and duplex scanning of their aneurysms. In addition to measuring diameter, degree of distortion was also measured. This was possible for patent aneurysms and for those thrombosed popliteal aneurysms which had been cleared, or partly cleared, with thrombolysis. The most proximal angle of distortion was measured with the angiogram in the anteroposterior plane.10
Natural history of asymptomatic popliteal aneurysms
Asymptomatic popliteal aneurysms develop symptoms at a rate of about 14% per year (range, 5–24%).11 Acute ischaemia occurs in about one-third of cases.12–18 This is usually due to thrombosis but thrombus from the aneurysm can embolise distally. About a quarter of popliteal aneurysms are associated with intermittent claudication, either from thrombosis, repeated micro-emboli or combined stenotic arterial disease. Pressure symptoms from large popliteal aneurysms can produce pain or discomfort behind the knee or swelling, with or without deep venous thrombosis, due to popliteal vein compression in about 5% of cases. Rupture nowadays is very rare.
Death and limb loss are both reported following elective popliteal aneurysms repair and about 1 % of patients will be left with residual symptoms.11 Five-year limb salvage and graft patency are about 90% and 80%, respectively. Despite this, approximately 25–50% of popliteal aneurysms which undergo operation are asymptomatic.12–18
Using a Markov decision analysis, we calculated that the average rate of developing symptoms was 14% per year and it was 16 months before a break-even point was reached such that the patient benefited from the operation.11 If aneurysms could be identified which are at high risk of developing symptoms, particularly acute ischaemia, then the time to break-even point would be shortened.
Risk factors for thrombosis
Popliteal aneurysms have been described as being ‘sinister harbinger of sudden catastrophe’.19 Paradoxically, the ‘sinister harbinger’ of acute thrombosis which we are now trying to prevent was the mainstay of treatment up until the start of the 20th century. In an attempt to predict those popliteal aneurysms, which are at high risk of thrombosis, a number of parameters have been examined.
Size
Elective repair of popliteal aneurysms greater than 2 cm in diameter is often advised although there is little evidence to support this suggestion. However, size does relate to symptoms.20 We found10 that the median diameter of asymptomatic popliteal aneurysms was 2.0 cm (range, 1.5–4.8 cm). This compared with 3 cm (range, 1.4–4.6 cm) for popliteal aneurysms producing acute ischaemia and 3.45 cm (range, 3.0–5.6 cm) for those producing compression (P = 0.0021 and P = 0.0045, respectively). There was no significant difference in size comparing asymptomatic with symptomatic aneurysms or those which had thrombosed or not thrombosed.
Distortion
As the popliteal artery increases in diameter, it increases in length. Since the artery is relatively fixed at the upper and lower ends it becomes distorted. As the diameter of the popliteal aneurysm increases so does the degree of distortion.10 We found that the median distortion of asymptomatic popliteal aneurysms was 0° (range, 0–90°). This compared with 60° (range, 0–180°) for popliteal aneurysms producing acute ischaemia and 45° (range, 45–90°) for those producing compression (P = 0.0012 and P = 0.012, respectively). Asymptomatic popliteal aneurysms showed less distortion than those producing any symptom (P = 0.0066) or acute ischaemia (P = 0.0012).
Combination of size and distortion
The best positive predictive values (i.e. number or thrombosed popliteal aneurysms correctly identified) and negative predictive values (i.e. number of non-thrombosed popliteal aneurysms correctly identified) were obtained by combining diameter greater than 3 cm with distortion greater than 45°.10
Thrombus within popliteal aneurysm and run-off
The larger the popliteal aneurysm, the more likely it is to contain thrombus within it.20 One study has shown a greater risk of complications developing in those popliteal aneurysms associated with absent distal pulses.21 It is postulated that embolisation from the aneurysm can gradually occlude runoff. Although the presence of thrombus within an aneurysm and poor run-off have been suggestive of indications for elective repair, there is no hard evidence to support this.
Management of non-thrombosed popliteal aneurysms
If the popliteal aneurysms were symptomatic, operation was advised providing the patient was fit.
Conservative management of asymptomatic popliteal aneurysms
LESS THAN 2 CM IN DIAMETER
There were 17 popliteal aneurysms in this group. Within a median follow-up of 19 months, five popliteal aneurysms enlarged to greater than 2 cm diameter and one other produced discomfort. A 1.5 cm popliteal aneurysm thrombosed but this was associated with a popliteal artery stenosis proximal to the popliteal aneurysm.
DIAMETER OF 2–3 CM
This is the important group as all would have had operation had a 2 cm diameter cut-off point been adopted. Provided there was no significant distortion (less than 45°), these patients were managed by ultrasound surveillance. Of 17 popliteal aneurysms followed up for a median of 34 months, none thrombosed. This was no worse than patency after bypass of non-thrombosed popliteal aneurysms (P = 0.064; Fig. 1). Three developed symptoms, all consisting of discomfort in the popliteal fossa and all underwent elective repair.
Figure 1.
Patency of 2–3 cm diameter popliteal aneurysms followed with surveillance compared with all bypass procedures on non-thrombosed popliteal aneurysms.
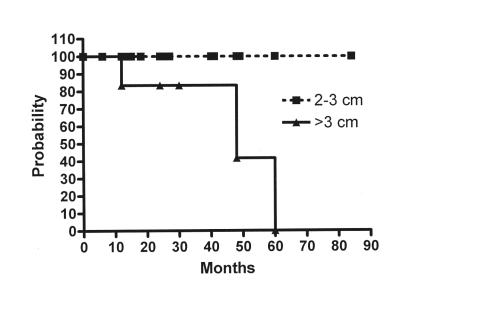
GREATER THAN 3 CM IN DIAMETER
Seven patients were unfit or declined operation. Of their seven aneurysms, three thrombosed and all became symptomatic with a median follow-up of 30 months. The likelihood of thrombosis or symptoms developing was significantly greater in these popliteal aneurysms than those in the 2–3 cm diameter group (P = 0.01 andP = 0.004, respectively; Figs 2 and 3).
Figure 2.
Freedom from thrombosis in popliteal aneurysms 2–3 cm diameter compared with >3 cm diameter.
Figure 3.
Freedom from any symptom in popliteal aneurysms 2–3 cm diameter compared with >3 cm diameter.
ULTRASOUND SURVEILLANCE INTERVALS
In a cohort of 24 popliteal aneurysms undergoing serial ultrasound scans, rate of growth increased with size and presence of hypertension.22 The upper 95% confidence interval for expansion of popliteal aneurysms less than 2 cm in diameter was 3 mm. Were 2 cm to be chosen as a cut-off point for consideration of operation then popliteal aneurysms less than 17 mm should undergo annual ultrasound scan and larger ones 6-monthly scans. Similarly, if 3 cm is the chosen cut off point then annual scans would be required for popliteal aneurysms less than 2.4 cm in diameter and 6-monthly scans if the aneurysm were larger.
Operative
A total of 28 operations were undertaken electively in non-thrombosed popliteal aneurysms. Of these, 12 popliteal aneurysms were totally asymptomatic. Symptoms in the remaining 16 were intermittent claudication, discomfort, embolus and leg swelling, each occurring in four cases. The median size of patent popliteal aneurysms undergoing operation was 2.5 cm (range, 1.5–5.6 cm) and the median distortion 30° (range, 0–180°). The aneurysm was ligated proximally and distally as close as possible to the aneurysmal sac and bypass carried out using a medial approach. Vein was used in 20 cases and all but three had popliteal-popliteal bypasses. There were no deaths, amputations or any other significant postoperative complications.
The 3-, 5- and 8-year patency was 78%, 78% and 51%, respectively. There was no difference in patency comparing asymptomatic with symptomatic popliteal aneurysms. During follow-up, no patient developed further symptoms from their ligated and bypassed aneurysm.
Endovascular repair of popliteal aneurysms has been suggested as an alternative to conventional repair despite initial results which were disappointing in terms of patency. However, more recently, a randomised trial25 and a large series of endovascular repairs24 have shown medium-term results, in selected cases, to be comparable with conventional bypass.
Management of thrombosed popliteal aneurysms
Forty-seven popliteal aneurysms thrombosed in 45 patients. This resulted in acute ischaemia in 31 and intermittent claudication in 16. One patient had bilateral metachronous thrombosis each producing claudication and another, who originally presented with claudication in one leg, subsequently developed acute ischaemia in the other.
Operation was undertaken for 38 of 47 popliteal aneurysms. The remainder were treated conservatively. Compared with elective repair of non-thrombosed popliteal aneurysms, more long bypasses (either from the femoral or to calf vessels) were carried out when the popliteal aneurysms had thrombosed (P < 0.0001). However, 3- and 5-year patency at 72% and 65%, respectively, was not significantly different from that achieved following bypass of non-thrombosed popliteal aneurysms (P = 0.56). Ligation and bypass with or without on-table thrombolysis was undertaken in 23 cases. Pre-operative lysis was undertaken in 15 cases. Seven patients experienced eight significant complications in association with pre-operative lysis. Four sustained bleeding from the groin and another patient had both bleeding from the groin and haematemesis. Lysis had to be abandoned in each case. There were three examples of acute deterioration during thrombolysis.
Overall, 16 of 38 patients with thrombosed popliteal aneurysms sustained 17 peri- or postoperative complications. In addition to those described above, two patients experienced footdrop, six wound problems, one a deep vein thrombosis and a fatal myocardial infarction. By contrast no patient had significant postoperative complications when undergoing elective repair (P < 0.0001 Fisher's exact test).
Four major amputations were required in patients presenting with thrombosis.
Acute deterioration of ischaemia during thrombolysis
General complications of intra-arterial thrombolysis include major bleeding (5% of cases) and, in particular, intracranial bleeding (1 %) which can be fatal.25 Acute deterioration of the limb during thrombolysis seems to be a particular problem when dealing with popliteal aneurysms. In a multicentre study involving 866 patients, we found that 13% of patients undergoing lysis for thrombosed popliteal aneurysms experienced this condition compared with about 2% of those undergoing thrombolysis of native arteries or blocked grafts.28 This may be due to destabilisation of a large volume of thrombus within the popliteal aneurysm and its subsequent embolisation. Since the aim of thrombolysis is to clear run-off, on-table appears to be a better option than pre-operative lysis. Under these circumstances, the lytic agent can be infused into the calf vessels whilst the upper dissection and anastomosis are being performed.27
Fate of the excluded aneurysm
There have been a few reports suggesting that following ligation and bypass up to one-third of popliteal aneurysms may retain or develop flow postoperatively. Symptoms have been described in 8–17%.13,15,28,29 However, we found that, following proximal and distal ligation and bypass, no popliteal aneurysm developed either symptoms or ultrasound-detected flow.30
Conclusions
The main controversies surrounding the management of popliteal aneurysms are when to carry out repair of asymptomatic popliteal aneurysms, what procedure to perform and how to manage acute thrombosis. The results of this paper show that asymptomatic popliteal aneurysms less than 3 cm in diameter with distortion of less than 45° can be safely managed by ultrasound surveillance.
Bypass, preferably using vein with proximal and distal ligation of the popliteal aneurysm is the operation of choice and provides 5-year patency of 70–90% with good exclusion of the aneurysm. However, endovascular procedures may be appropriate in some cases. Whether this form of treatment stands the test of time remains to be seen.
Patients with acute ischaemia due to thrombosis of popliteal aneurysms should be immediately treated with intravenous heparin and, following assessment, undergo operation, if appropriate. In addition to the recognised complications of thrombolysis, acute deterioration during treatment seems to be a particular problem when dealing with popliteal aneurysms. Thrombolysis should be restricted to attempted clearance of run-off on-table carried out whilst the upper anastomosis is being performed.
Acknowledgments
This paper is based on a Hunterian Lecture delivered at the Royal Society of Medicine, 7 December 2005.
References
- 1.Szilagyi DE, Schwartz RL, Reddy DJ. Popliteal artery aneurysms. Their natural history and management. Arch Surg. 1981;116:724–8. doi: 10.1001/archsurg.1981.01380170194034. [DOI] [PubMed] [Google Scholar]
- 2.Hollier LH, Stanson AW, Gloviczki P, Pairolero PC, Joyce JW, Bernatz PE, et al. Arteriomegaly: classification and morbid implications of diffuse aneurysmal disease. Surgery. 1983;93:700–8. [PubMed] [Google Scholar]
- 3.Trickett JP, Scott RAP, Tilney HS. Screening and management of asymptomatic popliteal aneurysms. J Med Screen. 2002;9:92–3. doi: 10.1136/jms.9.2.92. [DOI] [PubMed] [Google Scholar]
- 4.Barwell R Aneurism., editor; Ashurst J, editor. Encyclopaedia of Surgery. 1883;vol. 3:375–544. [Google Scholar]
- 5.Schechter DC, Bergan JJ. Popliteal aneurysms: a celebration of the bicentennial of John Hunter's operation. Ann Vasc Surg. 1986;1:118–26. doi: 10.1016/S0890-5096(06)60712-7. [DOI] [PubMed] [Google Scholar]
- 6.Stevenson LG. The stag of Richmond Park: a note on John Hunter's most famous animal experiment. Bull Hist Med. 1948;22:467–75. [Google Scholar]
- 7.Matas R. The radical cure of aneurysm. JAMA. 1906;47:990–4. [Google Scholar]
- 8.Ramesh S, Michaels JA, Galland RB. Popliteal aneurysm: morphology and management. Br J Surg. 1993;80:1531–3. doi: 10.1002/bjs.1800801212. [DOI] [PubMed] [Google Scholar]
- 9.Galland RB, Magee TR. Management of popliteal aneurysm. Br J Surg. 2002;89:1382–5. doi: 10.1046/j.1365-2168.2002.02221.x. [DOI] [PubMed] [Google Scholar]
- 10.Galland RB, Magee TR. Popliteal aneurysms: distortion and size related to symptoms. Eur J Vasc Endovasc Surg. 2005;30:534–8. doi: 10.1016/j.ejvs.2005.05.021. [DOI] [PubMed] [Google Scholar]
- 11.Michaels JA, Galland RB. Management of asymptomatic popliteal aneurysms: the use of a Markov decision tree to determine the criteria for a conservative approach. EurJ Vasc Surg. 1993;7:136–43. doi: 10.1016/s0950-821x(05)80753-4. [DOI] [PubMed] [Google Scholar]
- 12.Bowrey DJ, Osman H, Gibbons CP, Blackett RL. Atherosclerotic popliteal aneurysms: management and outcome in forty-six patients. EurJ Vasc Endovasc Surg. 2003;25:79–81. doi: 10.1053/ejvs.2002.1781. [DOI] [PubMed] [Google Scholar]
- 13.Ebaugh JL, Morasch MD, Matsumura JS, Eskandar MK, Meadows WS, Pearce WH. Fate of excluded popliteal artery aneurysms. J Vasc Surg. 2003;37:954–9. doi: 10.1067/mva.2003.258. [DOI] [PubMed] [Google Scholar]
- 14.Mahmood A, Salaman R, Sintler M, Smith S, Simms M, Vohra R. Surgery of popliteal artery aneurysms: a 12-year experience. J Vasc Surg. 2003;37:586–93. doi: 10.1067/mva.2003.141. [DOI] [PubMed] [Google Scholar]
- 15.Kirkpatrick UJ, McWilliams RG, Martin J, Brennan JA, Gilling-Smith GL, Harris PL. Late complications after ligation and bypass for popliteal aneurysm. Br J Surg. 2004;91:174–7. doi: 10.1002/bjs.4426. [DOI] [PubMed] [Google Scholar]
- 16.Blanco E, Serrano-Hernando J, Monux G, Martin A, Rial R, Reina T, et al. Operative repair of popliteal aneurysms: effect of factors related to the bypass procedure on outcome. Ann Vasc Surg. 2004;18:86–92. doi: 10.1007/s10016-003-0062-6. [DOI] [PubMed] [Google Scholar]
- 17.Aulivola B, Hamdan A, Hile C, Sheahan MG, Skillman JJ, Campbell DR, et al. Popliteal artery aneurysms: a comparison of outcomes in elective versus emergent repair. J Vasc Surg. 39(6):1171–7. doi: 10.1016/j.jvs.2003.12.023. [DOI] [PubMed] [Google Scholar]
- 18.Laxdal E, Amundsen S, Dregelid E, Pedersen G, Aune S. Surgical treatment of popliteal artery aneurysms. Scand J Surg. 2004;93:57–60. doi: 10.1177/145749690409300112. [DOI] [PubMed] [Google Scholar]
- 19.Guvendik L, Bloor K, Charlesworth D. Popliteal aneurysm: sinister harbinger of sudden catastrophe. Br J Surg. 1980;67:294–6. doi: 10.1002/bjs.1800670422. [DOI] [PubMed] [Google Scholar]
- 20.Varga ZA, Locke-Edmunds JC, Baird RN. A multicenter study of popliteal aneurysms. J Vasc Surg. 1994;20:171–7. doi: 10.1016/0741-5214(94)90003-5. [DOI] [PubMed] [Google Scholar]
- 21.Dawson I, Sie R, van Baalen JM, van Bockel JH. Asymptomatic popliteal aneurysm: elective operation versus conservative follow-up. Br J Surg. 1994;81:1504–7. doi: 10.1002/bjs.1800811035. [DOI] [PubMed] [Google Scholar]
- 22.Pittathankal AA, Dattani R, Magee TR, Galland RB. Expansion rates of asymptomatic popliteal artery aneurysms. Eur J Vasc Endovasc Surg. 2004;27:382–4. doi: 10.1016/j.ejvs.2004.01.014. [DOI] [PubMed] [Google Scholar]
- 23.Antonello M, Frigatti P, Battocchio P, Lepidi S, Cognolato D, Dall'Antonia A, et al. Open repair versus endovascular treatment for asymptomatic popliteal artery aneurysm: results of a prospective randomized study. J Vasc Surg. 2005;42:185–93. doi: 10.1016/j.jvs.2005.04.049. [DOI] [PubMed] [Google Scholar]
- 24.Tielliu I, Verhoeven ELG, Zeebregts CJ, Prins TR, Span MM, van den Dungen JJ. Endovascular treatment of popliteal artery aneurysms: results of a prospective cohort study. J Vasc Surg. 2005;41:561–7. doi: 10.1016/j.jvs.2004.12.055. [DOI] [PubMed] [Google Scholar]
- 25.Berridge DC, Makin GS, Hopkinson BR. Local low dose intra-arterial thrombolytic therapy: the risk of stroke or major haemorrhage. Br J Surg. 1989;76:1230–3. doi: 10.1002/bjs.1800761204. [DOI] [PubMed] [Google Scholar]
- 26.Galland RB, Earnshaw JJ, Baird RN, Lonsdale RJ, Hopkinson BR, Giddings AEB, et al. Acute limb deterioration during intra-arterial thrombolysis. Br J Surg. 1993;80:1118–20. doi: 10.1002/bjs.1800800914. [DOI] [PubMed] [Google Scholar]
- 27.Thompson JF, Beard J, Scott DJ, Earnshaw JJ. Intraoperative thrombolysis in the management of thrombosed popliteal aneurysms. Br J Surg. 1993;80:858–9. doi: 10.1002/bjs.1800800714. [DOI] [PubMed] [Google Scholar]
- 28.Jones WT, Hagino RT, Chiou AC, Decaprio JD, Franklin KS, Kashyap VS. Graft patency is not the only clinical predictor of success after exclusion and bypass of popliteal artery aneurysms. J Vasc Surg. 2003;37:392–8. doi: 10.1067/mva.2003.30. [DOI] [PubMed] [Google Scholar]
- 29.Mehta M, Champagne B, Darling C, Roddy S, Kreienberg P, Ozsvath K, et al. Outcome of popliteal artery aneurysms after exclusion and bypass: Significance of residual patent branches mimicking type II endoleaks. J Vasc Surg. 2004;40:886–90. doi: 10.1016/j.jvs.2004.08.029. [DOI] [PubMed] [Google Scholar]
- 30.Box B, Adamson, M, Magee TR, Galland RB. Outcome following bypass and proximal and distal ligation of popliteal aneurysms. Br J Surg. 2007;94:179–182. doi: 10.1002/bjs.5577. [DOI] [PubMed] [Google Scholar]